Abstract
Background:
Atypically developing children often present with a variety of sensory processing difficulties which have been proposed to reflect abnormal development of pathways integrating sensation and action. A brain system in which the process of sensorimotor integration is particularly well understood is the oculomotor system, but no studies to date have used computerised eye tracking to assess eye movements in children with sensory processing difficulties.
Method:
Ten children with sensory processing difficulties completed a battery of oculomotor tasks comprising pro-saccades, anti-saccades, smooth pursuit tracking and sustained fixation. Eye movements were recorded using a high-resolution eye tracker.
Results:
Compared to age-matched controls, children with sensory processing difficulties were found to make more directional errors in the anti-saccade task and less-accurate smooth pursuit and sustained fixation.
Conclusion:
Consistent differences were found in oculomotor ability in children with sensory processing difficulties which are likely to impact children’s ability to process and respond to visual information within home and school contexts. Further research is needed to understand the relationship between oculomotor deficits in children with sensory processing difficulties and the presence/absence of neurodevelopmental diagnoses. Eye tracking may be of value in the future for assessment and objective evaluation of interventions for sensory processing difficulties such as sensory integration therapy.
Keywords
There is increasing awareness in research and practice of the importance of sensory processing difficulties (SPD) in understanding atypical development in children. Although there is debate about whether SPD should be considered a separate disorder (Borkowska, 2017; Mcarthur, 2022; Walbam, 2014), existing criteria acknowledge sensory problems as an important feature of common neurodevelopmental conditions (American Psychiatric Association, 2013). In practice, therapists consider SPD as encompassing a broad range of issues manifested within different domains (home, school) and modalities (auditory, visual, tactile, and vestibular). These are typically assessed using interviews with parents, direct observation and standardised assessments such as the sensory profile and sensory processing measure (SPM; Brown, 2021; Dunn, 2014; McIntosh et al., 1999; Tomchek and Case-Smith, 2009).
An influential perspective in practice around SPD is Ayres sensory integration (ASI®; Ayres, 1979). Ayres proposed that atypical development had its root in early dysfunction of brain systems integrating multi-modal sensory input and its transformation into appropriate action. She identified three different stages within which problems can occur: sensory registration (detection of sensory signal), modulation (inhibition and enhancement of sensory input) and motivation (initiation of actions in response to sensory stimulation). The theory has been criticised for its lack of grounding in neuroscientific evidence (Leong and Carter, 2008; Smith et al., 2015). For example, Ayres suggested that the vestibular nuclei may play an important role in attributing ‘meaning’ to stimuli (Kilroy et al., 2019; Lane et al., 2019), whereas contemporary research identifies the ventral processing stream in the cerebral cortex as critical for elaboration of object identity and meaning (De Haan and Cowey, 2011). Another suggestion put forward by Ayres (based on cluster analysis of symptomology) was that aspects of SPD arise due to lack of coordination of function between the two cerebral hemispheres (Ayres, 1977; Mailloux et al., 2011), although direct evidence for this proposal is also lacking and the nature of hemispheric specialisation is still debated in contemporary neuroscience (Beaumont et al., 1984; Nielsen et al., 2013). Nevertheless, it should be considered that Ayre’s theory was developed at a time when functional neuroimaging was in its infancy and more recent brain imaging studies have offered broader support for ASI® in the form of differences in white matter connectivity between cortical regions subserving visuomotor integration in children with and without SPD (Chang et al., 2014; Owen et al., 2013).
A brain network for which the processes of integrating sensation and action are particularly well understood is the oculomotor system. Humans make approximately 200,000 gaze-shifting saccades each day (Carpenter, 1988). These fast eye movements can direct the central fovea of the retina towards visual stimuli in a reflexive manner but can also be subject to modulating cognitive influences (Hallet and Adams, 1980; Yarbus, 1967). In tandem with saccades, smooth pursuit eye movements serve to maintain gaze on continuously moving objects by smoothly tracking them as they move across the visual field. Many decades of behavioural and neuroscientific research in humans and animals have mapped separate brain regions for the control of different aspects of saccades and smooth pursuit within the brain stem, midbrain (superior colliculus), cerebral cortex and cerebellum (Carpenter, 1988; Munoz and Coe, 2011; Thier and Ilg, 2005).
Given that the neuroanatomy of eye movements is well understood, eye movement tests have the potential to provide a window onto a widespread network of brain areas underlying sensorimotor integration and transformation (Hodgson et al., 2019; Hutton and Ettinger, 2006; Leigh and Kennard, 2004). In occupational therapy practice, oculomotor examination forms an important part of the approach to assessment. Typically, this involves noting presence/absence of resting nystagmus; observation of the ability to voluntarily shift gaze along cardinal and oblique axes; assessing the integrity of the vestibular-ocular reflex (whereby gaze is maintained on an object during head movement) and quality of smooth pursuit eye movements (Blanche et al., 2021). The assessment of saccades is typically limited to testing the ability to quickly ‘localise’ a stimulus, although the rate and size of saccadic intrusions in smooth pursuit are implicitly considered within overall ratings of pursuit quality (Gilligan et al., 1981).
Modern computerised eye-tracking devices allow precise measurement of the timing and accuracy of eye movements in tasks that place different demands on visual and cognitive processing. Eye tracking has been applied successfully across a wide range of neurological disorders as well as in neurodevelopmental conditions including autism spectrum disorder (ASD), attention deficit hyperactivity disorder (ADHD) and developmental coordination disorder (DCD; Johnson et al., 2012; Karateki, 2007; Munoz et al.,2003; Sumner et al., 2021). A typical battery of eye movement tasks applied in this way includes
If processes of sensorimotor integration were disrupted in children with SPD, then impairments in eye movement control would be expected. Consistent with this, Ayres’ research indicated that one group of children who might particularly benefit from therapy were those who showed impairments in smooth pursuit across the mid-line (Ayres, 1977). More recently, Walker et al. (2019) have applied the North-eastern State University College of Optometry (NSUCO) oculomotor assessment battery (Maples et al., 1992) to compare groups of children with and without SPD. In this test, children make voluntary saccades between two fixed coloured balls in response to verbal commands from the assessor. Smooth pursuit is then assessed by moving one of the balls in a circular pattern and asking the child to follow it with their eyes. Children with SPD were found to exhibit poorer accuracy for both saccades and pursuit relative to typically developing children. As is the case with other clinical and occupational therapy assessments of oculomotor control (see above), the NUSCO relies on observer ratings of eye movement speed and accuracy rather than eye tracking. The NSUCO also does not include tasks to assess inhibitory control of reflex eye movements such as the sustained fixation or anti-saccade tasks (Hodgson et al., 2007; Sumner et al., 2021; Walker et al. 1998).
In the current study, children’s eye movements were recorded using an eye tracker whilst they completed a battery of eye movement tasks including anti-saccades and sustained fixation. The performance of a group of SPD children was compared to a sample of age-matched controls. An aim of the study was to determine whether eye movement tasks and measures were consistently impaired across all the children with SPD regardless of other diagnoses. This might indicate that there may be commonalities in the neural pathways affected and therefore offer support for aspects of ASI® and the concept of SPD as a specific diagnostic entity. However, given the essential heterogeneity and multifactorial nature of SPD in practice (Mulligan et al., 1998), together with the variety of specific diagnoses of the children we assessed, we equally expected to see diverse patterns of oculomotor functioning within the SPD group relative to typically developing children.
Methods
The study adopted a between-groups design to compare quantitative performance measures derived from computerised eye-tracking tasks between children with and without SPD. The sample represented a ‘real-world’ cross section of children with SPD who had been referred to a paediatric occupational therapist for SPD prior to initiating a programme of ASI®-based therapy. An experienced paediatric occupational therapist (Hippisley) was involved in conceiving and designing the research study and in recruiting participants in collaboration with the academic researchers.
Participants
The SPD group comprised 10 children aged 4–15 years old (all male), with a variety of clinical diagnoses ranging from no other diagnosis (2 participants) to multiple comorbid neurodevelopmental conditions (ASD + ADHD, 2 participants). A control group of 9 typically developing children (5 male) were recruited through personal contacts of the researcher and therapist in the local community and schools. The mean age for SPD participants was 9.10 ± 2.89 (standard deviation) and 9.67 ± 2.55 years for typically developing controls. Children were provided with an age-appropriate information sheet to read before completing the tests and parents/guardians gave informed consent for their children to participate. The research was reviewed and approved by the University of Lincoln Research Ethics Committee (Ref: 2022_0190).
Measures
Standardised assessments
All children were assessed by a paediatric occupational therapist using the SPM standardised assessment tool. The SPM involves primary caregivers and teachers completing a 75-item questionnaire which generates a standardised T score describing how sensory problems impact social participation, vision, hearing, touch, body awareness, balance, motion and planning (Brown, 2021). Children’s overall sensory processing is summarised as being ‘typical’, ‘some problems’ or ‘definite problems’. All the children in the SPD group were judged to have some or definite problems with sensory processing within either their home or school environments or both. Other tests carried out for the majority of children as part of their pre-therapy assessment included the
Procedure
Eye-tracking
An
Eye-tracking tasks were programmed using
Eye movement tasks
The eye movement tasks were presented to the children as a series of eye-tracking ‘games’ in a fixed presentation order as follows:
Each trial started with the presentation of a cartoon bee at the centre of the screen. Following a period of 800 ms, the bee disappeared and simultaneously reappeared at 6° eccentricity to either the left or the right side of the screen. The participant made a saccade away from the centre of the screen to ‘follow busy bee’. Once a saccade to the bee had been detected by the eye tracker a ‘beep’ sounded and the bee disappeared (Figure 1(a)). If a saccade was made in the wrong direction (i.e. away from the bee), a low-pitched tone sounded. The next trial then began after a delay of 500 ms with the presentation of the bee once again at the central location. After 24 trials, the task finished and the sound of a buzzing bee played along with a picture of the smiling cartoon bee saying thank you.
The anti-saccade task was identical to the pro-saccade task except for the participant instructions. At the start of the test, children were told this time not to follow busy bee but to make a saccade in the
In this task, a cartoon dinosaur was displayed on either the left or right (horizontal pursuit trials), top or bottom (vertical pursuit trials) of the screen. Once fixated the dinosaur was replaced by a red dot which moved in a sinusoidal pattern with amplitude 12° either horizontally or vertically for 10 cycles. Each movement direction was repeated at slow or high speed (frequency 0.2 Hz or 0.5 Hz) giving a total of four trials. At the end of the task, a roaring dinosaur sound effect played along with a picture of a smiling cartoon dinosaur saying ‘Well done!’.
In this task, a small picture of a bull’s-eye target was displayed in the centre of the screen. Children were told to ‘keep your eyes on the target’ without looking away. The bull’s eye appeared for 2 periods of 30 seconds each. At the end of each 30 second period, the sound effect of an arrow hitting a target was played and the message ‘Well done, you hit the bull’s eye!’ was displayed.
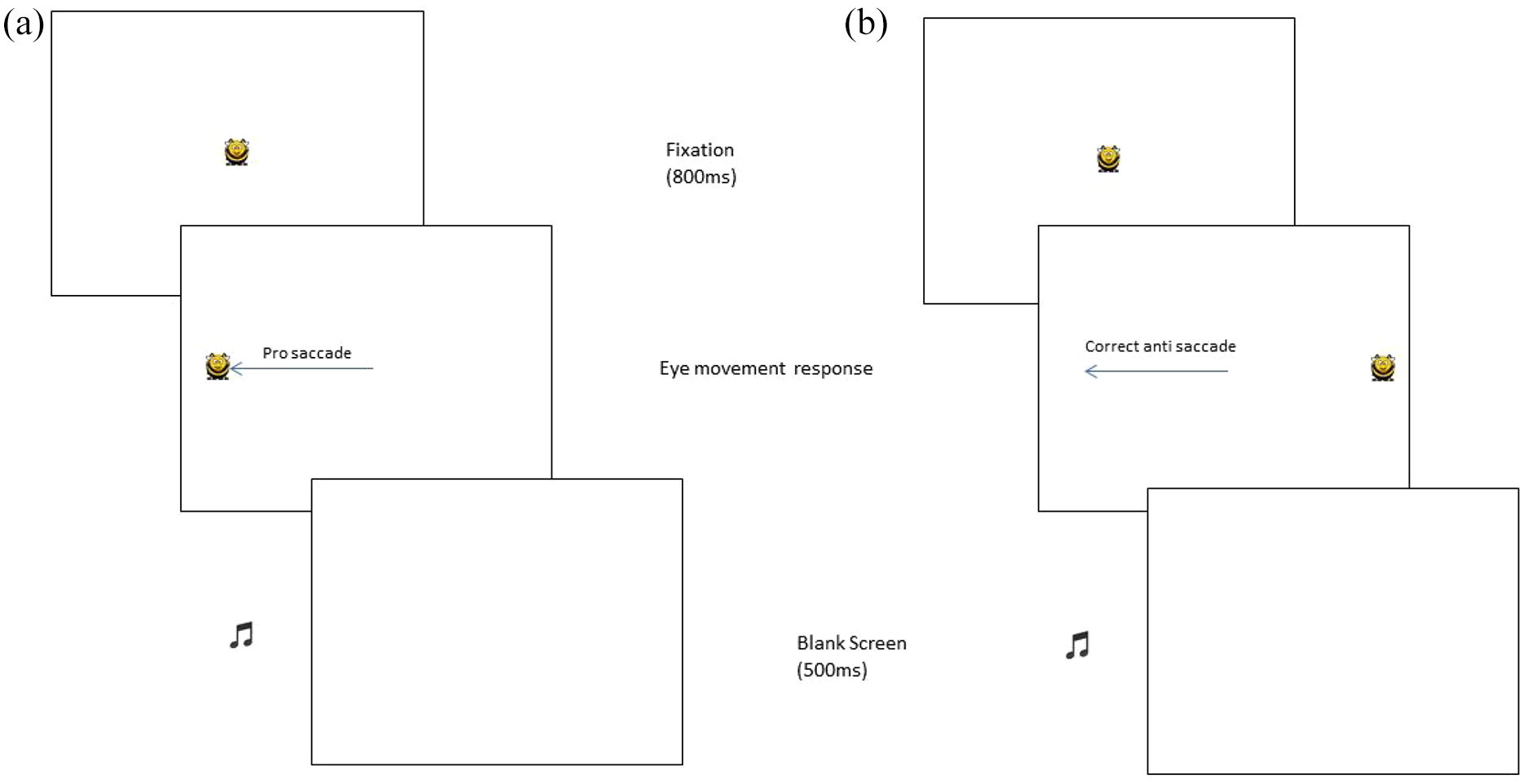
Schematic illustration of the sequence of events in a single trial for (a) pro-saccade task (b) anti-saccade tasks (target appeared with equal likelihood to the left or right side on each trial, but for simplicity, left-side target example is shown in both examples).
The entire battery of tasks took around 15 minutes to complete. Data for the anti-saccade task were not available for one of the SPD participants due to poor eye-tracking calibration and recording.
Eye movement measures
Key metrics extracted from the eye movement tasks were
The time in milliseconds (ms) between the onset of the saccade target (busy bee) and the start of a correct saccade response either towards (pro-saccade) or away from (anti-saccade) the target.
Percent of trials in the pro-saccade task where a saccade was directed away rather than towards the target.
Percent of trials in the anti-saccade task where the first saccade after target onset was directed towards rather than away from the target.
Size of saccades (in degrees of angular rotation of the eye).
Average spatial distance of fixation compared to target position during smooth pursuit tracking.
Eye velocity as a proportion of target velocity during smooth pursuit tracking.
Number of saccades measured during smooth pursuit.
Total number of saccades measured during the fixation task.
Analysis
Independent sample
Results
Pro- and anti-saccade tasks
For the pro-saccade task, no significant differences were found between the control (Con) and SPD children for any of the eye movement measures. Neither SRT (Con:
SPD children made significantly more errors in the anti-saccade task (Con: 45 %± 28% versus SPD: 79% ± 24%;
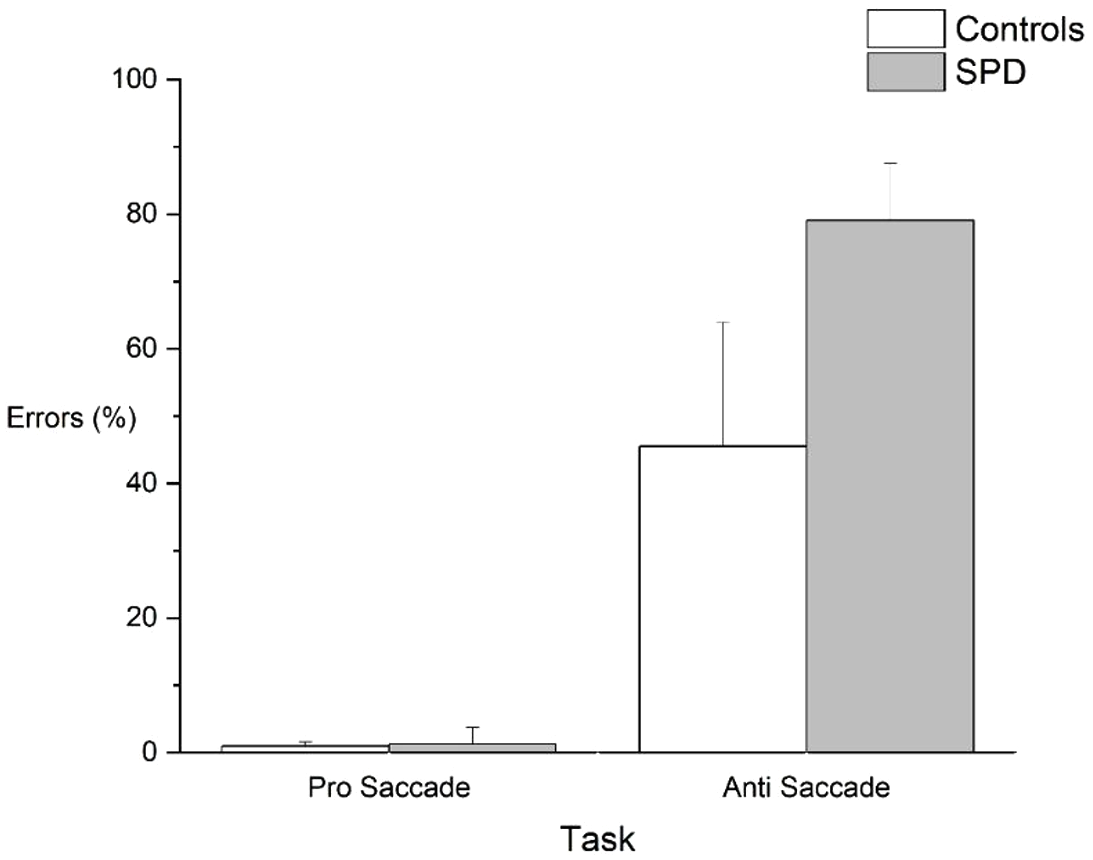
Rate of directional saccade errors in the pro and anti-saccade task for the two participant groups.
Smooth pursuit task
SPD children made significantly larger amplitude saccades on average during horizontal (Con: 3.35° ± 2.21° vs SPD: 6.02° ± 3.14°;
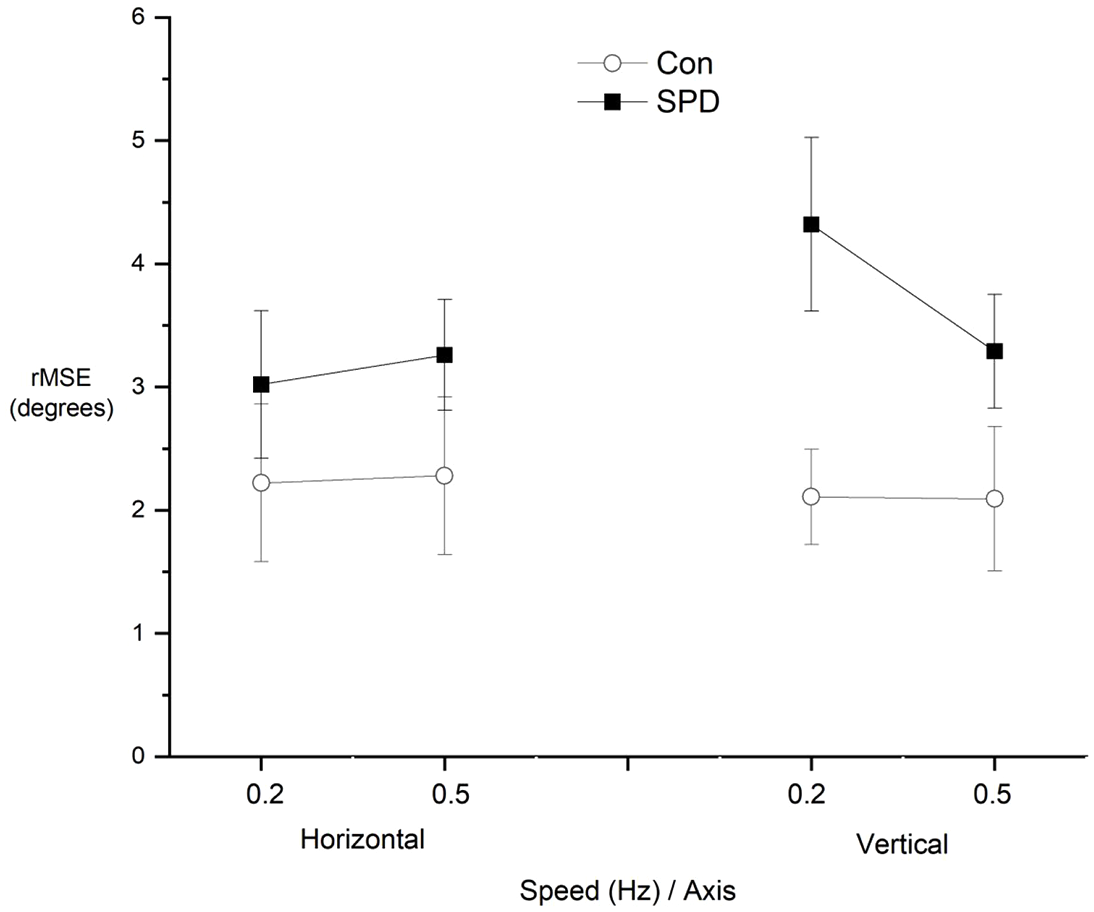
Accuracy of smooth pursuit eye movements (rMSE) along horizontal and vertical axes for SPD and control (Con) children at the two different speeds.
Sustained fixation task
A significant difference was observed in N-saccades measured during the sustained fixation task, with SPD children making significantly more saccades away from the bull’s-eye target (Con: 10.67 ± 7.80, SPD: 22.89 ± 14.27;
Correlation with age and diagnosis
Participating children in the SPD and control groups varied in ages between 7 and 15 years. Pearson’s correlations were therefore conducted to assess the relationship between age and eye-tracking task measures. Age was found to have a significant negative correlation with anti-saccade errors (ρ = −0.62,
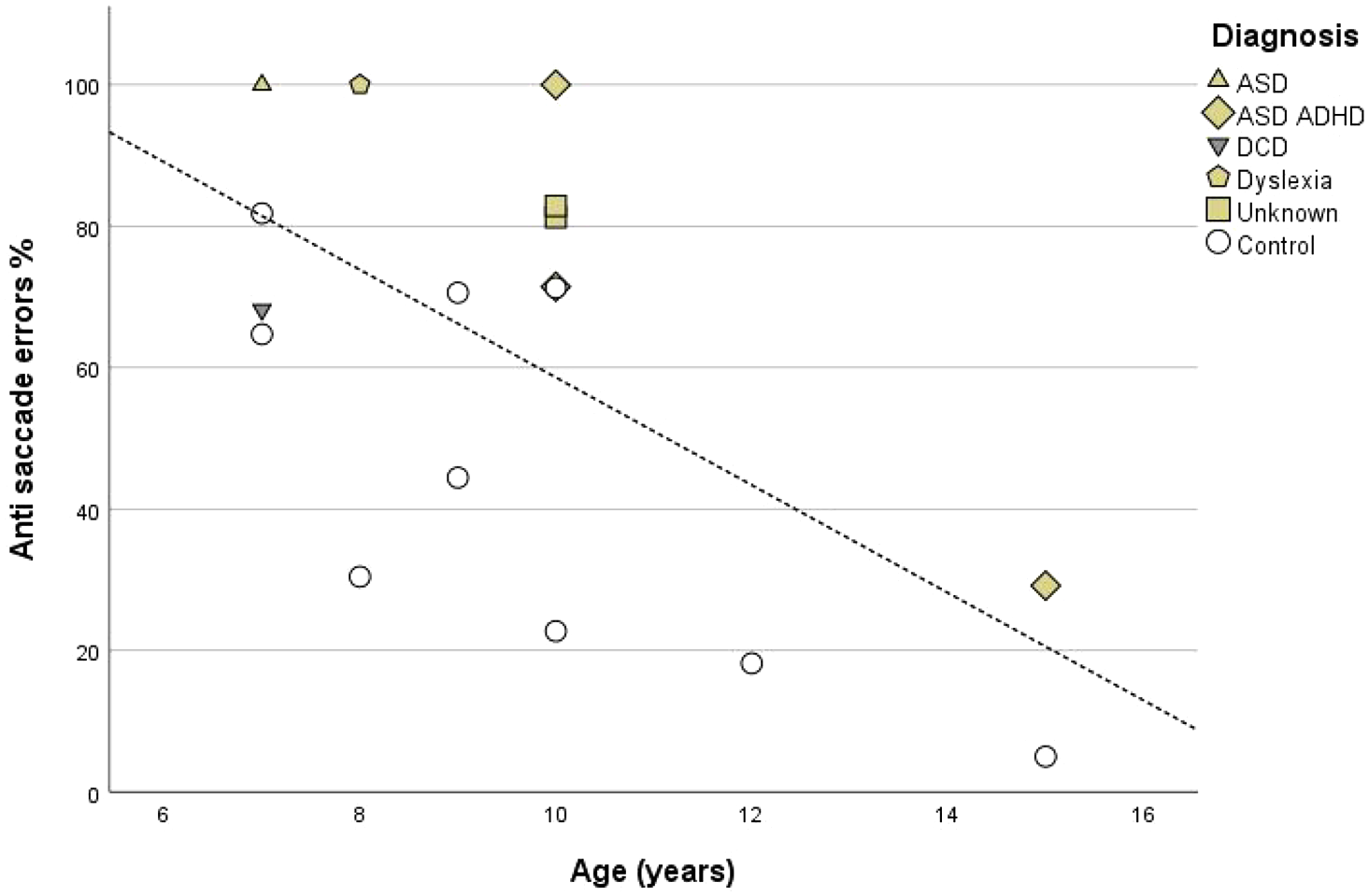
Anti-saccade errors plotted against participant age. Open circles indicate control group participants. Filled symbols indicate children with SPD and diagnostic status.
Gender
All of the participants in the SPD group were male, whereas the control group was of mixed gender (four female, five male). In order to confirm that the effects of SPD were not confounded by gender, all the analyses of eye movement measures described above were repeated but including only the male participants from the control group. Statistically significant differences were found between male children with and without SPD for all the measures previously found to be significant between groups, including anti-saccade errors (Con: 52% ± 29% vs SPD: 79% ± 24%;
Discussion
This study used a computerised eye-tracking device to examine oculomotor function in a group of children with SPD. It was found that saccades directed towards peripheral visual target onsets (pro-saccades) had normal amplitude and timing in children with SPD, but SPD children made significantly higher error rates in the anti-saccade task compared to controls. Differences were also found in smooth pursuit and sustained fixation. Increased amplitude of intrusive saccades were apparent in the SPD group whilst viewing dynamic and static fixation targets and pursuit was significantly less accurate for targets moving along the vertical axis. These oculomotor differences can be seen as broadly consistent with theoretical perspectives that propose disrupted sensory motor integration as an important aspect of developmental psychopathologies. The findings also have implications for the type of day-to-day problems experienced by children with SPD and future approaches to assessment and evaluation of outcomes of therapeutic interventions.
The anti-saccade task tests the ability to inhibit a reflexive saccade towards a stimulus as well as the initiation of voluntary saccades towards a cognitive goal as opposed to a sensory goal. Children with SPD made more anti-saccade errors, in which a saccade is directed towards the target location, with some children making 100% errors in a block of 24 trials. One interpretation of this finding is that SPD children had more difficulty understanding the task instructions. The instructions were presented to children on the computer screen at the start of the test and also explained verbally by the researcher. The great majority of errors were also corrected with secondary saccades in the opposite direction, making it unlikely that children failed to understand the task instructions. Instead, the overall pattern is suggestive of increased oculomotor impulsivity and poorer cognitive control of eye movements and fixation in the SPD group.
It is important to note that the children with SPD in the current study were a ‘real world’ sample of children with sensorimotor problems referred for occupational therapy and as such the majority had diagnoses of one or more developmental conditions. Previous studies have used similar eye movement tasks to examine groups of children with isolated diagnoses of ASD, ADHD and DCD (Johnson et al., 2012; Munoz et al., 2003; Sumner et al., 2018, 2021). Munoz et al. (2003) reported that both anti-saccade errors and SRTs were increased in ADHD, whereas we found that SRTs for correct anti-saccade were significantly faster in SPD children. Sumner et al. (2018) found that children with DCD also made significantly more anti-saccade errors but did not differ compared to controls in terms of SRTs. The same researchers report no consistent difference in anti-saccade performance in children with ASD compared to typically developing controls (Sumner et al., 2021). Out of the three children in the current study who made 100% anti-saccade errors, one had a diagnosis of ASD, another had a diagnosis of both ASD and ADHD and the third had a specific learning difficulty (dyslexia). The participant with the lowest anti-saccade error rate also had diagnoses of both ASD and ADHD, but is also the oldest child in the sample, such that when the effect of age on error rates was considered they still made relatively high numbers of errors in the anti-saccade task (Figure 4). Given these differences between the findings of studies of eye movements in children with singular diagnoses, it is notable that a more consistent pattern of increased errors and reduced SRTs was seen for the anti-saccade task within our small sample of children with SPD and a variety of specific diagnoses.
Fixation stability and smooth pursuit have also been studied in children with ASD (Takare et al., 2004), ADHD (Caldani et al., 2019) and DCD (Sumner et al., 2018). Both ADHD and DCD have been reported to be associated with increased rates of intrusive saccades during fixation tasks. However, previous studies have shown that in the case of ASD, deficits in both sustained fixation and smooth pursuit are associated with scores on motor skills assessments (e.g. MABC-2) and hyperactivity ratings (Sumner et al., 2021), suggesting they are affected by the extent of sensorimotor dysfunction. In the current study, statistically significant differences in rMSE accuracy of smooth pursuit eye movements were found between children with and without SPD. As with the findings of the anti-saccade task, this suggests consistent deficits in oculomotor control within our SPD group independent of diagnosis.
It should be noted that the SPD group in this study comprised entirely male children, whereas the control group was of mixed gender (four female/five male). This is likely to reflect a male bias in the prevalence and/or diagnosis of neurodevelopmental conditions (Gershon, 2002; Loomes et al., 2017). Because of this gender imbalance, we repeated all the reported analyses, but including only male children from the control group. Statistically significant differences were still found to be present between groups for the anti-saccade, sustained fixation and pursuit tasks, suggesting that gender is unlikely to be a confounding factor in the comparison between SPD and non-SPD children.
It is interesting to consider the current findings in the context of neuroimaging studies of children with SPD and in relation to theoretical perspectives such as ASI®. Owen et al. (2013) used the diffusion tensor magnetic resonance imaging technique to assess the structural integrity of brain white matter tracts connecting different areas of the cerebral cortex. This revealed a pattern of differences relative to controls in children with SPD that was distinct from that seen in other neurodevelopmental conditions. In both ASD and ADHD, differences in frontal and temporal cortical connectivity are found (Tamm et al., 2012; Travers et al., 2012), whereas in SPD, more widespread changes are seen including within posterior tracts connecting occipital and parietal cortices. Such a pattern might explain some of the distinct features of oculomotor control in children with SPD. Pathways connecting the frontal cortex with sub-cortical regions have been most commonly implicated in inhibitory control of reflexive saccades in the anti-saccade task (Munoz and Everling, 2004), but posterior parietal regions are also involved in eye movement and fixation control (Ptak and Muri, 2013), notably the ability to voluntarily disengage attention in order to shift fixation elsewhere. Within the framework of ASI®, these processes would most closely align with the concept of sensory modulation, by which inhibition and enhancement of sensory input is applied to modify stimulus-elicited behaviour (Ayres, 1979).
The current findings also have implications for the types of tasks and settings within which children with SPD might experience problems, as well as pointing the way towards further research on the nature of SPD in children with neurodevelopmental pathologies. The findings indicate consistent problems with aspects of oculomotor control in children with SPD which might be useful in targeting occupational support for children. For example, the increase in errors in the anti-saccade task seen in SPD children would equate to increased visual distractibility and an impaired ability to efficiently switch eye gaze between one object and another. This would likely to have implications for children in educational settings, such as paying attention selectively to tasks or information in a classroom or learning new visuomotor skills and tasks. Smooth pursuit tracking was significantly less accurate in SPD children on the vertical axis, particularly at lower velocities. This might be of functional significance to children when engaging in sports involving catching and striking balls, as well as when avoiding obstacles such as moving vehicles.
It is important to note that we only studied a small group of children, most of whom had diagnoses of other neurodevelopmental conditions in addition to SPD. Future studies could examine eye movements in children with isolated diagnoses (ASD only) with and without identified SPD. For example, if high anti-saccade errors were specifically related to problems with sensory processing rather than ASD per se, then they would be expected to correlate with sensory difficulties scores. Another area of research would be to use mobile eye tracking to examine how children use eye movements during performance of everyday tasks and activities, rather than computerised eye movement tests (Hodgson et al., 2019, 2024). Tasks such as anti-saccades, smooth pursuit and sustained fixation might also be usefully applied as objective assessment and outcome measures, offering advantages over interview and questionnaire-based evaluations of therapeutic approaches such as ASI® (Ayres, 1979).
Eye tracking is still relatively expensive and is typically not available outside specialist research centres. There is also a lack of standardised norms available for eye-tracking tasks in children, limiting their application in occupational therapy and optometry assessment. However, it is possible that the technology may become more affordable and acceptable for use in these contexts in the future. For example, eye tracking might be usefully applied to identify children with particular difficulties (e.g. deficits in smooth pursuit) that might benefit most from therapeutic interventions. To this aim, future research could directly compare eye-tracking measures with scores derived from standardised assessments of oculomotor function within the same children (Blanche et al., 2021; Gilligan et al., 1981; Maples et al., 1992).
Overall, the research reported here can be seen as making an incremental contribution towards a better understanding of the place and meaning of oculomotor function and occupational performance in children with SPD. Further collaborative, inter-disciplinary research of this type will enhance the debate around the status of SPD in diagnostic manuals and whether it should be considered a separate disorder or as an important dimensional variable across a range of conditions (McArthur, 2022; Miller et al., 2009).
Key findings
Computerised eye tracking revealed that children with SPD had deficits in saccadic and pursuit eye movements
Eye tracking may be useful in the future for oculomotor assessment in occupational therapy.
What the study has added
The study is the first to use computerised eye tracking to assess oculomotor deficits in children with a variety of diagnoses (ADHD, ASD) and sensory processing deficits.
Footnotes
Acknowledgements
The authors wish to thank the children for their enthusiastic participation in the study and parents/guardians for their time and interest in the research.
Research ethics
The research was reviewed and approved by the University of Lincoln Research Ethics Committee (Ref: 2022_0190).
Consent
Parents/guardians gave written informed consent for their children to participate.
Patient and public involvement data
During the development, progress, and reporting of the submitted research, Patient and Public Involvement was included in the conduct of the research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) declared no financial support for the research, authorship and/or publication of this article.
Contributorship
Hippisley conceived the study, recruited participants, assisted in data collection and commented on draft manuscript. Hodgson designed eye-tracking tasks, supervised data collection and drafted manuscript. Cartwright and Dodds assisted with data collection and analysis and drafting manuscript.
