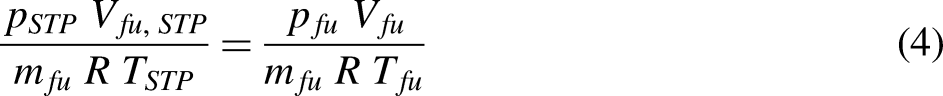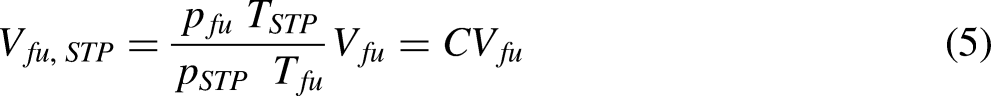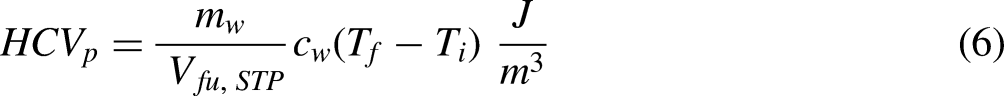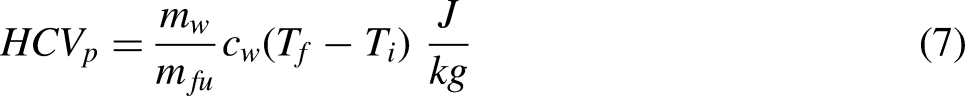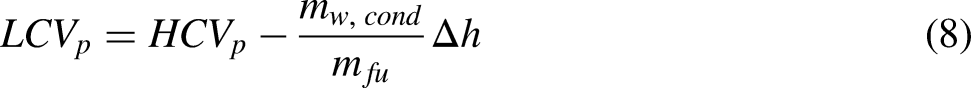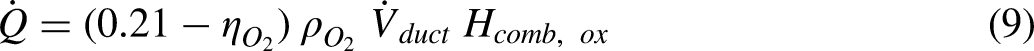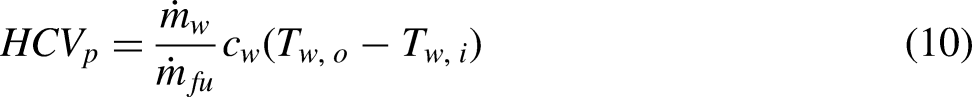Abstract
This paper describes an inexpensive calorimeter for measuring the higher calorific value of Liquified Petroleum Gas (LPG). The calorimeter is a hybrid of a Boys gas calorimeter and cone calorimeter. The calorimeter does not require a continuous supply of LPG. The mass flow rate of fuel is measured using a load cell. The hybrid calorimeter in its present form can measure the higher calorific value for LPG with a relative error of approximately 25%. Future projects to improve the accuracy of the calorimeter and extend its capability to measure the lower calorific value of LPG are considered. It is argued that the self-built calorimeter in its present form makes it a better laboratory experiment for the education of engineering students than an accurate commercially available apparatus.
Introduction
This paper discusses a novel gas calorimeter, developed as part of an undergraduate individual capstone project. Mechanical engineering undergraduate students at Heriot-Watt University undertake an individual project in the 4th year of their degree program over two semesters. The project is supervised by a lecturer with expertise in the area of interest of the student. The project brief is formulated by the student and supervisor. The project briefs are diverse and can span the full range of topics covered in an undergraduate mechanical engineering degree program. An ideal individual project is one that requires the application of knowledge gained over the first three years of the student's time at university. It is important to appreciate that the student is not assessed on achieving the objectives of the project. Assessment is based more on the student's path taken in trying to satisfy the project brief.
The novel gas calorimeter replaces an aging Boy's gas calorimeter used to support undergraduate studies. A commercially available calorimeter cost of the order of £20 K. Funds are not available for a commercial calorimeter so the design and fabrication of a self-build calorimeter as an undergraduate project is the strategy employed for replacing the old calorimeter. A further complication is the original Boy's gas calorimeter uses natural gas with a continuous supply of fuel. The new calorimeter is located in a modern laboratory space with no natural gas line. This means that a straight replacement apparatus based on a Boy's gas design is not possible. The limited availability of funds restricts the budget to a maximum of £1 K.
The output of the undergraduate project is a self-built calorimeter. A calorimeter designed and fabricated in-house is an ideal calorimeter for teaching undergraduate mechanical engineering students the principles of calorimetry, heat transfer and heat exchangers. The final design fits the design brief, and it has done that by producing a novel hybrid calorimeter. The novelty of the calorimeter is it includes elements of a Boy's gas calorimeter and a cone calorimeter. The self-built hybrid calorimeter as well as reduced cost compared to a commercially available calorimeter also does not require a continuous supply of fuel.
Review of calorimetry
In many fields of engineering, chemistry and biology
Bomb calorimeter
For constant volume calorimetry the bomb calorimeter is the calorimeter of choice.
4
A bomb calorimeter is suitable for measuring the calorific value of combustible liquid fuels and solid fuels. An application of the bomb calorimeter is the combustion characteristics of dusts that represent an explosion hazard.
5
The combustion takes place in a closed vessel, which is placed in a water bath, and everything is insulated from the surroundings. The combustion reaction is ignited by a thin wire spark igniter. The bomb calorimeter also has a stirrer inside the closed vessel to ensure complete combustion occurs. Typically, pure molecular oxygen is used as the oxidizer in the combustion reaction. The temperature of the water is measured using a thermometer. The temperature rise of the water is used to calculate the calorific value. The theoretical basis is the non-flow energy equation with the work and heat transfer terms set to zero.
Boys gas calorimeter
There are a number of different flow calorimeter designs for determining calorific value for the constant pressure combustion of gases and volatile liquids. The Boys gas calorimeter is an example of a flow calorimeter. The Boys gas calorimeter is a counterflow heat exchanger design. In the Boys gas calorimeter, a burner is surrounded by a cooling coil. The burner used can be a standard Bunsen burner or a specially designed two slot burner. The combustion process should ideally be on the lean side of the stoichiometric fuel: air ratio to ensure complete combustion of the fuel. The burner-cooling coil assembly is enclosed by a chromium steel enclosure to reduce heat transfer to the surroundings by radiation and convection. The rig is instrumented to measure the temperature of the cooling water stream at the entrance and exit of the cooling coil. The temperature of the combustion stream outlet is also measured. The mass of cooling water and the volume of fuel consumed over a given time interval is measured. The pressure of the fuel is also measured to make a correction to the volume of fuel consumed to an equivalent volume at standard temperature and pressure (STP). This means the volume of fuel must be adjusted to STP if the calorific value is expressed in units of J/m3. This is done using the ideal gas equation of state.
The higher calorific value is calculated using a version of the steady flow energy equation.
Cone calorimeter
The cone calorimeter in general is used to determine the calorific value of solid fuels. Its basis is oxygen depletion rather than some form of energy equation. The advantage of this approach is that the calorific value of a composite material can be measured. It is a very useful tool in material reaction to fire studies.6,7 A schematic diagram showing the components and instrumentation of a cone calorimeter are shown in Figure 1. A sample is placed on a load cell underneath a cone shaped hood which is used to collect the combustion products. The cone shaped hood is connected to a duct. The combustion products and additional surrounding air are drawn into the duct by a fan. The sample is initially heated using an electrical radiant heater. The volume flow rate through the duct and the oxygen volume fraction are measured. A foundation of the cone calorimeter is that for the combustion of organic materials the heat of combustion based on the mass of oxygen consumed rather than the fuel has a high degree of universality.
8
For many fuels,
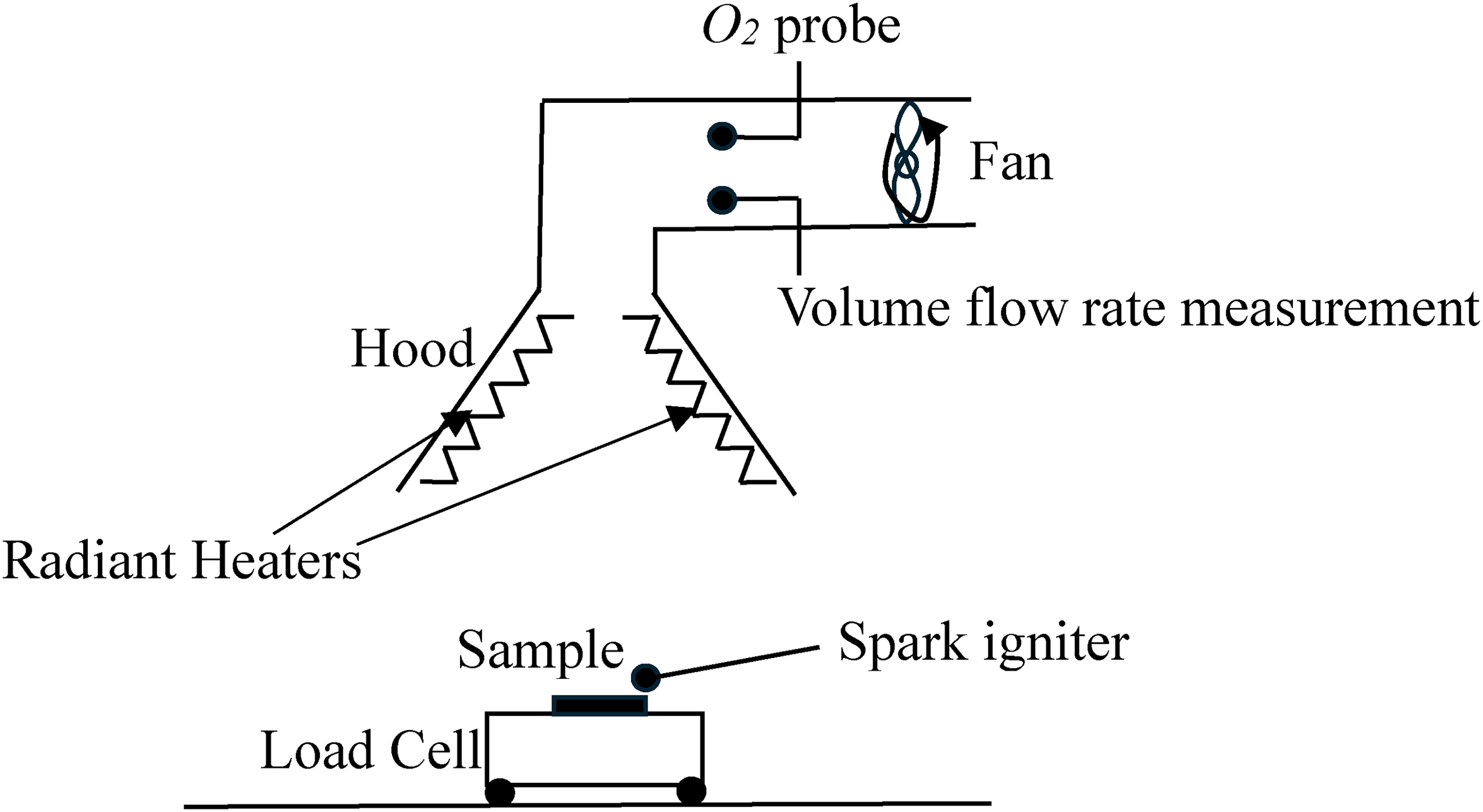
Schematic diagram of a cone calorimeter.
The mass loss rate from the sample can be determined as the gradient of the mass loss vs. time measurements taken from the load cell.
Student led energy related experiments and education
Verma et al. 9 identified a weakness in the teaching of thermal analysis in materials science and proposed a framework for addressing this issue. A survey of the literature on student led heat transfer / energy related experiments has identified a number of interesting papers. The educational experience of physics students is enhanced using a bomb calorimeter. 10 A bomb calorimeter can be used to analyze different types of plastic waste as alternative fuels. 11 The main finding of the investigation is the oxygen content in the polymer determines the viability of plastic waste as an alternative fuel. There is a body of work using the lumped capacitance model to measure thermal properties.12,13 In 12 the lumped capacitance model together with measured temperature vs. time for metal samples allows the student cohort to indirectly measure the specific heat capacity of aluminum. In 13 the lumped capacitance model is used to calculate the heat flux distribution surrounding a jet fire. This makes it possible to measure external heat flux distributions surrounding jet fires using thermocouples. Thermocouples are significantly cheaper than heat flux gauges. Mullisen14,15 published two linked papers where the specific heat capacity of aluminum is measured using an electrically heated calorimeter. In 14 the student cohort designs measured the specific heat capacity of aluminum with a relative error of 20%. In the second paper 15 a second cohort of students managed to improve their calorimeter designs to reduce the relative error to 8%. In 16 a design project to design and operate an ice calorimeter for determining the enthalpy of fusion for water is investigated.
Student led experimental design has value in enhancing the student experience. To date no one has investigated the design, build and test of an inexpensive combustion calorimeter for improving the mechanical engineering student experience.
Calorimeter design
The design specification is as follows. The calorimeter must be inexpensive to fabricate, not require a fuel line, with sufficient fuel to achieve steady state, and where possible it should use modern instrumentation and log measurements. As a minimum the calorimeter should approximately measure the HCVp of a gas or volatile liquid and be extendable to the measurement of the LCVp.
The solution to the overall design specification given above is a calorimeter that can be thought of as a hybrid between a Boys gas calorimeter and a cone calorimeter. The hybrid calorimeter has a cold-water cooling coil and uses a load cell to measure the mass loss of fuel. Assuming all of the thermal energy liberated by combustion enters the water stream through the wall of the cooling coil gives a statement of energy conservation,
Measuring fuel flow rate
The fuel flow rate is to be measured using a load cell. The load cell selected is a 2 kg Tedea Huntleigh compression load cell. This gives a mass flow rate rather than a volume flow rate. Initially the fuel considered was methylated spirits as the burner can be placed on the load cell directly. Ultimately methylated spirits as a fuel was rejected on the grounds of safety. The final choice of fuel / fuel delivery system is Liquified Petroleum Gas (LPG) in a small-scale gas cannister combined with a Trangia burner, where the burner and cannister are separated and the flame is both controllable and visible, see Figure 2. The fuel cannister holder on the load cell is 3D printed. The 3D printed cannister holder is designed to allow the ease of placement of the cannister to prevent shock loading of the load cell as the sides are flanged to allow a ‘smoother’ locating of the canister. Mechanical stoppers /bolts mean that the load cell cannot be overloaded. The cannister holder is as light as possible and is sufficiently stiff to prevent deformation due to the high bending moment between the load cell and the fuel cannister. The final design for the gas cannister holder is shown in Figure 3.

A Trangia burner.

CAD representation of the fuel cannister, cannister holder base and load cell.
Cooling coil sizing
There are physical limits placed on the cooling coil related to the size of the burner and rig stability. The helical copper cooling coil has an inner diameter of 76.2 mm such that it can be formed around a standard size length of pipe. The bore of the cooling coil is 6.8 mm with a wall thickness of 0.6 mm. To ensure a stable rig the height of the helical coil is restricted to less than 0.5 m. The helical pipe has 30.5 revolutions. The strategy for achieving the required duty is the water flow rate and fuel flow rate are to be adjusted to measure the higher calorific value for LPG. The precision of instrumentation also places limits on the mass flow rate and water flow rate.
Often the composition of LPG is not known. It is often a mixture of propane and butane, or it can be pure propane or pure butane. Pure butane gas canisters have the advantage that butane is stored at a quarter of the pressure of propane and is less expensive than propane. In terms of cooking characteristics, the difference between propane and butane is marginal. In a camping scenario it is more common to see mixtures of propane and butane as it widens the ambient temperature range that ignition and sustained combustion is possible. The LPG composition for the gas cannister used is 70% butane and 30% propane. The higher calorific value using a composition weighted average is,
Figure 2 shows the Trangia burner. The holes in the burner allow air to be entrained into the hot stream. Figure 4 shows a schematic diagram of the calorimeter with all of the important mass flow rates labelled.
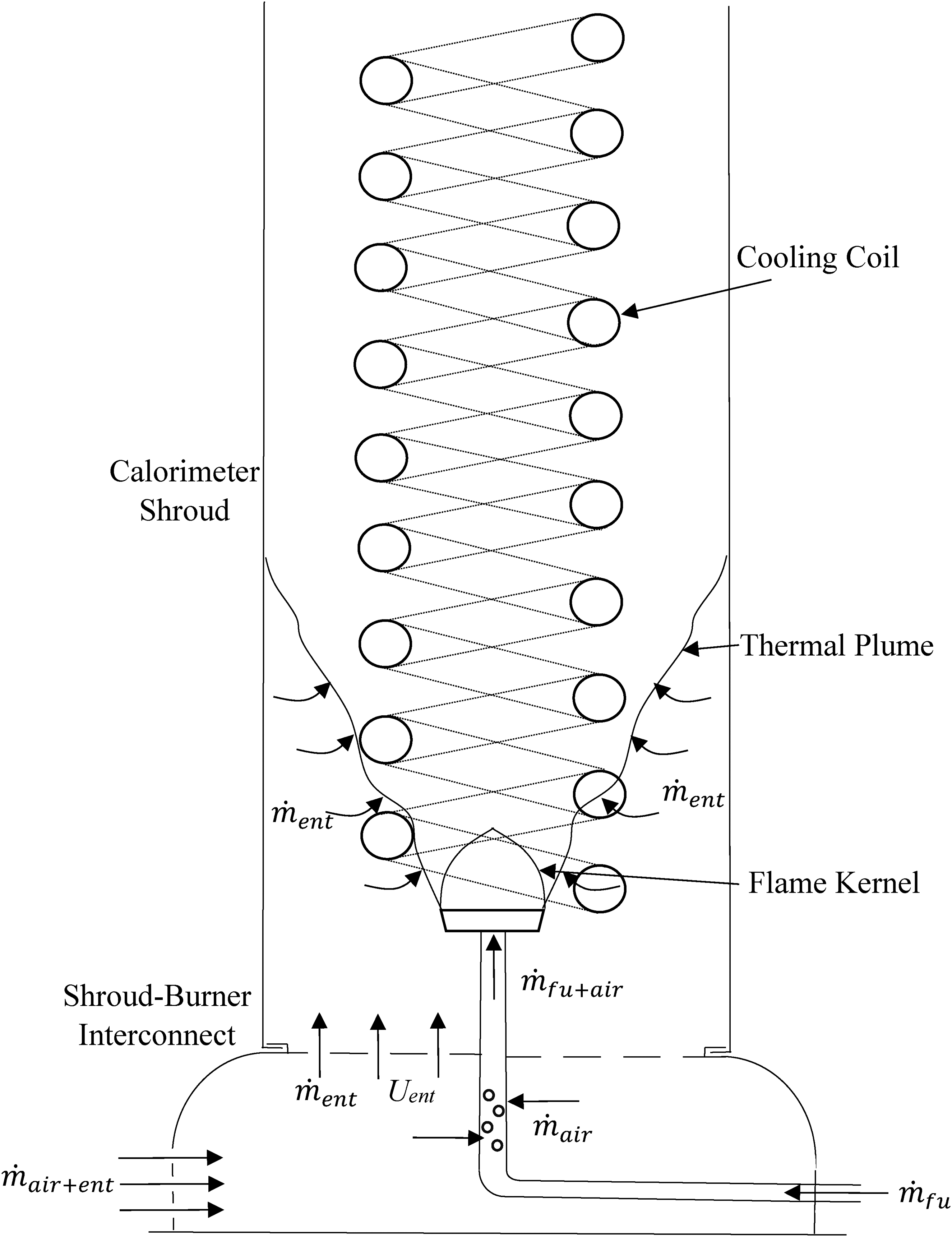
Schematic diagram of the calorimeter showing the different mass flow rates.
The design of the cooling coil is more complicated than a standard heat exchanger as the hot stream has a small, forced convection component and a more significant natural convection component for the heat transfer process. The hot stream inlet condition is also not well defined. The hot stream inlet condition consists of a stream of high temperature combustion products and a stream of cool entrained air. The mass flow rate of entrained air is not well defined. A superficial analysis suggests a sensitivity analysis of the heat exchanger characteristics to the mass flow rate of entrained air might be possible.
To make progress in the analysis you must assume all the entrained air mixes with the hot combustion products at the height of the burner. This approach leads to an unrealistic temperature at the hot stream exit of the calorimeter. The reason for this is the entrained air introduced into the base does not mix instantaneously with the combustion products. Initially the hot combustion products behave in a similar way to a free thermal plume with increasing axial height. A proportion of the introduced air will mix with the thermal plume. The thermal plume spreads out until the wall of the calorimeter affects the dynamics of the plume. This means the cooling coil will be emersed in a heterogenous flow of higher temperature hot stream with a lower mass flow rate and the air not yet entrained into the plume at ambient temperature.
This is an ideal flow for investigating using computational fluid dynamics, but that is beyond the scope of this paper.
The final assembly for the calorimeter is shown in Figure 5.
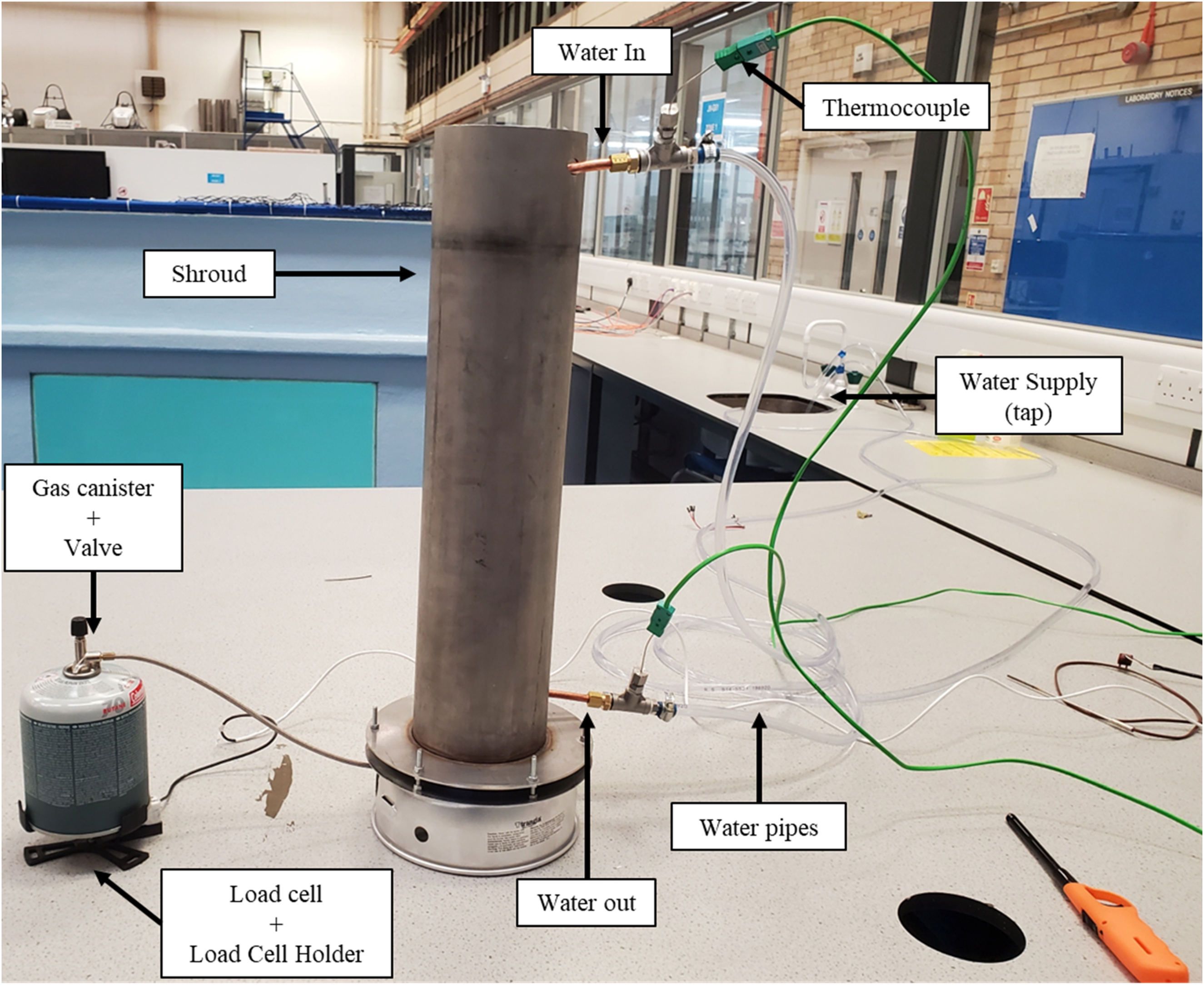
The final calorimeter assembly.
Instrumentation and data logging
The instrumentation for the calorimeter measures the load cell data and thermocouples measuring the temperature of the water inlet and water outlet. A National Instruments (NI) USB-6001 14-bit datalogger is used to capture the measurements. A 14-bit datalogger is used as it gives a balance between cost and precision. A schematic block diagram for the connectivity of the electronic units is shown in Figure 6. For the load cell there is a signal conditioning unit between it and the data logger. Signal conditioning is used to convert the signal into a much more easily readable form for the datalogger and further processing. 17 For the thermocouples the signal passes through an amplifier to give more precision over a narrower range of temperature as the expected temperature range is 5–50°C. 18 The water flow rate is measured using a measuring cylinder and stopwatch to keep the costs low.
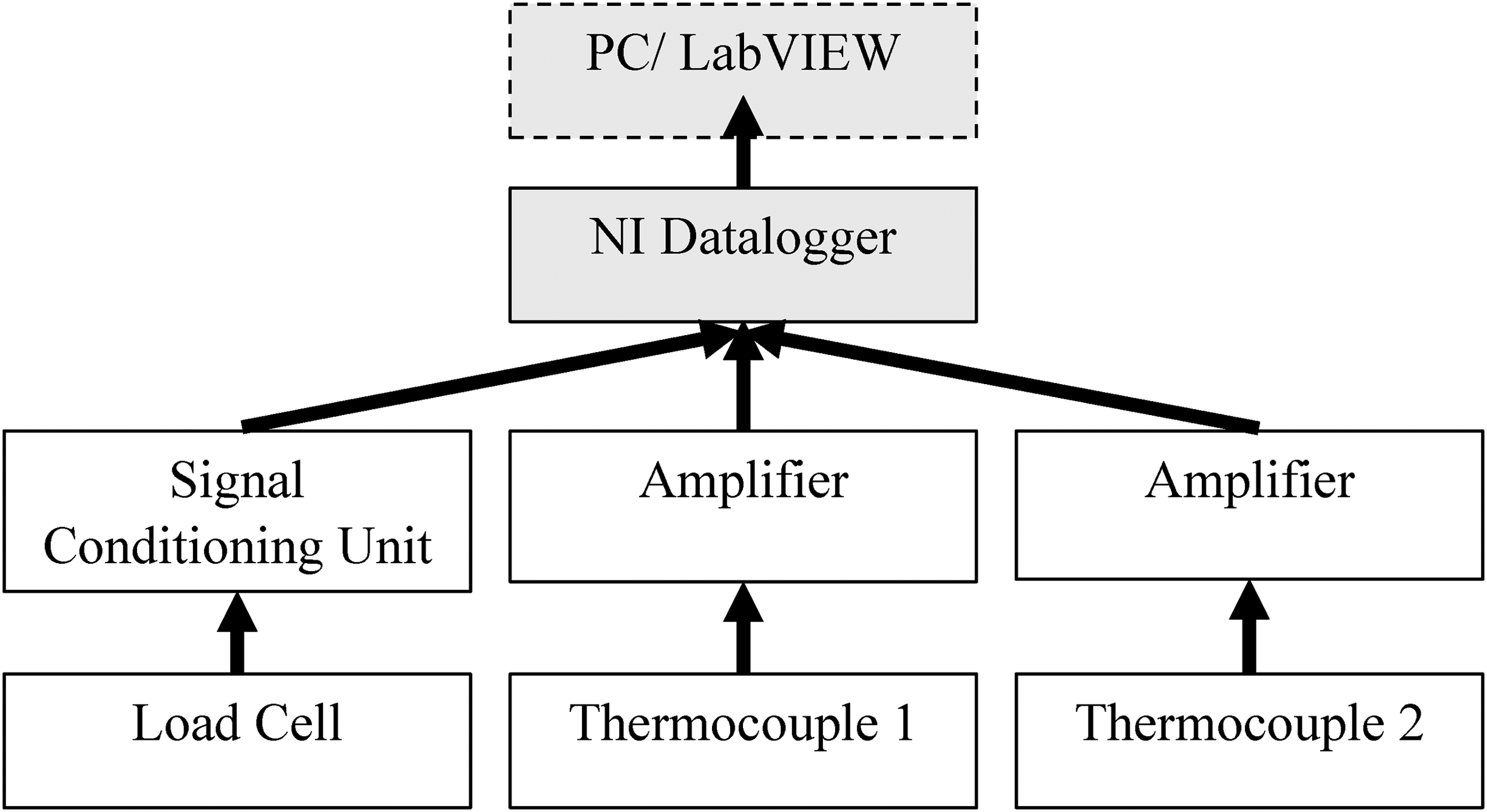
A schematic diagram of the electronic units of the logging system.
A table of the components and costs that make up the calorimeter are given in Table 1. The steel shroud and cooling coil dimensions are given in CAD drawings in the Appendix.
Cost breakdown of components of the hybrid calorimeter.

Measuring the HCVp
The designed rig logs the temperature of the water inlet and outlet as well as the fuel mass loss rate. There are some issues to do with operability that need to be investigated. Can the change in mass of the gas canister over time be measured with sufficient accuracy? As the fuel mass flow rate is calculated from the load cell data as a gradient of the mass loss vs. time, it must be linear. The theory assumes the system is at steady state. It needs to be confirmed that the rig achieves a steady state in a reasonable time.
In this section two sets of measurements are presented. For both experiments the water flow rate is set to 1.16 l/min. The two experiments are differentiated by the fuel mass flow rate. For Test 1 the fuel mass flow rate is
Figure 7(a) shows the temperatures of water inlet and outlet for Test 1. The time interval between data points is 60 s. The average temperature for the two streams neglecting the first 60 s of data is Tw,i = 18.6°C and Tw,o = 27.0°C. The mass loss data for the gas cannister is shown in Figure 7(b). A linear fit to the measured data is also plotted on the graph and has a correlation coefficient of R2 = 0.9907. Using the measurements taken and the assumption that all of the thermal energy enters the cold-water stream,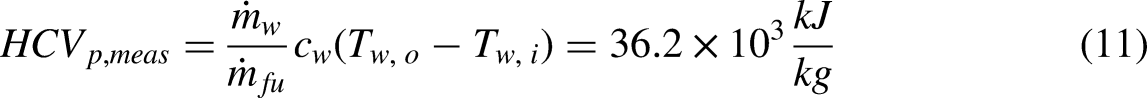
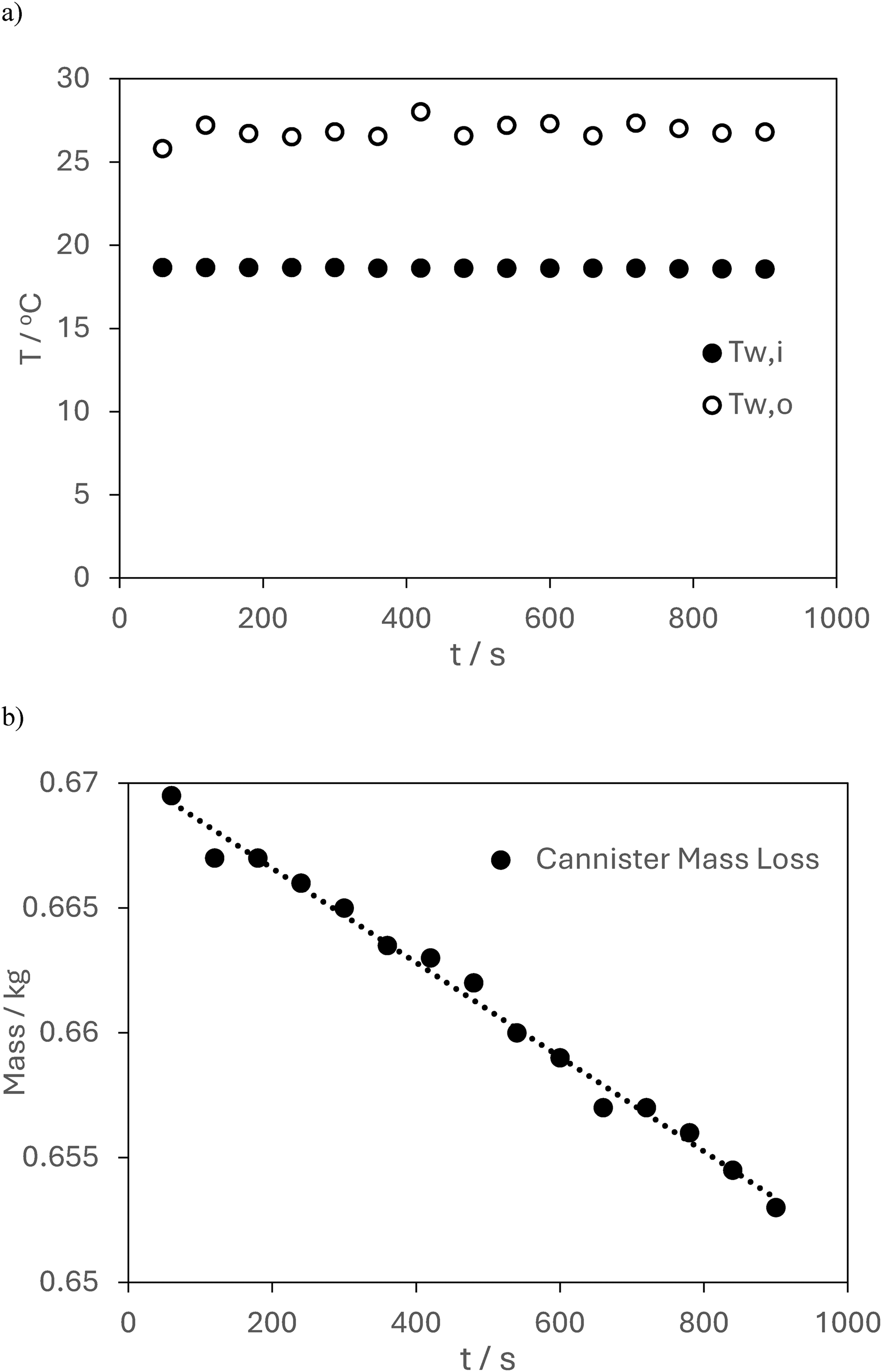
Test 1, measurement of a) temperature for entry and exit of the water stream, b) mass of gas cannister.
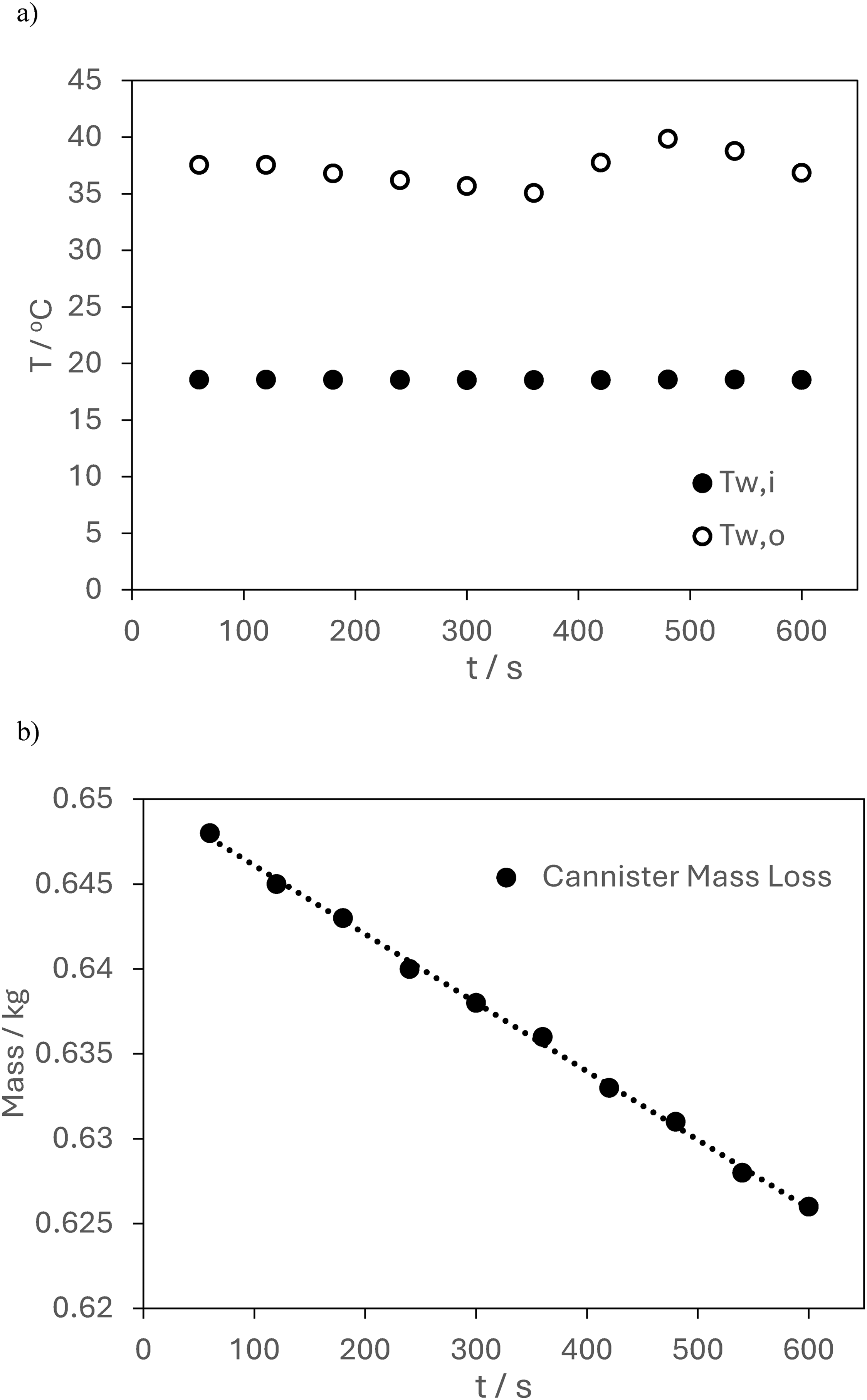
Test 2, measurement of a) temperature for entry and exit of the water stream, b) mass of gas cannister.
Compared to the HCVp for LPG of
For Test 2 the water inlet / outlet temperature measurements are shown in Figure 8(a). The average temperature for the two streams is Tw,i = 18.6°C and Tw,o = 37.4°C. The mass loss data for Test 2 is shown in Figure 8(b) together with a linear fit. The linear fit has a correlation coefficient of R2 = 0.9985. Using the measured data the higher calorific value can be calculated.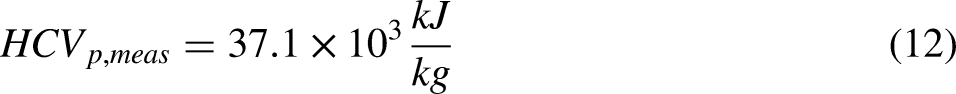
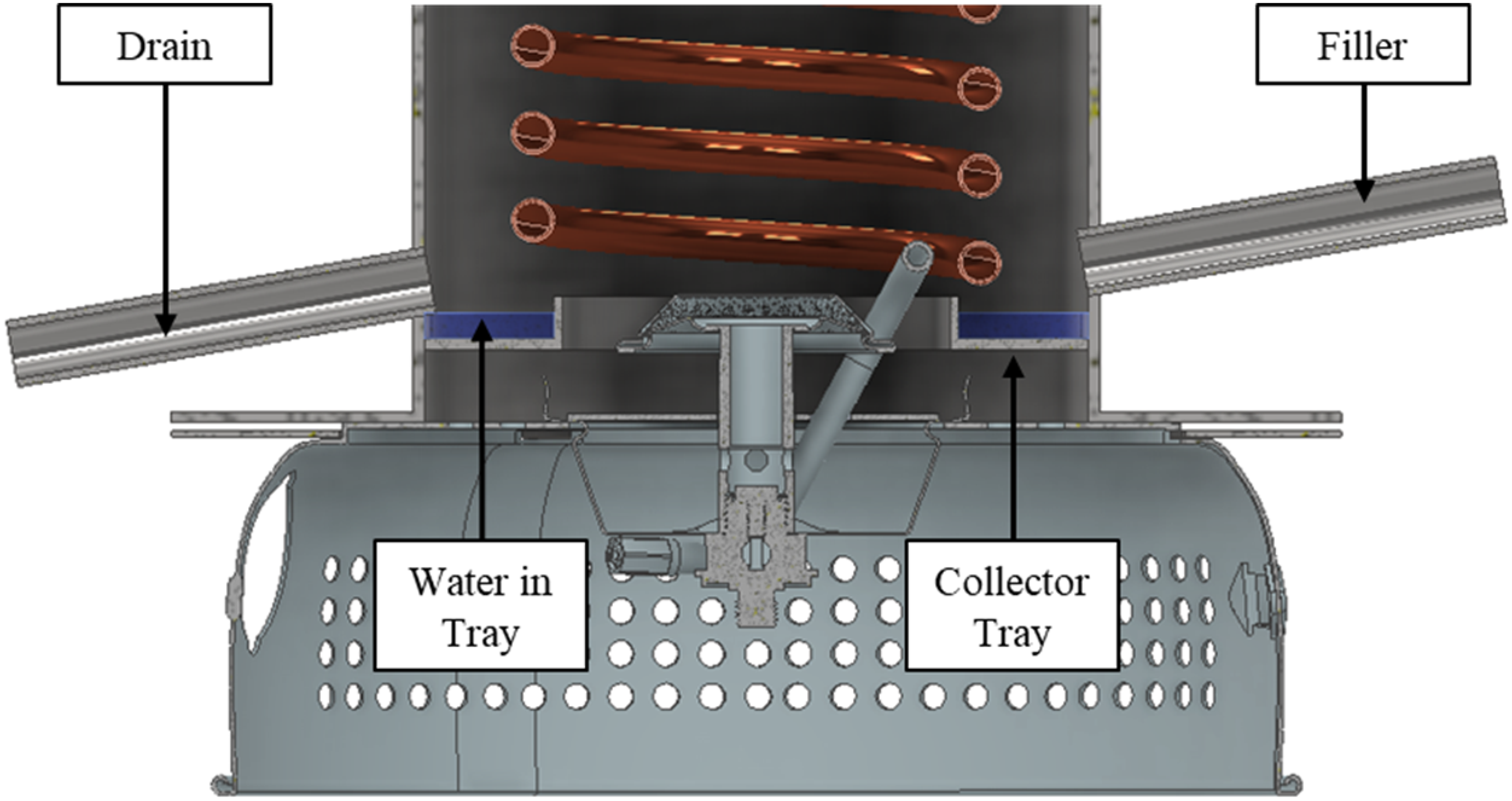
CAD rendering of the proposed water condensate collection system.
This has a relative error of 25%. It is very encouraging that the two tests demonstrate that the calorimeter produces repeatable results.
This is at the lower end of acceptable accuracy for a thermodynamics laboratory but given the simplicity and low cost of the rig is viable. For an educational rig the characteristics of a low accuracy / inexpensive self-built calorimeter is of value as it prompts discussion within the student cohort as to what is causing the discrepancy? How could the calorimeter be improved? Indeed, a second undergraduate project in the future will look at these issues and develop the calorimeter further to allow it to produce two sets of results, with and without any improvements.
Future developments
In this section any potential improvements to the rig are considered.
Increasing the accuracy of HCVp, meas
The analysis and experiments presented above indicate a more complete energy equation for the calorimeter is,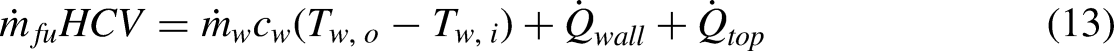
The calorimeter could also be extended by adding a pump and water reservoir to produce a truly independent bench top laboratory experiment. The instrumentation could be extended to something more sophisticated for measuring the water flow rate than a measuring cylinder and stopwatch.
Extension to LCVp measurement
To calculate the lower calorific value the water vapor in the combustion product stream must be collected and measured. The lower calorific value is then calculated using (8). In the current calorimeter the water vapor condenses on the cooling coil and then drips onto the base of the burner. To capture the condensate, it is proposed to introduce a collector plate with a ridge and an outlet drain such that the condensate can empty into a measuring cylinder This is similar to a Boys gas calorimeter. The design also includes an inlet to make it simpler to fill the collector plate to ensure the collection time for the condensate is kept to a minimum. A CAD design of the burner/ collector plate is shown in Figure 9.
Conclusion
The findings of a design/ build undergraduate capstone individual project is presented. The motivation for the project is a requirement for an inexpensive combustion calorimeter that demonstrates the principles of calorimetry. The calorimeter is to be used as a second-year thermodynamics laboratory to support the delivery of a course on thermodynamic systems and cycles. The solution is a hybrid calorimeter for measuring the higher calorific value of LPG. The calorimeter is considered a hybrid calorimeter as it has elements of a Boys gas calorimeter and a cone calorimeter.
The measurements presented demonstrate proof of concept of the design with the measured higher calorific value having a relative error of approximately 25%. This is at the lower end of acceptable for an educational thermodynamics experiment. There are a number of possible improvements/ extensions that could be explored as part of another undergraduate capstone individual project. It could be argued that it is a much stronger student experience using a self-built calorimeter compared to a commercially available calorimeter as the question “How can this be improved?” and the follow-on question “What simulations or experimental and theoretical analysis are required?” are useful questions. Consider asking the analogous question of a high accuracy calorimeter, “Why is this calorimeter so accurate?”, such a question does not have the same educational impact. It is far more interesting to a student cohort to look at the possibilities of the improvement of the apparatus compared to a situation where the analysis is complete.
An educator wishing to use the findings of this paper could consider the fabrication of a similar calorimeter either as a student / design project or with the intent of expanding the thermodynamic laboratory activities on their degree program. The different components of the calorimeter as well as costs are stated in Table 1. It would be advisable to look to improve the calorimeter design as suggested in Section - – Further Developments.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.