Abstract
Chills are emotional peaks especially in response toward acoustic stimuli. In the present study, we examined facial expressions associated with pleasant and unpleasant chill experiences during music and harsh sounds by measuring electromyographic activity from facial corrugator and zygomatic muscles. A rubber bulb could be pressed by the participants to report chill intensities. During harsh sounds, increased activation of both corrugator and zygomatic muscle was observed. Zygomatic muscle activity was even more pronounced when a chill experience was reported during such sounds. In contrast, pleasant chill experiences during music were associated with slightly increased corrugator activity compared with absent chills. Our data suggest that harsh sounds produce a painful facial expression that is even intensified when a chill experience is reported. Increased corrugator activity during chills toward music might refer to states of being moved. The results are discussed in the light of a proposed role of the chill in regulating social behavior. Our results suggest that recording facial muscle activity can be a valuable method for the examination of pleasant and unpleasant peak emotions induced by acoustic stimuli.
Chills are described as transient physiological reactions associated with bodily manifestations like shivers down the spine, goose bumps, and lump in the throat (Goldstein, 1980; Kunkel et al., 2008; Sloboda, 1991) reflecting emotional peak experiences (Grewe et al., 2010). While the majority of work focused on such reactions toward music stimuli (Benedek & Kaernbach, 2011; Blood & Zatorre, 2001; Harrison & Loui, 2014; Laeng et al., 2016), chills can also occur toward harsh sounds, gustatory stimuli, pictures, film scenes, and physical touch (Blake, 1986; Boyd, 1959; Grewe et al., 2010; Halpern et al., 1986; Wassiliwizky, Jacobsen, et al., 2017). Therefore, chills were suggested to constitute markers of increased autonomic arousal independent of valence (Grewe et al., 2010).
Interestingly, experiencing chills was also shown to affect an individual’s social behavior. In fact, listening to chill-inducing music resulted in increased altruistic behavior in a dictator game (Fukui & Toyoshima, 2014), higher willingness to donate money (Strick et al., 2015), and higher prosocial motivation (Ji et al., 2019). Likewise, Haar et al. (2020) found increased ratings of empathy in an experimental condition where chill experiences toward film stimuli were enhanced by artificially induced shivers down the spine. Hence, chills are not only characterized by unmistakable subjective experiences and autonomic changes which can be reliably assessed, for instance, by measuring skin conductance (Grewe et al., 2009), there is also evidence that these experiences have direct implications for regulating behavior in relation to the social surrounding (Maruskin et al., 2012).
In turn, facial expressions are one of the most prominent nonverbal ways to communicate emotional states to the social environment (Fridlund, 1991; Schmidt & Cohn, 2001). Among facial muscles, especially the zygomaticus major muscle and the corrugator supercilii muscle have been examined in the context of emotion processing (Dimberg, 1990; Overduin et al., 1997; Ree et al., 2020; Sato et al., 2008), including emotional responses elicited by short excerpts of film music (Fuentes-Sánchez et al., 2021). Actually, the zygomatic muscle of the cheek region raises the corners of the mouth which can result in a visible smile (Rubin, 1974). Increased activity of the zygomatic muscle was found during viewing of pleasant pictures (Cacioppo et al., 1986; Lang et al., 1993) and during listening to pleasant music (Fuentes-Sánchez et al., 2021; Witvliet & Vrana, 2007), but also during the imagery of pleasant scenes (Witvliet & Vrana, 1995). In contrast, the corrugator muscle, situated at the medial end of the eyebrow, produces facial frowning by pulling the eyebrow downward (Isse & Elahi, 2001). Activity of the corrugator muscle linearly rose when individuals viewed increasingly unpleasant pictures (Lang et al., 1993), while also an increase in activity was consistently found during imagery of personally unpleasant episodes (McTeague et al., 2012) and toward both unpleasant sounds (Bradley & Lang, 2000) and music (Fuentes-Sánchez et al., 2021; Witvliet & Vrana, 2007).
Only a handful of studies have examined facial expressions during chills. Weth et al. (2015) filmed facial expressions of participants listening to music stimuli and applied an emotion detection software. They found that self-selected sad music caused not only more chills and tears compared with unfamiliar sad and happy music but also evoked stronger facial expressions of sadness. In a further video-based approach with experimenter-selected audiovisual stimuli, more smiles were registered during the artificial induction of shivers down the spine compared with a control condition while also more chills were reported (Haar et al., 2020). Wassiliwizky, Koelsch, et al. (2017) played recordings of poems and especially found stronger corrugator muscle activity during chills and piloerection compared with passages without chills and piloerection. Similarly, participants showed the strongest increase in corrugator and zygomatic muscle activity while watching self-selected film scenes when they felt moved to tears and simultaneously showed piloerection (Wassiliwizky, Jacobsen, et al., 2017). More recently, increased corrugator but not zygomatic muscle activity was found in participants in response to an experimenter-selected film scene rated as especially arousing and moving (Kimura et al., 2019). In contrast to that, Zickfeld et al. (2020) found an increase in zygomatic muscle activity toward experimenter-selected videos of moving scenes. However, Grewe et al. (2007a) rarely found changes in facial muscle activity during preselected chill-evoking music stimuli of various types.
Although chill experiences have been examined in different contexts using a variety of measures, work on facial expressions during chills is sparse so far while results were inconsistent. Furthermore, to the authors’ knowledge no work on facial expressions during unpleasant chills has been realized to date. Therefore, the present study was conducted to examine facial expressions during pleasant and unpleasant chill responses evoked by acoustic stimuli. As subtle facial expressions are difficult to detect using a video-based approach (Tassinary & Cacioppo, 1992), we obtained electromyographic recordings of the corrugator and zygomatic muscles. A rubber bulb was applied which could be pressed by the participants to report the occurrence and intensity of the experienced chill response.
For inducing pleasant chills we chose experimenter-selected excerpts from classical music pieces because it has been shown that such music is a reliable chill trigger (Grewe et al., 2010). Also, the applied music excerpts have been successfully used in previous work (Guhn et al., 2007; Klepzig et al., 2020). Hence, we ruled out that chill responses might be associated with individual memories which could interfere with the examined response and complicate interpretations as it can be the case with music stimuli selected by participants (Guhn et al., 2007; Schulkind et al., 1999). Our analyses focused on certain passages (durations of 7.2–11.5 s) which have been identified as especially prone to evoke chills (Guhn et al., 2007). We decided to apply excerpts of longer durations (90 s) to allow the participants to develop expectations about how the music evolves over time and, hence, to increase the possibility of experiencing chills (Grewe et al., 2007b). For inducing unpleasant chills we used recordings of harsh scraping sounds (durations of 7.5–9.9 s) which have been shown to be especially apt to evoke such experiences in past work (Ely, 1975; Grewe et al., 2010; Halpern et al., 1986; Klepzig et al., 2020). To achieve durations similar to the music stimuli, the harsh sounds were inserted in a sound environment of the same acoustic domain (simple drum sound with a length of 90 s). As music has been shown to be effective at altering the perception of aversive stimulation (Shpaner et al., 2019; Silvestrini et al., 2011), we combined the latter two conditions in an exploratory approach where harsh sounds were also inserted in further excerpts of classical music, to examine whether playing music affects the susceptibility to unpleasant chill experiences.
Considering the findings by Grewe et al. (2010), we expected that more chill responses would be elicited by harsh sounds relative to those evoked by music stimuli. Also, as it has been shown that unpleasant acoustic stimuli were more effective in evoking highly arousing states than pleasant acoustic stimuli (Bradley & Lang, 2000; Grewe et al., 2010), chills during harsh sounds should be experienced as more intense than chills during music. As chills during music are seen as a sign of enjoyment (Blood & Zatorre, 2001), it would be expectable that zygomatic muscle activity during chills is greater than zygomatic muscle activity during absence of chills. Likewise, we predicted that the reduction in corrugator muscle activity was more pronounced in association with chills compared with when chills were absent. However, in the light of findings by Wassiliwizky Jacobsen et al. (2017), an increase in corrugator activity during pleasant chills could be also possible, which would underline that such states indicate mixed emotions. As chills during harsh sounds were described as highly unpleasant states (Ramachandran, 1996) we hypothesized a stronger increase in both corrugator and zygomatic muscle activity during harsh sounds when chills were reported than when chills were absent. A synchronized increase in activity in these facial muscles have been typically found during pronounced unpleasantness evoked by acoustic (Bradley & Lang, 2000) and visual stimuli (Lang et al., 1993).
As it was shown that playing music reduces the perceived unpleasantness of harsh sounds (Standley, 1991), less chills and lower chill intensities during harsh sounds inserted in music than during harsh sounds inserted in drums would be expectable.
Method
Participants
Twenty-six students were tested. They did not report any mental or somatic disorders. All participants were right-handed (assessed via self-reports) and provided written informed consent for the protocol approved by the Ethics Committee of the German Society for Psychology and received course credits. The study was realized in accordance with the Declaration of Helsinki. One participant constantly showed excessive facial activity and was therefore not considered for analyses. Due to technical problems, data of three participants were not available. Finally, data of 22 participants (19 women; age: M = 24.23, SD = 2.94) were analyzed. As research on chills and facial electromyography (EMG) is scarce, no profound power analysis could be conducted. However, our sample size is similar to those from previous studies (Kimura et al., 2019; Wassiliwizky, Koelsch, et al., 2017; Weth et al., 2015).
Auditory stimuli
Six excerpts of classical music pieces (90 s; see Table 1) were used to evoke pleasant chills (music condition). Furthermore, we applied six recordings of harsh scraping sounds (7.5–9.9 s; see Table 2). Each harsh sound was inserted in drum beats (90 s) created with d-lusion DrumStation software (d-lusion interactive media, Leonberg, Germany) at a random position (harsh sounds and drum beats). In a second condition, each harsh sound was inserted in a further classical music excerpt (90 s) in such a way that the sound’s onsets coincided with climaxes in the music such as dynamic or tonal changes (harsh sounds and music). Thus, 18 auditory stimuli were applied with 6 stimuli for each condition. All stimuli were equipped with fade-in/out effects. An amplitude normalization was realized to achieve a uniform sound level. Editing was realized with Audacity (https://www.audacityteam.org) and Cool Edit 2000 (Syntrillium Software Corporation, Scottsdale, AZ, USA). Stimuli were stored and played in WAV format.
Music Condition.

Harsh Sound Stimuli.

Procedure
Participants took seat in a comfortable chair in a dimly lit, sound-attenuated room shielded against electrical noise. Prior to the attachment of sensors for recording facial muscle activity skin sites were cleaned with wipes and purified water to reduce inter-electrode impedance. Cables of sensors were fixed to the participant’s head and neck using tapes to reduce cable motion. A rubber bulb was introduced to the participants with the instruction to indicate each chill experience by pressing (dominant hand), while the amount of pressure should be used to express the intensity of the chill experience. To obtain reference values, participants were instructed to press it 10 times with medium strength. Afterward, a practice trial was realized using an excerpt of the track “The Black Pearl” from the film Pirates of the Caribbean: The Curse of the Black Pearl to allow the participants to get used to the experimental setting. The experiment started with the presentation of the 18 auditory stimuli. Participants were randomly assigned to one of two pseudo-randomized presentation orders with no more than two stimuli of the same condition played in a row. After each stimulus a resting period followed (30 s of bird chirps). Information about the current stimulus (composer or nature of the harsh sound) was displayed on a screen at a distance of 2 m (e.g., “Mozart” or the German translation for “cutting styrofoam”) and also presented by a male voice prior to each stimulus onset to increase the participants’ susceptibility to chills (Ely, 1975). Stimulus presentation was realized using Presentation software (Neurobehavioral Systems, Berkeley, CA, USA) and closed headphones (AKG K60, Los Angeles, CA, USA) at a standardized volume. After the experiment participants evaluated the pleasantness of chills during music and harsh sounds.
Apparatus and data acquisition
The rubber bulb was connected to a Varioport Portable Recorder (Becker Meditec, Karlsruhe, Germany) which converted air pressure changes into a digital signal. Recording was realized with PhysioMeter (Ritter, 2004). The pleasantness of chill sensations was assessed using a paper-and-pencil version of the 9-point valence scale of the Self-Assessment Manikin (Bradley & Lang, 1994) varying from 1 (highly pleasant) to 9 (highly unpleasant). One participant showed problems with the rating format and was therefore not considered.
EMG was recorded over the left corrugator supercilii muscle and the left zygomatic major muscle following the recommendations by Fridlund and Cacioppo (1986). Two Ag/AgCl miniature surface electrodes (4-mm diameter; SensorMedics, Yorba Linda, CA, USA) were filled with an electrolyte paste (Marquette Hellige, Freiburg, Germany). A Coulborn S75-01 bioamplifier with a 30 Hz high-pass filter and a 400 Hz Kemo KEM-VBF8-03 low-pass filter were used to amplify the raw EMG. The signal was then rectified and integrated via a Coulborn S76-01 contour-following integrator. The time constant was set at 500 ms. Sampling rate of the integrated signal was 10 Hz. Signal quality was visually inspected throughout the entire recording session.
Data reduction and response definition
The digitized signal of the rubber bulb was processed with MATLAB-based scripts (The MathWorks, Natick, MA, USA). A detection routine identified peaks in the signal to assess the occurrence of chills and their intensities. As the baseline pressure within the rubber bulb slightly changed over time, intensity values were corrected for the baseline pressure of the corresponding section. Separate peaks within 1 s were treated as representing a single chill experience (Mori & Iwanaga, 2014). The largest peak value was then scored as the corresponding chill intensity. Furthermore, chill intensities were divided by the participant’s largest peak value recorded at the beginning of the experiment to control for inter-individual differences (Dodds et al., 2014). Analyses of frequency and intensity of pleasant chills (music condition) focused on preselected passages in the music stimuli (Guhn, 2003; Guhn et al., 2007) (see Table 1). Also, frequency and intensities of unpleasant chills during the harsh sounds were scored. Absolute numbers of chills were converted to chills per minute (numbers of chills / duration of the corresponding passage) (Rickard, 2004). Finally, means of chills per minute and chill intensities were computed, respectively, for each stimulus condition. Confirming Guhn et al. (2007) during the excerpts by Albinoni and Barber almost no chills were reported. Hence, these two music excerpts were not considered for analyses.
Digital values of facial muscle activity were converted to microvolts and reduced offline to 1/4-s bins. The time course of the facial EMG during the preselected music passages and harsh sounds was assessed by computing 1/2-s mean values (14 values) and subtracting the mean activity during the 3 s preceding the onset of the music passage or harsh sound (baseline). In addition, the mean change in EMG activity averaged across the 7 s during the preselected music passages and harsh sounds were computed (again, corrected for baseline). Mean values of EMG data (1/2 s means, 7 s means) were computed, respectively, for each condition regardless of reported chills. Furthermore, we calculated mean EMG activity for trials in which chills were reported (chill) and for trials in which no chills were reported (control) for each condition. When chills were reported just shortly after the offset of the music passage or harsh sound the corresponding EMG mean values were treated to be associated with a chill. Facial EMG was not affected by pressing the rubber bulb alone supporting previous data (Cacioppo et al., 1984; Smith & O’Connor, 2003).
Data analysis
Repeated measures analyses of variance (rmANOVAs) with Condition (music, harsh sounds and drum beats, harsh sounds and music) as within-subjects variable were conducted to compare frequency and intensities of chills. For evaluating chill intensities at least one chill report during each of these conditions was required (nine participants). Similarly, rmANOVA with Condition as within-subjects variable was conducted to examine ratings of pleasantness while data of those participants were considered who reported at least one chill in each condition (eight participants).
First, the time course of facial EMG activity irrespective of chills was examined using rmANOVAs with Condition and Time (14 half-second mean values) as within-subjects variables.
Second, facial EMG activity was examined with regard to chills. However, chill-associated and control means were not available for all participants for all conditions as some reported either no chills or chills in all trials of a condition, thus lacking any control. Hence, the distribution of participants considered for analyses differed across conditions. Therefore, rmANOVAs with Chill (chill, control) and Time as within-subjects variables were computed separately for each condition to examine facial EMG during chills and absent chills. A sensitivity analysis following recommendations by Mehr (2019) was applied to examine whether outlying values have a substantial influence on results regarding chill-associated facial EMG. Visual inspection of individual 7 s means was realized to detect potential outlying values (see Figure 5). Such values were then replaced with the next-highest or next-lowest values (Yale & Forsythe, 1976). Afterwards, paired t-tests were conducted (see Supplementary Materials online). Non-normality was observed for several measures in our data. However, the statistical method (ANOVA) applied here was shown to be robust regarding non-normality (Blanca et al., 2017). To provide a comprehensive report of the results, nonparametric tests (Friedman tests, Wilcoxon tests, Wilcoxon post-hoc tests with adjusted Alpha [i.e., Alpha/3]) were additionally conducted to compare measures of subjective experience (chill frequency and intensity, ratings of pleasantness) and 7 s EMG mean values (see Supplementary Materials online).
To include also those participants with incomplete data (i.e., no means associated with chills or with absent chills), linear mixed models with fixed effects (Duricki et al., 2016) were computed, respectively, for each muscle (see Supplementary Materials online for results). Following the protocol by Duricki et al. (2016), three types of error covariance structure were tested (compound symmetry, unstructured, first-order autoregressive model). Restricted maximum likelihood estimation was applied. An unstructured error covariance structure was finally chosen because it offered the best fit to the data (indicated by Akaike’s information criterion). Chill (chill vs. control) and Condition (music vs. harsh sounds and drum beats vs. harsh sounds and music) served as within-subjects factors. It must be noted that testing the model with unstructured error covariance did not achieve convergence. Therefore, the model with the second best fit (compound symmetry) was additionally reported.
All statistical analyses were conducted using Statistical Package for the Social Sciences 22 (IBM Corporation, Armonk, NY, USA). Alpha was set at .05. In cases of a significant main effect post-hoc comparisons with Bonferroni corrections were conducted. If the assumption of sphericity was violated, Greenhouse–Geisser corrections were applied. Nominal degrees of freedom and effect sizes are listed. Confidence intervals (CI, 90%) for effect size estimates of main effects were computed using the effect size calculator by Uanhoro (2017). Swarm and line plots were created with R (R Development Core Team, 2019), beeswarm (Eklund, 2019), and ggplot2 packages (Wickham, 2016).
Results
Frequency and intensity of chills
The number of chills per minute differed between conditions, F(2, 42) = 29.086, p < .001,
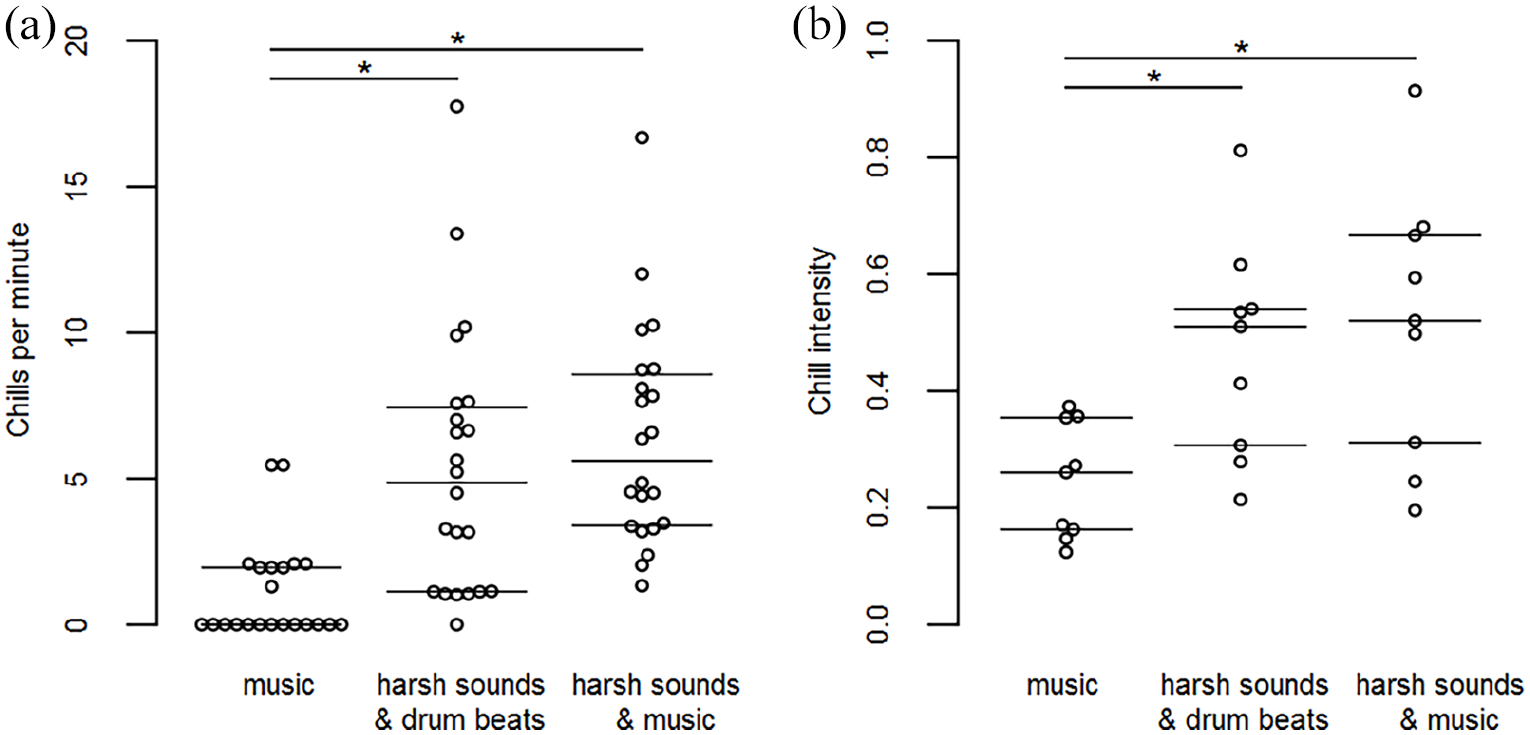
Swarm plots showing (a) individual chills per minute values and (b) chill intensities for the music passages and harsh sounds.
Reported chill intensities also differed between conditions, F(2,16) = 10.906, p = .006,
Pleasantness of chill sensations
Chills during music and chills during harsh sounds differed significantly regarding rated pleasantness, F(2, 14) = 28.745, p < .001,
Facial EMG irrespective of chills
Corrugator activity differed between conditions, F(2, 42) = 5.030, p = .021,
Zygomatic activity differed between conditions, F(2, 42) = 10.181, p = .002,
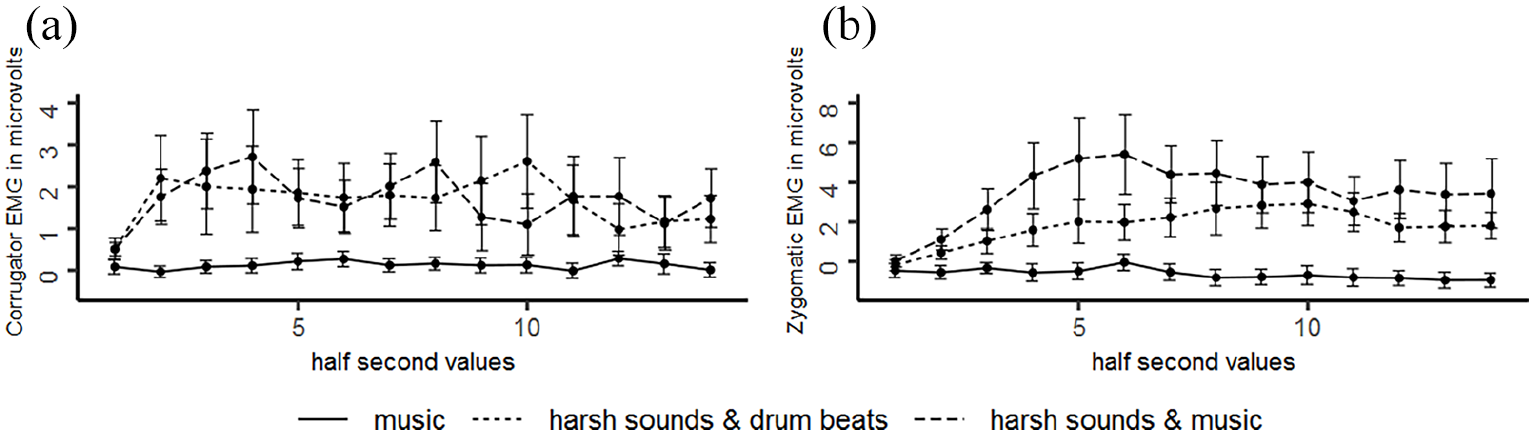
(a) Time course of corrugator and (b) zygomatic EMG activity during music passages and harsh sounds, irrespective of chills.
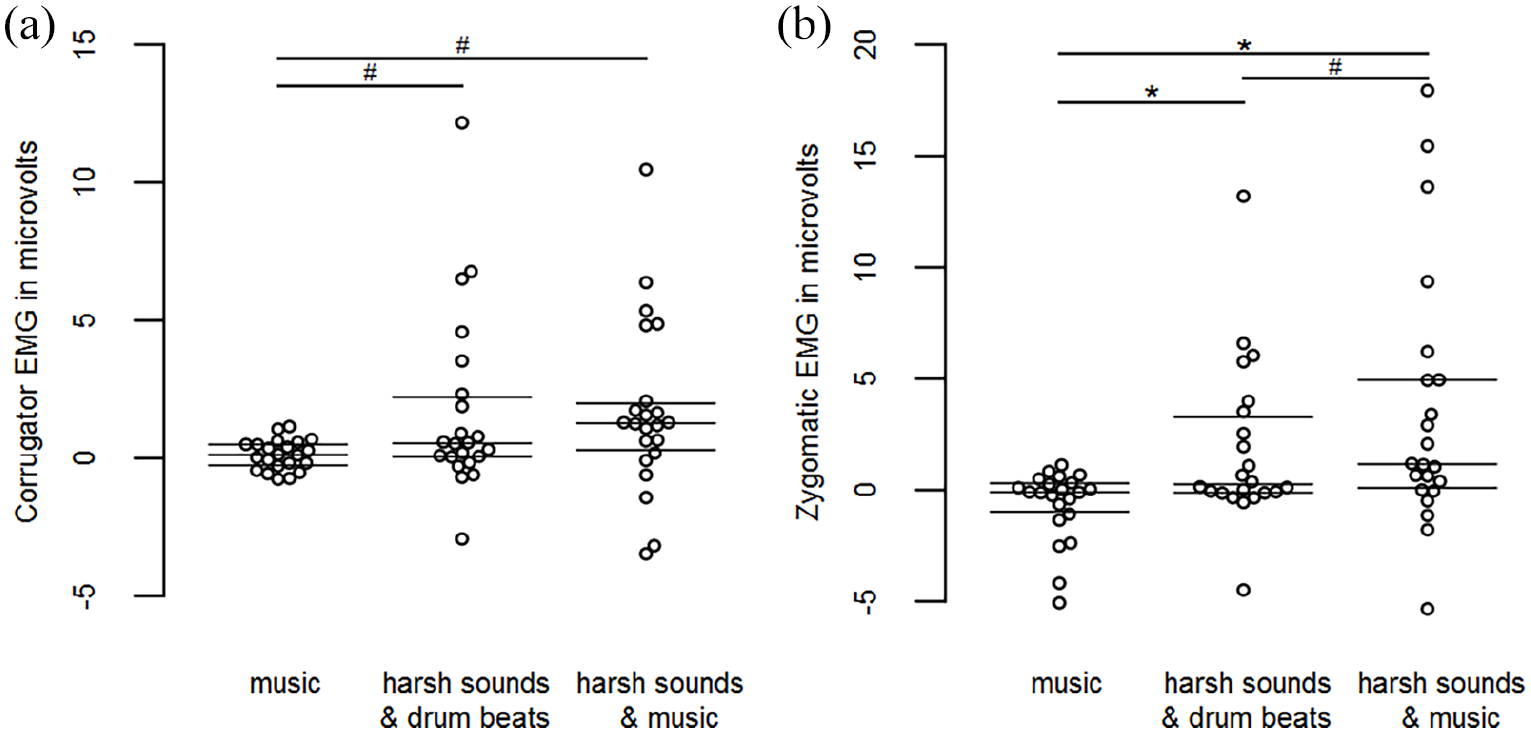
Swarm plots showing (a) individual 7 s means of corrugator and (b) zygomatic EMG activity during music passages and harsh sounds, irrespective of chills.
Facial EMG during chills
Corrugator muscle activity during music passages was slightly higher when chills were reported compared with absence of chills, F(1, 8) = 4.238, p = .074,
Zygomatic muscle activity during music passages did not differ between chills and absence of chills, F(1, 8) < 0.001, p = .991,
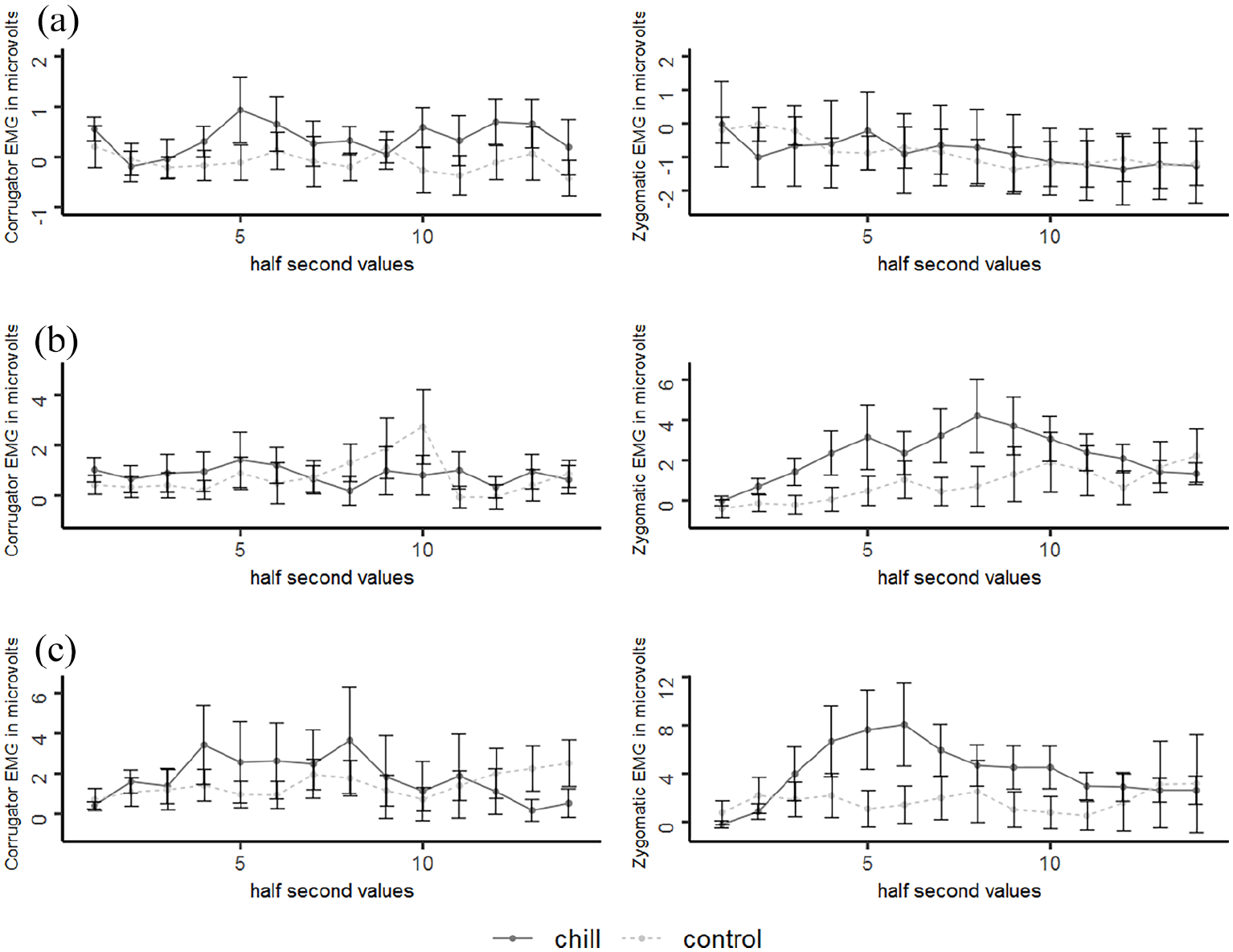
(a) Time course of corrugator and zygomatic EMG activity during music passages, (b) during harsh sounds inserted in drum beats and (c) during harsh sounds inserted in music, respectively, for chills and absent chills (control).
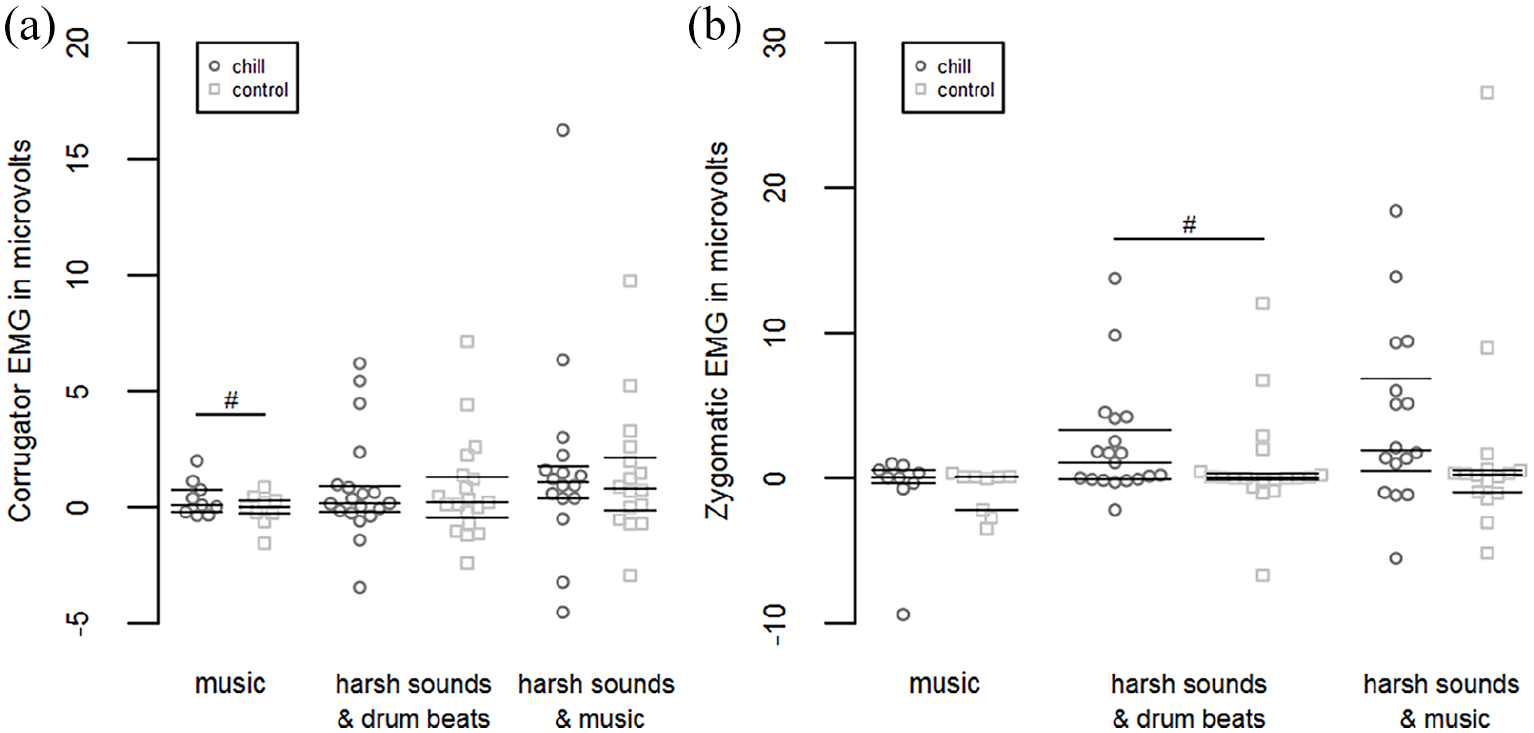
Swarm plots showing (a) individual 7 s means of corrugator and (b) zygomatic EMG activity during music passages and harsh sounds, respectively, for chills and absent chills (control).
Discussion
The present study examined facial muscle activity during both pleasant and unpleasant chills evoked by music and harsh sounds. As expected, harsh sounds reliably elicited chill experiences that were more frequent than pleasant chills elicited by music passages. This finding is consistent with Grewe et al. (2010), who found that even the most successful music stimulus evoked chill responses only in less than half of the participants while playing a harsh sound resulted in a chill response in two-thirds of all participants. Hence, unpleasant chills seem to be rather reflex-like and less individually shaped (Grewe et al., 2010). Moreover, chills evoked by harsh sounds were experienced as more intense compared with chills toward music supporting previous work, which showed that unpleasant acoustic stimuli generally induce states of higher arousal compared with pleasant acoustic stimuli (Bradley & Lang, 2000). However, we did not find any influence of the harsh sounds’ surrounding (i.e., drum beats and music) on frequency and intensity of chill reports. Interestingly, Standley (1991) attenuated aversive reactions toward a subsequent drill sound by playing music from the new age genre, which was described as relaxing. In our study, the music excerpts surrounding the harsh sounds were partly of sad character and depicted moments of grief, tragedy, loss, and longing which might have evoked states of arousal themselves. In fact, states of arousal were shown to be positively associated with an increased susceptibility toward (pleasant) chills (Mori & Iwanaga, 2014) which might be an explanation why no decrease in chill frequency and intensity was detectable when harsh sounds were inserted in music pieces. In contrast to Standley (1991), Matsuo et al. (2019) could not find an effect of relaxing music on the experience of unpleasant acoustic stimuli. Although chills were reported during the excerpts by Barber and Albinoni in line with previous work, no passages could be identified where chills occurred more frequently (Guhn et al., 2007). This might be due to the fact that these excerpts lack characteristics which have been especially related to the induction of chills such as new chords or a sudden increase in volume (Sloboda, 1991).
Generally, corroborating Grewe et al. (2007a) listening to the music passages rarely resulted in responses of the examined facial muscles. In contrast to that, Fuentes-Sánchez et al. (2021) found increased corrugator activity during music of unpleasant character and increased zygomatic activity during music of pleasant character, although the observed increases were of small size. This group applied music from famous films as stimuli and it is not unlikely that the examined cohort knew these films. As familiarity was shown to affect the processing of music stimuli (Pereira et al., 2011; Van den Bosch et al., 2013), using film soundtracks as stimuli could be an explanation why facial responses were observable in this study but not in our work. What is more, Fuentes-Sánchez et al. (2021) used highly pleasant and unpleasant music stimuli which could have contributed to observable variations in the facial responses, while we choose music stimuli primarily with regard to their capacity to elicit chills.
However, when looking at the data in more detail, we could find a slight increase in corrugator activity during the music passages when participants reported chills compared with absent chills. An increase in corrugator activity associated with chills has been also observed by Wassiliwizky, Koelsch, et al. (2017), who applied poems as stimuli. They suggested that such an increase reflects negative emotions of sadness. Although this interpretation appears hardly plausible in the first place given chills are seen as indicators of enjoyment, it was shown that music of sad character evoked pleasurable states in the listeners (Taruffi & Koelsch, 2014; Vuoskoski & Eerola, 2017) and appeared to be especially apt to evoke pleasant chills (Grewe et al., 2007b; Panksepp, 1995). In fact, Wassiliwizky, Jacobsen, et al. (2017) suggested that positive affect during the exposure to sad and moving film scenes (e.g., a scene of separation) was due to the anticipation or realization of a social reunion. Following this perspective, our finding of increased corrugator muscle activity during the experience of pleasant chills might also refer to the simultaneous induction of positive and negative affect in the context of aesthetic stimuli, which has been termed “being moved” (Wassiliwizky et al., 2015). In contrast, we found no chill-associated increase in zygomatic muscle activity. Likewise, in a study by Grewe et al. (2007a), those music pieces which were most successful in inducing goose bumps and shivers rarely resulted in laughing in the participants. As laughing is mainly realized via the zygomatic muscle (Ruch & Ekman, 2001) this result is in concordance with our finding of rare zygomatic activity during music passages. Similarly, Wassiliwizky, Koelsch, et al. (2017) found only a weak association between zygomatic activity and chills underlining that chills were rather associated with negative states (e.g., sadness). Facial expressions indicative of sadness during pleasant chills as observed in the present work and in the previous literature could signal the need for help and, hence, potentially support affiliation (Crivelli & Fridlund, 2018; Seidel et al., 2010) which has been linked to pleasant chills (Maruskin et al., 2012).
In line with a suggested social relevance, the pattern revealed here of facial response during chills toward music (i.e., increase in corrugator activity) could also reflect cognitive disfluency. Such states can be generally evoked by events discrepant to one’s expectations and are associated with increased corrugator activity (Topolinski & Strack, 2015). Interestingly, chills toward music have been primarily reported during unexpected harmonic turns and sudden tonal changes (Grewe et al., 2007b; Guhn et al., 2007). An association between cognitive disfluency and pleasant chills would be plausible as cognitive disfluency is suggested to broaden information processing and to inhibit impulsive responses which in turn support social affiliation (Alter, 2013).
In accordance with the findings of Bradley and Lang (2000) and Dimberg (1990) the applied unpleasant harsh sounds evoked a reliable increase in corrugator activity which mirrors the unpleasantness experienced by the participants. This increase, however, did not differ between chills and absent chills. In addition, we found an increase in zygomatic activity during harsh sounds, which has been also found by Bradley and Lang (2000) when playing highly unpleasant stimuli. This increase was slightly stronger during harsh sounds inserted in music than during harsh sounds inserted in drum beats, which suggests an increased unpleasantness when music and harsh sounds are simultaneously played. Importantly, the increase in zygomatic activity during harsh sounds was more pronounced when chills were reported compared with absent chills, which was especially observable during harsh sounds inserted in drum beats. Sensitivity analyses, however, showed that our null finding in chill-associated zygomatic activity during harsh sounds inserted in music was mainly due to one atypically high mean value. After adjusting this value a significant chill-associated increase in zygomatic activity could be observed also for this condition. Parallel activity of the corrugator and zygomatic muscles toward highly unpleasant acoustic stimuli has been previously shown by Bradley and Lang (2000) and such a response pattern is typically seen during felt pain (Kunz et al., 2008; Prkachin & Mercer, 1989). Therefore, it seems that harsh sounds can be experienced as truly painful, hence, confirming previous work on responses toward such stimuli (Boyd, 1959; Zald & Pardo, 2002). Experiencing a chill during these sounds may reflect an especially strong discomfort, which is also accompanied by an even more pronounced facial reaction as indexed by a larger zygomatic muscle activity. Increased activity of the zygomatic muscle as especially indicative of highly unpleasant states is in line with the work of Wolf et al. (2005), who found that responses of this muscle first appeared when a laser stimulation exceeded the pain threshold, while corrugator muscle activity was observable during all stimulation intensities. Generally, pain is seen as an indicator of an actual or potential injury (Merskey & Bogduk, 1994), while facial expressions of pain communicate information about the experienced distress to fellow beings which are then able to adapt their behavior and provide support (Craig, 1992, 2004). Hence, the pronounced subjective and expressive reaction revealed here could be critical for indicating an aversive context and for initiating avoidance-related behavior which have been both associated with unpleasant chills (Maruskin et al., 2012). Hence, our results corroborate pioneering work by Blake (1986), who interpreted chills toward harsh sounds as a vestigial reflex dating back to flight and group warning behavior in our ancestors. A further explanation could be that facial responses during highly unpleasant sounds and associated chills may act as a part of a response pattern of the whole body, which is realized to get away from the evoking unpleasant stimulus or simply as a strategy to divert attention from the experienced discomfort (Frijda, 2002). Similarly, one could also argue that a profound facial response reflects an attempt to reduce the sensory exposure to chill provoking unpleasant acoustic stimuli (Jäncke et al., 1996). Actually, it was found that activity in the facial muscles coincided with activity in the middle ear muscles, which in turn are known to affect the sensory intake (Djupesland, 1965).
The results presented here were further supported by additionally conducted nonparametric tests. Linear mixed models also confirmed the findings drawn from rmANOVAs. However, it must be stated that for models with the best model fit, convergence has not been achieved which might be due to our small sample size. Therefore, validity of the model fits appears uncertain and, hence, significance must be treated with caution.
Limitations
The applied music and unpleasant sound stimuli differ in various aspects such as their waveforms (Roederer, 2009). Hence, a confounding effect of stimulus modality on the examined measures cannot be ruled out. As a consequence, comparisons regarding chill reports and facial responses should be interpreted with caution, as noted by Grewe et al. (2010). However, to assure that pleasant and unpleasant chills could be reliably induced the applied stimuli were selected with regard to their chill evoking capacity. As we were primarily interested in whether reactions associated with chills differ from reactions not associated with chills separately for each valence condition (pleasant, unpleasant), we accepted heterogeneous natures of the applied stimuli (i.e., music and sounds). To draw comparisons more adequately, stimuli could be applied which systematically vary in valence and arousal within one acoustic modality, as previously used in research on affective responses toward sounds (Bradley & Lang, 1999) and music (Eerola & Vuoskoski, 2011; Witvliet & Vrana, 2007). However, it must be noted that it is challenging to find pleasant and unpleasant chill stimuli of similar arousal (Grewe et al., 2010).
Our sample size was comparable with previous research in the field (Haar et al., 2020) and appears adequate in the light of an extensive peripheral physiological experiment. However, a rather small number of included participants might have hampered the detection of chill-associated facial responses and could explain why mainly just trend significant differences could be observed. The problem of a small sample size was also reflected by wide confidence intervals associated with the effect size estimates. We suggest that future approaches should include a larger sample size to increase power and precision of estimates and to provide further evidence for the results shown here.
Conclusion
A vast amount of research dealing with chill reactions, associated symptoms, and elicitors have been conducted in the past; however, only few studies have evaluated facial responses associated with chills. Also, it is surprising that although highly unpleasant sensations toward harsh acoustic stimuli seem to be well known all over the world (Cox, 2008), chills during such contexts have received much less attention than chills toward esthetic stimuli so far. This lack might be due to ethical concerns about inducing deliberately such unpleasant sensations (Halpern et al., 1986). However, examining facial expressions associated with pleasant and unpleasant chills offers the opportunity to shed further light on a unique and fascinating human sensation for which everyday relevance has been proposed (Blood & Zatorre, 2001; Fukui & Toyoshima, 2014; Maruskin et al., 2012). In the current study we could elicit both pleasant and unpleasant chills, thus corroborating the idea of the chill as a nonspecific physiological phenomenon which indicates different psychological states of high intensity such as pleasure and pain (Grewe et al., 2010; Maruskin et al., 2012). By recording EMG activity of two facial muscles we found chill-associated facial reactions for both pleasant and unpleasant chills. These findings constitute further evidence for the idea that chills play a role in regulating interpersonal relationships by promoting approach and avoidance behavior (Maruskin et al., 2012). Hence, chill responses might have lost certain adaptive functions in humans such as thermoregulation (Tansey & Johnson, 2015); however, in line with recent findings our study contradicts the claim that the chill experience constitutes a “blind alley” nowadays (Konecni et al., 2007). The results of the current study suggest that measuring facial muscle activity can be a valuable approach toward an objective reference of acoustically induced pleasant and unpleasant chills.
Supplemental Material
sj-docx-1-pom-10.1177_03057356221122607 – Supplemental material for Written in the face? Facial expressions during pleasant and unpleasant chills
Supplemental material, sj-docx-1-pom-10.1177_03057356221122607 for Written in the face? Facial expressions during pleasant and unpleasant chills by Kai Klepzig, Katharina Stender, Martin Lotze and Alfons O. Hamm in Psychology of Music
Footnotes
Author contributions
K.K. worked on measuring participants, data evaluation, and writing of the manuscript. K.S. helped with study design, data evaluation, and writing of the manuscript. M.L. worked on the study design and writing of the manuscript. A.O.H. worked on the study design and writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been funded by the DFG (German Research Foundation; LO795/22-1).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
