Abstract
Introduction:
Upper limb dysfunction is common in people with Parkinson’s. This study aimed to pilot and establish the initial psychometric properties of the timed upper limb assessment (TULA), a 10-item timed functional upper limb assessment tool.
Method:
A prospective observational study was conducted with 381 participants (202 with Parkinson’s, 179 controls). The TULA was administered to both groups, with inter-rater reliability assessed in a subset (n = 47). Principal component analysis examined structural validity, while convergent validity was explored through correlations with cognitive and motor assessments.
Results:
The TULA demonstrated high internal consistency (Cronbach’s α = 0.81 for Parkinson’s group) and high inter-rater reliability (intraclass correlation coefficient (ICC) > 0.9). People with Parkinson’s took significantly longer to complete all items (mean (SD) 22.2 ± 7.5) compared to controls (16.8 ± 3.7, p < 0.001). The tool showed moderate correlation with the timed up and go test (r = 0.56, p < 0.001), a significant low correlation with the Hoehn and Yahr Scale (r = 0.44, p ⩽ 0.001) and a significant low correlation with both the Montreal Cognitive Assessment (r = −0.37, p ⩽ 0.001) and the Mini Mental Status Examination (r = −0.31, p ⩽ 0.001).
Conclusion:
This study provides an alternative functional upper limb assessment tool for people with Parkinson’s attending occupational therapy. Initial data have been provided to support the reliability and validity of the TULA.
Introduction
Parkinson’s is a neurodegenerative disorder, typically characterised by bradykinesia, tremor, postural instability, and muscle rigidity (Lees et al., 2009). The risk of developing Parkinson’s increases as a person ages; however, approximately 10% of those diagnosed are under 45 years of age (Lees et al., 2009). Parkinson’s is the second most common neurodegenerative condition with an increasing prevalence rate (Zhu et al., 2024). Changes to upper limb functioning are common, with many people experiencing impairments in coordination, range of movement and dexterity as a result of the underlying motor changes associated with the condition (Agostino et al., 2003; Ponsen et al., 2008; Proud et al., 2021). Changes in hand and upper limb function can restrict participation in activities of daily living, even in the early stages of the condition (Choi et al., 2017; Ponsen et al., 2008; Proud et al., 2021). As upper limb dysfunction significantly impacts function, it is critical that therapists assess abilities and limitations accurately and provide intervention to (a) remediate, (b) adapt or (c) compensate for upper limb challenges (Welsby et al., 2019).
A recent systematic review identified 18 measurement tools used for upper limb evaluation in people with Parkinson’s and found few high-quality studies to support the measurement properties of existing tools (Proud et al., 2015). Capato et al. (2023) reported finding no specific outcome measures to assess the functional capacity of the upper limbs in people with Parkinson’s. Physiotherapists and occupational therapists who completed a survey of current practice reported a nonuniform approach to clinical upper limb measurement (Proud et al., 2013, 2015). They reported using a range of tools including generic manual dexterity tests and tools developed for other clinical populations, and some survey respondents were unable to identify upper limb measures suitable to their needs (Proud et al., 2013, 2015).
Existing upper limb assessments and outcome measures have known limitations including poor high-quality studies to support the psychometric properties of existing tools, particularly the validity and responsiveness; lack of standardisation (Giovannoni et al., 1999; Proud et al., 2013, 2015); or standardisation with different populations, that is, stroke (Adams et al., 2014; Proud et al., 2013, 2015); not performance based or incorporating functional tasks relevant in occupational therapy (Corona et al., 2018; Earhart et al., 2011; Proud et al., 2015); decreased clinical utility; and are often assessing a single movement pattern. Assessments used with a Parkinson’s population, such as a ‘hand to mouth’ task (Corona et al., 2018), have looked at quantitative analysis of upper limb movements using kinematic measures, rather than timing of tasks. Furthermore, there is a need for portable measures that can be administered outside of the clinical setting that is, in a patient’s home.
Occupational therapists working in a Parkinson’s Clinic have aimed to address this identified gap and develop a quick, timed assessment that can be utilised to track upper limb function over repeat clinic visits. An expert working group was formed to identify the components and constructs of a performance based timed upper limb assessment (TULA) for this population. The assessment, known as the TULA, was developed, includes 10 items and can be completed in approximately 20 minutes (Table 1). The aim of this research was to pilot the TULA and to establish the initial psychometric properties of the TULA mapped to the COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN; Mokkink et al., 2010).
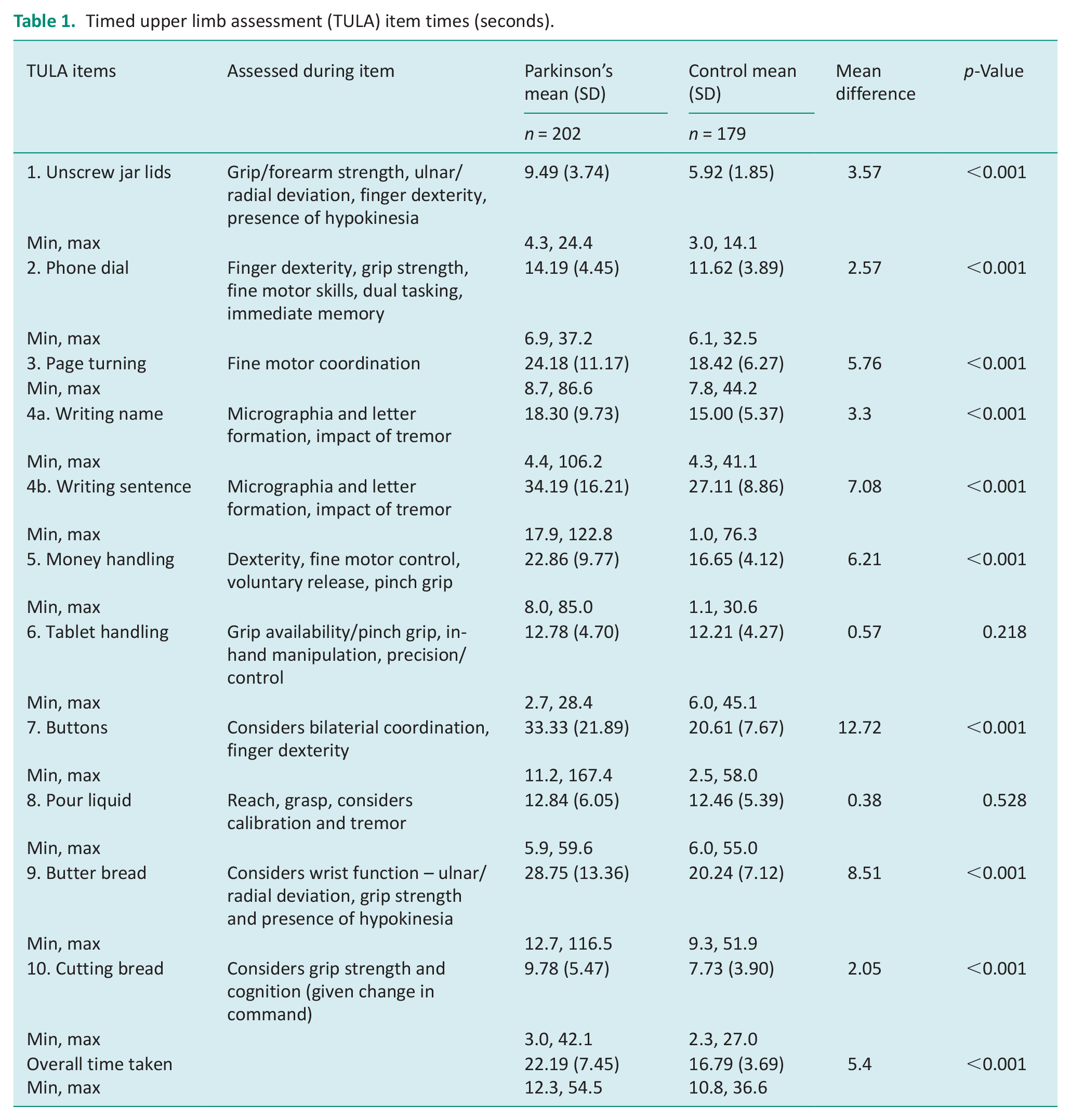
Timed upper limb assessment (TULA) item times (seconds).
Method
This was a prospective observational study to pilot and psychometrically evaluate the TULA with two populations consisting of participants with and without Parkinson’s.
Setting
The study was conducted at an outpatient Parkinson’s clinic at a specialist rehabilitation and aged care hospital between 2015 and 2024. Convenience sampling was utilised to recruit patients with Parkinson’s and participants without the condition to pilot the TULA.
Participants and recruitment
People with Parkinson’s
Those included were 55 years of age or older, attending the Parkinson’s clinic at the hospital, proficient in English and able to follow test instructions. Patients were seen by the Parkinson’s Clinic Senior Occupational Therapist.
Control group
A control group consisting of people without the condition was recruited via social media and through word-of-mouth and agreed to participate in the TULA. Controls were excluded if they had a diagnosed cognitive impairment. Additionally control patients were excluded if they had any known or apparent musculoskeletal impairment in their upper extremities or neurological conditions, including dementia.
Outcome measures
Initial descriptive data were recorded for all participants including gender, age and hand dominance. For patients with Parkinson’s additional data was collected including medication use and years post diagnosis regarding Parkinson’s, using a piloted data collection sheet.
Timed upper limb assessment
The TULA was initially developed by occupational therapists working in the Parkinson’s Clinic as a means of assessing upper limb function in a standardised manner for this population over time. This was in part driven by a service need, with Medical Consultants requesting standardised timed assessments of patient upper limb function, to complement timed data obtained by physiotherapists on mobility including the Timed Up and Go (TUG; Podsiadlo and Richardson, 1991). Benchmarking with other movement disorder clinics nationally revealed a paucity of functional upper limb assessments, with basic standardised assessments, such as the 9-Hole Peg Test (Mathiowetz et al., 1985) being the routine assessment administered. Clinicians with specialist expertise working with people with Parkinson’s selected a variety of items which aimed to evaluate functional upper limb tasks common in daily life. Through this expert consultation with the panel, a final group of 10 items (Table 1) were selected for the TULA.
The TULA comprised of functional tasks, including buttering bread, tablet handling and pouring liquids (Table 1). The therapist administering the TULA reads from a developed and piloted script to ensure instructions are delivered in a standardised manner. Instructions for use are included, which explains how the environment should be set up for each task, including where items should be placed in relation to the participant. The time taken to complete each task is recorded, and comments are made regarding quality of movement and other symptoms observed, such as tremor, rigidity, akinesia or bradykinesia. Additionally, fatigue, decreased active range of movement, patient’s level of familiarity with task (i.e. noting level of assistance normally required), relevant co-morbidities, need for repeat attempts or repeat explanations are recorded. The items were subjected to face validity checks by the panel of experts. The panel was asked to comment on format, clarity and relevance with all feedback incorporated. Overall, the TULA was designed for use with people with Parkinson’s presenting to an outpatient clinic, to track changes in upper limb function over time.
Each participant in this study completed the TULA once, which was video recorded with signed consent. For a subset of patients (n = 47) with and without Parkinson’s, two assessors consisting of Senior Occupational Therapists, evaluated the TULA video independently to establish inter-rater reliability.
Hoehn and Yahr Scale
The assessment of Parkinson’s signs and symptoms was completed via the Hoehn and Yahr Scale (Permlutter, 2009). Hoehn and Yahr Scale was used to stage the motor symptoms experienced by the patients with Parkinson’s, reflecting the level of severity and marking the progression of the condition from stage 0 (no clinical signs of the condition) to stage 5 (Wheelchair-bound or bedridden unless aided; Permlutter, 2009).
Timed up and go
The TUG test is one of the most frequently used tests of balance and gait and is often used to assess fall risk in older patients (Barry et al., 2014; Herman et al., 2011). The TUG is a timed test of general mobility, where the patient stands from a chair with arm rests (approximate seat height: 46 cm), walks 3 metres to a line on the floor and returns to sit in the chair (Podsiadlo and Richardson, 1991). Shorter times indicate better performance, and test times of up to 13.5 seconds are considered normal (Bridenbaugh and Kressig, 2015; Podsiadlo and Richardson, 1991). Test–retest reliability and inter-rater reliability of the TUG are reportedly high in patients with Parkinson’s (intraclass correlation coefficient (ICC) = 0.87–0.99) (Morris et al., 2001).
Cognition
The Mini Mental Status Examination (MMSE) (Folstein et al., 1975) or the Montreal Cognitive Assessment (MoCA) (Nasreddine et al., 2005) were used to assess cognition in patients presenting to the Parkinson’s Clinic. These are well-established, reliable, valid and brief cognitive screening tools. The total score (out of 30 points) places the individual on a scale of cognitive function defined as mild (19–23 points), moderate (10–18 points) or severe (⩽9 points) cognitive impairment in the MMSE (Folstein et al., 1975). The MoCA scores were classified as mild (18–25 points), moderate (10–17 points) or severe (<10) cognitive impairment (Nasreddine et al., 2005). Scores were adjusted for patients with <12 years of education. Tool selection was dependent on clinical reasoning at time of review in the clinic.
Data analysis
Data were summarised using frequency distributions for categorical data and means and standard deviations or medians, interquartile range (IQR) and range for continuous data-dependent on the normality of the data. Group comparisons for sample characteristics including age, gender and time to complete TULA items were reviewed using independent t-tests and Mann–Whitney U tests for continuous data and Chi-squared or Fisher Exact tests for categorical data. One-way between group analysis of variance was used to investigate the differences in time taken to complete TULA items dependent on severity of Parkinson’s via the Hohen and Yahr Scale. Linearity between TULA items were evaluated using correlation matrices, with variables exhibiting r > 0.3 retained for analysis (Field, 2018). Principal component analysis (PCA) was employed to examine the structural validity of the instrument, utilising Kaiser–Meyer–Olkin (KMO) measure of sampling adequacy and Bartlett’s test of sphericity (Mehdizadeh et al., 2020). Internal consistency was assessed using Cronbach’s α, with values above 0.70 considered acceptable (Lohr, 2002). Inter-rater reliability was evaluated through ICC, with values above 0.9 indicating high agreement (Ostelo et al., 2004). Convergent validity was explored through Pearson’s correlation coefficients between TULA scores and cognitive assessments (MoCA and MMSE) and motor progression scales (Hoehn and Yahr and TUG). Subgroup analysis was completed for people with Parkinson’s regarding the diagnostic accuracy of the TULA related to cognitive and motor function. TULA scores were summarised using median, IQR and ranges and compared between MMSE and MoCA cognitive impairment cutoffs (⩽24 and ⩽22 respectively) (Fiorenzato et al., 2024) and TUG score falls risk cutoff (⩾11.5) (Shirley Ryan Ability Lab, 2013) using Mann–Whitney U tests. Receiver operator characteristic (ROC) curve analysis was used to determine TULA score cutoffs for cognitive impairment and falls risk against the MMSE, MoCA and TUG cutoffs, with results summarised as area under the curve (AUC) with 95% confidence intervals, Youden’s Index cut-point, sensitivity and specificity. Statistical analysis was conducted using IBM SPSS version 24.0 (Armonk, NY, USA). p-values of <0.05 were considered statistically significant.
Ethical considerations
The study was approved by the Sir Charles Gairdner Osborne Park Health Care Group Ethics Committee (RGS0000001735) and the Edith Cowan University Ethics Committee (12368). All patients provided informed and written consent. All patients’ cohort data was de-identified and analysed in aggregate form.
Results
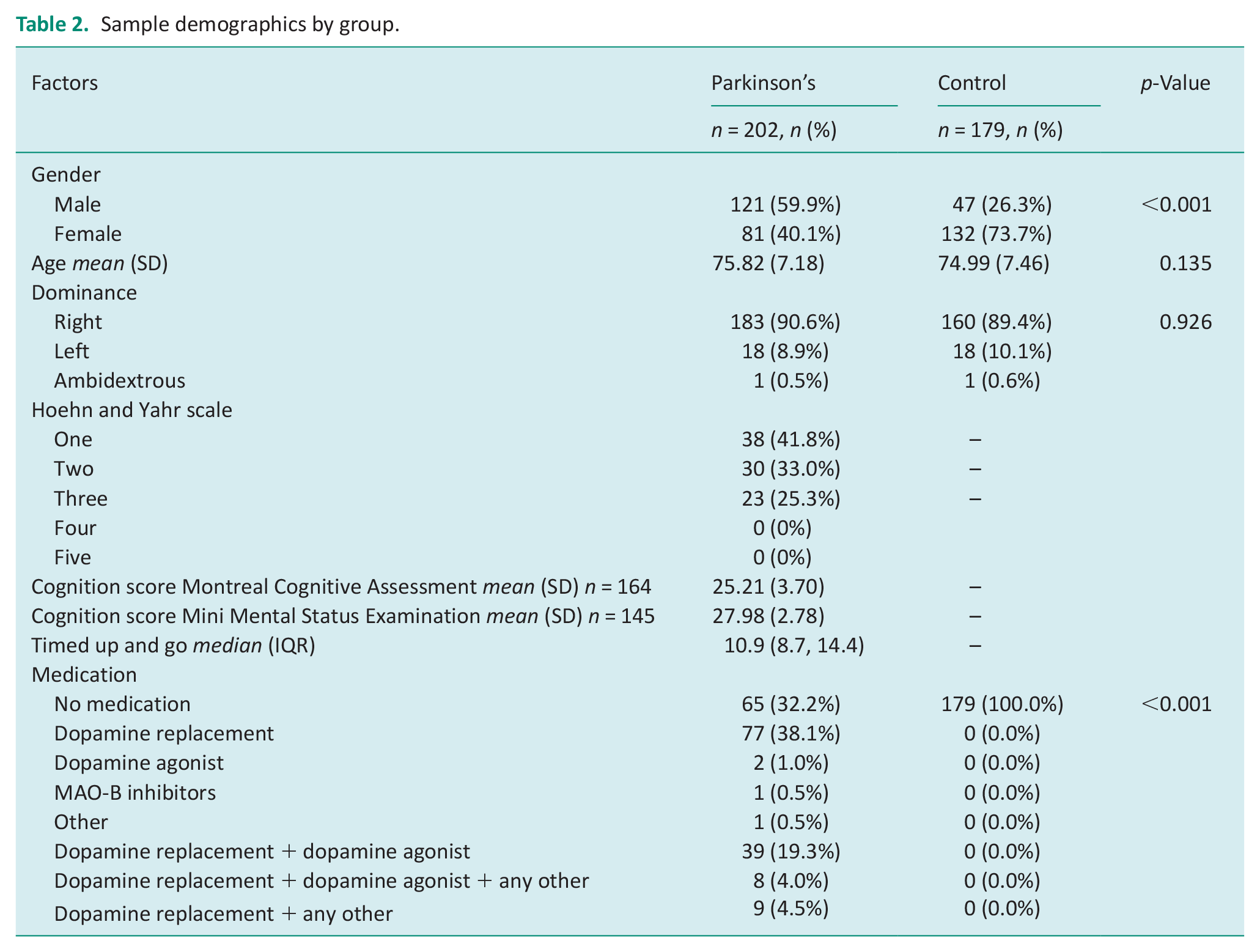
A total of 381 patients were recruited, consisting of 202 people with Parkinson’s, and a control group of 179 people. Table 1 outlines the general characteristics of the study population. Overall, the study population had a mean (SD) age of 75.2 (7.3) years consisting of 168 (44%) males. In the people with Parkinson’s group 41.8% (n = 38) had a Hoehn and Yahr of one, indicating unilateral involvement with minimal or no functional disability. Additionally, the people with Parkinson’s group had intact cognition with a mean (SD) MoCA score of 25.2 (3.7) and a MMSE score of 28.0 (2.8). For people with Parkinson’s the median TUG was 10.9 (IQR 8.7, 14.4) and 67.8% (n = 137) were taking medication for Parkinson’s management.
TULA and group comparisons
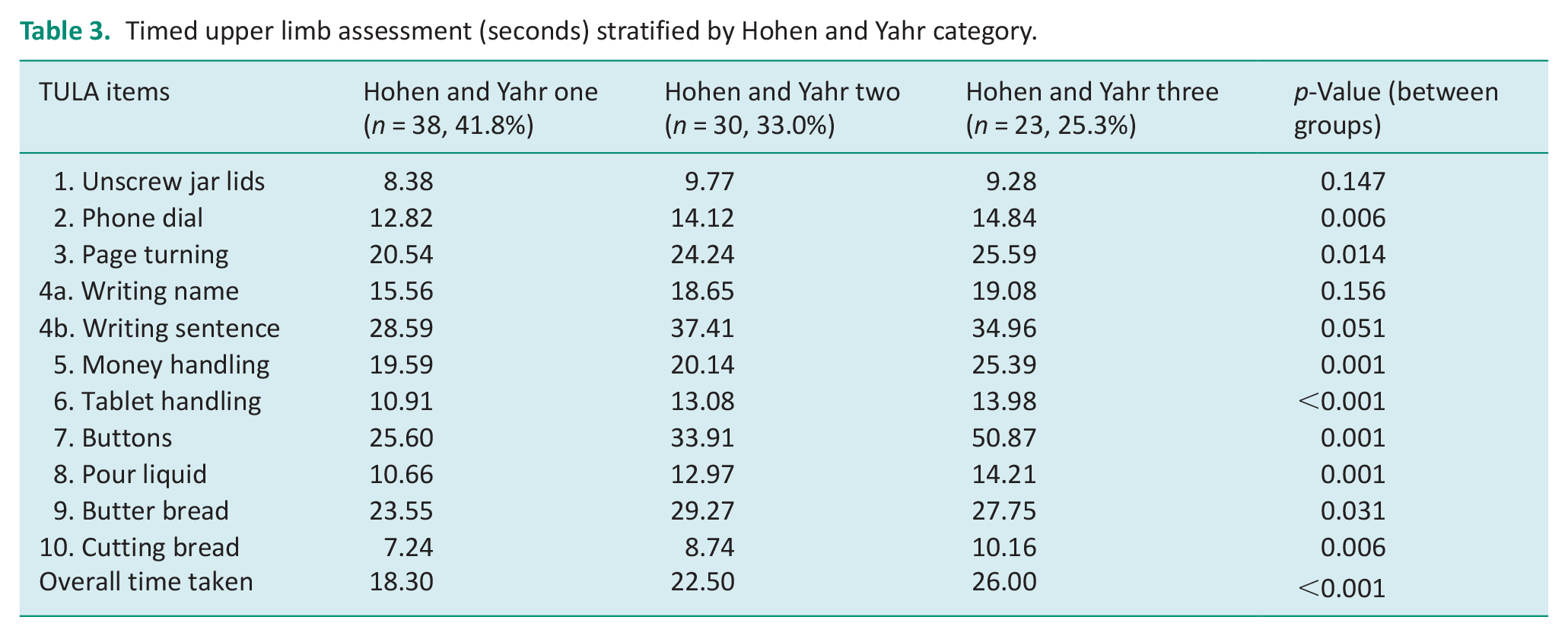
The mean time per item for both groups are outlined in Table 2. The overall mean (SD) time for people with Parkinson’s was 22.2 (7.5) seconds compared to 16.8 (3.7) seconds in the control group (p ⩽ 0.001). There were significant differences between groups for all items except for pouring liquid (p = 0.528) and tablet handling (p = 0.218). The mean difference was greatest between groups for the managing buttons item with a difference of 12.7 seconds, followed by buttering bread with a difference of 8.5 seconds. Table 3 highlights the differences in the TULA dependent on the Hoehn and Yahr category, demonstrating a change in upper limb function with progression of Parkinson’s. All items except for item one (unscrewing jar lids) demonstrated significant differences with progression of Parkinson’s.
Sample demographics by group.
Timed upper limb assessment (seconds) stratified by Hohen and Yahr category.
Structural validity
A PCA was performed on the 10 items (n = 202) for the people with Parkinson’s group. KMO measure of sample adequacy for the scale was 0.90 and Bartlett’s test of sphericity was significant (p ⩽ 0.001), further supporting the plausibility to conduct factor analysis (Kaiser and Rice, 1974). Two items were generated with eigenvalues exceeding one (eigenvalues = 5.67, 1.11), accounting for 64% of variance of upper limb function. All 10 items had loadings of 0.40 and above (Table 4). Based on the present analysis, all 10 items were retained for the final version of the TULA as they loaded exclusively on their respective component with loadings above 0.40.
Factor structure (oblique rotation) of the timed upper limb assessment (10 items) – Parkinson’s and non-Parkinson’s groups.
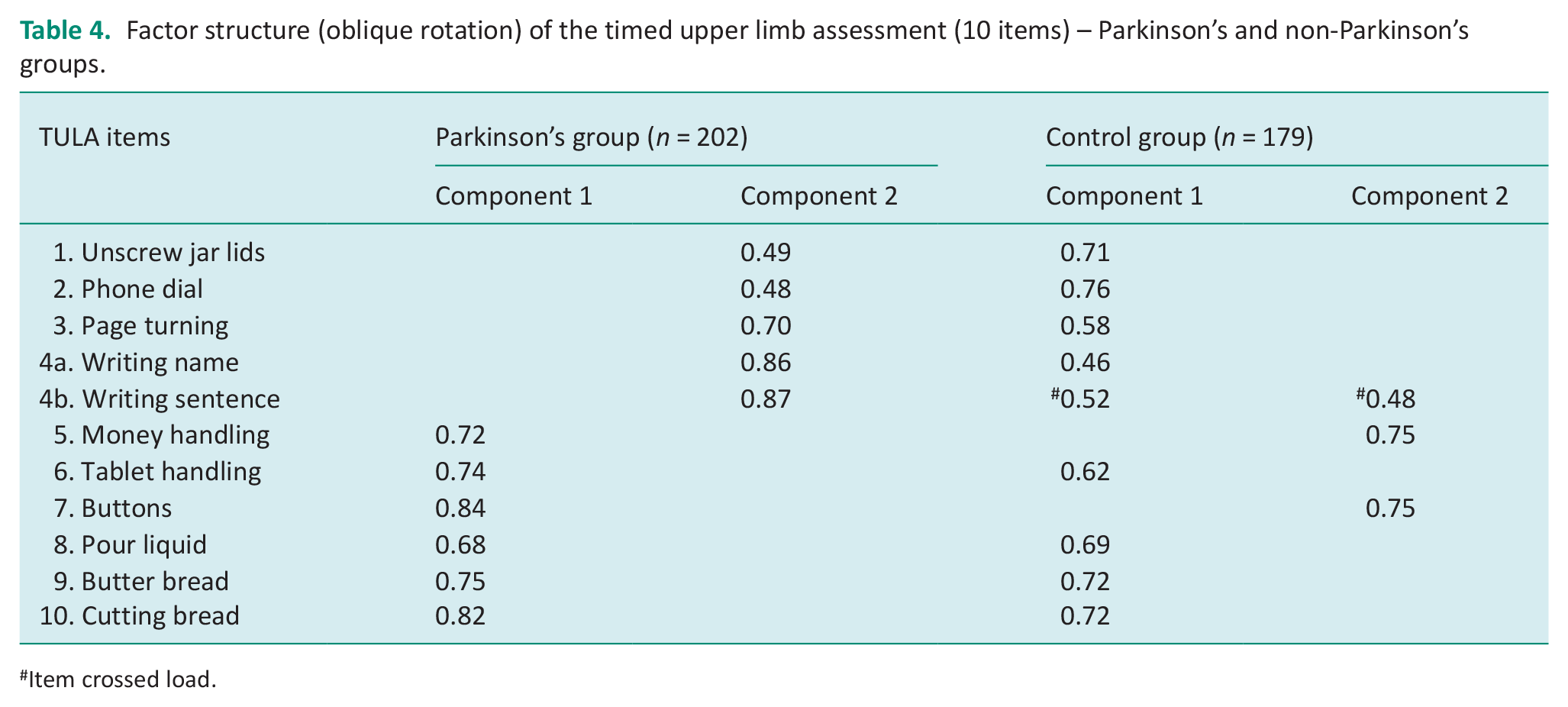
Item crossed load.
Replication of the factor structure of the TULA
A PCA was performed on the control sample (n = 179). The KMO measure of sampling adequacy was 0.84 and Barlett’s test of sphericity was significant (p ⩽ 0.001). Two components were generated with eigenvalues exceeding one (eigenvalues 3.94, 1.27), accounting for 52% of variance. Item 4b (writing sentence) cross loaded. The factor structure was not replicated with items 1–4a and items 5 and 7 loading on different components, although all loadings were above 0.4.
Internal consistency
Data showed that the TULA carried high internal consistency with the Chronbach’s α for the group with Parkinson’s at 0.81 and in the control group this was 0.76. All measures were above the minimum acceptable rate of 0.70. Additionally inter-item correlations are provided in the Supplemental Data (Appendix 1) and ranged from 0.83 to 0.94.
Inter-rater reliability
A subset of patients (n = 47) was reviewed by two different assessors to explore inter-rater reliability. Inter-rater reliability of the TULA was explored per item with high ICC above 0.9 for all items, indicating high agreement between different assessors.
Convergent validity
The overall TULA score had a significant low negative correlation between both the MoCA (r = −0.37, p ⩽ 0.001) and the MMSE (r = −0.31, p ⩽ 0.001) (Appendix 2). Items consisting of using the phone (r = −0.37, p ⩽ 0.001 with both the MMSE and MoCA) and writing a sentence (r = −0.32, p ⩽ 0.001 with both the MMSE and MoCA) had a higher level of correlation with the cognitive assessment tools. Overall, TULA score had a significant low positive correlation with the Hoehn and Yahr Scale (r = 0.44, p ⩽ 0.001) and a significant moderate positive correlation with the TUG (r = 0.56, p ⩽ 0.001).
Subgroup analysis regarding diagnostic accuracy
The TULA scores were significantly higher in patients with cognitive impairment compared to those without, as indicated by both MMSE (p = 0.003) and MoCA (p < 0.001) cutoffs (Appendix 3). Similarly, patients at risk of falls had higher TULA scores than those not at risk (p < 0.001) via the TUG. ROC analysis showed good discrimination for TULA scores with AUC values of 0.767, 0.787 and 0.777 for MMSE, MoCA, and TUG cutoffs, respectively, with corresponding Youden’s Index cut-points providing acceptable levels of sensitivity and specificity (Appendix 4).
Discussion
This study aimed to pilot the TULA and establish its initial psychometric properties for assessing upper limb function in people with Parkinson’s. Our results indicate that the TULA is a reliable and valid tool for measuring upper limb function in this population. Construct validity, internal consistency, inter-rater reliability and convergent validity were confirmed. The TULA was tested with people with Parkinson’s and in a control group, where overall significant differences were found in timed completion of upper limb functional tasks. Furthermore, the TULA was also able to discriminate between people with Parkinson’s with and without cognitive and motor impairment.
The TULA differentiated between changes in upper limb function with progression of Parkinson’s. Therapists have identified the need for normative data regarding task completion to provide an indication of mild, moderate or severe hand and upper limb impairment (Valevicius et al., 2018). The initial data have provided the mean times for completion of the TULA dependent on Hoehn and Yahr category. This can support clinical practice and monitoring of upper limb function over time.
Previous research has highlighted the limitations of existing upper limb assessments for people with Parkinson’s (Capato et al., 2023). For example, Capato et al. (2023) completed a systematic review including 50 studies and continued to note a large shortage of specific tests to assess the functional capacity of upper limbs in people with Parkinson’s. The TULA was specifically developed to support functional upper limb assessment and provide an efficient way to monitor progression over time through a Parkinson’s Clinic and communicate changes with the multidisciplinary team. Overall, the TULA demonstrated good internal consistency and high inter-rater reliability. The TULA’s inclusion of tasks such as managing buttons and buttering bread reflects real-world activities, aligning with the need for assessments that capture functional limitations relevant to daily living, as emphasised in a systematic review by Welsby et al. (2019).
The divergent factor structures between the Parkinson’s and control groups underscore the impact of Parkinson’s on upper limb function. In the control group, items such as writing a sentence cross-loaded, indicating that in a normative population, tasks involving fine motor skills may cluster differently compared to those in people with Parkinson’s. This suggests that the condition affects the coordination and execution of tasks differently, highlighting the necessity for condition-specific assessments.
The development of the TULA addresses a critical gap in the assessment of upper limb function in people with Parkinson’s. The tool’s ability to differentiate between different stages of the disease (as shown by the significant differences across Hoehn and Yahr categories) and its reliability makes it a possibly useful addition to clinical practice. Therapists can use the TULA to track changes in upper limb function over time, providing a basis for targeted interventions aimed at improving daily functioning and quality of life.
Limitations and future research
While the initial psychometric properties of TULA are promising, further research is needed to validate these findings in larger and more diverse populations. Longitudinal studies could explore the sensitivity of TULA to changes in upper limb function over time, which is crucial for monitoring disease progression and treatment efficacy. We used a convenience sample with all participants recruited from one site in a western population with patients aged between 58 and 93 years of age, impacting on result generalisability. Bias needs to be reported in that the senior occupational therapist completing the assessment measures including the TULA also recruited all patients. Although medication use was collected, further exploration could be completed to determine impact on performance, and time of day for testing was not standardised. Other psychometric properties require review including test-retest reliability and discriminant validity for example. Additionally, no subgroup analysis has been completed on different types of Parkinson’s presentations which could be completed in future research.
Conclusion
The TULA is a reliable and valid tool for evaluating upper limb function in people with Parkinson’s. The assessment’s ability to capture the nuanced differences in upper limb function between those with Parkinson’s and healthy controls highlights its potential use in clinical practice. Further validation studies are required.
Key findings
TULA demonstrated high reliability and internal consistency for assessing upper limb function in people with Parkinson’s.
People with Parkinson’s took significantly longer to complete the TULA compared to controls.
What this study adds
The TULA provides clinicians with a reliable and validated assessment tool specifically designed for efficiently measuring functional upper limb performance in people with Parkinson’s.
Supplemental Material
sj-docx-1-bjo-10.1177_03080226251335226 – Supplemental material for Development and initial psychometric properties of the timed upper limb assessment in older adults with Parkinson’s
Supplemental material, sj-docx-1-bjo-10.1177_03080226251335226 for Development and initial psychometric properties of the timed upper limb assessment in older adults with Parkinson’s by Kristie Harper, Angela Jacques, Melanie Burton, Janet Richmond and Lisa Barry in British Journal of Occupational Therapy
Footnotes
Acknowledgements
We would like to acknowledge Jo Davey, Sarvenaz Takroosta, Emily Cheetham and Tammy Murray for project support.
Research ethics
The study was approved by the Sir Charles Gairdner Hospital Human Research Ethics Committee (RGS0000001735) (2015) and the Edith Cowan University (ECU) Human Research Ethics Committee (12368) (2015).
Consent
All patients provided informed and written consent.
Patient and public involvement data
During the development, progress and reporting of the submitted research, Patient and Public Involvement in the research was not included at any stage of the research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by a Curtin University, School of Allied Health, Teaching and Research Grant.
Contributionship
LB, MB and JR conceived the study and completed the ethics approvals. LB supported the data collection. AJ supported the data analysis. KH and LB reviewed all data output. KH drafted the manuscript, and all authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
