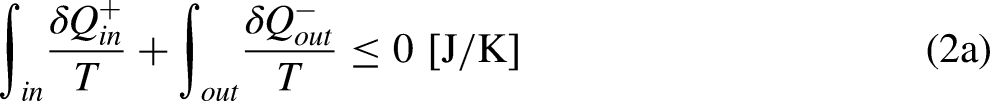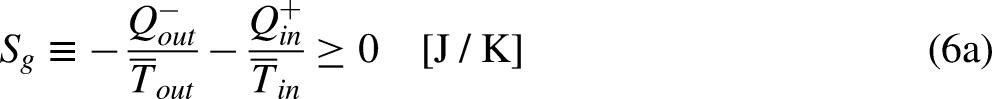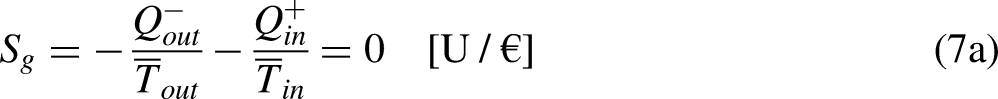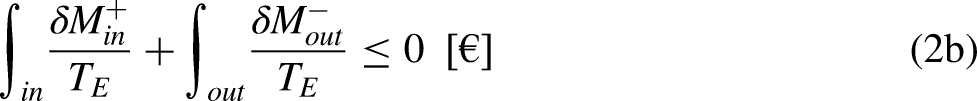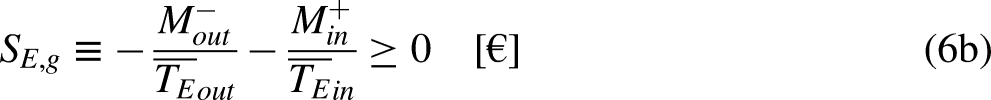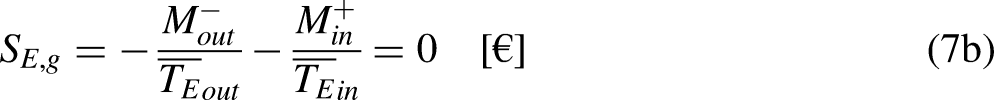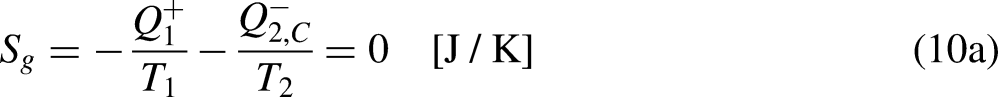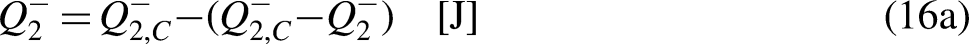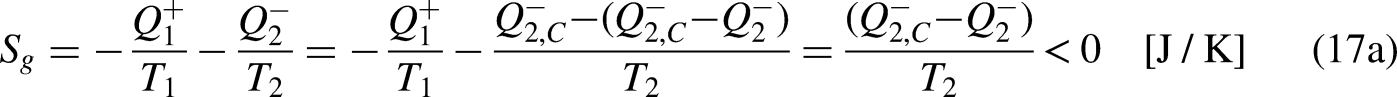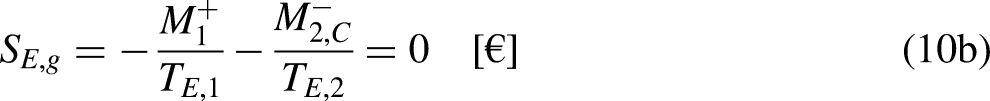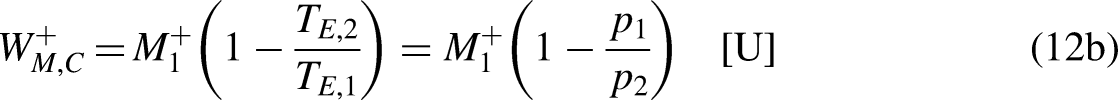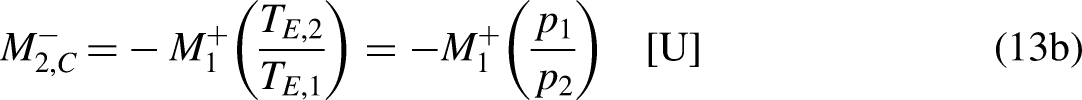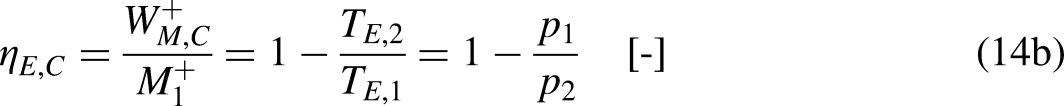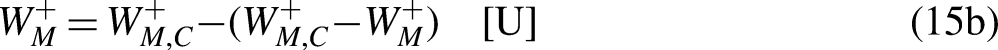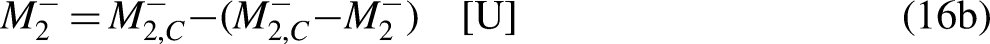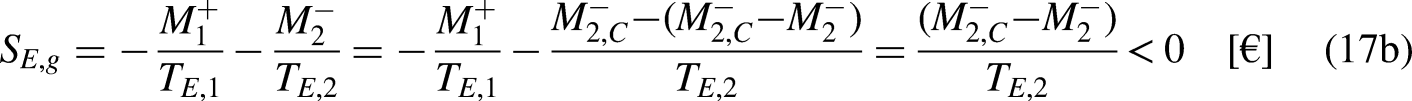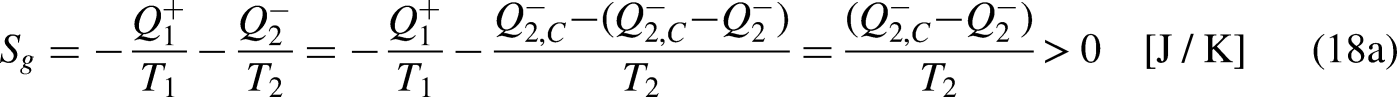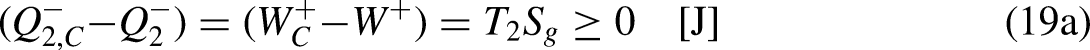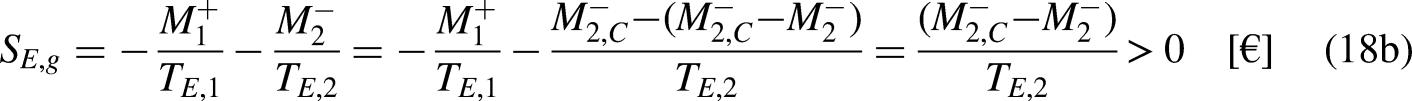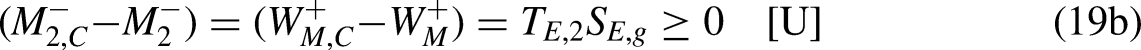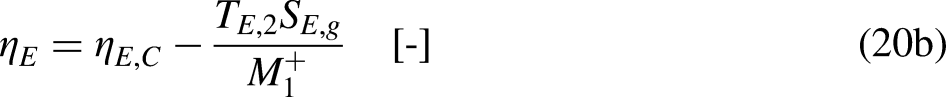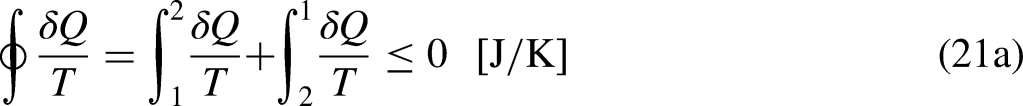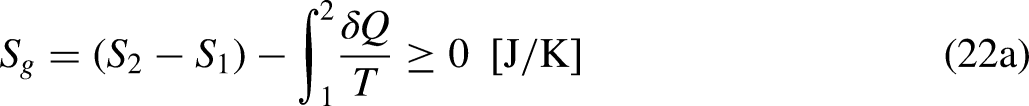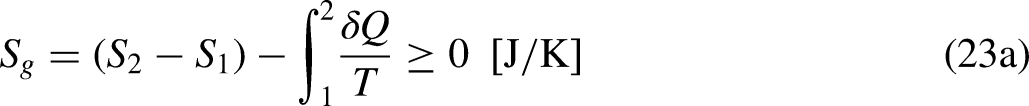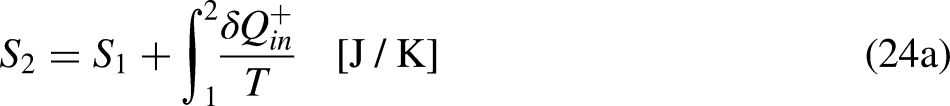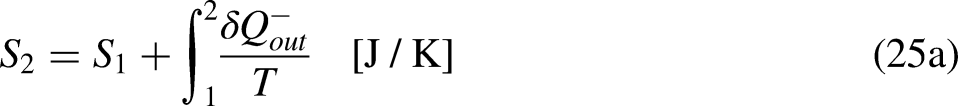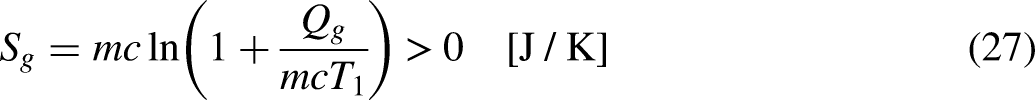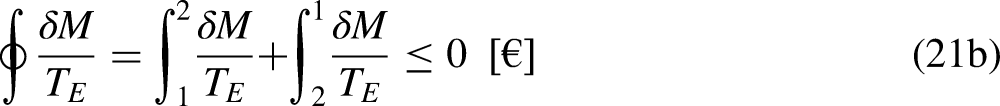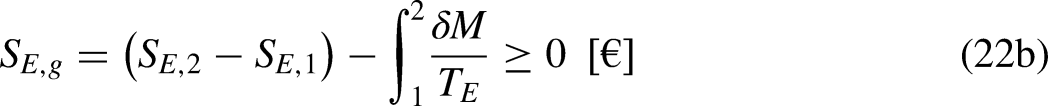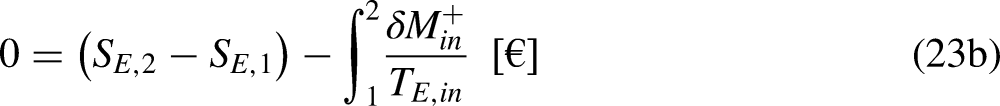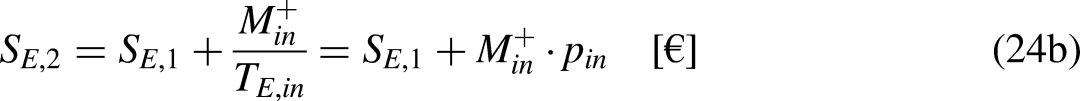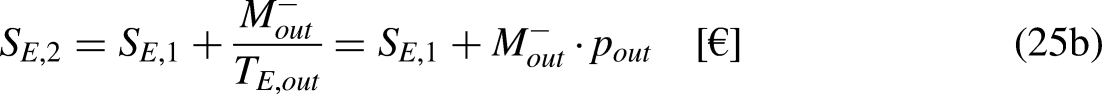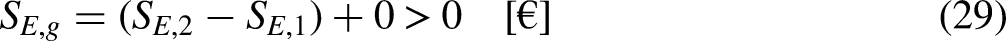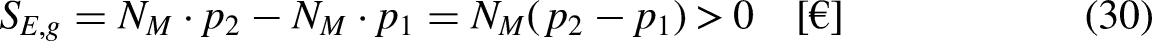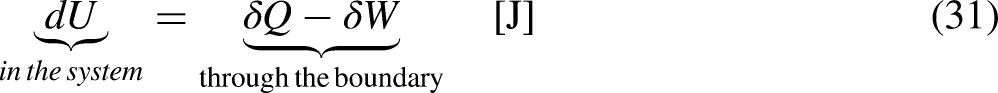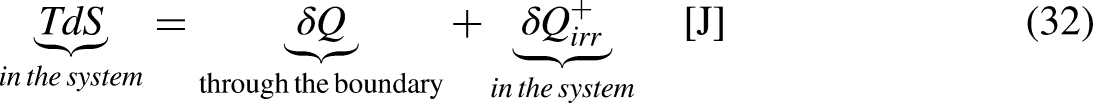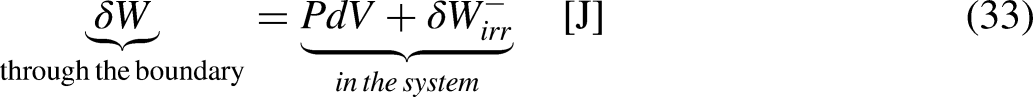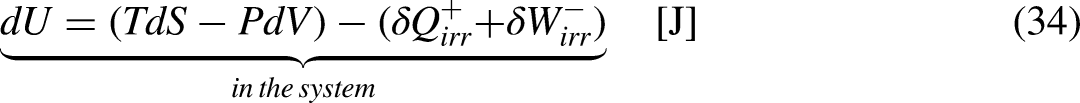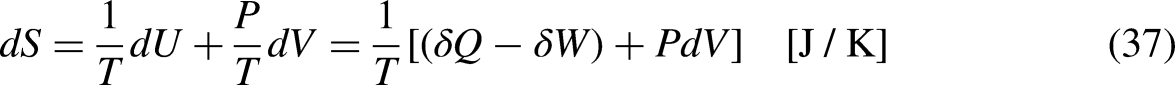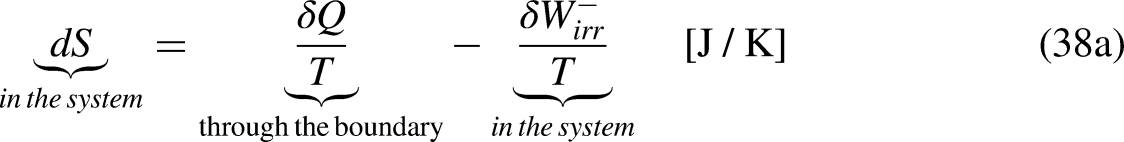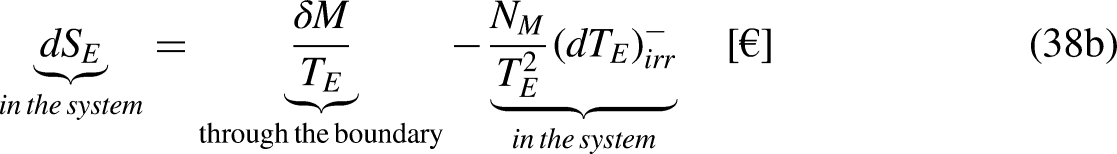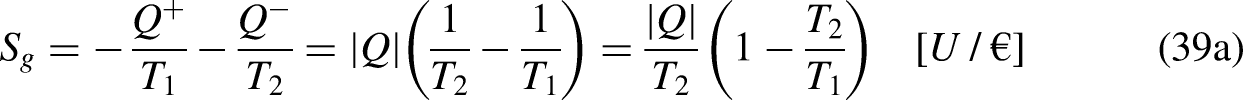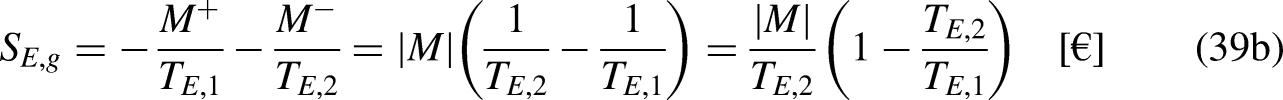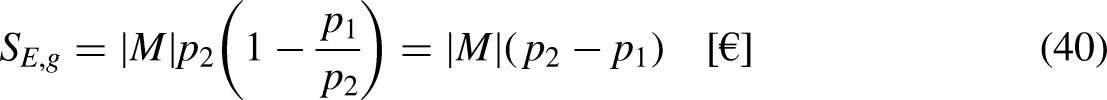Abstract
Teaching and learning Thermodynamics have challenges. Energy and the First Law as the Energy Conservation Principle are usually well accepted. This is not the case with the Second Law, entropy, entropy generation, and the consequences of the Second Law. Analogies can be used to help learn and understand the Second Law of Thermodynamics, entropy, entropy generation, irreversibility, and power cycles. The present work proposes a Thermodynamics-Economics analogy. The analog of heat transfer (flowing in the decreasing temperature direction, driven by a temperature difference) is the traded merchandise transfer (flowing in the increasing unit price direction) in normal trading operations. Developments start with the economic analog of Clausius inequality. The analog of entropy is the merchandise financial value, and the analog of the entropy generation is the financial value generation. Irreversibility is associated with entropy generation, and economic irreversibility is related to economic entropy (financial value) generation. The reversible and irreversible power cycles also have their economic analogs. The irreversibility associated with heat transfer through a finite temperature difference also has its economic analog. The economic Second Law developments are more familiar to students and more easily understandable, which can help them learn the Second Law of Thermodynamics and its implications.
Introduction
Teaching and learning Thermodynamics have their problems and challenges. Work in 1 analyzes why Thermodynamics is usually considered a difficult subject. That motivates research and experimental/classroom approaches in the search for improved methodologies and solutions to solve those issues. Mulop et al. 2 analyze the main difficulties of Thermodynamics learning and present a review and analysis of different approaches that can help. If the referred problems are general, they increase in relevance when related to entropy. 3 Brown and Sing 4 report how a survey indicated significant challenges for students’ understanding of Thermodynamics, thermodynamic processes, the First and Second Laws, and entropy. O’Connel 5 points out the main challenges of Thermodynamics teaching and learning. In, 6 Bain et al. review the research on Thermodynamics teaching and learning. A recent survey on research works on Thermodynamics teaching and learning can be found in. 7 In, 8 Yang et al. proposed some methods to overcome the challenges of heat transfer and Thermodynamics teaching and learning. Other efforts are made to simplify the students’ tasks, such as the recent work by Çengel and Kanoglu 9 proposing a new function for helping students deal with entropy calculations of ideal gases whose specific heats depend on temperature, and the work of Wang et al. 10 proposing a helping learning analytic system. In 11 Gaggioli gives his opinions based on his extensive experience in Engineering Thermodynamics teaching. Smith 12 emphasizes how students’ previous experiences can be intuitively related to energy, the Second Law, and entropy. With this in mind, analogies, invoking different but familiar domains, are also important to increase the students’ curiosity and interest, facilitating Thermodynamics learning and understanding.
The one-way direction set by the Second Law is usually well accepted, but not the Kelvin-Plack statement of the Second Law, contrary to the Clausius statement which seems obvious. It can be proved the equivalence between these two statements of the Second Law;13,14 however, the developments leading to the Clausius Inequality and the definition of the property entropy are based on the less understood and less accepted Kelvin-Planck Statement. 15 Even if energy is not tangible, its effects are well perceived and felt. Its consumption can be measured using counters, its consumption is paid, and the Energy Conservation Principle is accepted without major problems. This is not the case of entropy, a strange thing that can be generated from nothing. Once installed the entropy strangeness, discomfort, and disgust, these feelings extend to everything related to the Second Law and entropy.
Several approaches can be followed to attract students to Thermodynamics making it an interesting and challenging subject to learn, study, and apply.1,13,14 The applicability of Thermodynamics to virtually all domains and all kinds of problems13,14 is one of its major strengths, yet usually, students see that as an additional problem, as no typical problems or well-established typical solutions exist for all problems. References 13 and 14 (both 9th editions) result from successive pedagogical improvements based on the extensive experience of their authors in Engineering Thermodynamics teaching. Introduced novelties are mainly related to the pedagogical approach, trying to make Engineering Thermodynamics appealing and interesting for the students, highlighting how it is strongly related to, and helping understand, almost everything, technical and/or present in the student's everyday life. The usual Engineering Thermodynamics treatment in Mechanical Engineering, including that of the Second Law, entropy, entropy generation, irreversibility, and power cycles, are those proposed and followed in the referred textbooks.13,14
Analogies can be used to help Thermodynamics teaching and learning, using more familiar or even common-sense analogs to help understand the less familiar, and harder-to-understand, Thermodynamics issues. 16 This always bearing in mind that some care is needed when teaching/learning using analogies. 17 It is interesting to mention in this context the work of Norton, 18 dissertating on how ‘analogy helped create the new science of Thermodynamics’, and the seminal Carnot work 19 based on the caloric theory, analogously to what happens with the falling flowing fluids. If analogies may be useful in science and engineering teaching and learning, there are challenges when using analogies in teaching. As examples we may have situations when students do not identify the used analogy and its usefulness, they might not recognize or understand the analogy of the auxiliary used domain with the source domain effectively under study, or they are not able to understand (simultaneously) the power and the weaknesses of the used analogies. Due to that, analogies are double-edged swords. Instructors/teachers need to be aware that any analogy may fail at some point, opening the way for miscomprehensions. Even thus, given the difficulties usually related to Thermodynamics teaching and learning, it is tempting to try new approaches, in this case through the proposed analogy, as it may increase interest and understanding of the Second Law and entropy as analogs to a more familiar domain for everyone.
Costa20,21 proposed a parallel structure for looking at Economics through the eyes of Thermodynamics. These are far from being the first studies proposing an analogy for looking at Economics through the eyes of Thermodynamics, as pointed out in 20 and the references therein. However, to the best of the author's knowledge, they are the first ones based on the analogy between heat transfer (flowing in the decreasing temperature direction, driven by a temperature difference) and the traded merchandise transfer (flowing in the increasing unit price direction) in normal trading operations. The works by Costa20,21 emphasize the setting, analysis, and discussion of a parallel structure for looking at Economics using the eyes of Thermodynamics, and not in using the Thermodynamics-Economics analogy for helping the teaching and learning of the Second Law, entropy, and related subjects. This is the objective of the present paper: as there is an interesting and useful analogy between Thermodynamics and Economics, and as everyone is familiar with merchandise transfer and profit generation in trading operations, that familiarity can be advantageously used for a better understanding of the Second Law of Thermodynamics, entropy, and related subjects. An analogy exists not only for variables but also for the relevant mathematical expressions.
This work presents the developments for each subject, in each section, firstly in Thermodynamics (its main motivation), and secondly in Economics (in the present context, a more familiar domain for those learning Thermodynamics). Section 2 introduces and discusses the Clausius Inequality, entropy definition, and entropy generation. Section 3 analyzes the Carnot cycle, and Section 4 analyzes the power cycles arbitrary in terms of reversibility. In Section 5, the previous developments that apply to cycles are extended to apply to general processes, cyclic or not. Section 6 revisits the differential equation defining entropy and highlights the conditions in which it applies, and Section 7 is devoted to the irreversibility of the transfer through a finite temperature difference.
As many ideas, concepts, variables, and relations appear in the text, in two different domains, the units of the involved variables are indicated inside square brackets for accuracy and easier reading and understanding.
The Clausius Inequality, entropy, and entropy generation
The starting point of the presented developments is the Clausius Inequality, which can be obtained in both Thermodynamics13–15 and (its economic counterpart) in Economics, based on the observations from the thermal engines and the economic activity (specifically from the observations of normal merchandise trading),20,21 respectively. The developments leading to the Clausius Inequality start with the Kelvin-Plack statement based on those observations.13–15,20,21
The thermodynamic system under analysis is separated from the rest of the Universe by a boundary, an imaginary frontier separating what is under analysis from what is out of analysis. The thermodynamic system may exchange heat and work with its neighboring through the impermeable parts of the boundary, and mass and energy through the permeable parts of the boundary if it is an open system.13–15 These are the mass and energy interactions of the system with its neighboring. Exchanges may occur through the boundary, and processes may occur in the system, inside the boundary.
Analogously, the economic system under analysis is separated from the rest of the Universe by a boundary, an imaginary frontier separating what is under analysis from what is out of analysis. The economic system may exchange merchandise, wealth, and money with its neighboring through the boundary, which can be crossed by material (tangible, hardware) or immaterial (intangible, software, ideas, information, knowledge) species. The economic system can be an individual, a family, a set of individuals, an organization, a city, a county, a country, or a set of countries. Exchanges may occur through the boundary, and processes may occur in the economic system, inside the boundary.
In Thermodynamics
The developments are based on heat and temperature, and on the fact that heat flows spontaneously from higher to lower temperatures (which is an observed fact, imposed by nature).
The Clausius Inequality sets that, in a cycle
15
Considering now the heat entering the system in one cycle,
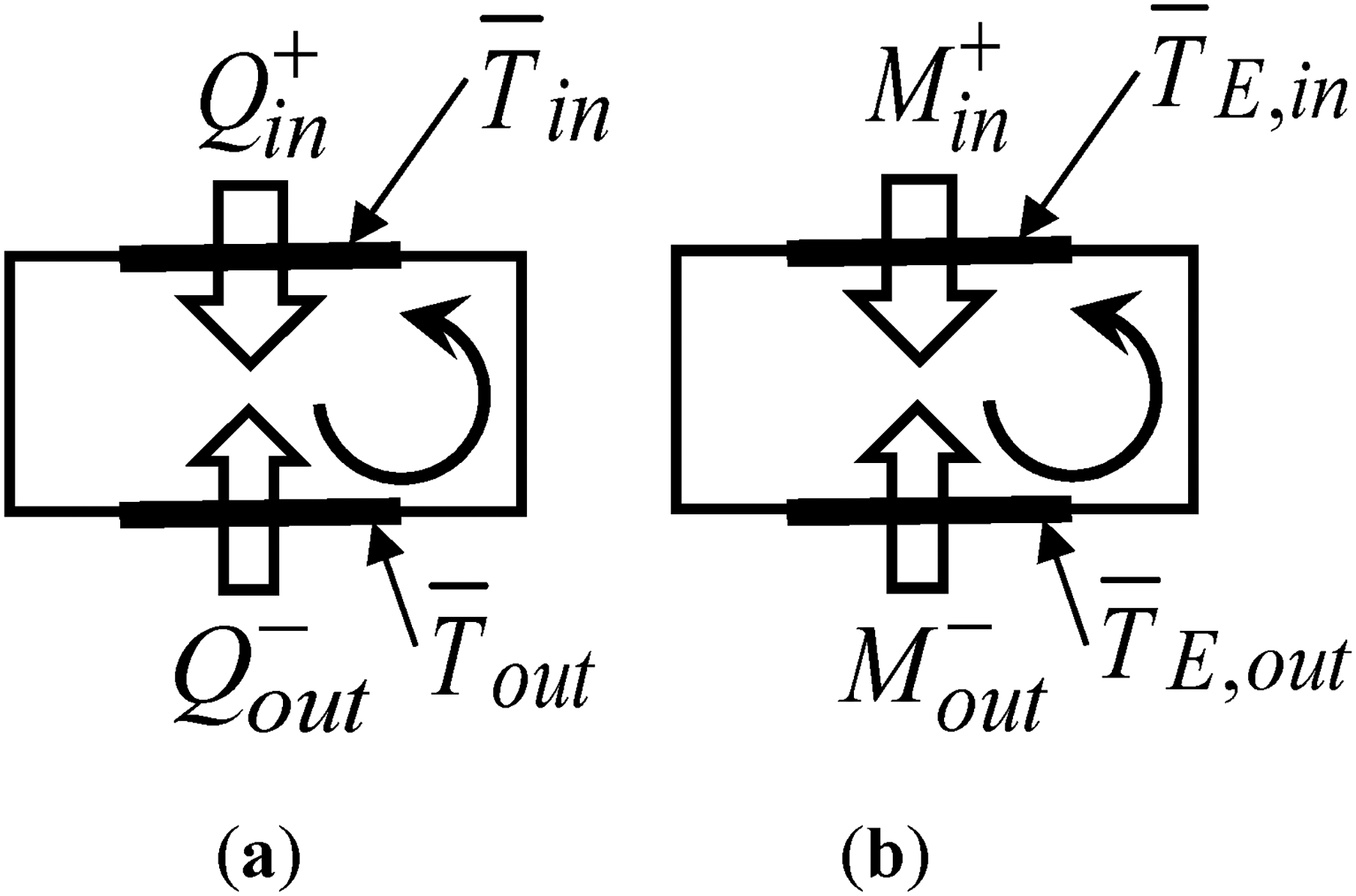
Auxiliary figure for a better understanding of the Clausius inequality: (a) In thermodynamics; and (b) in economics.
This result sets that, in a cycle, the output ratio
A reversible cycle can be reversed, that is, operated in the reverse direction, reverting its mass and energy interactions, without requiring other additional effects, namely without investing additional external energy. On the contrary, an irreversible cycle can be operated in the reverse direction only if other additional effects are provided.
The limiting equality situation of the Clausius Inequality corresponds to the equality in Eq. (4a), which must correspond to the absence of heat generation in the system due to irreversibility (lowest heat
It is well-known from Calculus that if the cyclic integral is null the integrand function is an exact differential. This is the basis for the definition of the new property entropy, through its differential expression, as
When the system operates irreversibly, some heat generation occurs in the system associated with that irreversibility, thus leading to entropy generation in the system. For the situation for which Eq. (3a) prevails, the entropy generation is defined as the strength of inequality, as
If no heat leaves the system in one cycle,
The mechanical work transfer interaction W [J] can be seen as heat crossing the system's boundary at an infinite temperature. In terms of energy, it continues being the mechanical work, W [J], and from the entropy viewpoint it has the associated null entropy
Only for a reversible cycle (this whose sense can be reversed, inverting its energy (work and heat) transfer interactions with no other additional effects)) it is
In Economics
The developments are based on traded merchandise and economic temperature, and on the fact that traded merchandise flows spontaneously in the increasing unit price direction in natural trading operations.20,21
The economic temperature
Temperature is a property of a thermodynamic system,13–15 and all the chemical species in the thermodynamic system share the same temperature value.13–15 The proposed economic temperature is not a property of the economic system but a property of one traded merchandise species in the economic system, as it depends on the context, which influences its unit price when crossing the economic system's boundary.20,21 Different merchandise species in the same economic system have different economic temperatures. A more complete and accurate definition of the economic temperature must refer to merchandise species i,
Additionally, the presented developments apply to situations when there is no generation or destruction of merchandise species in the economic system.
The economic Clausius Inequality sets that, in a cycle
21
A reversible economic cycle can be reversed, that is, operated in the reverse direction, reverting its merchandise, wealth, and money interactions, without requiring other additional effects, namely without investing additional external merchandise, wealth, or money. On the contrary, an irreversible economic cycle can be operated in the reverse direction only if other additional effects are provided.
The limit equality situation of the economic Clausius Inequality corresponds to the equal sign in Eq. (4b), which must correspond to the absence of merchandise financial value generation in the economic system due to irreversibility, that is, it must correspond to the reversible situation. The reversible situation is the one for which the cycle is perfect and can be reversed by inverting its merchandise (traded merchandise and merchandise wealth) transfer interactions.
It is well-known from Calculus that if the cyclic integral is null the integrand function is an exact differential. This is the basis for the definition of the new property economic entropy, through its differential expression, as
21
From Eq. (5b) it is anticipated that the economic entropy of an economic system is its merchandise financial value. Thus, if the economic system is composed of
If no traded merchandise leaves the economic system in one cycle,
Merchandise wealth is merchandise that is not in the market and is thus not transferred by trading reasons/motivations. Consider, for example, a loaded truck, the load being (tradable) merchandise but not the truck itself (which, in this sense, is merchandise wealth).20,21 In a different context, the truck can itself be tradable merchandise. In common language, it is usual to say that (true) wealth corresponds to things that are not on the market, are not to be purchased or sold, and have no price, or, in the present context, have a null unit price, which is the same as saying that they have an infinite economic temperature. Analogously as for mechanical work in Thermodynamics,13–15 the merchandise wealth transfer interactions
The merchandise wealth
Only for an economic reversible cycle (this whose sense can be reversed, inverting its merchandise (traded merchandise and merchandise wealth) transfer interactions with no other additional effects)) it is
The Carnot cycle
The Carnot cycle is crucial in Thermodynamics.13–15 In this section, it is analyzed as usual, and its analog, the economic Carnot cycle, is proposed and analyzed.
In Thermodynamics
For the (reversible) Carnot cycle in Figure 2a, it is null the entropy generation
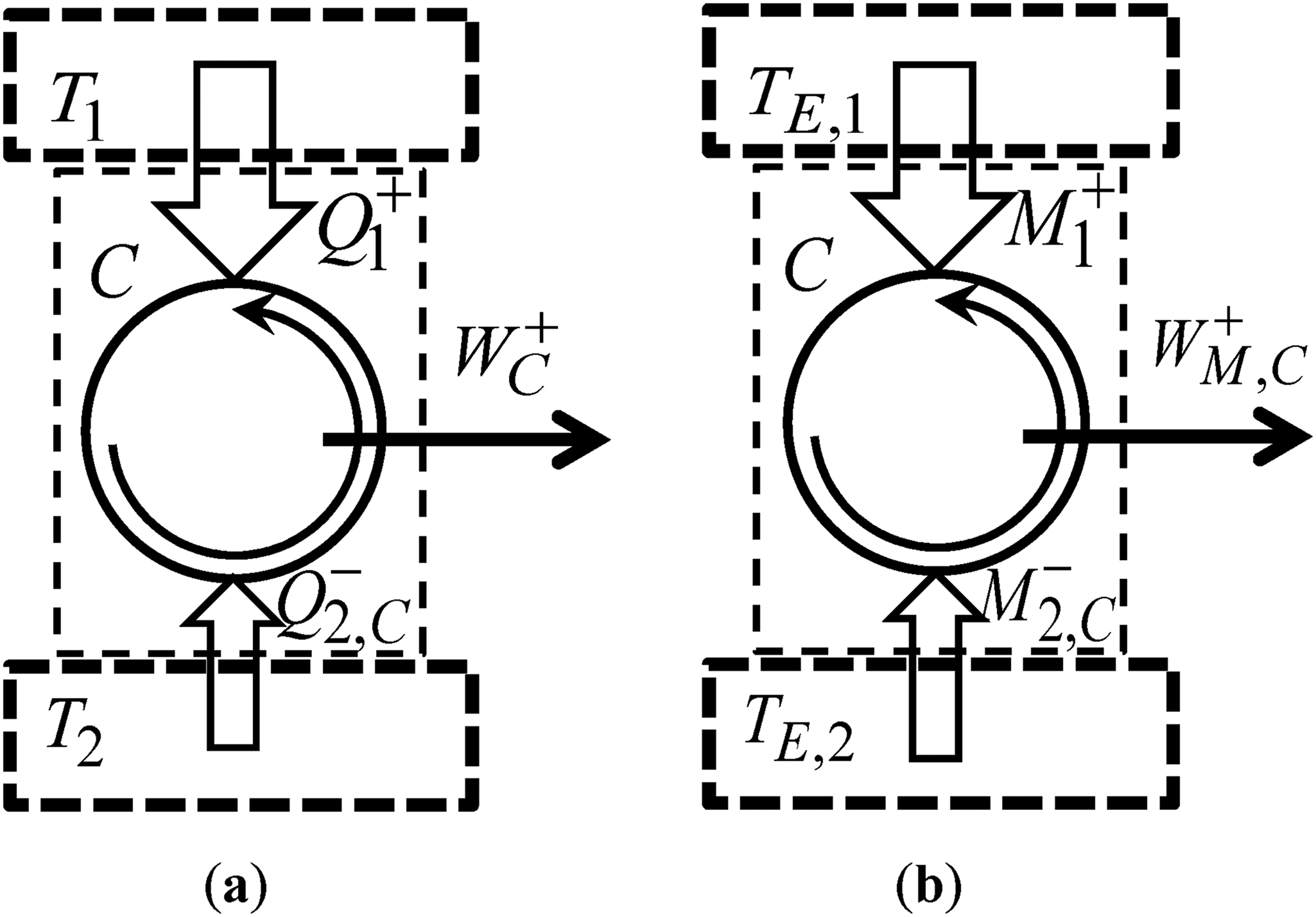
Graphical representation of the Carnot cycle: (a) In thermodynamics; and (b) In economics.
For a given
At this point, it can be easily understood why even the Carnot thermal engine, the best thermal engine, is unable to integrally convert heat into mechanical work. If that would be possible, for such a situation it would be
In Economics
For the (reversible) economic Carnot cycle in Figure 2b, it is null the economic entropy generation
At this point, it can be easily understood why even the economic Carnot engine, the best economic engine, is unable to integrally convert traded merchandise into merchandise wealth in normal trading operations. If that were possible, in such a situation it would be
At this point, it can be understood how the economic Carnot cycle partially converts traded merchandise into merchandise wealth,20,21 which helps understand how the Carnot cycle partially converts heat into mechanical work.13–15 Considering Figure 2b, the traded merchandise
Cycles arbitrary in terms of reversibility
The irreversibility concept and entropy generation are crucial in Thermodynamics.13–15 In this section are analyzed cycles arbitrary in terms of reversibility and the associated entropy generation, and their economic analogs, the economic cycles arbitrary in terms of reversibility and the associated economic entropy generation.
In Thermodynamics
For a fixed
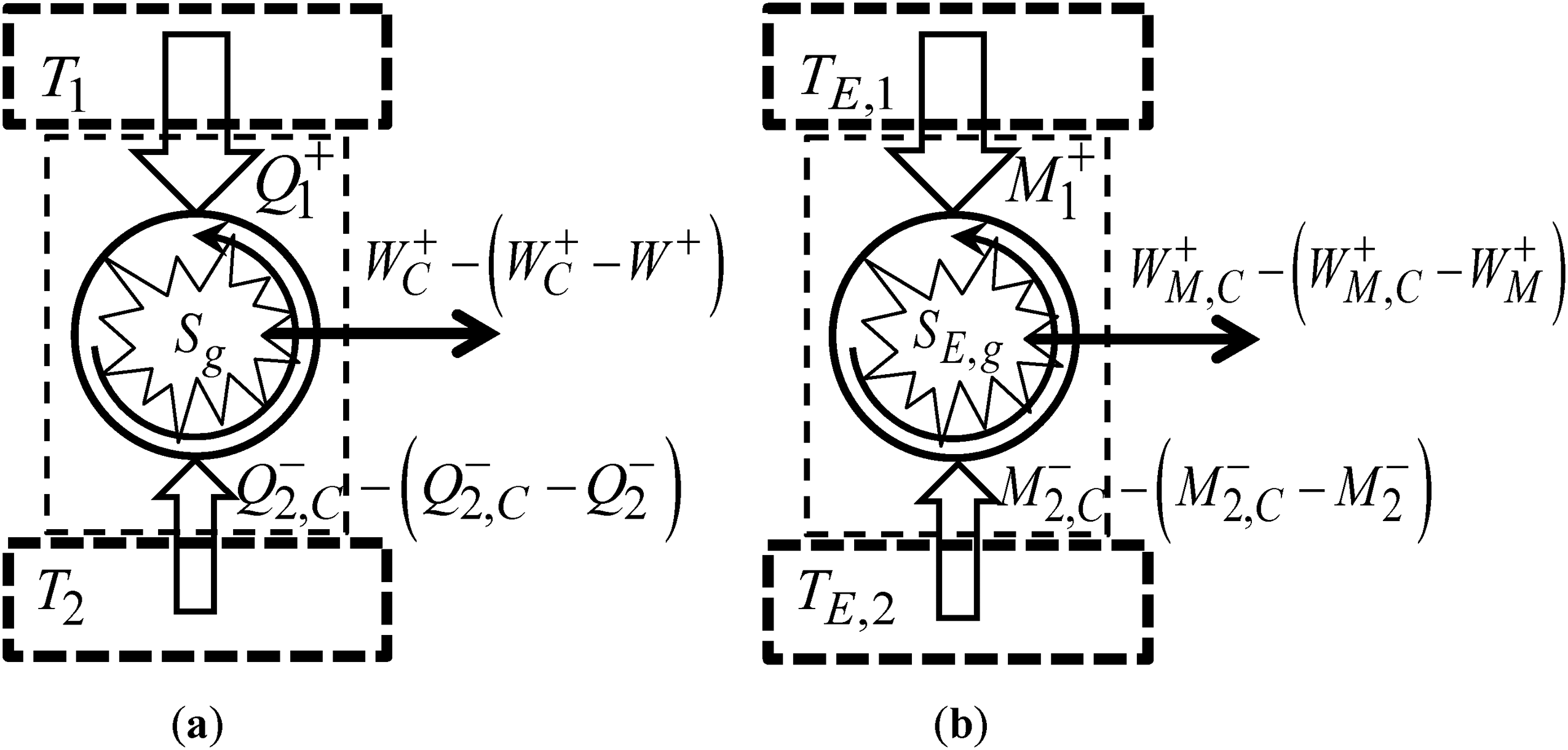
Graphical representation of a cycle arbitrary in terms of reversibility: (a) In thermodynamics; and (b) In economics.
As given by Eq. (18a), the entropy entering the system
In turn, the efficiency of the irreversible thermal engine,
In Economics
For a fixed
In turn, the efficiency of the irreversible economic engine,
The normal economic activity is to create merchandise wealth and introduce it into the market, giving it a unit price, to generate economic entropy (to generate financial value). The economic normal is not to have an economic engine operating cyclically to obtain the maximum merchandise wealth from normal merchandise trading.20,21 However, the economic cycles and the associated concepts are of major relevance to the Second Law developments and understanding.13–15,20,21
Processes in general
Sections 2–4 were devoted to cyclic processes. The analysis is now extended to general processes, cyclic or not. As illustrated in Figure 4, any cycle can be decomposed into two processes, process 1–2 being arbitrary in terms of reversibility and process 2–1 being reversible.
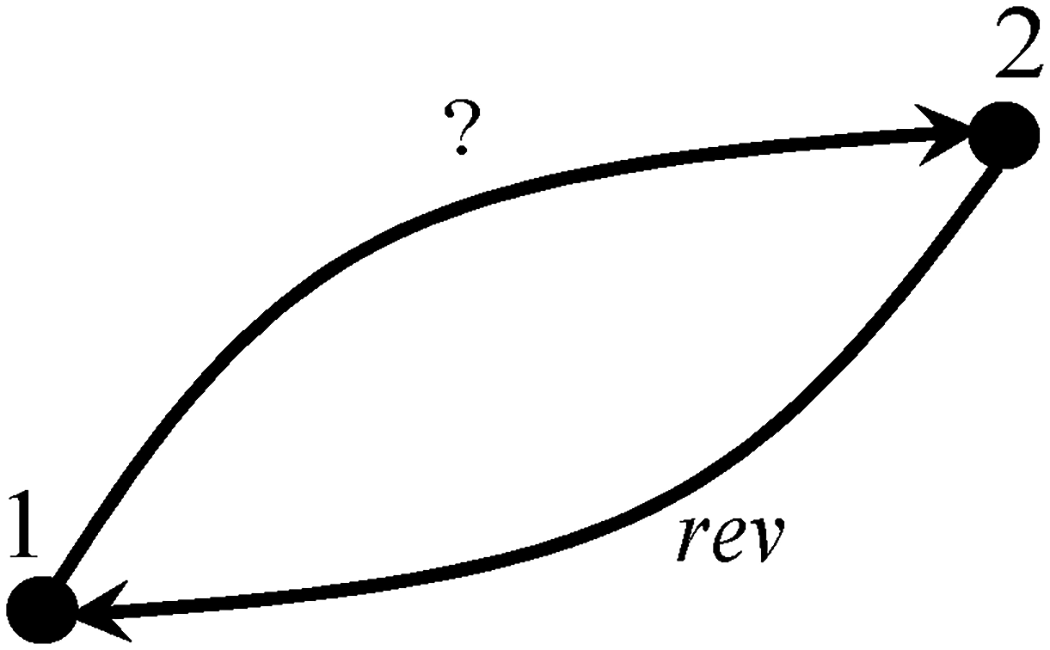
A cycle as composed of two processes, process 1–2 being arbitrary in terms of reversibility and process 2–1 being reversible.
In Thermodynamics
Given Figure 4, Eq. (1a) can be written as
For example, if we have the reversible heating of a body, Eq. (22a) gives
Consider now the irreversible process of water heating in an electric water heater, where electrical resistance generates heat
In Economics
Given Figure 4, Eq. (1b) can be written as
For example, if we have the
If we have the irreversible process of increasing the unit price of the
In general, we prescribe the economic entropy generation (the financial value generation) to be obtained from the economic systems’ operation, the merchandise interactions required for that being a consequence. In this case, we act based on the economic Second Law (financial value), the results (merchandise units transfer interactions) being an economic First Law consequence of that.
Entropy is generated (appearing from nothing) in irreversible processes. The same happens with the economic entropy (the financial value), which is generated (appearing from nothing) in irreversible economic processes, as is the case of the processes increasing the merchandise unit price.
Revisiting the equation defining entropy
The property entropy is defined through a differential equation for the special case of an internally reversible process.13–15 Such an equation and the requirement of internal reversibility are revisited. Its economic analog, the differential equation defining the merchandise economic entropy, and the requirement of the internal economic reversibility involved in its definition, are detailed.
In Thermodynamics
The First Law of Thermodynamics (the energy balance equation) for a closed system sets that13–15

Graphical representation for a better understanding of the fundamental relation of thermodynamics.
Inserting Eqs (32) and (33) into Eq. (31) leads to
The Fundamental Relation, Eq. (36), can be rewritten as
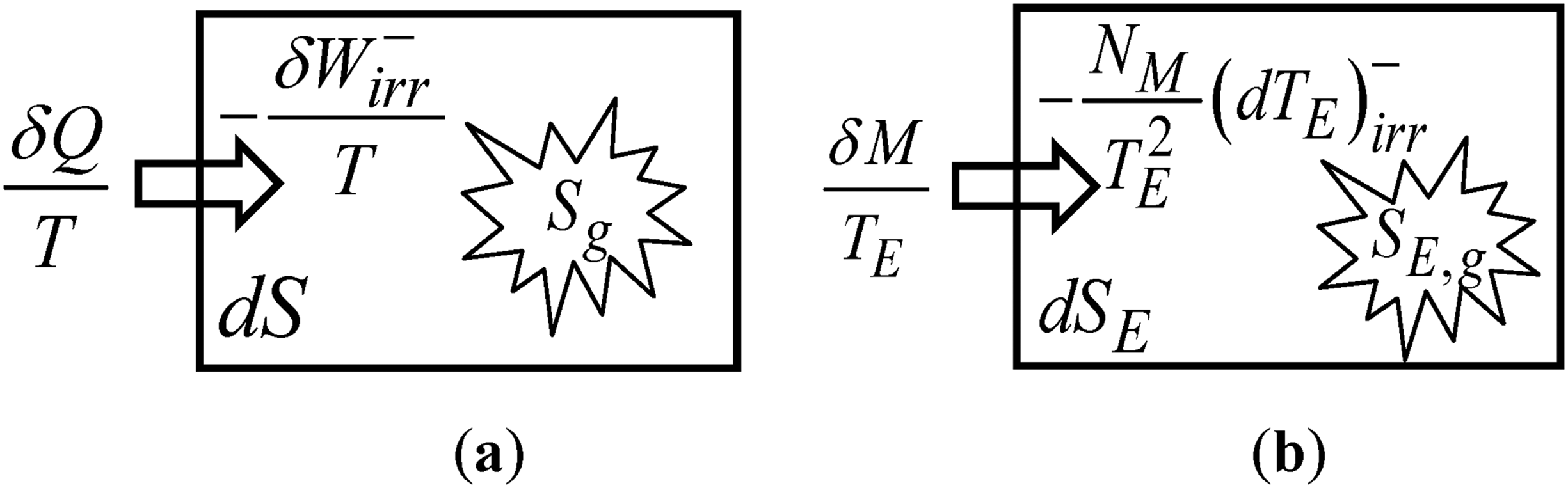
Graphical representation of the equation relevant to defining entropy: (a) In thermodynamics; and (b) In economics.
For an internally reversible process
In Economics
The proposed state equation (Eq. (9)) of the economic system composed of only one merchandise species is simpler than the state equation for thermodynamic systems.13–15
Differentiation of Eq. (9) leads to
For an internally reversible process
Transfer through a finite temperature difference
The heat transfer through a finite temperature difference and the associated entropy generation are of major relevance in Thermodynamics.13–15 They are analyzed in this section, as well as their economic counterparts of merchandise transfer through a finite economic temperature difference (through a finite unit price difference) and the associated economic entropy generation.
In Thermodynamics
The entropy balance equation for the thermodynamic system sandwiched between the heat reservoirs at temperatures
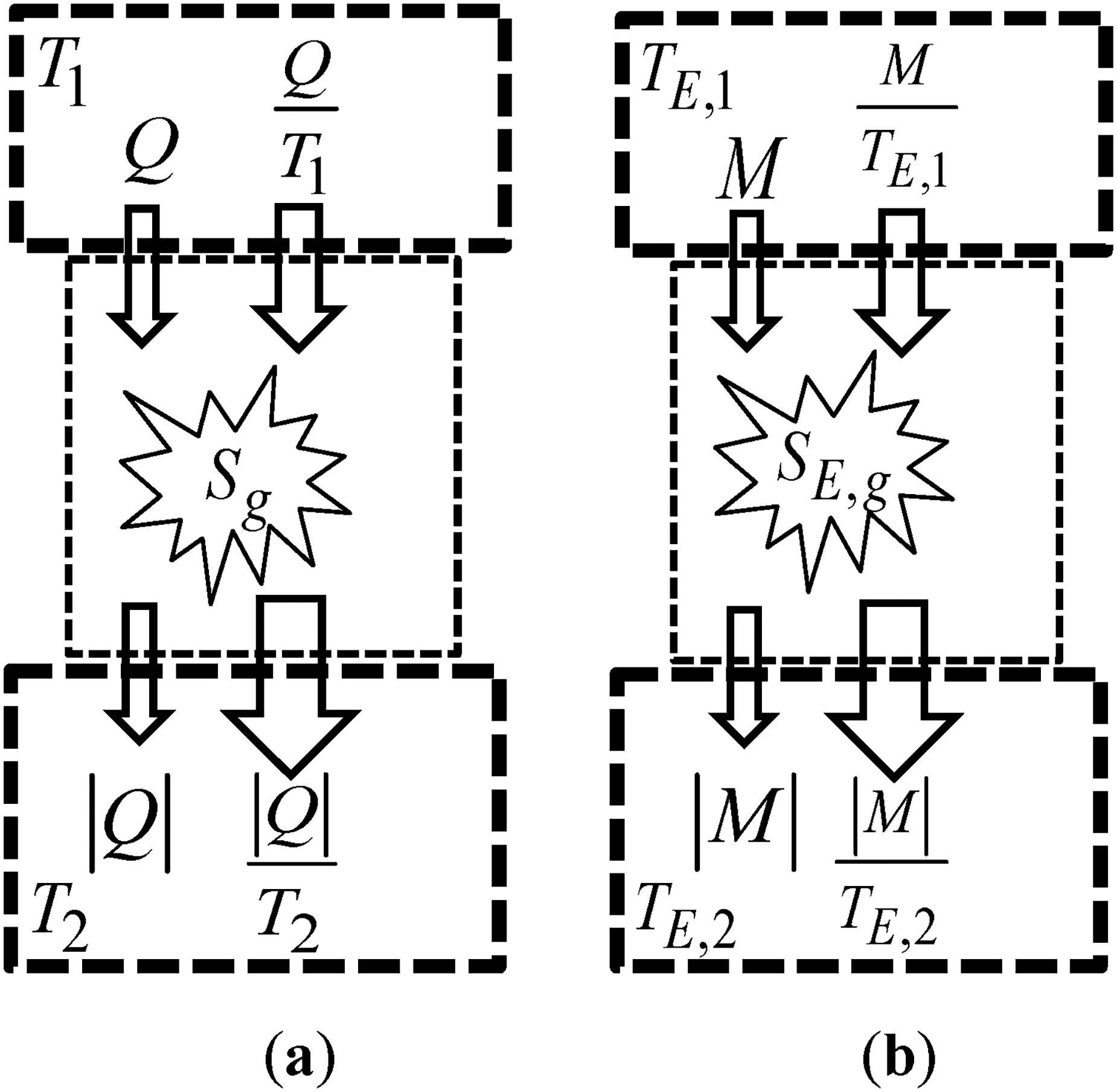
Graphical representation of the transfer through a finite temperature difference: (a) In thermodynamics; and (b) In economics.
Taking Eq. (12a) in mind, and as
In Economics
The economic entropy balance equation for the economic system sandwiched between the merchandise reservoirs at the economic temperatures
Taking Eq. (12b) in mind, and as
Equation (39b) can be rewritten as
Eq. (40) highlights also a very well-known result, even if it is not usually read as an economic entropy generation. Product
Conclusions
An analogy is proposed to help learn and understand the Second Law of Thermodynamics and related subjects. As a parallel structure of that of Thermodynamics can be set for Economics, and especially for normal trading operations, and everyone is familiar with the merchandise trading operations and the profit generation (the financial value generation) in normal trading operations, these economic processes can be taken as the analogs of thermodynamic processes. This analogy can be used advantageously to help learn and attract to the study of Thermodynamics, and especially to learn and study the Second Law of Thermodynamics and related subjects. The proposed and explored analogy exists for both variables and relevant mathematical expressions.
The main results of the proposed analogy include:
- The traded merchandise transfer (flowing in the increasing unit price direction) in trading operations as the analog of heat transfer (flowing in the decreasing temperature direction, driven by a temperature difference), - The differential equation defining the economic entropy as the economic analog of the differential equation defining entropy, - The merchandise financial value as the economic analog of entropy, - The financial value generation in normal trading operations as the analog of entropy generation, - The economic analog of irreversibility, - The economic irreversibility related to economic entropy (financial value) generation in normal trading operations as the analog of entropy generation in irreversible processes, - The (reversible)economic Carnot cycle, the analog of the Carnot cycle, - The economic analogs of the irreversible power cycles, and the associated economic entropy generation (the associated financial value generation), - The economic entropy generation in merchandise transfer through a finite unit price difference as the analog of the entropy generation in heat transfer through a finite temperature difference.
The proposed analogy is advantageous for better learning and understanding of the Second Law of Thermodynamics and related issues. It is also useful in giving new insights and promoting the use of new approaches for economic and financial analyses. It sets analogies of the (only apparently different and non-related) Thermodynamics and Economics domains.
Footnotes
Nomenclature
Greek symbols
Subscripts
Superscripts
Acknowledgements
This work was supported by the projects UIDB/00481/2020 and UIDP/00481/2020 - FCT - Fundação para a Ciência e a Tecnologia; and CENTRO-01-0145-FEDER-022083 - Centro Portugal Regional Operational Programme (Centro2020), under the PORTUGAL 2020 Partnership Agreement, through the European Regional Development Fund.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundação para a Ciência e a Tecnologia, (grant number UIDB/00481/2020).
Data availability statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.