Abstract
Wildlife field guide books present salient features of species, from colour and form to behaviour, and give their readers a vocabulary to express what these features look like. Such structures for observation, or observational grids, allow users to identify wildlife species through what Law and Lynch have called ‘the difference that makes the difference’. In this article, we show how these grids, and the characteristics that distinguish species, change over time in response to wider concerns in the community that use and make the field guides. We use the development of Dutch field guides for dragonflies to show how the ethics of observing wildlife, the recreational value of dragonfly observation, the affordances of observational tools, and biodiversity monitoring and conservation goals all have repercussions for how dragonflies are to be identified. Ultimately, this affects not only how dragonflies are to be observed and identified, but also what is taken to be ‘out there’. The article is based on a transdisciplinary cooperation between a dragonfly enthusiast with emic knowledge and access, and an STS researcher. We hope the articulation of our approach might inspire analyses of other observational practices and communities.
Keywords
In this article, we analyse the development of Dutch dragonfly field guides since 1964, to show how these guides have built on the experiences, concerns, and incentives of the community of Dutch dragonfly enthusiasts, and how in turn they supported the further development of the community. Among enthusiasts’ concerns were not only ethics of interacting with wildlife and nature conservation, but also the appeal of learning, recreational values, the development of the observational community itself, and even elements of a gaming logic. By analysing these guides, we show that these concerns shaped how dragonflies were to be observed and identified, what were to be considered identifying characteristics and, ultimately, which dragonflies were made observable.
Our analysis hinges on the notion of observational grids (Grasseni, 2004, 2007b), which are conceptual devices that highlight salient features of an organism or its behaviour and that articulate the terms by which these features can be expressed. For instance, a damselfly has a ‘sleek abdomen’, while the emperor dragonfly, Anax Imperator, ‘patrols its territory’. Observational grids shape and facilitate the development of skilled vision (Goodwin, 1994) in observational practices, by the naked eye, or supported by tools such as cameras, binoculars, wildlife traps, or combinations thereof. (Observational grids are not to be confused with the plastic frames biologists used to mark quadrats under investigation, although these too focus attention.) In addition, we will show that, beyond observational practices in the field, grids also structure how these observations are shared and assessed in the community of practice. Apart from the grid articulated in a field guide for the purpose of wildlife identification, further grid layers operate in the naturalist communities as they integrate observations in comprehensive accounts and databases (E. Turnhout & Boonman-Berson, 2011). These grids similarly identify key features of observations (such as place or weather) that are maintained in biodiversity records. As a conceptual infrastructure in a communal observation practice, these layered grids help to articulate what has been seen, by whom, when, where, and under which conditions. As these accounting practices feed into nature conservation, these grids ultimately contribute to the particular articulation of protection practices (Kasperowski & Hagen, 2022).
In their seminal article on observational acts in birdwatching, Law and Lynch (1988) demonstrate how field guides shape what can be seen out in the field. With the use of basic graphic pointers and by highlighting key features, such guidebooks alert users to the ‘difference that makes the difference’ in species identification: the distinctive characteristics that observers should learn in order to name wildlife species. Law and Lynch’s study pioneered the analysis of how field guides allow and structure observation ‘in the wild’, that is, how identifying features are presented, how the guides’ instructions support observation, and how they direct the attention of an observer in the fleeting presence of a bird.
In a sequel publication, Law and Lynch compare the affordances of drawings versus photographs, further improving our understanding of how pictorial representations in field guides support taxonomic identification. Naturalists consider stylized drawings to be more accurate than photographs because they are representations of key characteristics, and therefore superior renditions of the wildlife ‘out there’ (Lynch & Law, 1998), although new digital imaging possibilities have reshaped this debate. Clearly, field guide images are not unmediated reflections of nature, but idealized representations, shaped by concerns such as adequate identification in a taxonomic system—itself an idealized systematization of biodiversity’s complexity. However, before field guides shape observers’ attention (the original focus of Law and Lynch’s work), the representation of biodiversity in these books is itself shaped by the concerns of their users and producers. Much like the schematized representation of the London Subway map (Turnbull, 2007; Vertesi, 2008), biodiversity field guides are designed and idealized representational devices, bundling particular concerns embedded in specific practices. How such concerns and practices shape biodiversity field guides, and the ‘differences that make the difference’ within them, is the focus of this article.
The issues at stake reach well beyond identifying birds, or in our case, dragonflies. Not only wildlife identification, but observational practices more generally require observers to recognize, name and discern salient features of what is to be observed. Whether through the lens of a microscope or binoculars, or on photographs or computer-generated images, seeing and naming are epistemically complex feats (Goodwin, 1994; Lynch, 1988). Analysing and improving the accomplishment of identification is a concern of both practitioners and social scientists studying them. However, more is at stake than just a technical achievement of correct identification, because what is to be observed, why, and how, are part of the complex distributed cognition (Hutchins, 1995) developed by communities such as dragonfly enthusiasts. We will show how what counts as salient features in observational practices is readjusted to changes in the composition, goals, tools, ethics, and game-like logics (Lynch & Law, 1998) of these communities and their extended networks. We will show how the notions of the observational grid (Grasseni, 2004, 2007a) and the game can be expanded to analyse how observations are structured and curated in such networks. Understanding how and why readjustments occur, as well as their consequences for what can be seen and made present, allows for better epistemic appreciation of observational data, as well as a fairer and more respectful appreciation of observational communities of practice (Bowker, 2000; Ellis & Waterton, 2004; Hinchliffe, 2008; Wenger, 1998).
Law and Lynch analyse field identification from the perspective of two casual users, but further research on observational tools has expanded this focus. Field guides not only structure observation, but also project specific conceptions of their users, which becomes particularly clear as observational guides and tools move beyond books to apps or, for some species, even DNA ‘barcoding’ (Ellis et al., 2009). The observational tools and their instructions, particularly as they go beyond introductory guides for casual observers, encode assumptions about who is observing, and how and why they are, not only individually, but also as members of naturalist communities. Adding to ethnographic analyses of wildlife identification, historians have analysed the development of representational styles in response to changing conceptions of nature, such as with the introduction of birds in their natural setting in Audubon’s (1827–1838) famous Birds of America (Audubon, 1827–1838; Dunlap, 2011).
Law and Lynch briefly mention how some key identifying features have changed over time; as seen in, for example, the gradual disapproval of ‘shotgun ornithology’ that ruled out characteristics observable only with a bird in hand (Dunlap, 2011). But there are tangles of concerns woven into the articulation of defining features and observational practices in naturalist communities, including: ‘being out there’ (Ellis & Waterton, 2004), learning about nature (Ganzevoort et al., 2017), the skill and joy of instant recognition (Ellis, 2011; Lorimer, 2008), a sense of community, status, and authority (Liep, 2001), or care for nature (Hinchliffe, 2008) and data scaling for conservation purposes (Kasperowski & Hagen, 2022; E. Turnhout & Boonman-Berson, 2011), in a long tradition of natural history enthusiasm (e.g. Allen, 2001; Dunlap, 2011). By shifting the focus from observational efficacy in the field to concerns in the naturalist communities of practice (Wenger, 1998) that produce and use field guides, we can analyse how field observation tools and practices are rearranged over time, altering not only what can and should be seen, but even what wildlife we assume to be ‘out there’.
Our analysis can rely on a wealth of studies investigating observational practices published since Law and Lynch’s pioneering work, covering a wide range of domains, within and beyond biodiversity. 1 We begin with an explication of the key concepts that we borrow from this theoretical context, with pride of place to Grasseni’s notion of the grid (Grasseni, 2004). We end our conceptual framework with a description of our transdisciplinary approach that relied heavily on the emic understanding of one of us (Turnhout), a committed dragonfly enthusiast. The subsequent empirical body of the article investigates a succession of Dutch dragonfly field guides. We analyse their textual and graphical organization to reveal readjustments in the observational grid over time, and to relate shifts in this grid to recalibrations in the wider observational community of practice. In the conclusions, we describe the consequences of our perspective on biodiversity-observing communities for the understanding of biodiversity data and conservation, and for developing careful relations with these communities and their practices.
Observational grids in communities of practice
In her analysis of skilled vision among Italian Alpine cattle breeders, Grasseni (2004) investigates how breeders view, describe, recognize, and assess their cattle. Crucial in this vision is the ability to recognize significant traits, which implies identifying and articulating parts and shapes of cattle bodies and their particular characteristics. This includes udder morphology, rump angle, or hock and hoof traits in a refined conceptual scheme or grid, used to highlight distinctive features appreciated among these breeders. The grid helps to discern relevant body parts and discriminate the features that can be observed there, directing the eye of the interlocutor and enabling specialized communication among the breeders. While concepts and visual representations are essential grid elements, grids may have material supports, such as the quadrats used by ecologists to specify a sampling area, or material marks and labels to identify animals (Lynch, 1988).
Grasseni explains at length that this observational grid for cattle is not a self-evident tool, but a set of subtle concepts acquired over a long time, often beginning early in life. Discussing features cherished in the breeding community forms an important marker of membership. As trained members of the community, ‘one never simply looks’ (Grasseni, 2004, p. 47), but perceives particular features as part of a shared practice, valuing characteristics for further breeding, consequential for the cattle stock. This grid is therefore part of a community of practice (Wenger, 1998), beyond which its terms have little relevance and meaning. 2
Like cattle breeders, naturalists use conceptual grids to describe perceivable morphologies and behaviour of wildlife. For dragonflies, this means learning to name and recognize anatomical parts (distinguishing your mesepimeron from your metepimeron, see Figure 1), but also overall appearance, knowing what it means for body parts to have particular characteristics (such as ‘wide’, or ‘forked’), and being able to recognize them in the field even when the light is not perfect. Biodiversity-observing communities similarly connect skilful vision and respected community membership, as ‘vision itself emerges through associated rituals of participation and a sharing of socially aesthetic sensibilities’ (Ellis, 2011, p. 780).
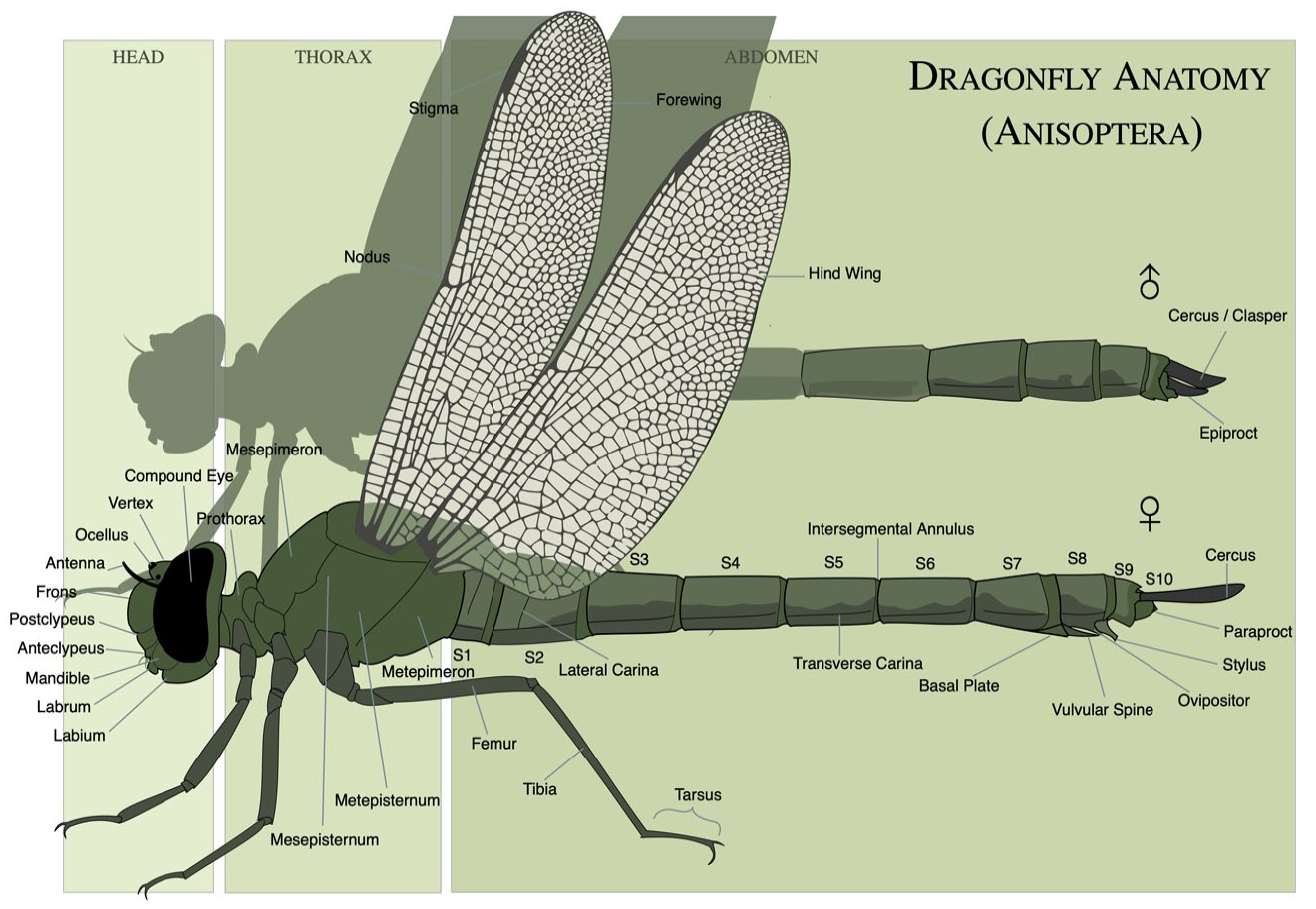
The anatomical dragonfly grid indicating body parts (but not their varying features, such as possible shapes or colours), using Comstock’s image of Boyeria irene (Wikipedia, M.A. Broussard, CC-BY-SA 4.0).
As observers become more skilled, applying the grid to recognition becomes instantaneous in virtuoso and joyful recognition (Ellis, 2011), although the grid remains available for explicit reference as a recourse (contra Ellis, cf. Macdonald, 2002, pp. 69–73). Grasseni describes how farm children play ‘recognize the cow’, much as naturalists play games of speedy identification—playful or in earnest competition, as with keeping spotted species lists in particular geographical or time frames (Obmascik, 2004). As a playful activity, wildlife observation can therefore be a game, not just in the sense of a language game with rules that have to be practiced to be understood (Wittgenstein, 1953), but also of an activity that is pursued with a game-like logic: calling it first, scoring on a list, or completing all the monitoring quadrants (known as ‘square bashing’ or ‘block busting’, when all the squares on a monitoring map are checked, even if no significant observations are expected, see S. Turnhout, 2020, p. 124).
Knowledge and application of the grid crucially revolves around comparison, as Law and Lynch (1988) already observe. Wildlife in the field has to be compared to an idealized representation to achieve identification, but this may involve comparisons with assumed previous observations. Eyes may be ‘further apart’ than in another species, wings ‘wider’, tibia ‘longer’. These descriptions imply prior knowledge and may even lead to backward correction: based on new knowledge, perhaps it was a different species that was observed before, after all. Such backward triangulation may even occur through improved taxonomies or through changing observational practices (E. Turnhout et al., 2016).
Although the knowledge of grids and how to use them is highly contextual to particular settings and communities of practice, these practices have long connections, extending far beyond an Alpine valley or a group of local odonatologists. Grasseni describes how the grid characterising Alpine cattle extends to cattle breeder organizations, cattle inspectors and markets, institutionalizing and quantifying standards that connect breeders world-wide, and that stretch back into the history of breeding practices (Grasseni, 2007a). Although breeding is geographically specific, the related practices of this community are dispersed. Learning as well as institutionalization has created hierarchies and authorities that can even overrule local assessments (Holloway & Morris, 2015). In the case of odonatology, this involves nationally and internationally organized communities in taxonomy or biodiversity recording, and conservation policies.
The skilled vision that registers dragonflies in the field is a distributed cognition (Hutchins, 1995), in the sense that it makes use of knowledge agglomerated in embodied observers, their observational grids, supported in the field by fellow odonatologists, and also tools such as modified binoculars, cameras, or guide books. However, it is also supported by the more extensively distributed cognition of reference collections, databases, and the scientific literature. In turn, the meticulously organized observational practices of committed biodiversity observers feed into these extended networks, correcting taxonomy, reappraising extinction rates, species distribution, or conservation policies—although not always successfully so (Ellis & Waterton, 2004, 2005; E. Turnhout & Boonman-Berson, 2011).
In the community of dragonfly enthusiasts, the observational grid is maintained, adjusted, and taught, but as the field observations are passed on and collected into databases, further grid layers are in operation. Here too, Grasseni’s grid notion may help clarify how the community of practice highlights particular features, here not only of the dragonfly, but also of its observation. Collected observations are in turn registered, evaluated on the basis of their evidence and the reliability of their observers, and compared to other observations (Kasperowski & Hagen, 2022). This datafication grid, which highlights and organizes key features of an observation, is not the one that supports skilled vision in the field. Rather, it is a ‘meta-grid’ that in turn allows skilled review of observations, articulated in terms of the features relevant for record-keeping, rather than field determination (S. Turnhout, 2020). We therefore suggest extending the grid notion from field observation to the post-hoc processing and assessment of observations, where similarly specific features are highlighted and named.
To form a functioning biodiversity observation network, these layered grids and their concerns need to be mutually adjusted: What counts as a particular dragonfly and how to skilfully observe it has to correspond to what counts as a valid observation in their databased records. At the same time, the grids need to be adjusted to the changing concerns of these communities. We describe such adjustments as reflected in observational grids, as presented in the text, figures, and format of dragonfly field guides. We chose field guides as an entry point into these readjustments since these guides are an important tool in the practice and provide a codified account of the grid that is periodically updated in consecutive editions.
Our analysis was written in a collaboration between an STS researcher (Halffman) and a dragonfly enthusiast deeply involved in the Dutch nature conservation community (Turnhout). This provided benefits and disadvantages. Among the benefits was exceptional access to sources and key informants, as well as intimate knowledge from Turnhout’s own experience, including what it means to stand thigh-high in a swamp while trying to name a flitting damselfly, or to realize the ramifications of retrospective doubt about an observation. This emic perspective, collecting information from within the practice’s self-understanding, alerted us to connections in these communities’ practices, such as the recreational logic of an under-twenty-five-only dragonfly youth camp. In conversations between the authors, the materials and experiences were jointly theorized from a more outside, STS perspective, in turn raising further empirical questions. In that sense, the STS concepts provided some etic analytic distancing.
However, the emic empirical approach can also be seen as a bias, shaped by this community’s self-understanding, potentially overly positive or uncritical. A more ethnographic ‘fly on the wall’ perspective might be more inclined to critically question routinized practices or implicit assumptions. Nevertheless, Turnhout’s perspective could at times also be surprisingly demystifying, shortcutting some of the flattering portrayal that dragonfly enthusiast might display to outsiders. For example, our analysis is at odds with the conservation community’s predominant self-understanding in terms of representational realism, which understands the contents of biodiversity records as a reflection of biodiversity ‘out there’.
Dutch dragonfly enthusiasts and their field guides
The community of Dutch dragonfly enthusiasts consists of hundreds of naturalists, ranging from the committed observers who regularly perform systematic counts, to a long tail of casual observers. While there is no exact boundary between who is ‘in’ and who is ‘out’, there is more clarity about who are the champions of its skilled vision, the respected authorities, a few dozen committed buffs. The core set of this community (Collins, 1981; Kasperowski & Hagen, 2022) is tight-knit and personal.
The community proudly traces back its lineage to the 17th century, when Jan van Swammerdam (1637–1680) mentioned some dragonflies in his Bible of Nature, describing their remarkable transformations and how to conduct field observations (Swammerdam et al., 1738, pp. 220–228). After an initial species inventory by the Dutch naturalist J. A. Herklots in the 19th century, based on Belgian and French taxonomical sources (Herklots, 1853), the first Dutch dragonfly taxonomical key was published by the educator and locally celebrated conservationist Eli Heijmans in 1918. In an article for a natural history journal, he classified species almost exclusively by their wing patterns (Heimans, 1918). 3 Apart from a sense of identity and pride that relates Dutch odonatologists to key historic figures among naturalists, this illustrates how taxonomic identification is a deeply rooted concern.
In the second half of the 20th century, dragonfly observation became an organized community centred on youth activities that combined nature education and recreational values in the Dutch Youth League for the Study of Nature (Nederlandse Jeugbond voor Natuurstudie) (Coesèl, 1997). This community relied on a core of committed volunteers and semi-professionals for instruction and support, fostering dragonfly interest beyond a handful of specialist odonatologists. This community was the driving force behind a network that today includes recording companies that curate observation data and contribute to biodiversity policy, and in return care for and educate voluntary observers (Lawrence & Turnhout, 2010). In fact, it was the initiative of dragonfly enthusiasts to develop a national dragonfly survey that made dragonflies a relevant organism for Dutch biodiversity policy. Today, the dragonfly enthusiasts operate in complex and occasionally tense cooperation with academic researchers, data companies, companies using data, government and government agencies such as Statistics Netherlands, and conservation organizations (detail in S. Turnhout, 2020, pp. 125–192). Growing from a few dozen original enthusiasts, dragonflies have even caught the attention of a nature-minded general audience.
Senior dragonfly enthusiasts developed a long series of field guides, initially to support Youth League field work. Of these guides, we will analyse four from between 1964 and 1997, each characteristic of what a prominent community member called attention ‘peak periods of Dutch odonatology’ 4 (Wasscher, 1995), as these guides reflect the evolving observational grid. We start with the first peak period and the 1964 issue of guides written by Jan Beukema in 1956, 1957, 1959, 1964, and 1968 (Beukema, 1964). The second peak period featured guides in 1974, 1975, and 1985, edited by Gerard Dutmer and Frans Duijm, from which we analyse the 1985 issue (Duijm & Dutmer, 1985). In the 1990s, the community started to discuss whether it was ethical to catch and collect dragonflies for the purpose of scientific research and nature conservation. After two limited edition ‘test issues’, in 1993 De Groot, Reinboud, and Wasscher published the first ODON key ‘for identifying dragonflies without catching them’ (Groot et al., 1993). The guide aimed to change odonatology practice, presenting itself at the start of the National Dragonfly Survey that started in the same year. In 1997, at the end of the project, the first full-colour guide to all Dutch species was edited by Bos and Wasscher, the fourth guide in our series (Bos & Wasscher, 1997), updated regularly since.
We will demonstrate that a changing ethic, from catching to watching dragonflies, shifting concerns and participants in the community of practice, and new technological affordances have led to adjustments in the dragonfly grid and specifically how identifying features are redefined in grid terms. These changes are reflected throughout the presentation of the guides, the organization of the information, their pictorial representations, the rules and objectives of the observation game, as well as the recommended tools. In a brief ‘Aftermath’ section, we contrast these guides with more recent identification-support technologies, such as automated recognition apps.
Guide contents and presentation
When put next to each other, the first striking difference is that the guides have expanded over time: from 32 to 256 pages. They contain increasing information on taxonomy, specific features, and morphology of adults and larvae, but also zoom out to cover ecology, habitats, and nature conservation. Later guides provide details on life cycle, food, enemies and parasites, geographical distribution (including preferred habitats), handling and identifying specimens, breeding of larvae, and how to conduct inventories or perform behavioural research. As the body of knowledge has evolved and grown, references to other guides increase. Contextual information about fieldwork, ecology and biology has grown from only 12.5 percent (four out of 32 pages) in the 1964 issue to 17 percent in 1985, 30 percent in 1993, to almost half of the 256 pages in 1997. The guides reflect how odonatology extended its interests beyond taxonomy to ecology and embraced a wider knowledge community.
As the guides have developed, more attention has been paid to the presentation of the material (Figure 2): The quality of the paper, the binding, the layout, and the drawings all improve. The 1964 issue was clearly edited on a typewriter and stencilled, with all the drawings and images inserted in the six middle pages. Later guides were computer-edited, culminating in the 1997 guide, which is a full-colour glossy hardback, rather than a stapled bundle. Consecutive guides contain more drawings, pictures, maps, and icons, which also became more integrated in the design. Instead of the separate centre pages of the 1964 edition, in the 1985 and 1993 guides drawings and pictures are placed near the relevant text and in the 1997 guide they are fully integrated. This, of course, is a sign of the increased possibilities of desktop publishing, but also serves to facilitate instant recognition: The text explains what is to be seen conjoined with the image.
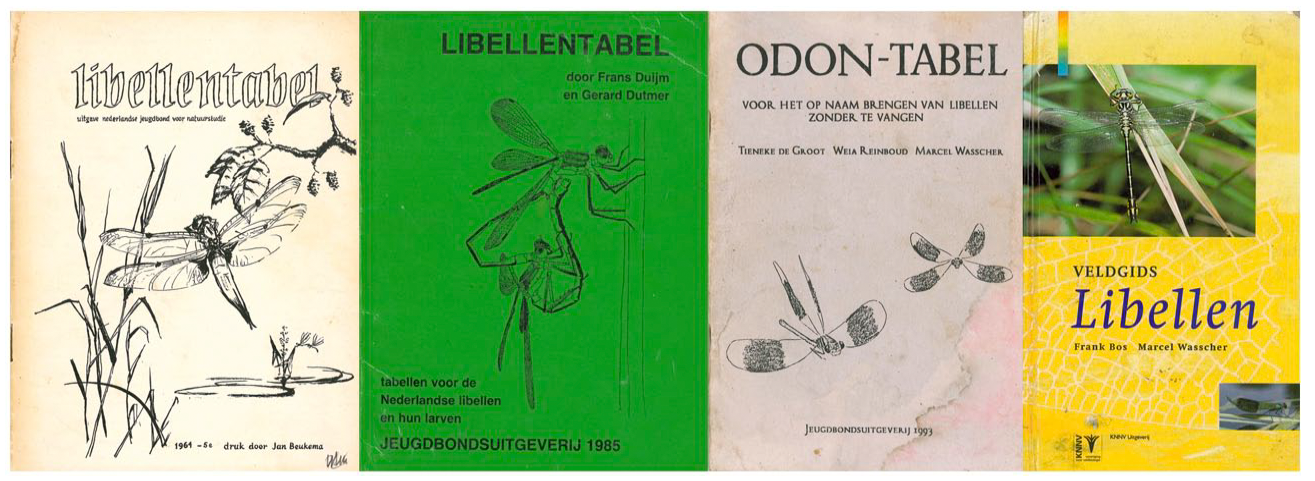
From left to right: dragonfly field guides of 1964, 1985, 1993 and 1997, evolving from black and white to full colour. Note the different representational styles that endorse specific observation strategies: detailed drawings based on caught dragonflies until 1985, shifting to impressionistic sketches that carry the remotely visible field marks (Odon-tabel, 1993), to colour photographs of species in their habitat (Veldgids Libellen, 1997).
As the field guides got ‘bigger and better’, the number of copies printed also increased. Only a few dozen of the Beukema editions were distributed, as opposed to a few hundred for the Dutmer & Duijm and ODON guides. Editions of the Bos and Wasscher guide sold 11,500 copies in just ten years. The 1997 guide is not only for members, but a product that can be bought in bookshops and hence features a barcode. The growing distribution reflects the growth of the odonatology community of practice, with a spectacular leap in the nineties; this was a growth in turn encouraged by the availability of a versatile guide. From a rather basic booklet, read by a handful of enthusiasts, the guides developed into information-rich and colour-illustrated books bought also by a more general audience.
Orientation, navigational structure, and grid codification
The first three guides use binary taxonomical keys, which rely on the proficient application of the observational grid. Navigating a binary key involves making a series of choices along a forking path, based on a description of distinguishing characteristics, in order to narrow down possible options until only one species is left. For dragonflies, observers usually are presented with about five to ten consecutive divisions before finalizing the identification process. With binary keys, it is easy to lose track along the way. Novices tend to end up with impossibly rare species. It is then difficult to determine where you went wrong and how far you have to backtrack to pick up the correct thread.
A more fundamental problem is that a binary key’s dichotomies are somewhat artificial and seldom universal. With the taxonomical keys of dragonflies, the very first division is the separation of dragonflies (Anisoptera) from damselflies (Zygoptera). As the 1964 guide states, with damselflies ‘the eyes are widely separated’, while with dragonflies they are ‘connected’. However, Clubtails (Gomphidae) are considered Anisoptera, but have separated eyes. A feature like this assumes prior knowledge, knowing what counts as ‘wide’ in separated eyes, weighing overall features against the exception. If you have not seen typical dragonflies (Anisoptera) and typical damselflies (Zygoptera), you would not know what to do with a specimen of the Clubtail family. Using binary keys therefore requires experience and familiarity with the options, the paths not chosen (see Lynch & Law, 1998).
The second feature in the 1964 binary key that is supposed to help you out, is both vaguely articulated and difficult to see. In damselflies, it says, ‘the front wings have the same shape as the hind wings’. With Anisoptera this is not the case, but this is far from self-evident: This is only observable for a sitting dragonfly, or if the observer catches a flying insect and takes it out of the net. With damselflies too, wing shape is a problematic feature. Usually damselflies fold their wings, but Calopterydea and Lestidae do not. How do you judge the hind wing size of folded wings? With folded wings, you might just see the bigger outer wings that cover the smaller ones, as is the case with some grasshoppers and wasps. The conditions for comparing front and hind wings may simply not be present.
These difficulties result from the fundamental difference of perspective between writing a binary key and using it. Binary keys are written from an overview of all regionally known species, laid out on a hierarchical grid that supports linear navigation: You go from A to B, median lines on the thorax are either diverging or non-diverging, and the difference can be compared in the physical presence of collected reference species. Taxonomical traits and identification features in the field are not hierarchical, nor systematic: The community’s distributed cognition is not equally distributed everywhere. When a guide states that an abdomen is dark, how dark is ‘dark’ (Goodwin, 1997)? In the field, comparisons are not made between species, but between living animals and the guide’s description, at best with the support of bystanders’ knowledge.
Even the basic distinction in Odonata between dragonflies and damselflies can be tricky without comparative reference, but the guides gradually attempt to compensate. The 1985 guide provides three pictures for two options, supported by more, and more specific, text right above the illustration (Figure 3a). To make sure no mistakes are made with the very first binary choice in the key, the second identification feature, wing shape and size, is illustrated and clarified with little arrows that show you where to look (Figure 3b). The challenge presented by species that resist binary classification are met with a more specific, detailed, and precise observational grid, supporting binary keys.

(a) First question in the 1985 guide distinguishing the suborders of Zygoptera and Anisoptera, ‘eyes further apart than the width of an eye’ or not, with Gomphus (the head on the right) being the intermediate, ‘displaying a bit of both’. (b) Distinguishing the wing shape feature in the 1987 field guide, supported by arrows to guide observers’ attention.
What stands out in the 1985 guide is that some species descriptions are printed in bold, while others are not. This is explained in the preface: Novices are advised to skip the choices printed in a small typeface, thereby avoiding the rare species. Apparently, the authors thought it better to rule out mistakes concerning rare species, than to rule out mistakes concerning more common species. With this guide, if you want to see rare species, you will have to muster the confidence to plunge into the guide’s small print. The 1993 guide provides a ‘quick and dirty’ key to common species for novices and a complete key including rare species for experienced odonatologists (Figure 4). With this division, the guide accommodates both the use as learning device for novice odonatologists and the use by experienced odonatologists, such as for making an overview of species occurrence.
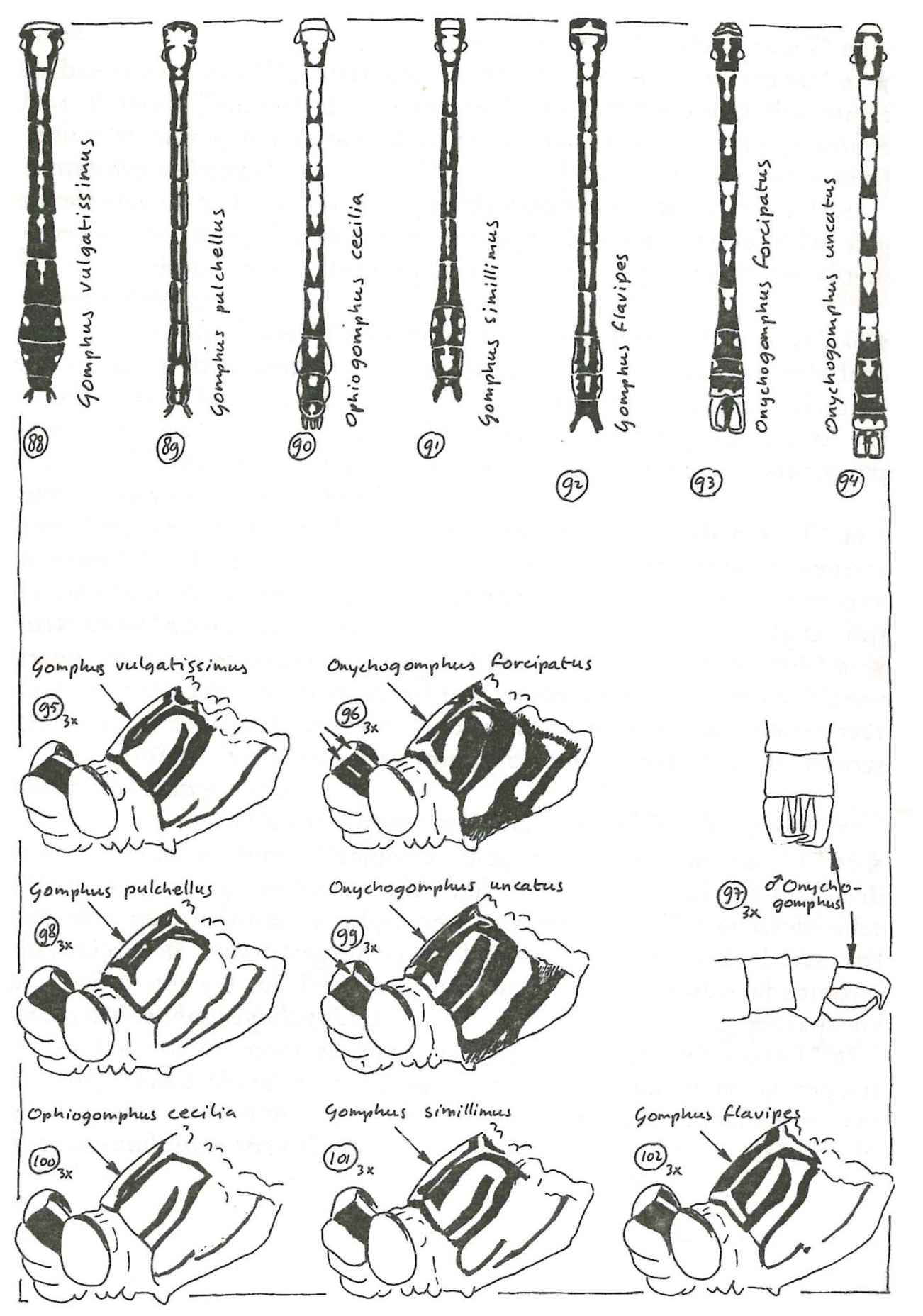
The ODON field guide (1993), aiming for visual identification without catching dragonflies. Related species are presented in close proximity. Again, note the arrows highlighting the ‘difference that makes the difference’.
In field conditions, it takes time to recognize all the necessary identification features. The observed animals often fly away before observers have finished the identification process. Looking at flying insects while leafing through the guide and answering questions and comparing the flying animal with the detailed drawings is very hard. In order to really guide the practice of odonatologists away from catching dragonflies, a guide was needed that supported instant gestalt recognition (cf. Ellis, 2011). The 1997 guide more radically abandons the taxonomical key for full-colour photographs of both males and females, accompanied by a range of graphical tools: a colour bar indicating rarity, drawings of the most important features of the species and its varieties, abbreviated information on size, seasonal habits, and habitats, and maps indicating occurrence and geographical distribution. The information is presented in an at-a-glance format and on the same page, to improve usability in the field and avoid the problems of using binary keys (Figure 5).

The 1997 field guide: photographs and drawings to distinguish species from closely related ones; ecology, occurrence and symbols for phenology.
Strategies to keep novices from identifying rare species have also disappeared from the guide. By 1997, the National Dragonfly Survey Project had not only attracted more odonatologists, it also showed that many rare species had returned and/or were much more abundant than previously expected. Even non-specialists could find interesting surprises in areas that were considered ‘boring’. After some spectacular (re)discoveries and ‘climate migrants’ discovered during the National Dragonfly Survey Project (1993–1997), anything could be expected and excluding rarities no longer seemed a viable identification strategy.
Changing pictorial representation
Leafing through the different guides, we see changes in pictorial representation related to improved technological affordances, as well as normative changes in the community of practice. The shift from black and white drawings to colour photos was also a matter of commerce: In 1997, an economically risky colour field guide for insects other than butterflies was only possible because of increased attention to dragonflies, such as generated by the National Dragonfly Survey Project. With a larger audience, a more expensive guide became possible, while in turn this community provided the necessary data and photos.
However, besides the more mundane material developments, the 1997 guide also reflects the growing objection in this community to catching dragonflies in order to identify them, as well as changing reasons for identifying dragonflies. If dragonfly identification depends on catching them, they are portrayed in taxonomic detail with features that require close inspection. The very first key for Dutch dragonflies of 1918 was based almost exclusively on wing shapes (Heimans, 1918). If dragonflies are to be left undisturbed, more impressionistic drawings must suffice, and new identification features and identification strategies appear: The grid is further articulated, while different grid elements become the key distinguishing features. Such differences in pictorial representation can already be noted when comparing the 1985 and 1993 covers (Figure 2): The 1985 cover shows wings in detail, implicitly endorsing catching dragonflies to count the number of cross-veins in the wings; the 1993 guide displays impressionistic wings that endorse visual identification from a dragonfly-friendly distance.
As for differences in identifying traits, the 1964 and 1985 issues present dragonfly drawings with detailed precision. Technical terms referring to body parts are explicitly specified with arrows and dotted lines, highlighting specific elements. Difficult damselflies of the Coenagrionidae family are reprinted from Lieftinck (1926). These detailed and precise features, that were added in the 1985 guide, do not always match its stated aim to establish a practice of identifying dragonflies without catching them. Usually, you cannot see if a mouth is ‘largely covered with lobes’ without a specimen in hand, but the guide attempts to replace such keys with clear, visual characteristics. This shifts the underlying logic of classification, as well as a redefinition of what counts as distinguishing characteristics. For example, species are clustered by colour, instead of taxonomic relations: There is a ‘blue abdomen sub-table’, a ‘red abdomen sub-table’, and so on. However, in many cases, identifying species still involves detailed identification features that are difficult to recognize (Figure 4), still sending observers down the wrong path.
In the 1993 issue, the impressionistic style of representing dragonflies can also be found in the text. In order to strengthen gestalt recognition, the guide includes the impression a body makes, rather than the number of wing cross-veins. The authors explicitly state that new drawings were made to accommodate this new practice, portraying dragonflies ‘as they are’ from ‘real live’ examples. However, the transition is not complete: The Coenagrionidae still are reprinted from Lieftinck’s 1926 drawings. Also anomalous is the continuation of the ruler in the back of the 1993 edition, since measuring wings or abdomen is impossible without catching dragonflies. Nevertheless, the changing pictorial representation is also part of the new coding scheme that highlights different features in the observational grid, reflecting the disapproval of catching dragonflies.
In the 1997 issue, colour photographs have become the new gold standard, while impressionistic drawings are only included in the chapter on behaviour. Detailed identification features are presented in stylized, schematic drawings, and in order to portray dragonflies ‘as they are’, colour photographs are included for all species (Figure 5). This presented some problems, especially in the first print, as for some rare species only one picture was available, while some images were cropped during editing or too small to recognize distinctive features. This was resolved in later editions, with better photography, and more material as dragonfly popularity grew and their ecological relevance was recognized.
Nevertheless, photographs remained but one identification tool. The 1997 preface mentions that it might be tempting to identify dragonflies using the included pictures of males and females for all species, but users are strongly advised to ‘let the descriptions in the texts prevail’ because photographs alone might easily lead to mistakes that are difficult to correct afterwards. (The ‘afterwards’ refers to the idea that, with visual identifications, recorded observations would be more difficult to correct than with collected specimens—databasing had become more prominent in the practice.) Apparently, the photographs, which are so prominent to this guide, should not be trusted entirely and the authors still hesitate a little about instant recognition and visual identifications (see Lynch & Law, 1998).
The dragonfly identification game
If the act of observing biodiversity can be seen as a literary language game (Law & Lynch, 1988; S. Turnhout, 2020), the goal and rules of the game are specified by the field guide. The original purpose of the game is to identify dragonflies correctly. All four guides explain this purpose in the preface, usually with explicit instructions. To quote the 1964 guide: Read very carefully and do not make any mistakes in the numbers. Base your identification on all given traits. If in the text a number is given between [round brackets], you can find the given trait in a drawing. It is necessary to compare the drawing with the dragonfly. Do not try to identify young animals: these have not acquired their colour yet. At the back of the guide a centimetre index can be found. If you get stuck, are in doubt, or are under the impression of having found something very special, the editor of this guide will be more than happy to check your identification. (p. 6)
The elements of ‘reading carefully’, taking ‘all given features into account’, using drawings, and the difficulty of young animals are mentioned in all guides in one way or another. The core objective of the observation game is identification and, in the 1964 guide, conditions for achieving this goal are articulated as a series of identification hurdles on the forking path of the binary key.
The instructions for ‘playing the game’ extend to how to behave in the field. The 1964 guide explains that it is of no use to run around after dragonflies and that it is better to sit still and observe them. The guide also states that, fortunately, most people do not kill dragonflies, but should the reader be a person who does so, a lot of space is devoted to instructions on how to do it properly. The 1985 guide repeats the remarks about sitting rather than running, but it also explains how to catch and hold dragonflies without damaging them. This is relevant because a freshly caught animal is under stress and might not be in the mood to cooperate with the identification process. Dragonflies are not poisonous, nor strong enough to do you any real harm, but they are not docile either.
In the 1985 edition, the section on killing, cutting, and collecting dragonflies has been shortened and the fourth paragraph presents a dilemma. The guide disapproves of killing dragonflies, but states that it can be useful for science and nature conservation to kill and collect rare species, although a series of clear pictures taken with a macro lens might also be sufficient. In the 1993 guide there is no more dilemma: Do not disturb or catch, let alone kill, dragonflies. The authors present three arguments in the preface. First, it might be easier to identify dragonflies by catching them, but ‘everyone who has ever caught dragonflies must admit that every now and then something goes wrong with the animals’. The authors state they wanted to make a ‘field guide for a clear conscience’ and refer to the code of conduct of bird photographers. 5 The second argument they present is an interesting quote from the Danish biologist C. Wesenber-Lund, who wrote in 1913(!): ‘After I had learned to identify the fauna of dragonflies by their flight image, the use of a net became superfluous’ (Groot et al., 1993, p. 6). Acquiring field knowledge is the virtuous path to harmless observation. The third argument is that bird field guides have put an end to the habit of killing species as proof of their sightings and the authors would like to achieve the same for dragonflies. Besides arguments against catching and disturbing dragonflies, the authors also instruct their users to be reluctant about entering natural areas and fragile habitats. You should ask yourself the question what is more relevant: ‘to see a certain animal and destroy its habitat or to leave both unspoiled?’
The guides suggest courses of action if identification is in doubt. In the 1964 quote above, the author acts as a verification institution, a mediator between the amateurs of the Youth League for the Study of Nature and the official, authoritative body of knowledge: He is ‘more than happy to check identifications’. The 1985 guide suggests sending specimens to the Dutch Knowledge Centre for Insects (EIS-NL) or the Natural History Museum if verification is required. In contrast, the 1993 guide does not mention sending in collected specimens at all, in line with the new ethic against catching dragonflies. The 1997 guide mentions the Butterfly Conservation Society and EIS-NL as institutions that collect data and are able to help out with identifications, including instructions on what equipment to use. Instead of sending in specimens, users are urged to send in photographs. The 1997 guide calls a photograph ‘the best way to preserve a dragonfly’, supporting the moral high ground with the argument that dead ones tend to lose their colours. This new identification practice is therefore also part of a broader shift from bringing dead animals to a museum as proof of rare sightings to providing photographic evidence, with a different appreciation of what counts as evidence (cf. Lynch, 2011).
Reasoning from the perspective of the users, we can recognize different intentions. Learning (Ganzevoort et al., 2017, pp. 2824, 2832) and being outdoors (Lawrence, 2006; Lawrence & Turnhout, 2010) are important drivers of dragonfly observation. In the guides these more personal motives can be recognized in a particular form of the ‘highlighting’ that Goodwin (1994) describes: In the game of observing biodiversity, highlighting works both ways, when users highlight or mark species with information on where they saw it for the first time, thereby extending the grid of observation by annotating the given info on geographical distribution of the species. Simultaneously, they make the guide into a personal list of experience and, for some, a proof of status in the community. How many crosses have you got in your guide? So, via the guide, the observation grid is extended with both personal and spatial elements, and even with elements of improved species identification, as information about key features can be extended and/or corrected through the invitation to participate. By sending in observations, the information on occurrence will be adjusted in consecutive prints, but also taxonomical information on traits and identification features will be adjusted to new insights in the community of practice.
The identification game expands to protection
The guides are closely informed by the developing community and its practices. Abma, the author of the first issue in our series, mentions that he improved the 1964 print based on feedback about how the guide was used in practice. The 1964, 1985, and 1993 guides were edited by the Dutch Youth League for the Study of Nature and were distributed through field trips and summer camps, with short lines between authors and users. In fact, dragonflies were deliberately chosen as a suitable year theme for the Youth League, because of their relative abundance and visual appeal (details in S. Turnhout, 2020, pp. 126–134).
During the 1990s, new concerns and new game objectives gained prominence in observation practice. The 1993 and 1997 guides were clearly improved with data collected in the National Dragonfly Survey Project and in monitoring practices. Gerard Dutmer, one of the 1985 authors, recalled that in the 1980s observers had to make do ‘without recent data on occurrence of species, barely accessible scientific literature, uncategorized and very few colour prints of dragonflies’ (cited in Wasscher, 2006, p. 24). From these communities, experience of the guides’ usability and knowledge about dragonflies were relayed to authors to improve new editions.
As the community of practice expanded, new considerations informed the guides’ contents. In the 1997 preface, a new target group was recognized in nature conservation professionals. Over time, developing legislation protecting endangered species created a small market in which protected species information represented some economical value. Subsequently, policy makers, ecological consultants, and site managers with formal responsibilities for dragonflies entered the community of practice. The guide addressed these new concerns with information on how surveys are conducted. Thus, dragonfly field guides not only reflected a changing animal ethic and a changing appreciation of evidence, but also changes of why and how observations were made: The game was no longer exclusively about identification, but also about surveying and conservation.
The utility of dragonfly observations for conservation purposes sharpened the dilemma of whether or not to kill rare species. In contrast to butterflies, dragonflies are not collected for aesthetic reasons and odonatologists do not keep a private reference collection, as is more common with larger species groups such as beetles and flies. However, protecting a rare species population may require definitive evidence of its occurrence to warrant protection measures. The 1985 guide comments on this dilemma: If the necessary equipment is not available, it might be useful to remember that locally rare species could be abundant someplace else. Implicitly, this condones killing one or two specimens if it benefits the protection of a rare species, reinforced by the suggestion that a single rare specimen (or, if numbers allow it: a male and a female) can be killed and sent in by mail. Paradoxically, this is also the section where nature conservation is first mentioned in the field guides and it immediately confronts the difficult choice between protecting all individuals of a rare species and sacrificing some for the protection of the species. With the 1993 and 1997 guides, this balance shifts, as watching and photographing become more explicitly preferred strategies over catching and killing dragonflies. This evolution is not linear, however; as late as 2008, a new guide by Dijkstra still included field and handheld characteristics, which the author claimed was for the sake of keeping the guide as accessible as possible by allowing for different identification strategies: catching, viewing, taxonomical, flipping pages, et cetera (Dijkstra interview).
From species protection, the game expanded to envelop wider environmental concerns. In the 1997 guide, dragonflies evolved into ‘indicators of their habitat’s environmental quality’. By now, they had become animals that should be ‘monitored’, rather than just identified or counted, with due attention to their natural environments. Readers were encouraged to discover the ‘fascinating world of water insects’ and to discover new habitats of rare or otherwise important species. Although some texts on how to hold dragonflies without damaging them reappeared, the sections on how to make a net, how to kill, collect, and cut up species were removed entirely. Identification dilemmas were not discussed: The authors simply state their preference for modified binoculars as observation instruments, and photographs as proof.
The shifting goals of observation practices have consequences for how knowledge is appreciated, as the game expands from species identification as a goal in itself to collecting data to conserve species and eventually to protect habitats. However, even at the beginning of our series, the practice was not exclusively taxonomical. For the 1964 guide, the stated goal is identification only: This guide merely sets out to be a means to gain knowledge of species, which, although often valued less, is the first precondition for studying a certain group of species. (p. 2)
Although identification is the objective, knowledge of species is ‘valued less’, although it is not clear what the implied ‘higher’ knowledge is. Presumably, taxonomical knowledge was seen as ‘lower’ than ecological, perhaps comparative or fundamental knowledge, with species knowledge as ‘a first, but necessary step one must take’. In the 1985 guide, the primary focus still lies on identification, but the increasing amount of contextual information also guides users in how to conduct a search. Thereby we see the grid of observation expanding: by describing and specifying preferred habitats, observers are instructed on where to look and what species to expect in certain areas. This function is enhanced by placing the information on habitats directly below the text that bears the identification features. Thus, species are connected more explicitly to habitats.
By 1997, the observation practice had evolved from a game of searching and identifying dragonflies into a game ultimately focused on gathering dragonfly occurrence data to inform their protection. This required not only rules on how to gather and integrate data (a new game of ‘databasing’), but also a new conceptual apparatus to describe and qualify observations. This datafication grid articulated what counted as an observation and its qualifying features of who saw what, where, when, and how (for details see S. Turnhout, 2020). The reorientation of the observation practice from taxonomy towards conservation also required a different kind of field guide.
The 1997 guide flags these changes immediately in one of its introductions, describing dragonflies as excellent environmental quality indicators, since they quickly respond to habitat changes caused by recreational activity, pollution, agriculture, water management, or climate change. The preface advocates the importance of monitoring dragonfly occurrence and dispersion patterns: ‘a purpose that suits this guide very well’. However, identifying and learning about dragonfly species and their habitat is not abandoned, since readers are encouraged to discover the ‘fascinating world of water insects’ and to identify new habitats of rare or otherwise important species, in turn a ‘marvellous contribution’ to dragonfly preservation. The preface not only updates the tone, but also provides a lot of new terms (indicators, to monitor changes, contribute to preservation). To support protection, readers are challenged to discover rare species instead of being instructed to ‘skip the rare ones’ to avoid mistakes.
The increased importance of dragonflies in nature conservation encouraged interest from conservation organisations, as well as a growing community of dragonfly enthusiasts. In the third introduction to the 1997 guide, the authors sought an explanation for the growing attention in the wonders of dragonflies themselves: That is no miracle. Dragonflies are the biggest insects in Europe. They have beautiful colours, can fly amazingly well and have a fascinating way of life. No wonder more and more people are starting to look at dragonflies, want to recognize the species and want to know more about their behaviour. (p. 8)
However, dragonflies probably have been big and colourful since the Carboniferous period and their ways of life and flying abilities have not changed very much between 1964 and 1997. The success of conservation organizations in advocating for their ecological importance was a more likely contributor to the growing naturalist attention. The authors acknowledge that ‘fortunately also governmental bodies have recognized the indicating function of dragonflies, and the worries about the on-going decline of dragonflies are transformed into action and policy measures’.
As dragonflies become indicators in environmental protection policies, field guides adapt to accommodate such concerns. The 1997 authors even state: ‘To meet this growing demand for identification and information about ecology of dragonflies, this guide was made’ (p. 8), identifying nature conservation policy as one of the main purposes for issuing a new edition. Furthermore, the authors hope that the guide will help increase the number of people who look at dragonflies and that the occurrence of dragonflies can be mapped more precisely, so that the necessity of conservation policy will present itself more clearly.
Observation tools
All four guides devote some attention to what instruments one should use. The 1964 guide discusses in some detail how to make a net, on how to make a papillote in which one can conserve collected species, and how to cut, dry, and preserve dead animals. These instructions can also be found in the 1985 guide, but a section on the use of a magnifying glass has been added. In the 1993 guide, no instruments other than binoculars are mentioned and these are discussed extensively. In the 1997 guide, besides a substantial section on binoculars, the authors include one on photography, cameras, and lenses.
In the early 1990s, parallel to the development of the ODON field guide based on visual traits, people started bringing small binoculars to the field, especially developed for enlarging objects close by. Binoculars came in handy when identifying small damselflies in the reeds. The quality and range of lenses in optical equipment improved and evolved considerably with the rise of digital photography. A 200 mm lens on a digital camera became a standard tool for the odonatologist. The lens could be used to magnify the necessary trait, while the actual photographs were usually discarded after the specimen had been identified.
The shift from the ‘obligatory’ magnifying glass in the 1985 guide towards binoculars in later editions is only logical in light of the shift from catching to watching. However, a pair of binoculars is not a one-to-one replacement of the magnifying glass: It is an instrument that fits another type of use and another way of looking at animals, that is, at flying insects instead of at hand-held ones. As Goodwin (1995) has shown, instruments have their effects on how observations are performed and what eventually is seen. The choice of instruments influences the observation strategy and, with that, it rearranges the observational grid: what species will be seen, how species are identified, and how they are made present when playing the game of observing and recording biodiversity.
Aiming well and making sure that the lens frame corresponds to the naked eye is a complication. A flock of damselflies in the reeds spotted with the naked eye might have taken off before the photograph was taken, or replaced by another species altogether. As photographic evidence is discussed among observers, this gap between the damselfly in the reeds and the one on camera offers leeway to settle potentially damaging disagreement and avoid the suggestion of incompetence: ‘Well, you must have another one in focus’. In the community this is known as the ‘second bird theory’ (Davidse, 2006).
Inversely, photographic evidence may allow species to be spotted that were overlooked by the naked eye. In 2016, Dutmer wanted to take a picture of Ceriagrion tenellum and noticed a Nehalennia speciosa hidden in the photograph only afterwards. The species had been absent from the Netherlands, or perhaps overlooked, since 1912 (Dutmer, 2016, p. 16). It is a classic ‘blow-up’ scenario: an interest in one damselfly leads to the discovery of another, smaller one in the back, that otherwise would have been overlooked. Though the rediscovery of a species that had been absent for more than a century came as a sensational surprise, the process of how it was rediscovered was less surprising in hindsight. Nehalennias are very small and rarely seen. When they are spotted, they are usually seen by people in the business of catching flies: entomologists who focus on smaller insects and therefore apply different observational strategies. Entomologists might blindly draw their nets through interesting vegetation and only afterwards look at what they happen to have caught. Odonatologists try to detect species of interest first and then to either catch them or take a picture for a closer look. The Dutch Nehalennia found in 1912 were caught by Demeyere, an entomologist who did a lot of research on flies and probably would have only seen it when he found it in his net. As what can be seen is often also mediated by tools such as nets, cameras, or binoculars, these tools co-define what is seen and counted as an observation.
Aftermath: Apps and computer-supported identification redefine the grid
Since the publication of the 1997 field guide, new guides and tools have been developed in the community of odonatologists. The continued co-evolution of observational practices with their tools, goals and uses is also reflected in some of the more recent field guides, now supplemented with apps and even artificial intelligence. In order to fence off the problem of ‘getting lost’ with the binary keys and the problem of oversight when one has to flip through 70 species, taxonomical matrix keys were developed that can be used on computers and telephones. In overview charts, divisions between species and identification features are presented in a non-linear structure. Users can select wing patterns, colours, thorax, abdomen, or colours in random order. When ‘lost’, as can happen with binary keys, users fine-tune some of the features and the matrix will rearrange the possible outcome accordingly. The innovation is quite radical: Users can base their choices on what they think are important identification features, while the designers still maintain control of the presented features. The ‘difference that makes the difference’ has become a set of potentially crucial features, some of which the observer may miss or even misinterpret.
A new generation of apps even provides automated identification, such as the Dutch app ObsIdentify (Greeff et al., 2022). By comparing photos of identified species, combined with machine learning, such apps learn from experience and provide an identification along with an estimate of its accuracy. With mobile species, such as birds and dragonflies, or complex and variable species, such as mushrooms, this accuracy is low. However, with species that sit still (plants, night-active butterflies) or are killed (bees), scores are very high. Usually, these apps focus on patterned features of species, such as venation patterns in plant leaves or wings of bees and butterflies—or even fossils: Wilf et al. (2016), for example, used computer vision to classify fossil leaf images on the basis of leaf shape and venation with high accuracy. This proved not only to be an efficient protocol for classifying images, but also discovered previously unidentified morphological landmarks potentially useful for species identification and for understanding important evolutionary and ecological innovations not previously documented. (Hedrick et al., 2020)
A new leaf on the tree of representation, after digital photography, even puts living animals on a flatbed scanner without killing them. This technique was first used in an African field guide with detailed pictures of living animals, setting a new standard for representing animals ‘as real as possible’ (Tarboton & Tarboton, 2019).
The artificial intelligence involved works better than the human eye with such complex patterns. Automated dragonfly identification may well return to the distinguishing features of the early 20th century (Heimans, 1918), long considered undesirable. The ‘difference that makes the difference’ has once again shifted, as the grid develops from professional to computer vision, subtly altering the logic of making species present in biodiversity knowledge. As the game develops, new objectives, such as integrating databases on a global scale, and new game logics are introduced. These affect grid articulation, such as a push for standardization of identifying features (Kissling et al., 2018).
An even more radical development, suggesting the possibility of skipping visual observation altogether, concerns attempts to track the presence of species by means of analysing ‘environmental DNA’ (Bohmann et al., 2014), genetic traces that indicate the presence of a species. This already works for fish and some amphibians, but what this will mean for the practice of studying dragonflies remains to be seen.
Communities of practice readjusting observational grids
By analysing a series of field guides, we have traced adjustments and readjustments in the observational grid for dragonflies in the Netherlands. The changes in the guides are intimately connected to changes in the guides’ intended uses, the composition of their community of practice, the shift in animal ethics from catching to watching, and the affordances of new observation technologies. The guides evolve from a stencilled booklet, supporting a game of ‘identify the dragonfly’ (catching it if need be), played by a small in-crowd of enthusiasts, to full-colour glossy books that play a role in promoting dragonfly conservation and even environmental protection among a wider audience, including environmental professionals.
Our main attention in the succession of these guides has been for the redefinition of what counts as salient features of dragonflies, articulated through the observational grid. How to identify and make dragonflies present in recording, is recalibrated in function of the distributed cognition network maintained by the porous community and practice of dragonfly enthusiasts, including their composition, goals, tools, ethics, and even game-like logic. Choices in the guides reflect how and why the observation game is or should be played. Every guide embeds not only a pictorial theory of representation, but also a strategy of observation, with re-specifications of the observational grid. Articulated in terms of this grid, the ‘difference that makes the difference’ is in constant development, to match salient features with how the game is played. What counts as distinguishing features changes with how and why the community operates in the field, the observational methods and registration tools it endorses, the composition of the community of practice, and ultimately also how the practice extends its reach to connect with nature conservation and environmental protection regimes. Observers, species, time, place, instruments; all components evolve in the community of practice and the grid is recalibrated accordingly. As the guides change, we recognize the evolving strategy of observation, eventually affecting what will be seen, represented, and constituted.
From this angle, observing wildlife is more than what Law and Lynch refer to as ‘a hermeneutic reading of the world’(Law & Lynch, 1988, p. 273). It is more than guided vision alone, because it also concerns a community of practice and its technologically mediated, distributed cognition. This community constantly develops new conceptual schemata, rewrites the field guides, finds new ways to play the observation game, with new purposes and new players. Taking a guidebook to the field to investigate how it deploys the ‘difference that makes the difference’ and directs observers is but one moment in a complex weave of developing practices that redefine what is seen in function of more extended networks and concerns.
We believe that documenting these complex practices can inform a more careful appreciation for not just wildlife, but especially also their human enthusiasts. The blunt treatment of naturalists as ‘data drones’ (Ellis & Waterton, 2004, p. 98) or ‘ecological research tool’ (Dickinson et al., 2010), harvesting ‘raw data’, not only disregards what makes these communities tick, but also their complex recalibrations of wildlife knowledge, including the methods and conceptual devices by which this knowledge is gathered and curated.
We hope the analysis of other observational communities and their practices might be inspired by our approach and revision of key concepts. To summarize this, first, we have stretched the analysis of observational acts in time and place, beyond the usual ethnographic focus on accomplishing identification in situ. Analysing changes in how ‘the difference that makes the difference’ is articulated over decades leads to an investigation of what stabilizes or destabilizes seemingly essential characteristics. Second, beyond the place of observation, this recalibrates salient differences to the concerns, tools, and ethics of the observational community. This allowed us to describe these changes as mutual readjustments of grids and the networks in which they are used. Third, in this community, we have highlighted the game-like logic of observation practices, to analyse not just the act of observation (signalled in Grasseni, 2004; Lynch & Law, 1998), but also how the concerns of the network shape what the game is about and what are considered meaningful ways to play it, or to elaborate the game by adding new ‘levels’. Fourth, stretching the notion of grid beyond the observational act, the concept of layered grid allowed us to study how observations are articulated and appreciated throughout the network, in our case into conservation through a datafication grid (further elaborated in S. Turnhout, 2020, pp. 125–154). Last, our analysis would have been impossible without the transdisciplinary cooperation between an academic and a practitioner, for mundane purposes of access, but much more importantly for highlighting key processes in mutual conversations.
On a final reflexive note, to describe this complexity, we too have deployed a grid to ‘observe the dragonfly observers’, in our case using social science conceptual tools. Without wanting to push the metaphor too far, these tools have allowed us to highlight: the community of practice as it extended its reach, this community’s optical and literary tools including binoculars and guidebooks, the language games these practitioners play as objectives shifted from the joy of identification to embrace conservation and monitoring, the developing ethic from catching to watching, and how all these changes readjusted the grid of observation, articulating a new ‘difference that makes the difference’ to fit the changing practice. In this sense, our own grid also operates in a language game, involving the joy of recognising patterns from similar knowledge practices, to be shared and no doubt further readjusted by our own developing knowledge community.
Footnotes
Acknowledgements
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
