Abstract
This study investigates the influence of water vapour on the hydrogen reduction of iron ores, focusing on conditions closely aligned with industrial processes, including high temperatures and realistic gas compositions. Single-pellet reduction experiments were conducted isothermally in gas atmospheres with varying ratios of water vapour and hydrogen (30% H2O + 70% H2, 20% H2O + 80% H2, 10% H2O + 90% H2 and 100% H2) over a temperature range of 800°C to 1000°C. The results demonstrate a consistent increase in the reduction rate with rising temperature across all gas compositions, highlighting the impact of water vapour on reduction kinetics under near-industrial conditions. At 30% H2O, reduction was limited to wüstite, with no metallic iron formation observed. At 20% H2O, reduction degrees increased from 36% at 800°C to 77% at 900°C, where a thick metallic iron layer formed, and to 95% at 1000°C, leaving only trace wüstite. Under 10% H2O, reduction exceeded 97%, approaching completion. The study reveals that high water vapour content hinders hydrogen diffusion and blocks active sites on wüstite, impeding its reduction to metallic iron. These findings, supported by kinetic modelling, underscore a complex interplay of diffusion and chemical reaction as the controlling mechanisms. By examining the reduction process under conditions that closely replicate those in industrial hydrogen-based direct reduction, this study provides critical insights into optimising reduction efficiency for sustainable ironmaking.
Introduction
The iron and steel sector, a cornerstone of modern society, is currently heavily reliant on fossil fuels, accounting for 7% of global energy-related CO2 emissions. 1 The transition to fossil-free iron and steel production is of paramount importance, given the significant environmental impact of the current production methods.1,2 For instance, the production of steel, the most widely used metal on earth, consumes annually 27 EJ of coal, 3 EJ of gas and 5 EJ of electricity, resulting in an average emission of 2 tonnes of CO2 per tonne of steel. 1
A promising pathway towards decarbonising the steel industry involves the use of green hydrogen (H2) for the direct reduction of iron ore.1,3 This approach, which replaces fossil-based reductants with green H2, could potentially revolutionise the iron and steelmaking process. 3 Moreover, companies such as SSAB, LKAB and Vattenfall AB are aiming to create the first fossil-free value chain from mining to finished steel, underscoring the industry's commitment to this transition. 2 Thus, the shift to fossil-free iron and steel production is not only crucial for reducing CO2 emissions but also for ensuring the sustainability of the steel industry in the long term.1–3 Hydrogen can be utilised in three different ways in the iron and steel industry. First, hydrogen can be injected into the blast furnace. In this method, part of the coke (CO as the reducing gas) is replaced by hydrogen, which mitigates CO2 emission.4,5 However, since the blast furnace has been designed to operate with coke, it is not possible to replace all the coke with hydrogen.4,5 The second method is direct reduction (DR) using hydrogen as the reducing gas. This process can be carried out in shaft furnaces or fluidised bed reactors.6–8 This process can be done with only hydrogen, which can be fossil-free if the hydrogen is produced through methods like water electrolysis.6–8 The third method is plasma hydrogen reduction. In this process, iron ore can be melted and reduced simultaneously in a plasma arc zone.9–11 This method demonstrates the efficacy of hydrogen plasma in transforming metal oxides into metal.9–11
The process of hydrogen reduction in iron ores involves a sequence of steps, beginning with the reduction of hematite to magnetite, followed by the conversion of magnetite to wüstite (occurring above 570°C), and ultimately ending with the reduction of wüstite to metallic iron. Numerous researchers have delved into the intricate kinetics of hydrogen reduction of iron oxides, revealing a complex interplay of factors such as temperature, mineralogical composition, gas flow rate, pressure and porosity, all of which can significantly influence reaction rates. 12 These factors can interrelate and influence one another. Sadeghi et al. investigated the interplay between gas composition, temperature and porosity. H2 enhances early porosity and facilitates reduction at lower temperatures, but CO necessitates elevated temperatures for efficacy and initially generates less porosity. A mixture of 50% H2 and 50% CO preserves the initial advantages of hydrogen and the efficacy of CO at elevated temperatures. 13 A separate study examined the impact of temperature on porosity development. Elevated porosity evolution rates at increased temperatures and within larger pellets result in enhanced pore complexity and tortuosity. The heightened tortuosity may impede gas flow and diminish diffusion, hence extending the duration of reduction. 14 Cavaliere et al. discovered that porosity, pore size and pore tortuosity fluctuate according to certain processing parameters, with temperature exerting a more significant influence. 15 The reduction conditions and pellet structure also influence post-reduction processes like carburisation. According to Perrone et al., higher porosity and metallisation enhance pellet reactivity, making them more effective for carburisation. Additionally, a uniform distribution of SiO2, Al2O₃ and CaO is essential for ensuring a consistent carburisation process. 16
During hydrogen reduction reactions, water vapour emerges as a byproduct. Within the confines of the shaft furnace, this water vapour typically passes a distance of approximately 10 m before egressing as part of the top gas. Additionally, moisture inherent in the feed material or the inadvertent introduction of water vapour via other sources can also contribute to the presence of water vapour within the furnace environment. Moreover, the reaction between hydrogen and oxygen within the shaft or blast furnace can instigate further water vapour production through this exothermic reaction, thus facilitating the heat required for the reduction process.
The impact of water vapour on hydrogen reduction has been extensively studied, with research highlighting its varying effects across different temperatures and ore compositions. Kim et al. explored the impact of water vapour on the hydrogen reduction of magnetite powder, revealing a notable decrease in reduction rates, particularly within the temperature range of 500–700°C. 17 Lorente et al. conducted experiments on pure hematite, discovering that while water vapour did not impede the reduction of hematite to magnetite, a 5% concentration effectively hindered the formation of metallic iron. 18 Similarly, Ito et al. and Adam et al. investigated the reduction kinetics of crushed iron ore and magnetite, respectively, unveiling a diminishing rate of reduction as water vapour content increased, with significant inhibition observed at higher concentrations. Notably, the presence of water vapour was found to promote the formation of a dense magnetite layer, which hindered subsequent reduction steps.19,20 However, Sastri et al. presented contrasting findings, demonstrating a non-linear relationship between water vapour content and reduction rate in pure hematite systems. 21 Mao et al. reported this complexity, observing a decrease in reduction rate with increasing water vapour content, albeit with slight effects on the maximum reduction degree. 22 Further insights into microstructural changes during reduction were provided by Matthew et al. and John et al., highlighting the intricate interplay between water vapour content and pore structure evolution at different stages of the reduction process.23,24
Despite these valuable contributions, the existing literature appears fragmented, lacking a cohesive understanding of water vapour's influence on pellet regeneration, particularly within temperature ranges relevant to shaft furnace operations. Consequently, there is a pressing need for comprehensive research aimed at elucidating the multifaceted dynamics of water vapour-mediated regeneration processes in iron ore pellets. Given these complexities, it becomes imperative to investigate the impact of water vapour on both the thermodynamic equilibrium and the kinetics of the reduction process, notwithstanding the relatively sparse existing literature on this particular aspect.
Materials and methods
In this study, spherical industrial pellets crucially employed in the direct reduction (DRI) process were utilised. Each pellet weighed approximately 6 ± 0.1 g and measured 1.3–1.4 cm in diameter. The selection of these pellets was based on their significance as key feedstocks in the iron and steel production sector. The chemical compositions and physical properties of the pellets are detailed in Tables 1 and 2, respectively.
The chemical composition of DRI pellet.

The physical properties of DRI pellets.

To ensure the integrity of the samples, the pellets underwent preconditioning in an oven at 120°C for 1 h to eliminate any residual moisture. Subsequently, reduction experiments were conducted utilising the Linseis STA PT 1600, a state-of-the-art TG-DSC device capable of simultaneous mass and heat difference measurements which is illustrated schematically in Figure 1(a). This device offers optional mass spectrometer gas analysis capabilities and operates within a reducing atmosphere at temperatures up to 1600°C. The tubular device measures 40 cm in length, with an inner diameter of 30 mm and a wall thickness of 3 mm. The sample is positioned at the midpoint of the tube, while the gas is introduced from the bottom and exits from the top.
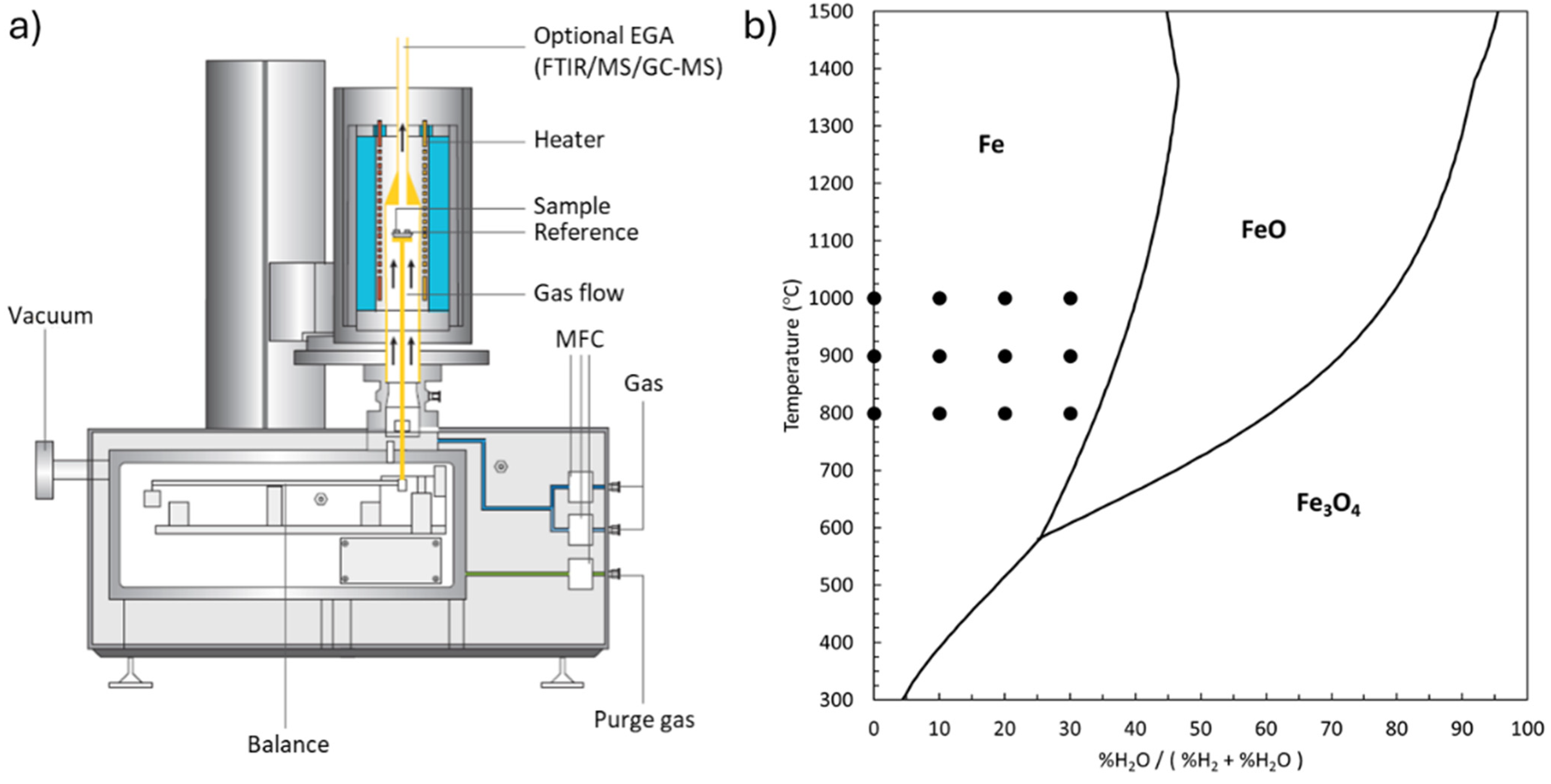
(a) Schematic of utilised TG-DSC device (Linseis STA PT 1600), and (b) Baur–Glaessner diagram. Each point represents one isothermal experiment (calculated with HSC Chemistry version 10.4.1.1).
Single-pellet hydrogen reduction experiments included 12 isothermal tests with gas composition of 100% H2, 90% H2 + 10% H2O, 80% H2 + 20% H2O and 70% H2 + 30% H2O at 800°C, 900°C and 1000°C. As illustrated in Figure 1(b), the gas compositions and temperatures have been selected in a way that the full reduction to metallic iron is thermodynamically possible. For all isothermal experiments with a constant gas flow rate of 2000 mL/min and after 110 min reduction, samples were cooled down to 20°C in N2 atmosphere. Due to the high inlet and outlet flow rate, it has been assumed that the gas composition remains constant during the experiment. To ensure uniform gas flow within the experimental setup, the gas inlet was designed with a diffuser to evenly distribute the gas before reaching the sample. The high gas flow rate of 2000 mL/min ensured minimal concentration gradients within the reaction chamber, allowing for stable and uniform gas exposure. Water vapour levels were precisely controlled by using a heated humidification system, in which the carrier gas (H2) was passed through a temperature-controlled water reservoir before entering the reaction chamber. The humidity level was adjusted by varying the temperature of the water reservoir and verified using a hygrometer at the gas outlet to ensure consistency throughout the experiments.
After high-temperature tests, the pellet specimens were embedded in epoxy, subsequently sectioned and polished for analysis. Subsequently, the polished samples were coated with carbon to enhance conductivity for electron microscopy examination. Morphological investigations of the reduced pellets were then conducted using a field emission scanning electron microscope (FESEM; Zeiss Ultra Plus, Carl Zeiss SMT AG, Germany), coupled with an energy-dispersive X-ray spectroscopy (EDS) device for elemental analysis.
Results and discussions
Reduction degree
Figure 2 illustrates the outcomes of isothermal reduction tests conducted within the temperature range of 800°C to 1000°C, varying the presence of water vapour from 0% to 30%. It is evident that as the temperature increases, the rate of reduction also increases. However, this effect varies depending on the gas composition and the stage of reduction. For instance, using pure hydrogen, the time taken to achieve maximum reduction degree decreases from 79 min at 800°C to 50 and 29 min at 900°C and 1000°C, respectively. The impact of temperature on the reduction rate becomes more pronounced in the presence of 10% water vapour, where the time required to reach the maximum reduction degree decreases further.
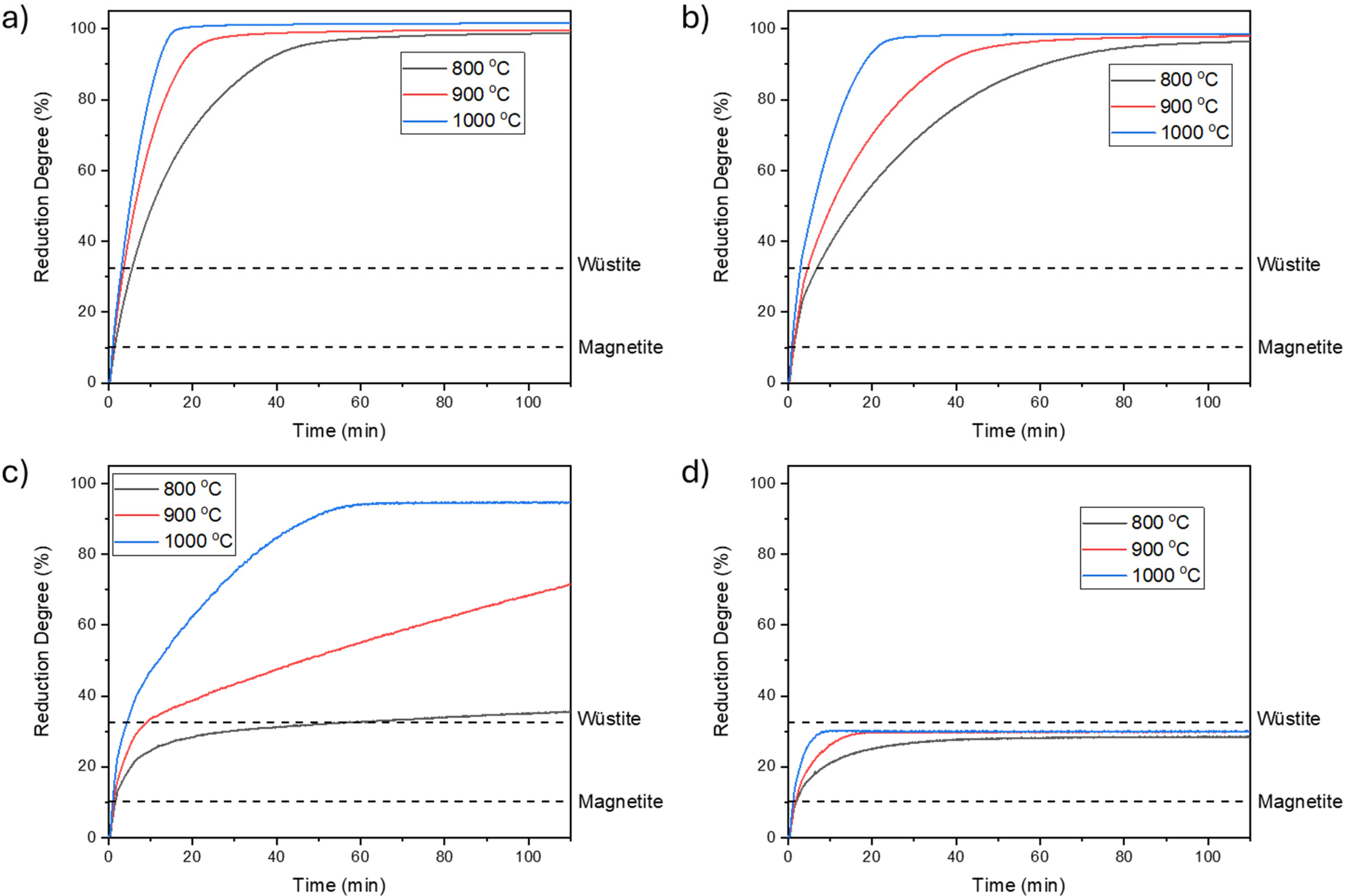
Hydrogen reduction curves with (a) 100% H2, (b) 90% H2 + 10% H2O, (c) 80% H2 + 20% H2O and (d) 70% H2 + 30% H2O at different temperatures.
Comparing Figure 2(a) and (b) indicates that increasing water vapour content decreases the reduction rate and prolongs reduction time, with the gas composition exhibiting a more pronounced effect at lower temperatures. Figure 2(c) demonstrates a substantial impact of temperature on the reduction rate when 20% water vapour is present. While the maximum reduction degree is achieved after 61 min at 1000°C, the reduction process does not reach equilibrium within 110 min at 900°C and 800°C, progressing at a slower rate. Figure 2(d) suggests that although reduction may appear halted after the second step, temperature still noticeably affects the reduction rate.
Furthermore, Figure 2 highlights that the first step of reduction (hematite to magnetite) is notably rapid and unaffected by temperature and water vapour content. However, both temperature and water vapour content influence the rate of the second step (magnetite to wüstite), with water vapour content's effect becoming more pronounced above 10%. Additionally, increasing water vapour content intensifies temperature's impact on the reduction rate. Figure 2(a) to (c) indicate that temperature's most significant effect on reduction rate occurs during the final stage (wüstite to iron).
Figure 3 depicts the maximum reduction degree under different reaction conditions. Notably, full reduction is achieved at all temperatures with pure hydrogen. Introducing 10% water vapour has a negligible effect on the reduction degree, aligning closely with results obtained using pure hydrogen. However, with 20% water vapour, a substantial decrease in reduction degree is observed, particularly at 800°C and 900°C. The reduction degree drop is less pronounced at 1000°C. With 30% water vapour, the maximum reduction degree remains around 30% for all temperatures, indicating that wüstite to metallic iron reduction fails to initiate, despite thermodynamic feasibility. Additionally, Figure 3 underscores that temperature's impact on reduction degree is significant only with 20% water vapour in the gas mixture.
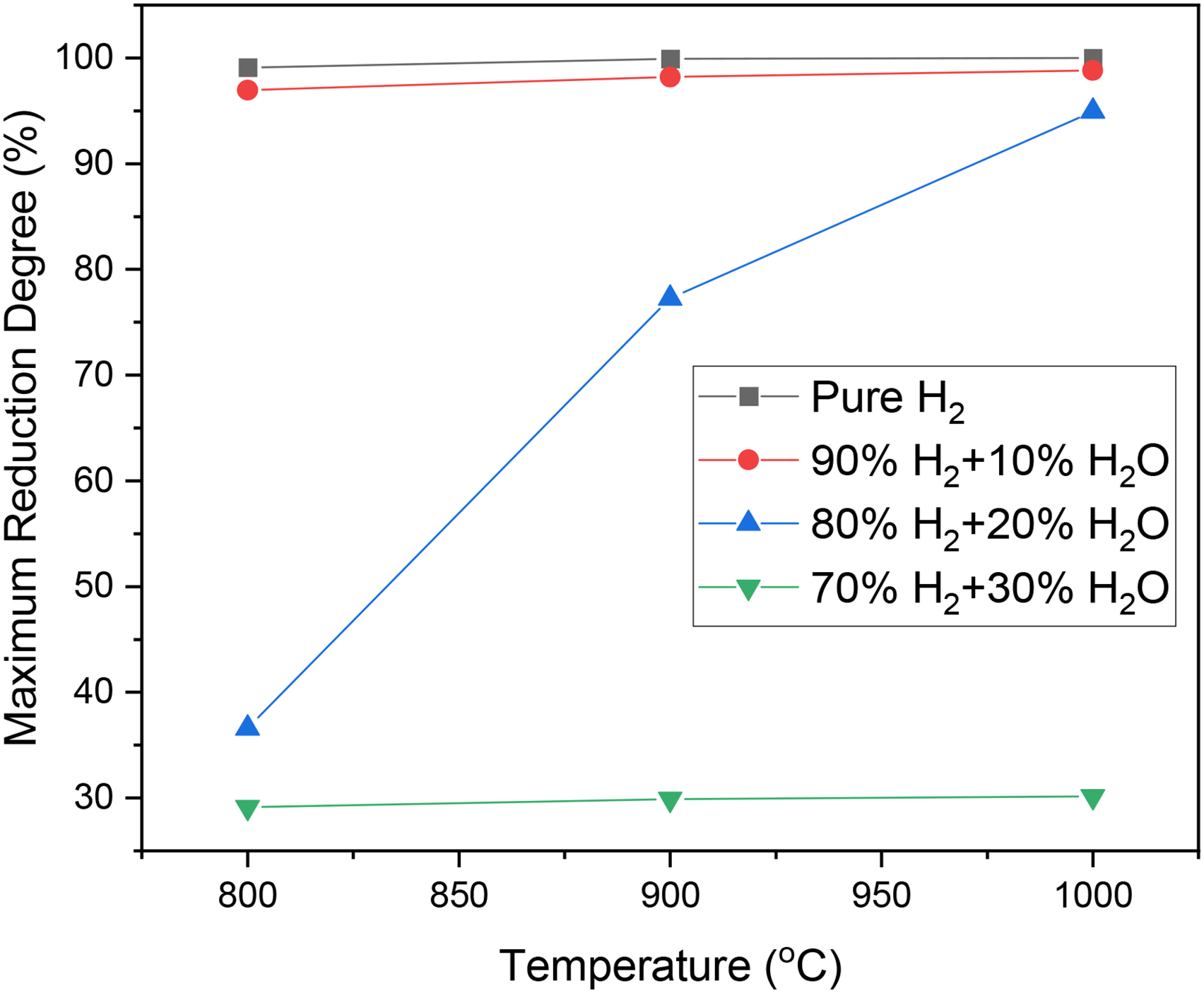
Maximum reduction degree at different temperatures and with different gas compositions.
Microscopic analysis
Figure 4 presents the initial microstructure of the unreduced pellets, revealing grains up to 20 μm in size surrounded by micropores. Energy-dispersive X-ray spectroscopy (EDX) elemental mapping in the SEM images confirms that hematite is the primary component of the pellets. Additionally, the main gangue minerals present include SiO2, Al2O₃, CaO and TiO2. While Al2O₃ and CaO are evenly distributed throughout the structure, SiO2 and TiO2 tend to concentrate in specific regions, particularly along grain boundaries.
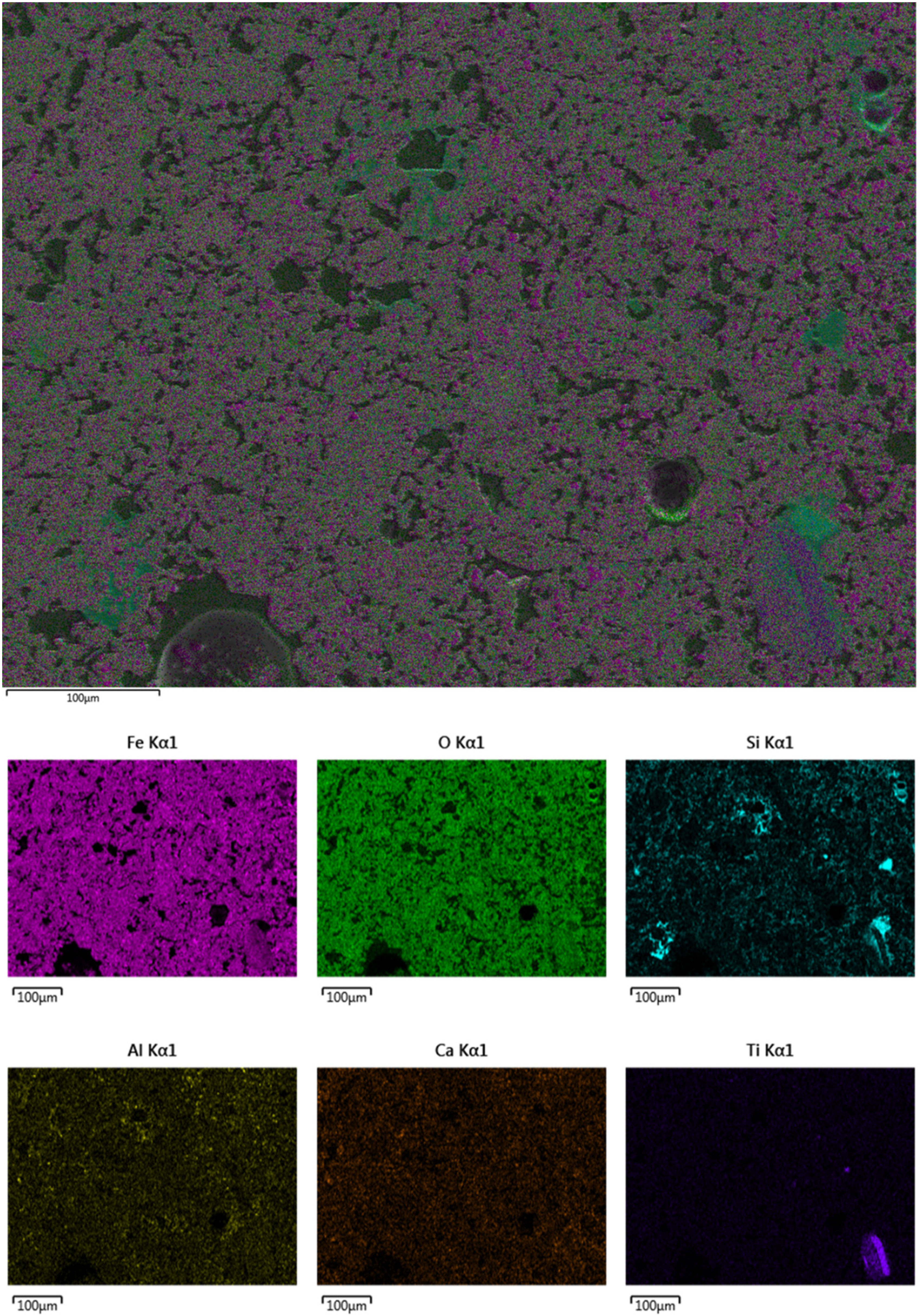
FESEM microstructure and map of initial unreduced pellet.
Figure 5(a) to (c) depict the FESEM images of pellets subjected to reduction using pure hydrogen at varying temperatures. At 800°C, although metallic iron predominates, pockets of unreduced wüstite are discernible within the core of certain larger grains. Conversely, at 900°C and 1000°C, the microstructure of reduced pellets reveals metallic iron as the sole phase, accompanied by darker grey gangue materials.

FESEM images of reduced pellets with 100% H2 at (a) 800°C, (b) 900°C and (c)1000°C, and reduced pellets with 90% H2 + 10% H2O at (d) 800°C, (e) 900°C and (f) 1000°C.
Figures 5(d) to (f) display FESEM results of pellets reduced with a mixture of 90% H2 and 10% H2O. At 800°C, the presence of unreduced wüstite is evident in some grains, with wüstite content notably more abundant compared to pellets reduced solely with hydrogen. At 900°C, metallic iron emerges as the primary phase, albeit with sporadic traces of wüstite occasionally observed within the core of larger grains. Notably, at 1000°C, no wüstite remains observable, suggesting a near-complete reduction of the pellet.
Figure 6(a) illustrates the compact microstructure of reduced pellets utilising a gas mixture of 80% H2 and 20% H2O, comprising predominantly wüstite and gangue oxides, with no detectable metallic iron. Contrastingly, Figure 6(b) demonstrates that at 900°C, metallic iron formation initiates from the grain edges towards the core, albeit wüstite remains the primary phase in larger grains. With a further temperature increase to 1000°C (Figure 6(c)), metallic iron becomes the predominant phase, with wüstite mainly present only in larger grains.

FESEM images of reduced pellets with 80% H2 + 20% H2O at (a) 800°C, (b) 900°C and (c) 1000°C, and reduced pellets with 70% H2 + 30% H2O at (d) 800°C, (e) 900°C and (f) 1000°C.
Conversely, Figure 6(d) to (f) illustrate that in the microstructure of pellets reduced with 30% water vapour in the reducing gas, metallic iron is absent at all temperatures. Instead, wüstite constitutes the main phase, with traces of other phases, mainly SiO2, observed in the dense microstructure of pellets.
Furthermore, Figures 5 and 6 collectively demonstrate that irrespective of water vapour content in the reducing gas, elevating the temperature enhances the formation and expansion of pores and cracks. The formation of cracks is attributed to the restructuring of the crystal lattice during phase transformations, resulting in stresses and lattice disruptions. 13 Increasing the water vapour content in the reducing gas significantly diminishes the porosity of reduced pellets, resulting in a denser microstructure, particularly when the water vapour percentage surpasses 10%.
This effect is detected for both macro pores between the grains and micro pores inside the grains. Although John et al. 25 reported the same results, Hessling et al. 26 did not detect any noticeable change in macro pores by adding 15% water vapour to the system. However, they observed wider but less numerous micro pores by increasing water vapour resulting in more dependence on solid-state diffusion. The difference in the results indicates the impact of other factors such as temperature, flow rate and structure of the initial pellet on the effect of water vapour. Nevertheless, higher reduction temperatures can mitigate this effect. Porosity plays a crucial role in facilitating gas penetration, increasing reaction rate and improving reduction performance. It influences gas diffusion, permeability and overall reduction kinetics by providing pathways for efficient gas flow and reaction sites. 14 Moreover, increasing both porosity and pore size provides more available surface for reaction development. 15
Figure 1(b) shows that even with 30% water vapour, complete reduction should be achieved at the thermodynamic equilibrium. However, the reduction curves and FESEM images show that there is a kinetic barrier that prevents the full reduction. Previous studies suggest that the reduction of wüstite to iron involves multiple kinetic steps: (1) diffusion of H2 molecules to the pellet surface; (2) reaction with oxygen atoms at the external surface of the Fe layer to form water; (3) removal of H2O from the surface; (4) diffusion of oxygen atoms through the Fe layer driven by an oxygen activity gradient, away from the FeO/Fe reaction front where the FeO ↔ Fe + O equilibrium exists; and (5) nucleation of Fe accompanied by the continuous release of oxygen at the internal reaction front. 27 As the partial pressure of water vapour (PH2O) increased, the grain size of the pellets grew larger, while the number and size of micro pores decreased. This led to greater diffusion resistance for hydrogen moving towards the core of the pellets and for the water vapour produced during the reaction moving towards the pellet surface. Additionally, as reduction progressed and a solid iron layer formed on the pellet surface, the distance hydrogen needed to travel from the surface to the iron/wüstite interface increased. Similarly, the distance water vapour had to travel from the iron/wüstite interface to the pellet surface also increased, further raising diffusion resistance. However, since no metallic iron was detected on the pellet surfaces when 30% water vapour was present in the gas mixture, these factors alone do not fully explain the effect of water vapour on the reduction process. Figure 7 shows hydrogen concentration at various locations within the pellet during the reduction process. Due to the high flow rate gas film resistance can be safely neglected [C(H2g)=C(H2s)]. The concentration gradient inside wüstite, which drives solid-state diffusion, likely plays a significant role.28,29 When water vapour content in the gas mixture increases while total pressure remains constant, the partial pressure of hydrogen decreases, effectively diluting the hydrogen in the reactive gas. This reduces the concentration gradient of hydrogen between the reaction interface [C(H2c)] and the pellet surface [C(H2s)]. Moreover, the higher water vapour concentration at the pellet surface lowers the gradient of water vapour between the reaction interface (where it is produced) and the surface, further impeding diffusion. So, a decrease in the concentration gradients of both hydrogen and water vapour is identified as the primary cause of the diminished reduction performance when water vapour levels in the system increase.
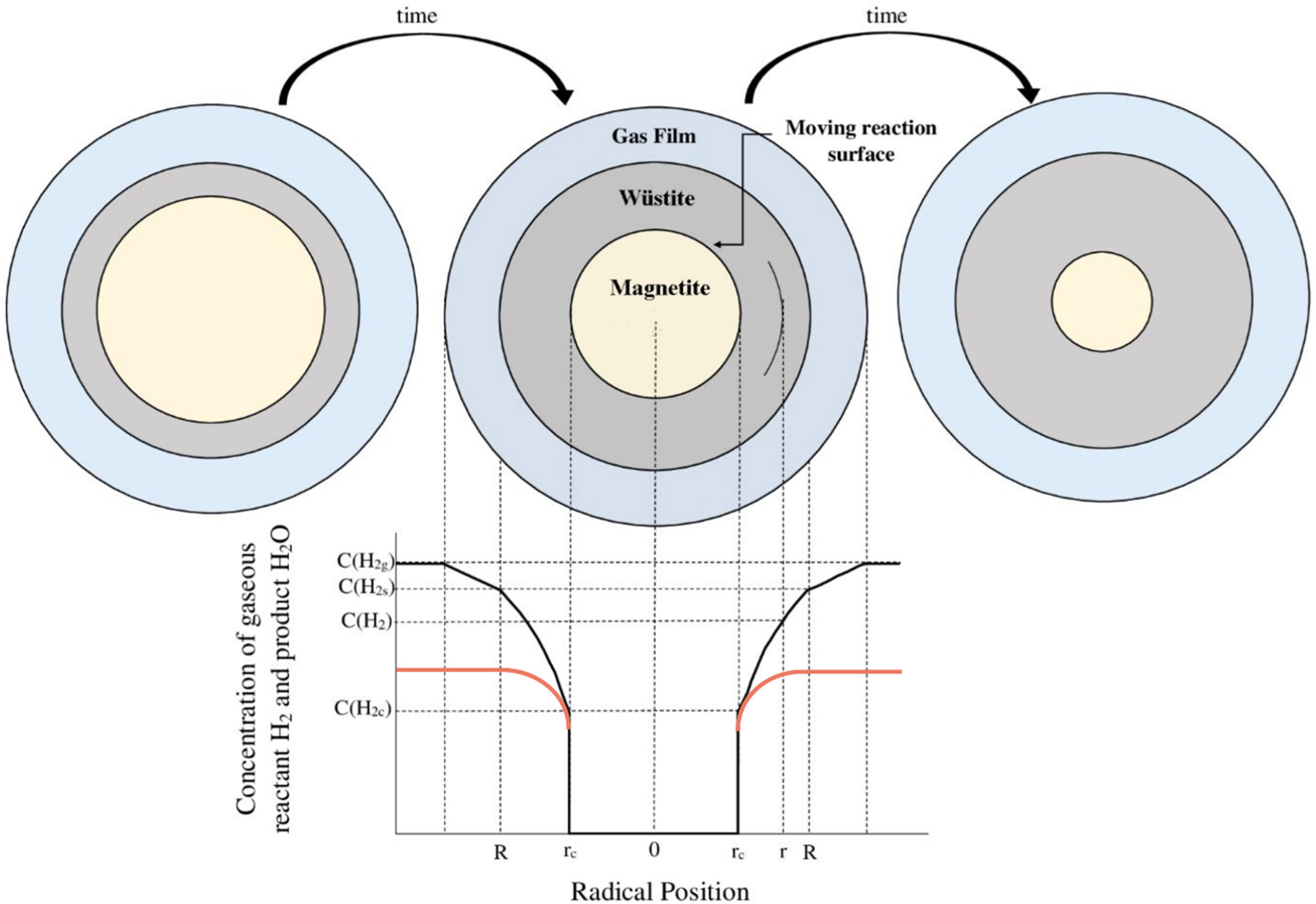
Hydrogen concentration at various locations within the pellet during the reduction process with pure hydrogen (black curves), and hydrogen plus water vapour (red curves) (modified). 12
Reduction kinetics
As it has been mentioned earlier, the reduction consists of three stages, reduction of hematite to magnetite, magnetite to wüstite, and wüstite to iron, so, the kinetics of each stage should be studied separately. However, since the reduction process of hematite to magnetite was very fast, it was not distinguished for kinetic analysis. So, the two first stages were merged and hematite to wüstite is considered as the first stage. A common method for determining isothermal kinetics parameters involves fitting experimental data to theoretical models. By comparing the experimental results with calculated data from various models, the best-fitting model can be identified based on the smallest difference in conversion rates. The rate of the reduction reaction is determined by differentiating the reaction conversion degree over time, with the kinetic equation expressed as follows:
In isothermal kinetics analysis, the integral method is another tool for processing data, as shown in equation (2):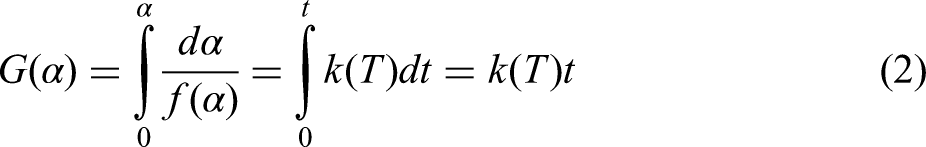
Here,
For each experiment, the values of
Gas–solid kinetic models.

It's crucial to note that gas resistance when passing through an ash layer is significantly higher than the resistance through the gas film surrounding the particle. Therefore, in cases where the reaction produces a solid, non-porous ash layer, like during hematite pellet reduction, the resistance of the gas film can be neglected, especially when using a reducing gas with a high flow rate. 34
The model fitting for the first stage of reduction is shown in Figure 8. As illustrated in Figure 8(a), when pure hydrogen is used at all temperatures, and when 10% water vapour is used at 900°C and 1000°C, the primary resistance to reduction is due to the chemical reaction. However, for reduction with 10% water vapour at 800°C, and 20% water vapour at 900°C and 1000°C, the controlling mechanism shifts to diffusion. In contrast, for reduction with 20% water vapour at 800°C and 30% water vapour at all temperatures, none of the models listed in Table 3 showed good linearity. This suggests that no single mechanism can fully describe the kinetics under these conditions, indicating that the process is likely controlled by a combination of factors (mix-controlled kinetics).
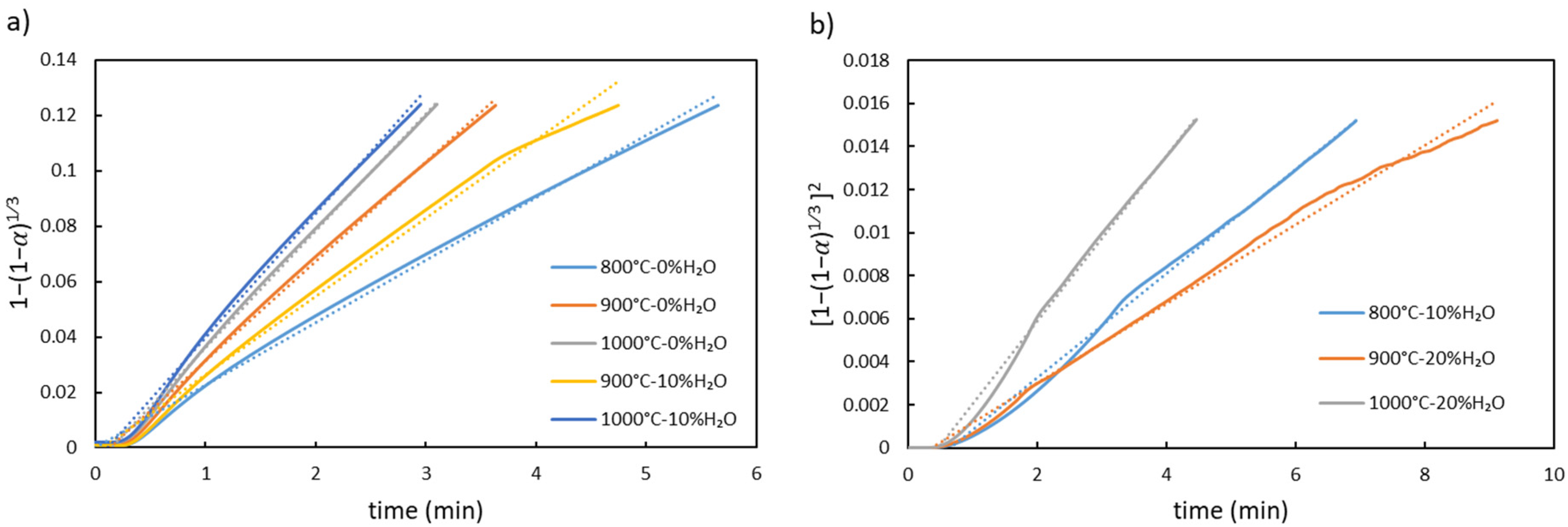
Model fitting for the first stage of reduction (a) 3D chemical reaction (R3), and (b) 3D diffusion (D3).
In other words, at low water vapour content, hydrogen diffuses easily into the pellets, and the reduction is primarily governed by the chemical reaction. However, increasing the water vapour content or lowering the temperature makes diffusion more difficult. As previously mentioned, higher water vapour reduces the concentration gradient, which drives diffusion. Further increases in water vapour also hinder the chemical reaction by blocking active sites at the reaction interface.8,36 The surface of wüstite is regarded as having a finite number of sites available for occupation by hydrogen and water vapour. Additionally, water vapour can be dissociated into oxygen ions and hydrogen at the wüstite surface, as per reaction (3). Consequently, alongside water vapour and hydrogen, the oxygen generated from reaction (3) can be adsorbed onto the surface sites of wüstite.
37
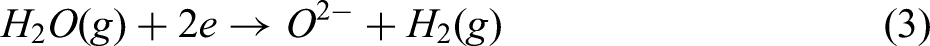
Equation (4) represents all the sites on the surface of wüstite:

The equilibrium constant 



Equation (9) shows that by increasing the partial pressure of water vapour, a higher fraction of sites are occupied by the activated complex means blocking the contact of hydrogen and wüstite and as a result hindering the reduction of wüstite by hydrogen. Consequently, the kinetics of reduction becomes a combination of both diffusion and chemical reaction by increasing water vapour in reducing gas, which prevents the initiation of reduction of wüstite to iron in the presence of 30% water vapour.
The relationship between the observable rate constant, 
Taking the natural logarithm of both sides gives:
In these equations,
As described in equation (11), the observable rate constant
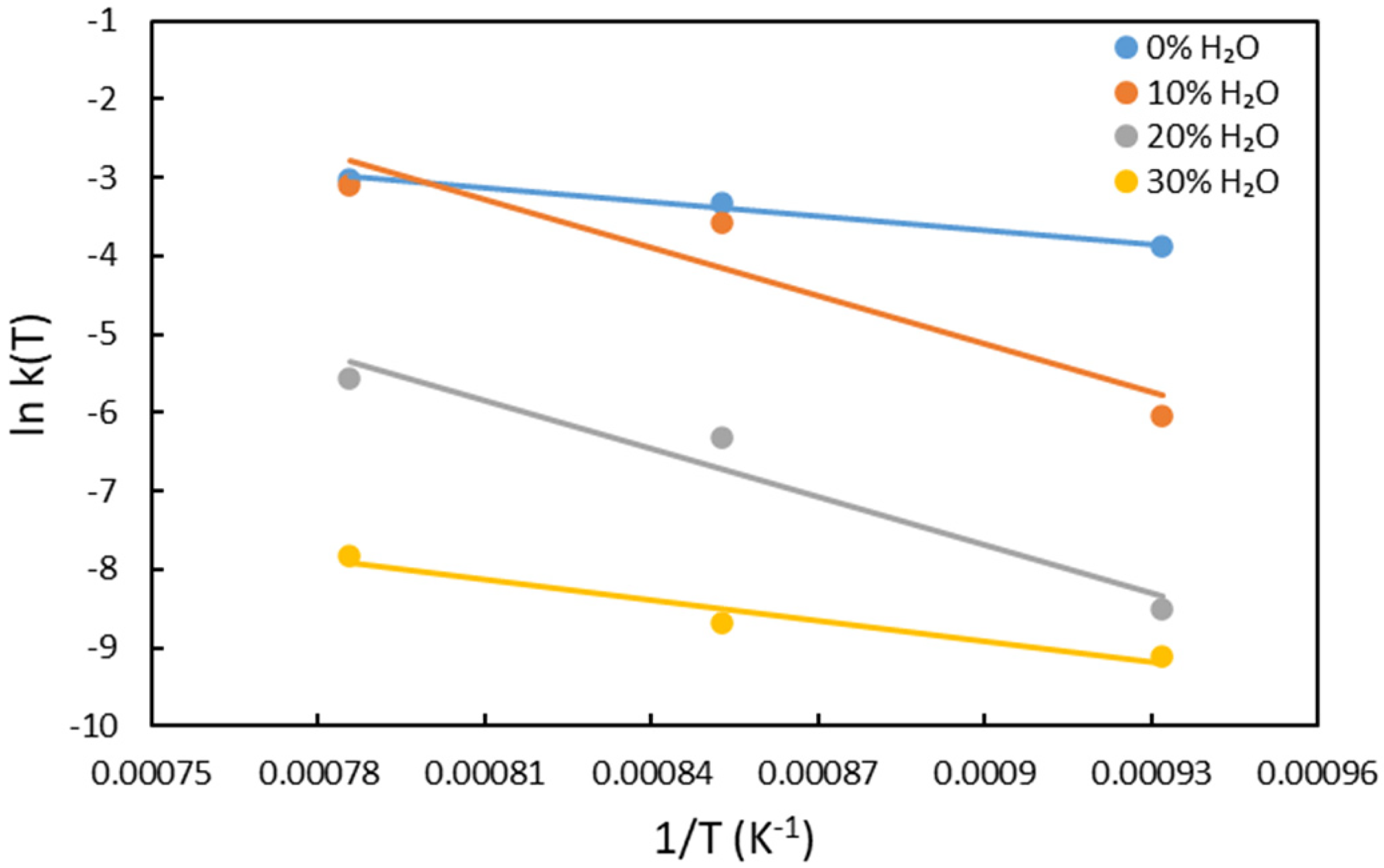
Arrhenius plots for the first stage of reduction.
Relationship between apparent activation energy and probable rate-controlling model.
A similar approach was used to identify the rate-controlling mechanism for the second stage of reduction. The model fitting results are presented in Figure 10. The 3D diffusion model (D3) showed the best linearity for reduction with pure hydrogen at 800°C and 900°C. This is due to the formation of solid metallic iron on the surface of the pellets, which impedes diffusion. However, at 1000°C, the rate-controlling mechanism shifted to a chemical reaction, as the higher temperature made hydrogen diffusion easier.
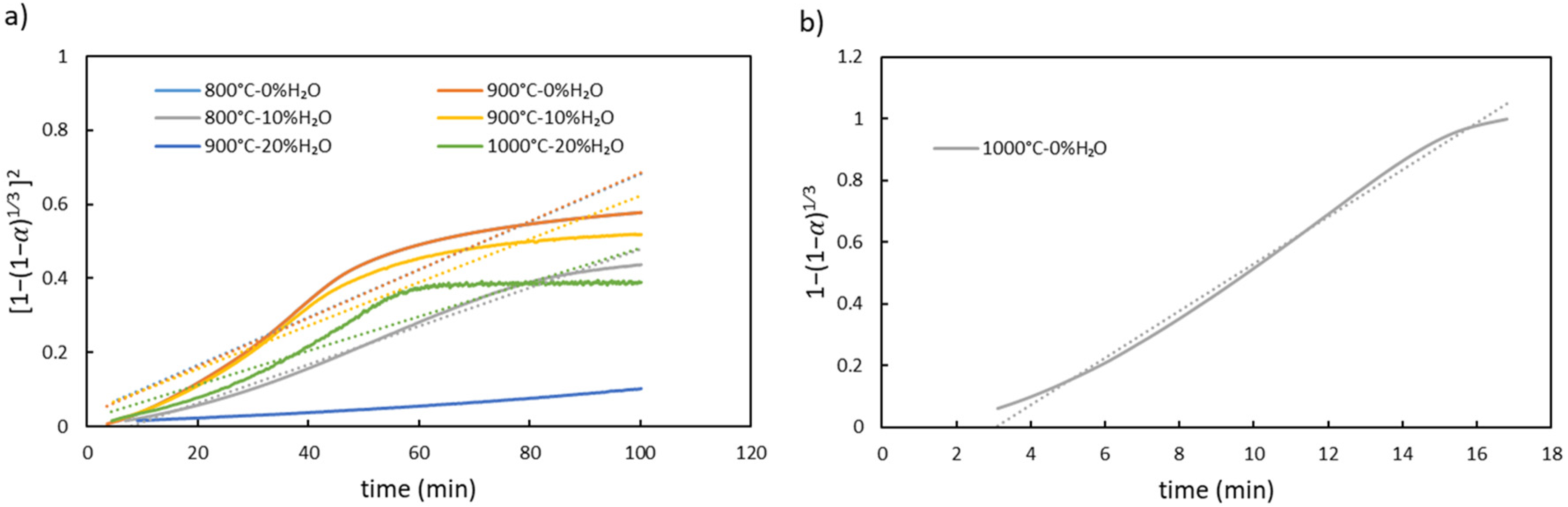
Model fitting for the second stage of reduction (a) 3D diffusion (D3), and (b) 3D chemical reaction (R3).
For reduction with 10% water vapour, diffusion was also the controlling mechanism at 800°C and 900°C, though none of the models from Table 3 fit perfectly for reduction at 1000°C, suggesting that the process is likely mix-controlled by both diffusion and chemical reaction. In the case of reduction with 20% water vapour at 900°C and 1000°C, diffusion remained the controlling mechanism. Although higher temperatures enhance hydrogen diffusion within the pellet, increasing the water vapour content reduces the concentration gradient, hindering diffusion. Therefore, even at 1000°C with 20% water vapour, diffusion continues to be the rate-controlling model.
The Arrhenius plots for the second stage of reduction are displayed in Figure 11. From the slope analysis, the activation energy was determined to be 134.65, 145.89 and 177.40 kJ/mol for reductions with 0%, 10% and 20% water vapour, respectively. This indicates that increasing water vapour content leads to a rise in activation energy. Additionally, when compared to the values in Table 3, the calculated activation energies fall within the range associated with solid-state diffusion, which can be attributed to the formation of a solid metallic iron layer on the pellet surface.
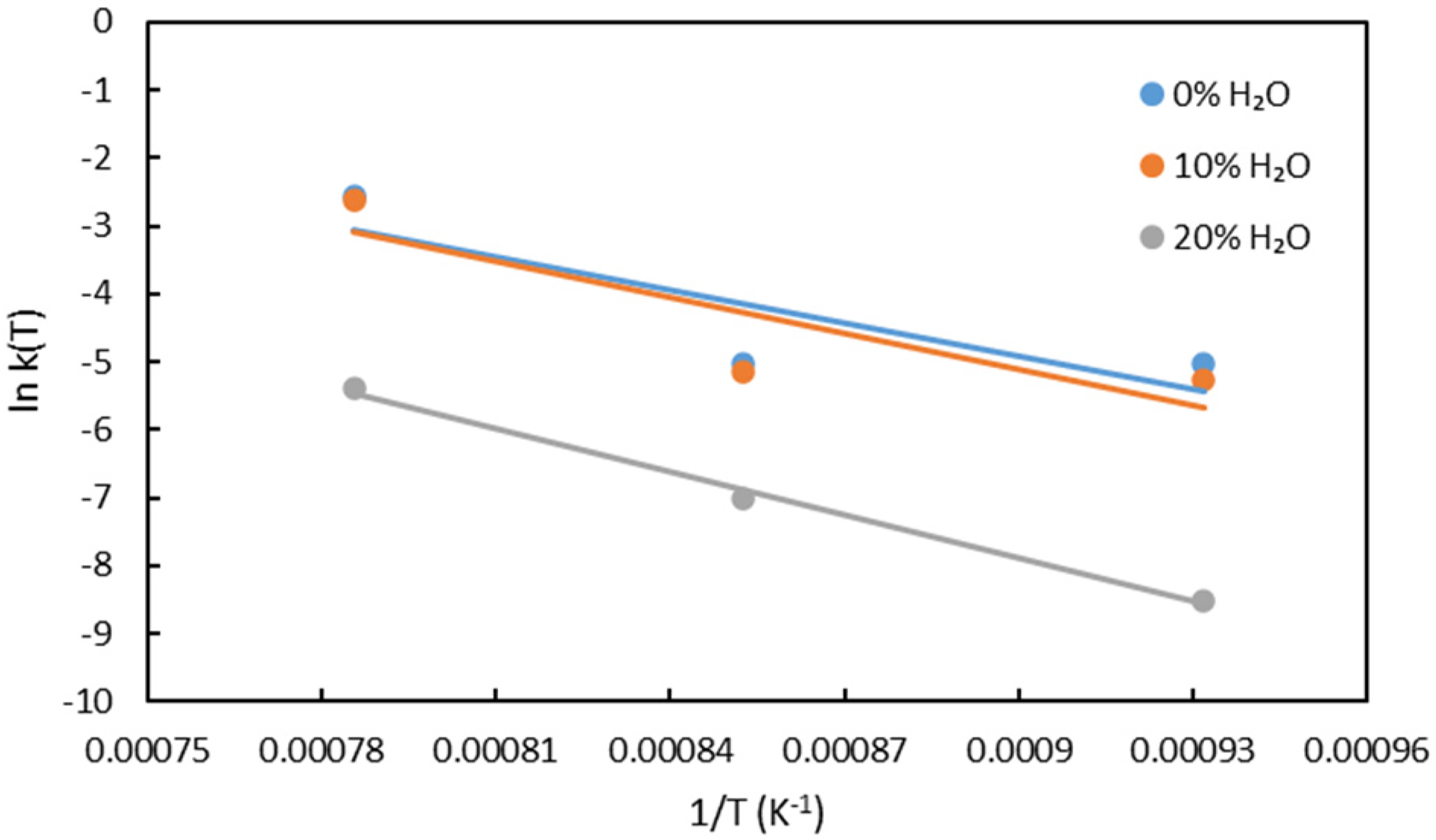
Arrhenius plots for the second stage of reduction.
Conclusion
In this study, we explored the impact of water vapour on the hydrogen reduction of iron ore pellets within the temperature range of 800–1000°C. Our findings reveal that introducing 10% water vapour into the gas mixture does not markedly affect the maximum reduction degree; however, it slows down the rate of reduction. At 800°C, the presence of 20% water vapour prevents the formation of metallic iron, while at 900°C and 1000°C, reduction degrees of 77% and 95% are achieved, respectively, with the same gas composition. Notably, with the addition of 30% water vapour, the reduction of wüstite to metallic iron fails to commence at all temperatures, and only reduction to wüstite is completed. Furthermore, as temperature increases, there is a notable rise in the formation and expansion of pores and microcracks, whereas augmenting water vapour content in the gas mixture results in decreasing porosity of the reduced pellets.
The primary factor contributing to weak diffusion and, consequently, poor reduction performance with increased water vapour in the system is the decrease in the concentration gradients of both hydrogen and water vapour. Additionally, higher water vapour levels impact chemical reactions by obstructing the active sites on wüstite, thereby inhibiting its reduction to metallic iron. These observations align with the kinetics modelling results, which indicate that the rate-controlling mechanism is a combination of diffusion and chemical reaction.
The interplay between temperature and water vapour content critically influences the efficiency of hydrogen-based direct reduction of iron oxide, making it essential for optimising the process in industrial settings. However, the interaction of water vapour with other parameters such as gas flow rate, pressure and mineralogy of iron oxide is still unclear and needs more study in the future.
Footnotes
Acknowledgements
This research was partially conducted with the assistance of the Centre for Materials Analysis at the University of Oulu, Finland. We would like to extend special thanks to the Sustainable Hydrogen – Potential for Bothnia Gulf Cluster project (20357962), funded by the European Regional Development Fund (ERDF). We also appreciate the support provided by grant number 5534/31/2023 from FFS2, Towards Fossil-Free Steel 2. Additionally, this work received funding from the Finnish Foundation for Technology Promotion (Tekniikan Edistämissäätiö). The authors are sincerely grateful for the financial support, which played a crucial role in enabling this research.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Tekniikan Edistämissäätiö, Interreg, Business Finland (grant number 10381, 20357962, 5534/31/2023).
