Abstract
At present, iron and steelmaking industry worldwide is going through the transition of decarbonisation to meet its goal of reaching net zero by 2050. In addition, Australian iron and steelmaking industry is facing its own challenge of processing lower grade ores with increasing gangue content. Two major pathways are direct reduction of iron – electric arc furnace pathway (DRI-EAF) and direct reduction of iron – electric smelter-BOF (DRI-electric smelter-BOF) pathway. In the present work, a mass and energy balance model of basic oxygen furnace (BOF) and electric arc furnace (EAF) have been developed using thermodynamic software. The EAF model showed that with 100 wt-% cold DRI, the specific electric energy requirement varied between 514 and 651 kWh/tls whereas in case of hot DRI, it varied between 399 kWh/tls and 510 kWh/tls. As the gangue content increased from 10.7 wt-% to 19.1 wt-%, yield decreased from 88 wt-% to 75.5 wt-% and slag weight increased from about 200 kg/tls to 630 kg/tls. The BOF model showed that the slag produced in a BOF varied between 63 kg/tls and 73 kg/tls for lower to higher grade ores reflecting different hot metal chemistry (P, Mn) coming from different ores. The results indicated that electric smelter-BOF is more compatible to process lower grade ores than EAF where quantity of slag and loss of yield are very high with increasing gangue content. CO2 emissions from H2DRI-EAF and H2DRI-electric smelter-BOF pathway for different types of ores increased with the increasing gangue content. For H2DRI-EAF pathway, as gangue content increases from 10.7 wt-% to 19.1 wt-%, CO2 emission rises from 0.10 t/tls to 0.19 t/tls as more limestone is needed to remove the gangue which also increases the production of CO2. In case of H2DRI-electric smelter-BOF pathway, CO2 emission increases from 0.12 t/tls to 0.19 t/tls with increasing gangue content. CO2 emissions from both pathways are significantly lower than the current BF-BOF pathway.
Keywords
Introduction
In recent times, the Australian iron and steelmaking industry is facing challenges due to the declining ore grade and increase in gangue content. At the same time, the industry is also charting its way for low carbon emissions to reach its net zero goal in 2050. The typical major routes for ironmaking and steelmaking are
2
Blast furnace (BF) – basic oxygen furnace (BOF) pathway – In this pathway, molten metal is produced in BF using iron ore, coke, and limestone. Then the molten metal is converted to liquid steel in a BOF. Direct reduced iron (DRI)/scrap – electric arc furnace (EAF) pathway – In this pathway, direction reduction process is used to produced DRI from iron ore. DRI and scrap are used in an EAF to produce liquid steel. Smelter-EAF route – Smelter is used to produce hot metal. Hot metal and scrap are used in an EAF to produce liquid steel.
For two distinct grades of iron ore, Wimmer et al.
1
has proposed two pathways for the future as shown in Figure 1. Lower grade ore can be processed using DRI-smelter-BOF pathway whereas higher grade ore can be processed via DRI-EAF pathway. When H2 is used as the reducing gas, the pathways are called as H2DRI-smelter-BOF pathway and H2DRI-EAF pathway.
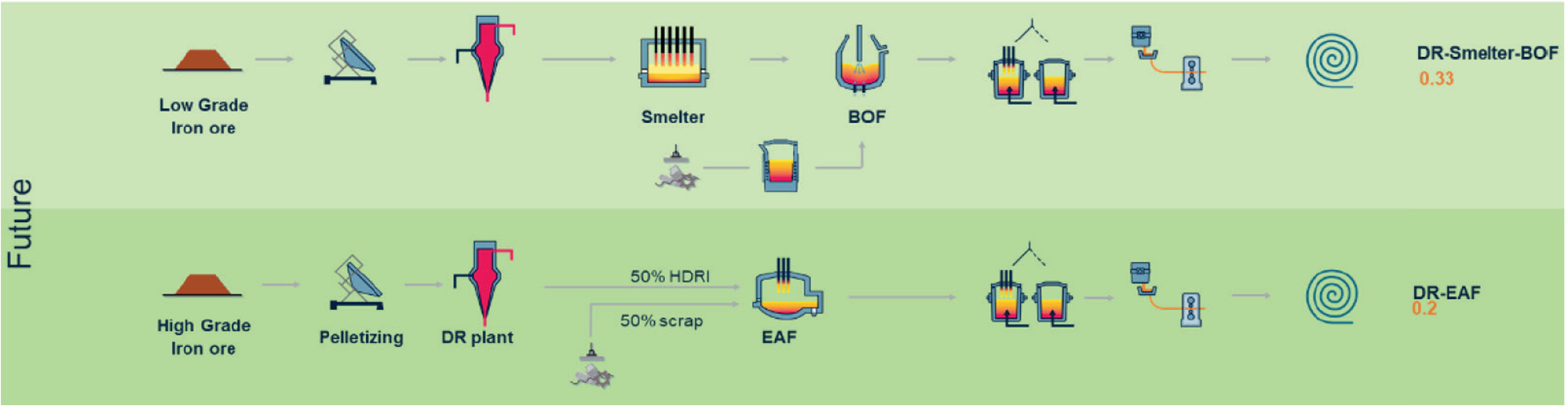
Two pathways of ironmaking and steelmaking process for two types of iron ore. 1
As shown in Figure 2, for all routes, for lower grade ores, the transformation cost (i.e. mainly costs of electric power, fluxes and gases) is higher, but the DRI cost is lower. Wimmer et al. 1 argued that for processing lower grade ores (i.e. Fe < 67 wt-%), the DRI-electric smelter-BOF pathway can be an economically feasible route and can also reduce carbon emission whereas DRI-EAF pathway is more appropriate for higher grade ores (i.e. Fe > 67 wt-%).
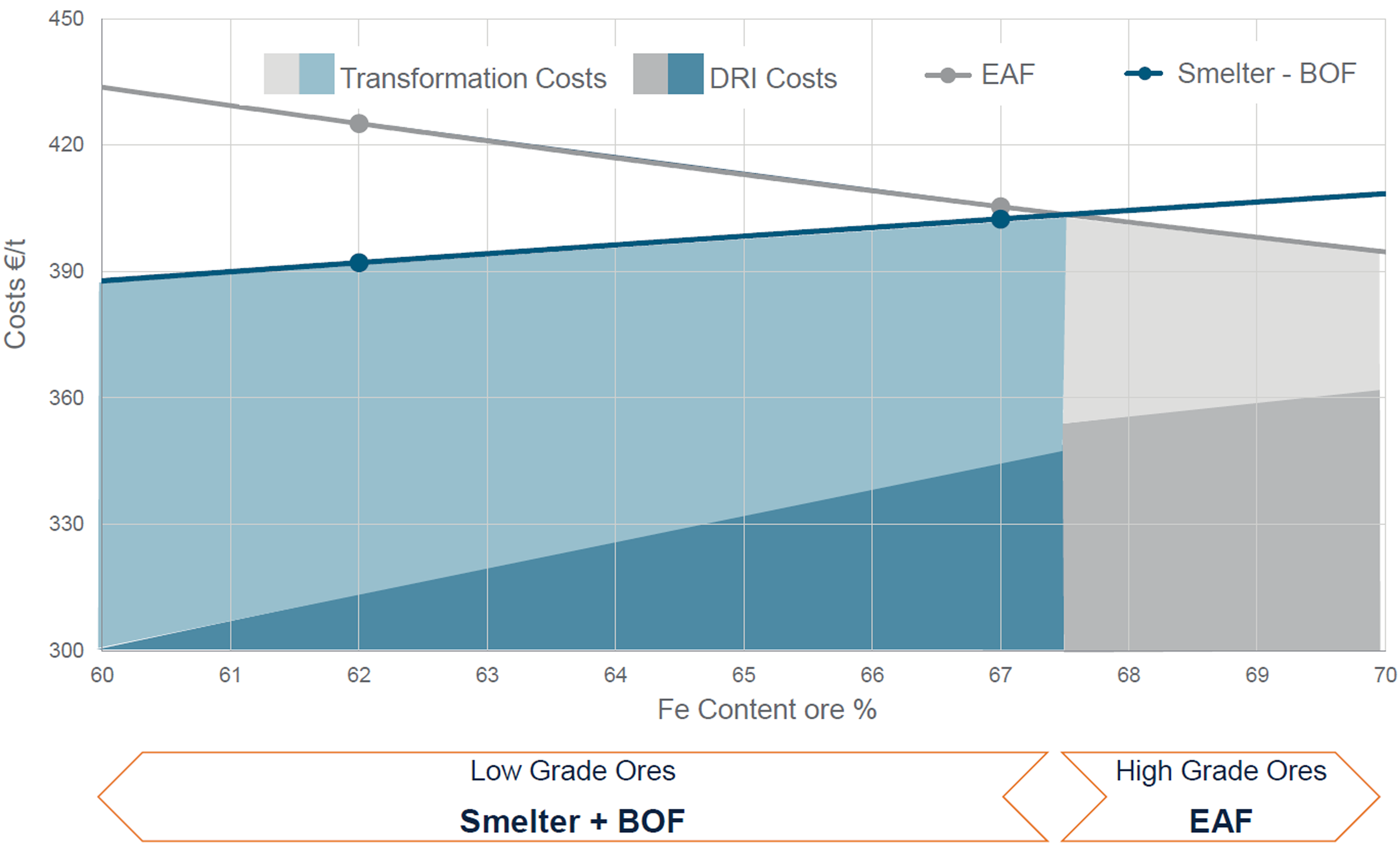
Transformation costs associated with future steelmaking pathways. 1
In this paper, a steady state mass and energy balance models were developed for BOF and EAF using different grade of iron ores as input. The models were developed in the ‘Flowsheet simulation’ module coupled with the ‘Equilibrium’ module in HSC Chemistry 10.0 which is widely used to analyse complex metallurgical processes.2,3 It takes a simplified approach to understand metallurgical reactions using chemical thermodynamics of mass and energy balance and does not consider the reaction kinetics of the process. In the input, all species with their specific phase, amount, temperature and pressure were specified. In the output, all likely species with their specific phase, temperature and pressure were specified. The model calculated the output at equilibrium condition using Gibbs free energy minimisation method. 3 HSC Chemistry only allows relatively simple models of activity to be included for solution behaviour (i.e. fixed activity coefficients), which means it is not intended to capture the detailed thermodynamics of complex reactions in steelmaking, such as the de-phosphorisation reaction where the actual behaviour is complex, and effect of temperature and the concentration of different species must be considered. This work will provide the overall general behaviour of the system with different iron ore grade inputs.
EAF steelmaking
The EAF furnace applies electrical energy through the graphite electrodes to produce steel from a mixture of scrap and DRI. Depending on the practice, hot briquetted iron (HBI), hot metal (HM) and solid pig iron (PI) are also put into the furnace. Lime (CaO) and Magnesia (MgO) are typically added as the fluxing materials. 4 A typical mass balance of an EAF is shown in Figure 3 which is based on 1 tonne of steel production from the EAF processes.
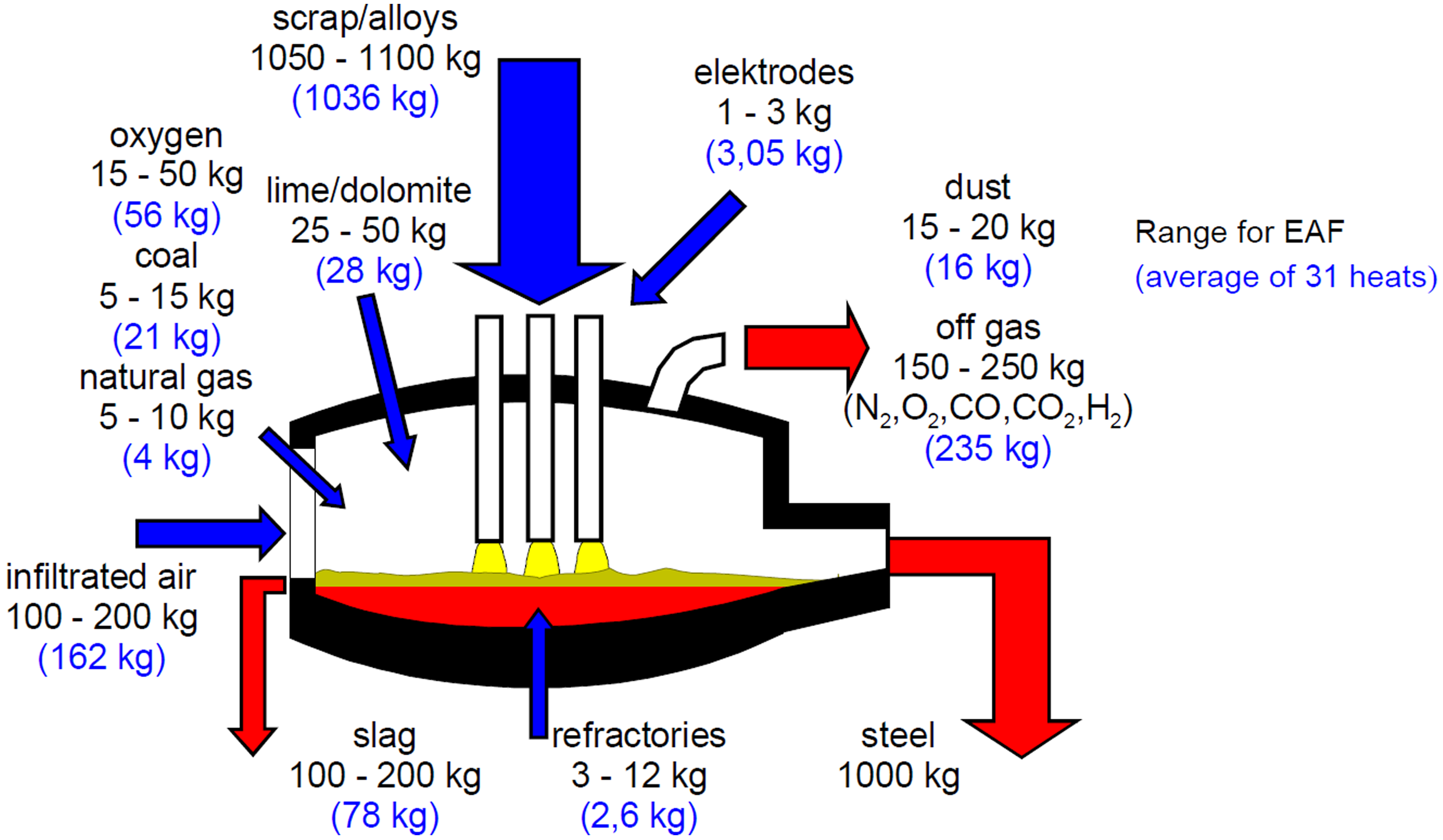
Typical mass balance of an EAF. 5
Carbon is an important element for producing steel. In an EAF, there is carbon coming from the DRI, HBI or any other ore-based metallics (in-situ C). Carbon is also injected into the furnace in the form of coke as well as coal depending on the requirement of the process. In-situ C has higher combustion efficiency (>95%) compared to injected C. After reducing FeO in the slag, the excess C generates CO which helps in providing additional energy, stirring of the bath, and forming foamy slag. Foamy slag can cover the electrodes, hence, improves the thermal efficiency of the process, and reduces the specific energy consumption (SEC).6,7 The primary source of energy in an EAF is electricity, corresponding to 56%. The remaining energy input is derived from various chemical processes, including the oxidation of carbon (23%), the use of burner natural gas (5%), metal oxidation (8%), the release of volatiles from scrap (7%) and electrode consumption (2%). 7
Description of the model
In the present EAF model, DRI and/or scrap, CaO, MgO, oxygen, infiltrated air and CH4 were taken as inputs (as shown in Figure 4). In the output side, molten metal, slag, and off gas (consists of CO2, CO, H2O) were specified. The temperature of the DRI was considered for both cold (25 °C) and hot DRI (600 °C) conditions. The pressure was taken as 1 atmosphere. Though there are EAF plants operating with preheated scrap (like the EAF's engineered by Tenova's Consteel® technology or Primetals Quantum EAF), in this model, scrap input temperature was taken as 25 °C for simplification. Four scenarios considered in this investigation were.
100 wt-% scrap 100 wt-% DRI 100 wt-% hot DRI and 50 wt-% scrap, 50 wt-% DRI
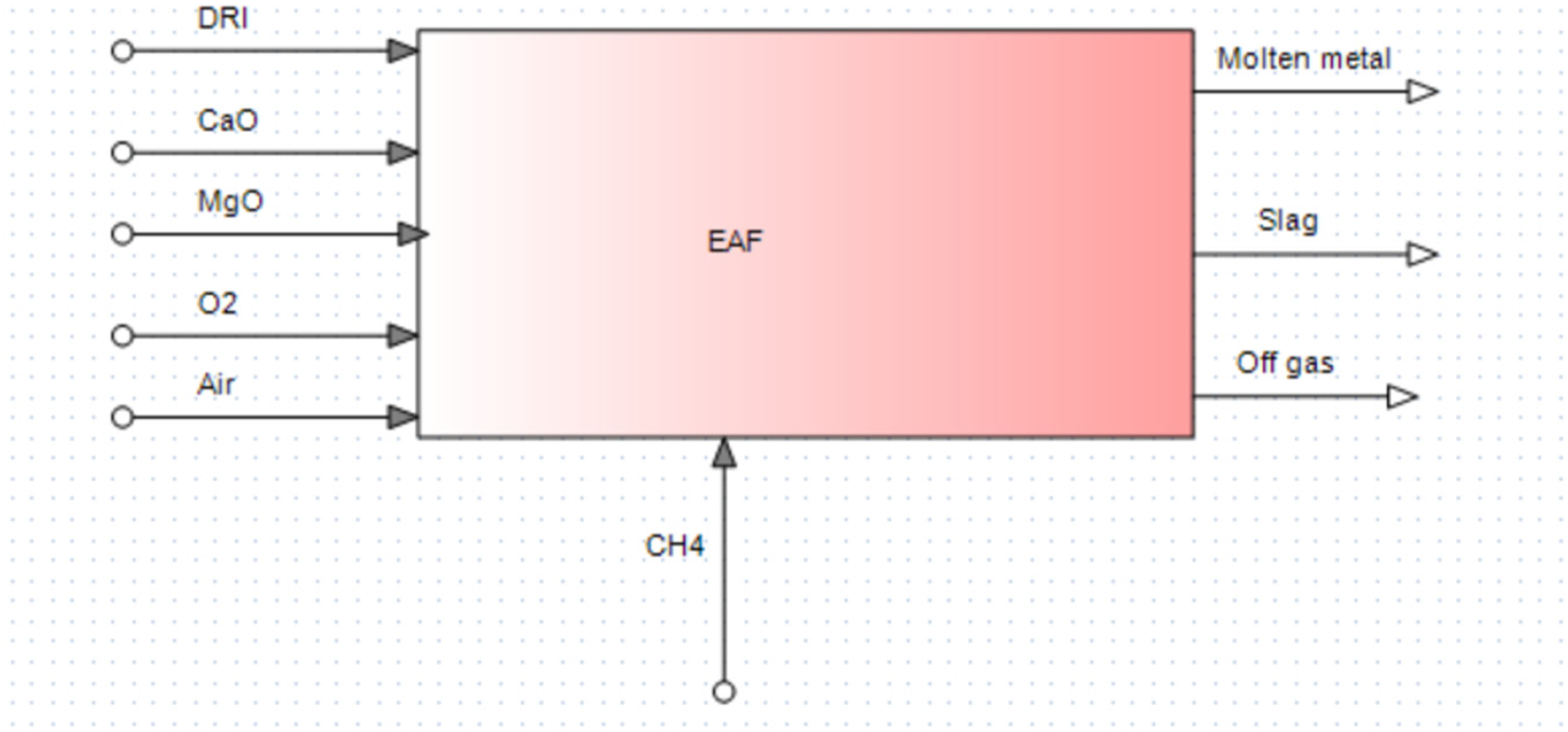
Inputs and outputs of the model (with 100% DRI as input).
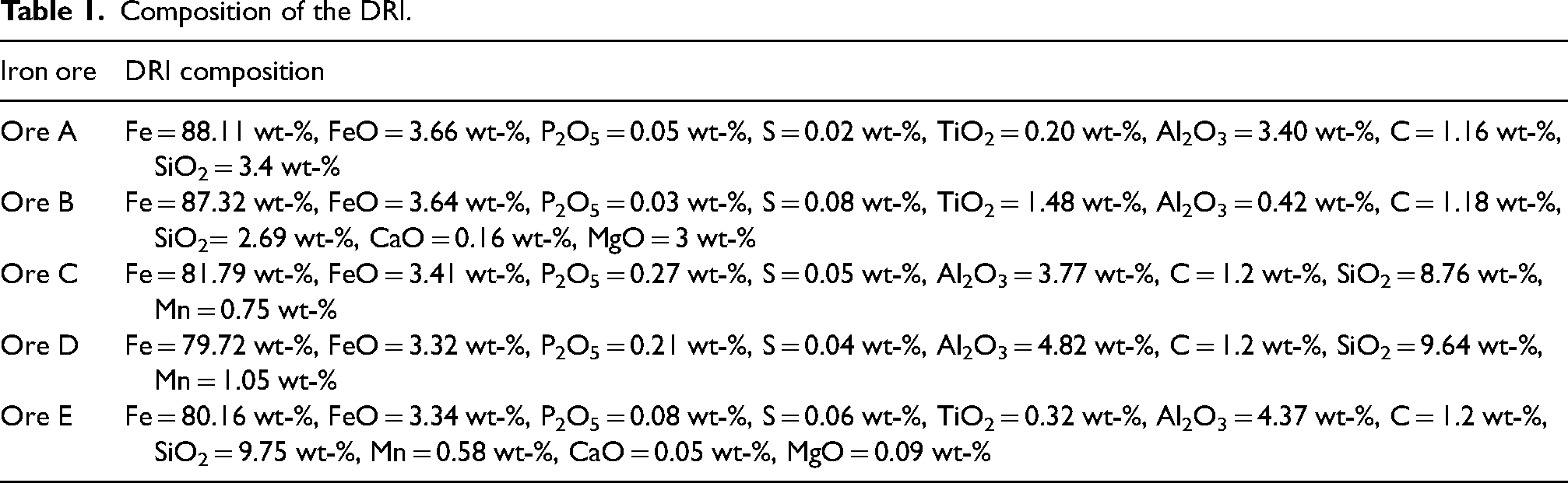
The five ores with different composition were taken as the input to the DRI (as shown in Table 1). Taking a 96 wt-% metallisation rate, amount of Fe and FeO of the DRI were calculated. This calculation assumed the iron oxide is in the form of ‘FeO’, and not a combination of Fe2O3 and Fe3O4 to simplify the calculations. C was assumed to be 1.2 wt-% of the DRI. Then rest of the DRI composition (SiO2, CaO, MgO, P2O5) was taken from the gangue composition of the five ores.
Composition of the DRI.
The amount of infiltrated air was kept at 100 kg/tls. The amount of entrained air in an EAF varies considerably and depends greatly on the furnace configuration and the details of the gas handling system. The 100 kg/tls was used as a reasonable value from the literature. 5 Natural gas (CH4) was varied between 6.4 kg and 6.7 kg for 100 wt-% DRI, 8 kg for 50% DRI and 12 kg for 100 wt-% scrap scenarios. Oxygen was varied from 15 kg to 39 kg depending on the ore type to achieve a specific mass fraction of 20 wt-% FeO in the slag phase. These input values are in the range of what reported by Pfeifer and Kirschen. 5
Table 2 shows the activity coefficients of different components based on the values reported by Kho et al. 3 The activity coefficients of the remaining components were kept at 1. This parameter is a function of temperature and the phase composition in which the component is dissolved. In this work, the following values are considered as an approximation, as the focus of this study is energy balance rather than exact distribution of minor elements.
Activity coefficients. 3

Assumptions
The following assumptions are applied to model the EAF process.
Level of metallisation
Typical metallisation level of a DRI process varies between 92 wt-% and 96 wt-%. 8 In this model, the level of metallisation in the DRI was taken as 96 wt-%.
Carbon content
Typical carbon percentage varies between 1% (HBI) and 4.5% (CDRI). Carbon percentage of the DRI in this model was kept at 1.2 wt-%.
Basicity
Basicity of the slag (B) is the ratio of CaO and SiO2 in the slag. In this model, basicity is kept at a constant value of 2 for all scenarios. This value may need to be set higher in case of DRI with high phosphorus content.
FeO
The amount of FeO was taken as 20 wt-% of the slag in the present model. This amount of FeO was based on the expected level of FeO for an EAF making a steel with around 0.10 wt-% carbon. 9 Lower carbon steels would generate more FeO and vice versa, affecting overall yield and energy but this value was chosen as a reasonable typical value.
MgO
In an EAF process, MgO is added as a fluxing agent and to protect the lining of the vessel from wear. The percentage of MgO in the slag varies between 7 wt-% and 12 wt-%. 9 In this model, the percentage of MgO is assumed to be 7 wt-% of the slag (except ore B where MgO is 18.21 wt-% of the slag).
Slag foaming
It is an important practice in EAF steelmaking. It increases thermal efficiency and slag reaction area while decreasing the tap to tap time and SEC. 7 The model uses 1.2 wt-% C (in-situ C) in the DRI. As the carbon is chemically bound to iron in the DRI, the carbon efficiency is 95% or more compared to carbon injected into the bath where carbon efficiency varies between 25% and 75%. Carbon efficiency implies the percent of potential energy the carbon influences on the system when oxidised. Greater carbon efficiency ensures foamy slag formation in the EAF furnace. 10 Therefore, it is reasonable to assume that some level of slag foaming has been considered into this model as there was 1.2 wt-% C (in-situ C) in the input DRI.
Tap temperature
The tapping temperature was assumed to be 1600 °C which is a reasonable value for EAF steelmaking.
Carbon in molten steel
The percentage of carbon in the molten steel was assumed to be 0.09 wt-% for all scenarios. Lower carbon will result in higher FeO, but we use this as a typical value.
Heat loss
The EAF process is subjected to heat loss mainly from roof, sidewalls, and other miscellaneous ways. The amount of heat loss varies from plants to plants. Heat loss at the rate of 49 kWh/tls is assumed in this research based on the values reported by Hornby and Brooks. 7
Off-gas temperature
Off-gas temperature was taken as 1600 °C.
BOF steelmaking
In a typical BOF furnace, molten PI and scrap is refined into steel of a desired carbon content and temperature using high purity oxygen jet. The aim of this process is to decrease the carbon amount from about 4% to less than 1%, to reduce the amount of manganese, silicon and phosphorus, and to increase the temperature of the liquid steel to approximately 1635 °C. 11 Typically, hot metal, scrap, flux, coolant and refractory are in the input of a BOF whereas liquid steel, flue gas, slag, dust and splash are the output of a BOF.
Description of the model
In the present BOF model (as shown in Figure 5), instead of PI, hot metal from an electric smelter, scrap, CaO, MgO, oxygen and FeSi were taken as inputs. FeSi has been added to increase the Si level of the hot metal from 0.40 wt-% to 0.60 wt-%. 12 In the output side, there were molten steel, slag and off gas (consists of CO2 and CO). Size of the scrap and oxidation of iron to fumes have not been considered.
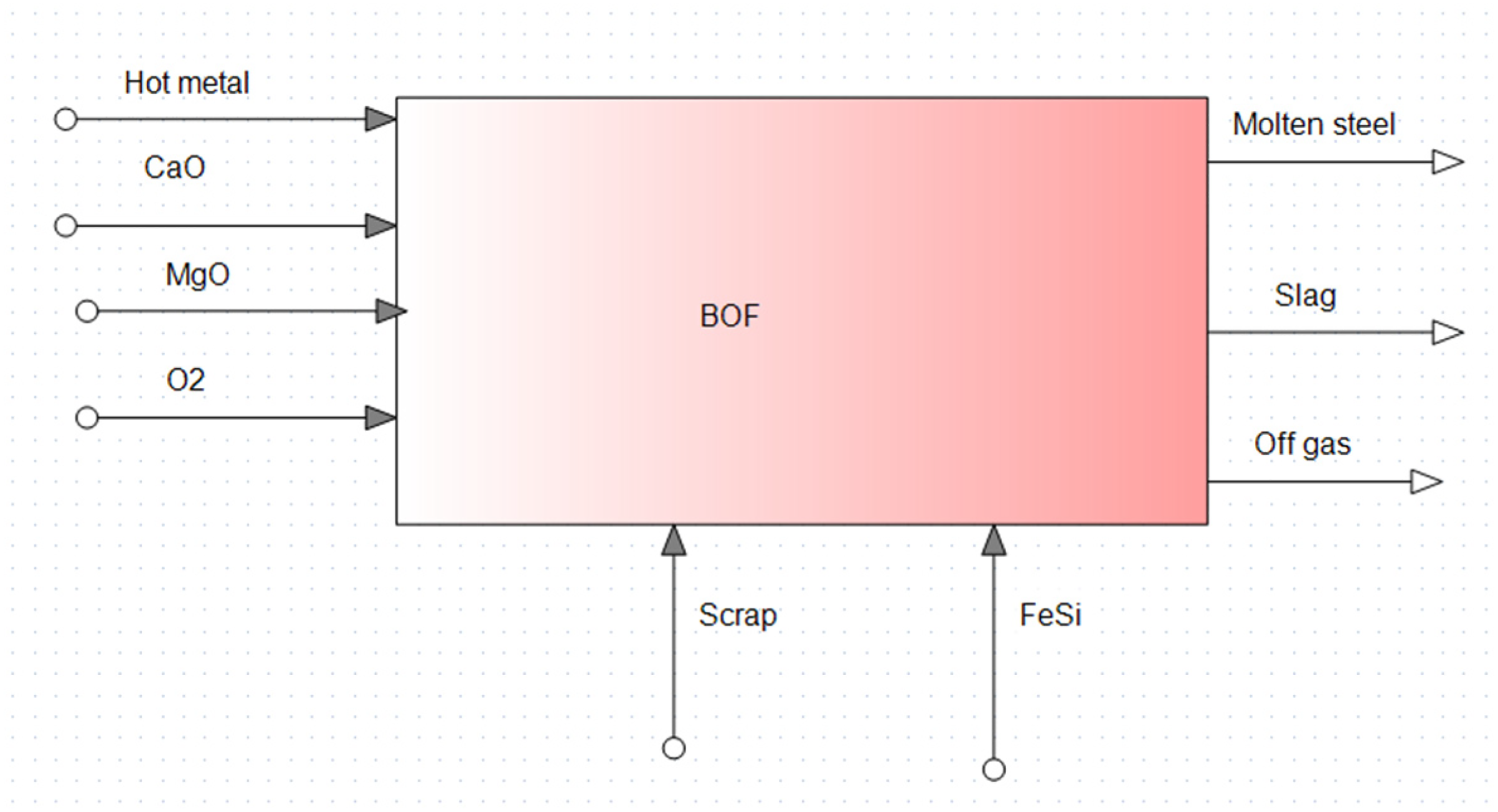
Input and output of the model.
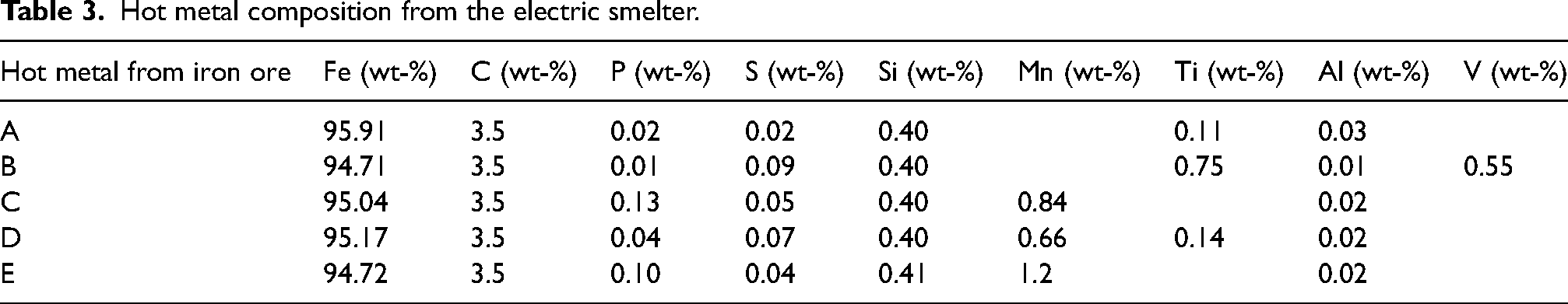
The hot metal charged into the BOF typically composed of 4.3 wt-% to 5 wt-% C, 0.3 wt-% to 1.5 wt-% Si, 0.05 wt-% to 0.2 wt-% P, 0.25 wt-% to 2.2 wt-% Mn and 0.03 wt-% to 0.08 wt-% S. 13 In this model, the hot metal produced from the electric smelter was fed into the BOF. The hot metal composition from the electric smelter for each of the ore is given in Table 3. The description of the electric smelter and its output have been discussed in a separate paper. 14
Hot metal composition from the electric smelter.
Assumptions
The major assumptions of the BOF model are as follows.
Slag basicity
In this model, basicity is kept at a constant value of 3.5. It is consistent with the calculation of Wimmer and Bohn. 15 This is a ‘typical’ lime/silica ratio in BOF steelmaking. This value, of course, varies depending on operating conditions, product requirement and scrap quality particularly the phosphorus value in the hot metal. 16
Carbon
Carbon in the liquid steel has been taken as 0.08 wt-% of the liquid steel. The activity coefficient of C in the steel phase has been adjusted to 0.004 to achieve this amount. In reality, the actual behaviour of carbon is a function of temperature and metal composition. A fixed activity coefficient is used in this model for the sake of simplicity.
MgO
Different plants use different percentage of MgO. Percentage of MgO in the slag varies between 5 wt-% and 20 wt-%.13,17 In this model, percentage of MgO is kept to 7 wt-% of the slag as a reasonable value.
Temperature
Temperature of the hot metal temperature was taken 1400 °C whereas temperature of the liquid steel, off gas and slag was taken 1650 °C. All other temperatures were taken as 25 °C. The pressure was kept at 1 atmosphere for all cases.
FeO
A typical BOF slag has 10 wt-% to 30 wt-% FeO.13,17 FeO was taken as 20 wt-% of the slag in the present model as a reasonable typical value.
Heat loss
It was assumed that 5% of the input energy was lost due to heat loss based on estimations from plant data. 16
Scrap
Typically, 15 wt-% to 25 wt-% of the metallic charge is put as scrap into the BOF. 13 In this model, scrap amount was varied between 25 wt-% and 27 wt-% of the metallic charge. Heat loss of 5% was considered for all scenarios and scrap amount was adjusted to keep it constant. The scrap composition was taken as 100 wt-% Fe.
Results
SEC as a function of gangue content in the EAF process
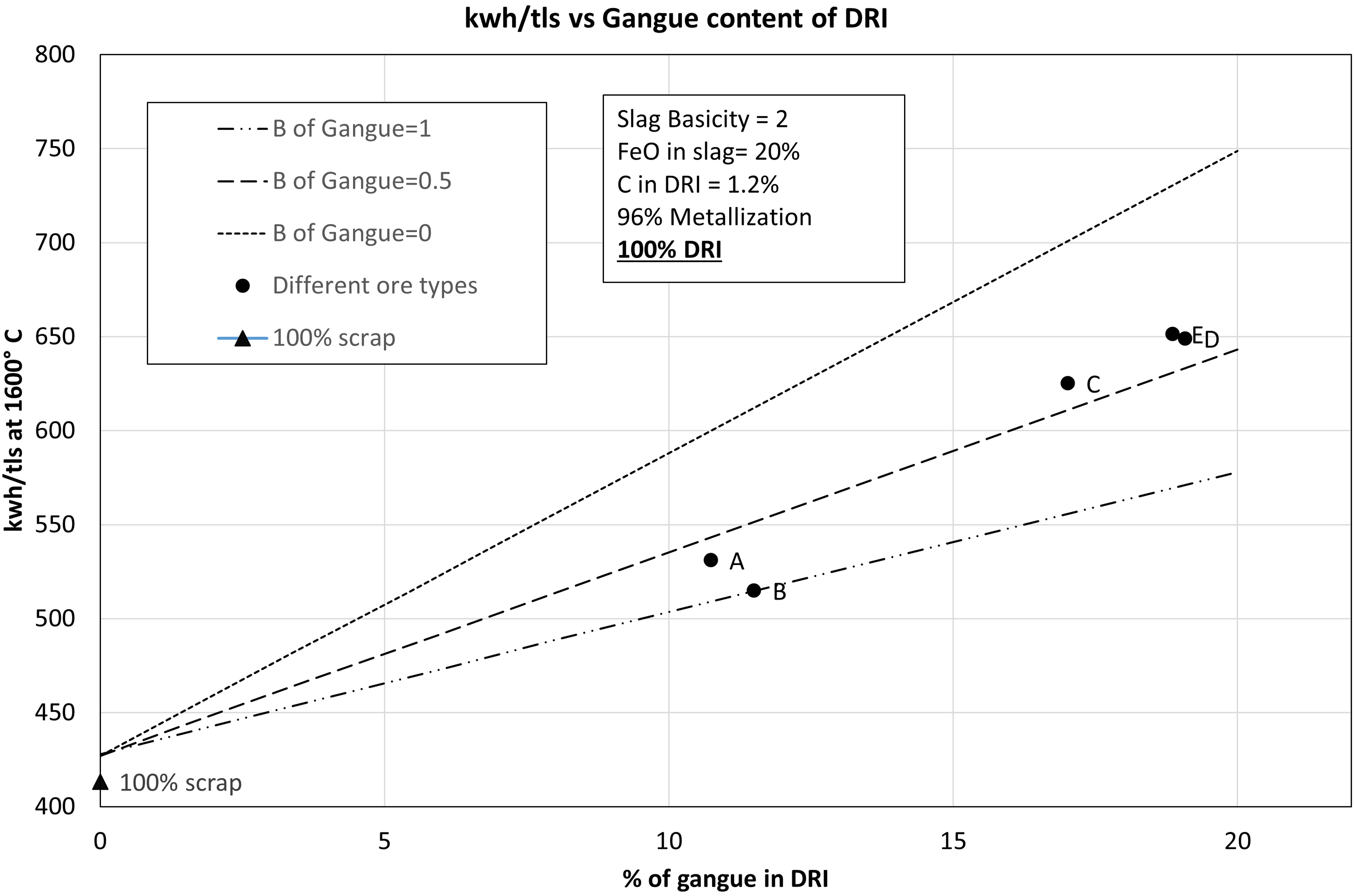
Figure 6 shows the comparison of SEC requirement for cold DRI, hot DRI and 100% scrap. As seen in the figure, the increase in the gangue content is associated with an increase in the SEC of the EAF process. Comparing between hot DRI (600 °C) and cold DRI (25 °C), there was 21% reduction in the SEC when hot DRI was used. When 100 wt-% scrap was used, SEC in the EAF was found to be 413.31 kWh/tls which is comparable to the value of 391 kWh/tls reported by Hornby and Brooks. 7 Figure 6 shows that SEC in case of cold DRI varied between 514 kWh/tls and 651 kWh/tls whereas in case of hot DRI, it varied between 399 kWh/tls and 510 kWh/tls with an increase of gangue from 10.74 wt-% to 19.08 wt-%. Overall, it shows that using hot DRI is profitable in terms of saving costs of electric energy in an EAF. For lower grade ores (C, D, E), the hot DRI was found to save about 142 kWh/tls of electric energy requirement compared to 160 kWh/tls reported by Hornby et al. 10
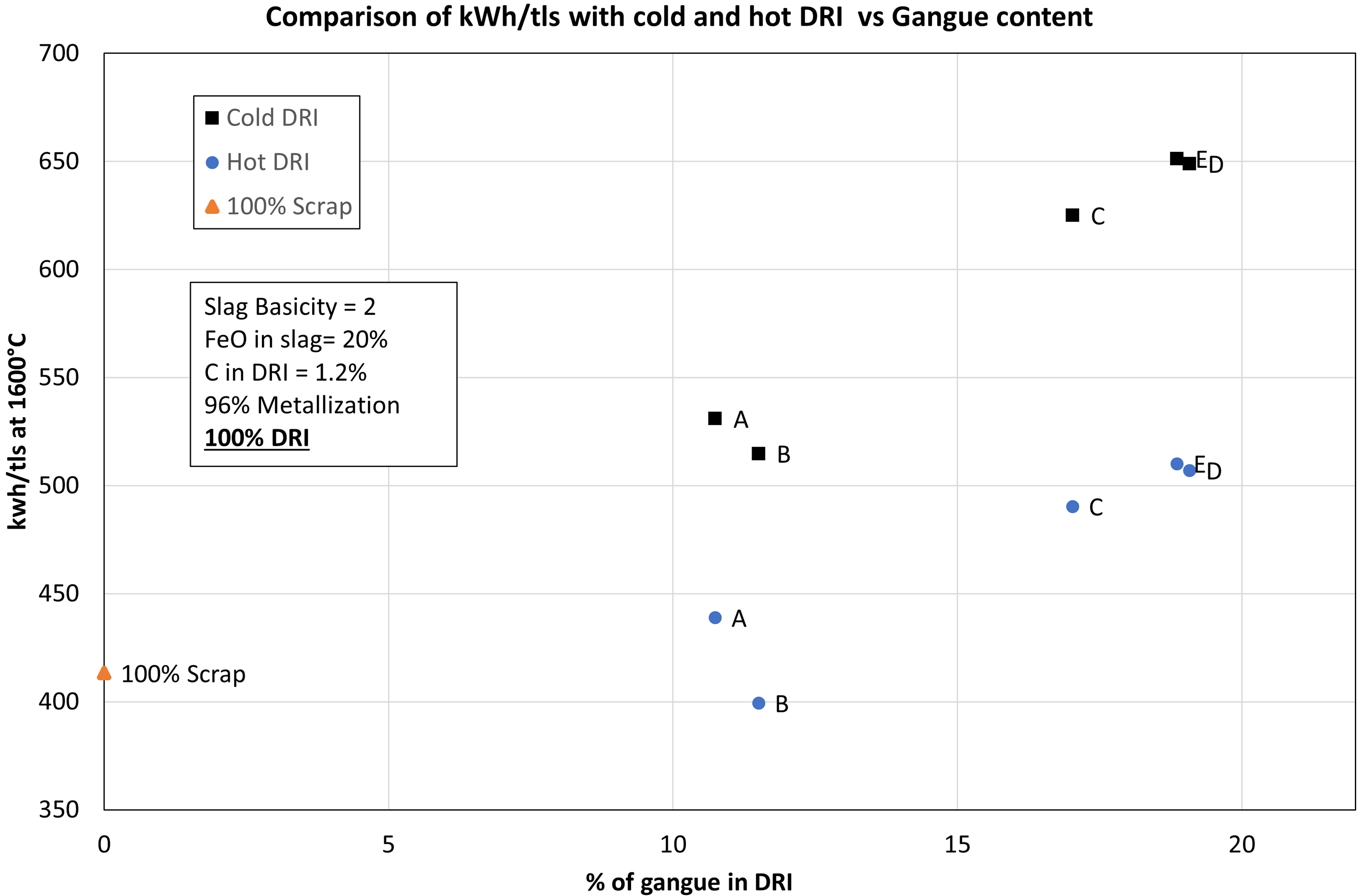
SEC as a function of gangue content for cold DRI, hot DRI, and scrap supply into EAF.
When a combination of 50 wt-% scrap and 50 wt-% cold DRI was used, SEC varied between 465 kWh/tls and 520 kWh/tls (as shown in Figure 7) for different types of ores. These values were still higher than that of 100 wt-% hot DRI.
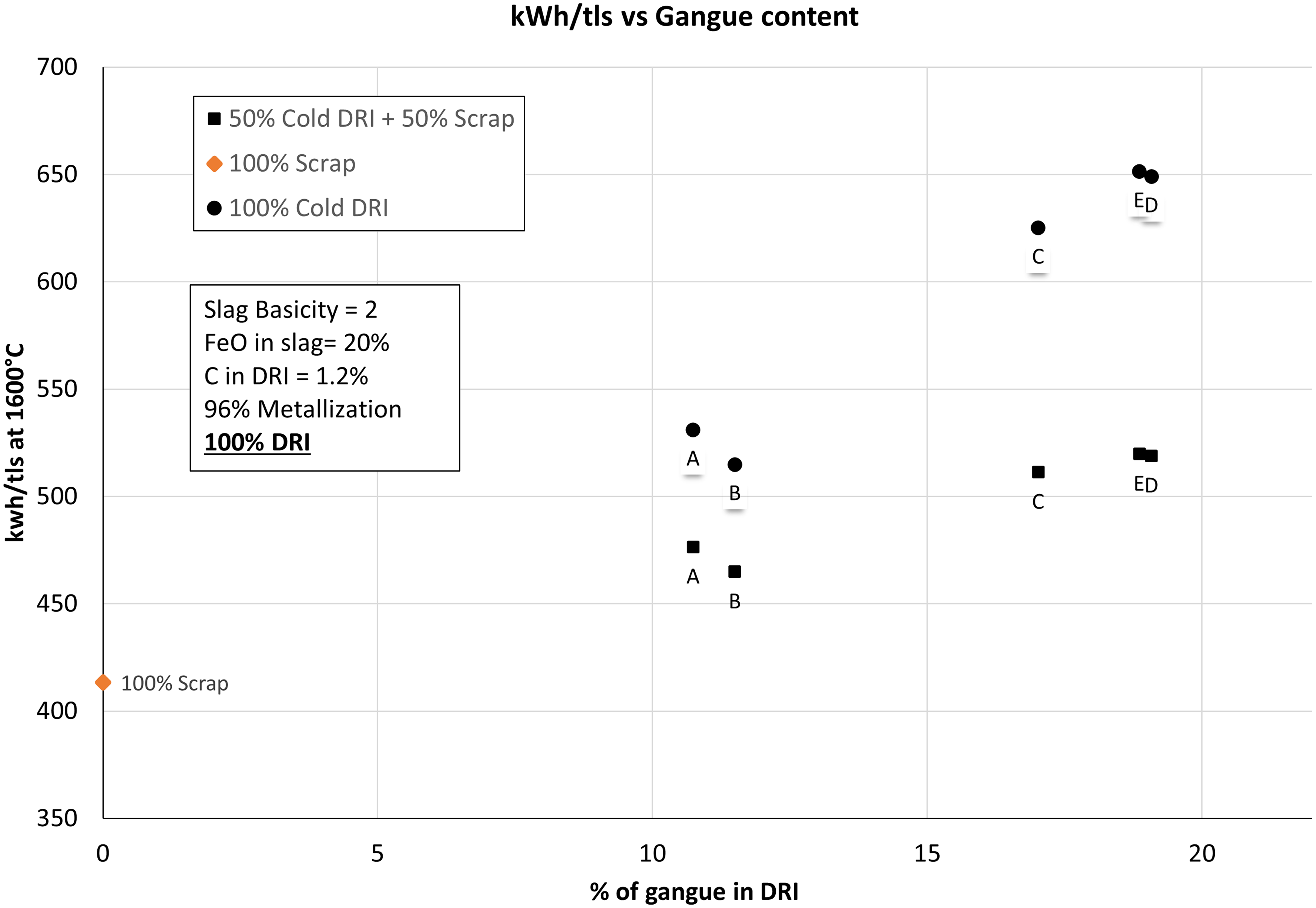
Comparison of SEC with 100 wt-% cold DRI and 50 wt-% cold DRI vs gangue content.
Yield as a function of gangue content in the EAF process
The yield (wt-%) of the EAF process decreased as the gangue content in the DRI increased. Less molten metal was removed as the lower grade ores were refined in an EAF. Figure 8 shows that increasing the gangue content from 10.74 wt-% to 19.08 wt-%, decreased the yield from 88 wt-% to 75.5 wt-%. This represents a significant loss of material and reflects the very high amounts of slag generated from high gangue ores.
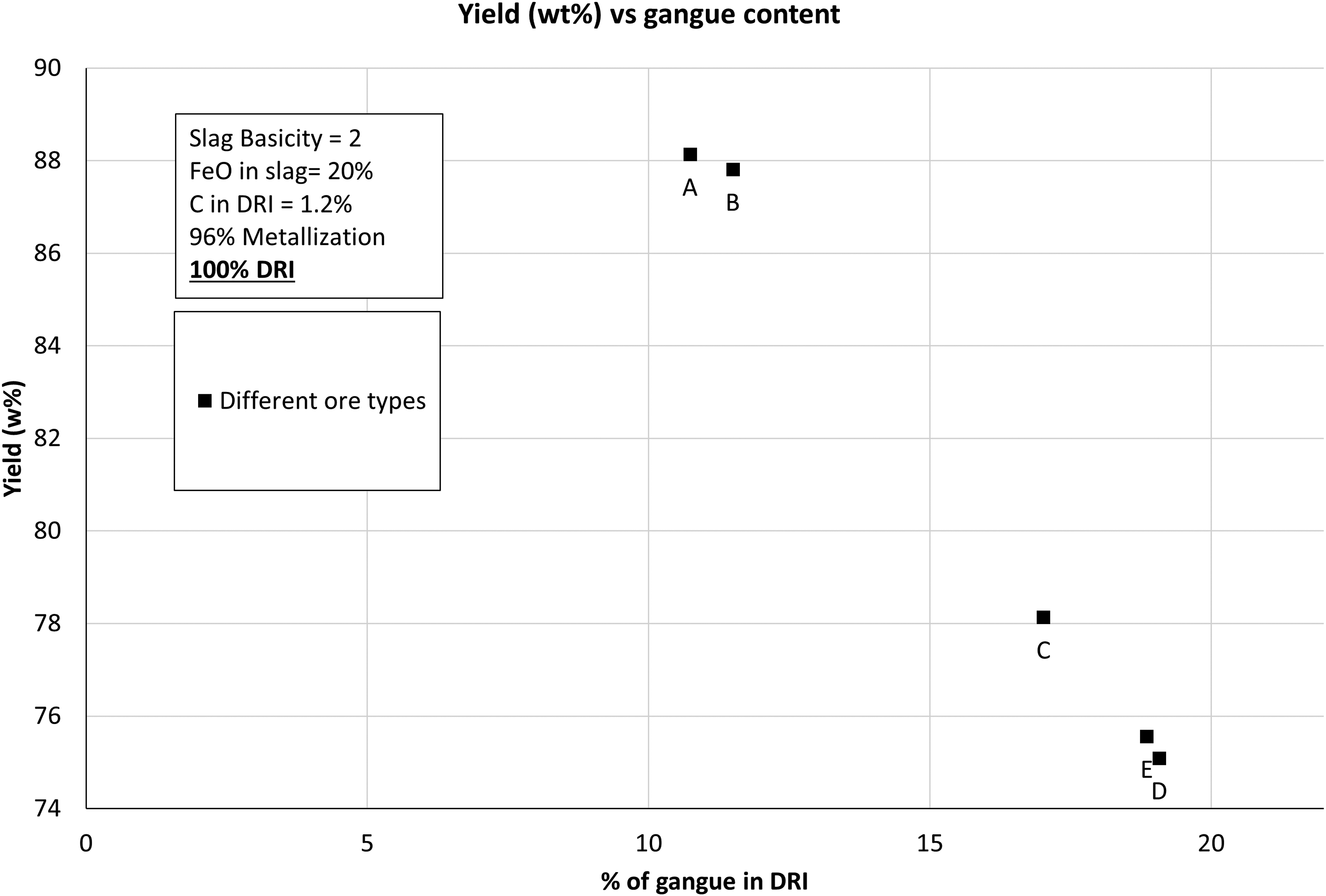
Yield (wt-%) as a function of gangue content.
A comparison of yield versus wt-% of gangue at different wt-% of FeO is presented in Figure 9. The empirical relationship by Turkdogan 9 has been used to relate FeO wt-% in terms of C. As a general trend, it was observed that a lower FeO in the slag corresponding to higher yield. However, for lower grade ores (i.e. C, D, E), an increase in FeO had a more pronounced negative impact on yield compared to higher grade ores (A, B). For higher grade ores, 10 wt-% increase in FeO reduced the yield about 1% to 2%. In case of lower grade ores, 10 wt-% increase in FeO reduced the yield about 4% to 5%. It implies that controlling the FeO in lower grade ores is more important due its significant effect on the yield of the EAF process.
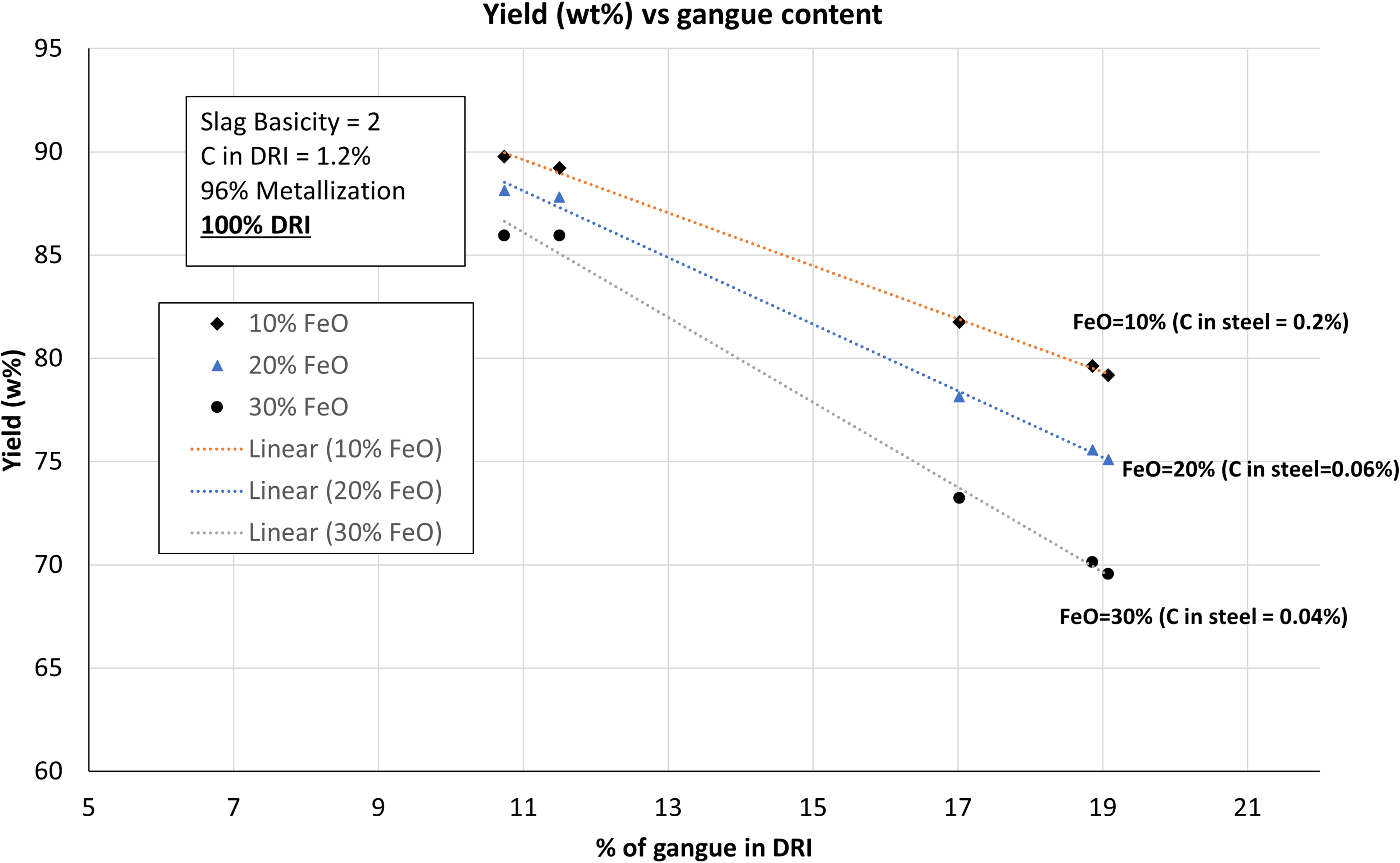
Yield (wt-%) as function of gangue content for DRI with different wt-% of FeO and C.
Slag as a function of gangue content in the EAF process
Not surprisingly, it was found that there was a strong correlation between slag weight and gangue content. As the gangue content of the DRI increased, the weight of slag significantly increased (as shown in Figure 10). For higher grade DRI (A and B), the slag weight was about 200 kg/tls which is compatible with reported value in literature. 5 In case of lower grade DRI obtained from lower grade ores (C, D, E), slag weight was found to be 543 kg/tls to 630 kg/tls which was quite large compared to typical values reported in the literature. Wimmer et al. 15 reported the slag weight to be high as 462 kg for DRI containing lower grade pellets (<67% Fe) considering slag basicity was 1.8. In cases involving lower grade DRI, a significant portion of slag weight was attributed to the presence of SiO2 and CaO. Ores C, D, E exhibited higher silica content which necessitated the use of greater amount of CaO in the input of the process to maintain slag basicity level of 2 in the slag. For this reason, the slag weights of the lower grade ores were significantly higher than that of a higher-grade ore. As previously mentioned, elevated phosphorus level in the ore will require more lime and would alter these calculations. 12
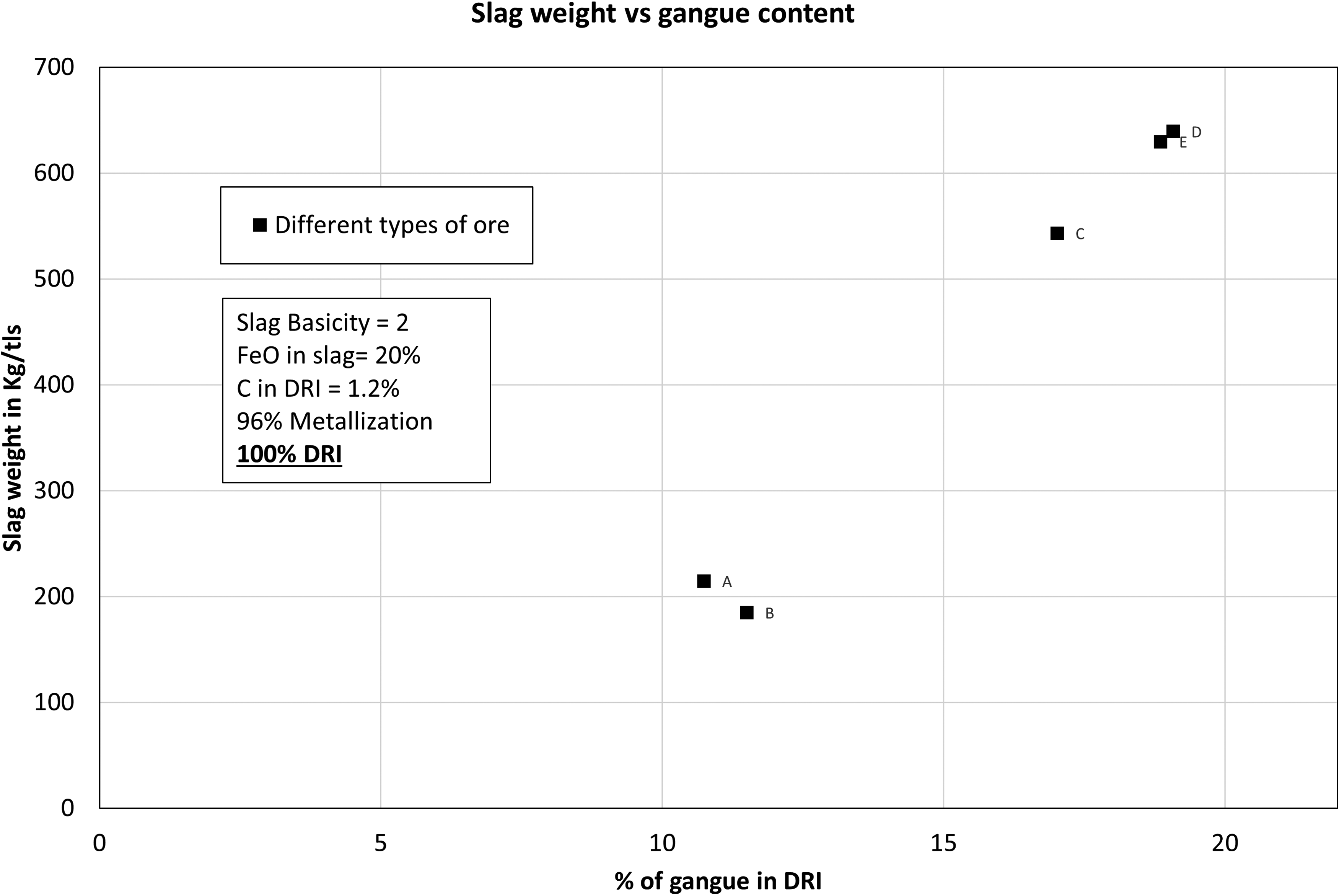
Slag weight as a function of gangue content.
Slag and flux as a function of gangue content in the BOF process
In general, the greater the amount of Fe (wt-%) in the iron ore, the lower the amount of slag produced in the output. The quantity of slag produced varied between 63 kg/tls and 73 kg/tls (as shown in Figure 11), which is similar to as reported by Wimmer et al. 1 Compared to the slag produced in the present EAF model (where slag varied from 200 kg/tls to 630 kg/tls with the grade of the ore), the quantity of slag did not vary a lot with the quality of the iron ore in a BOF, as much of the gangue in ore was removed in the electric smelter.
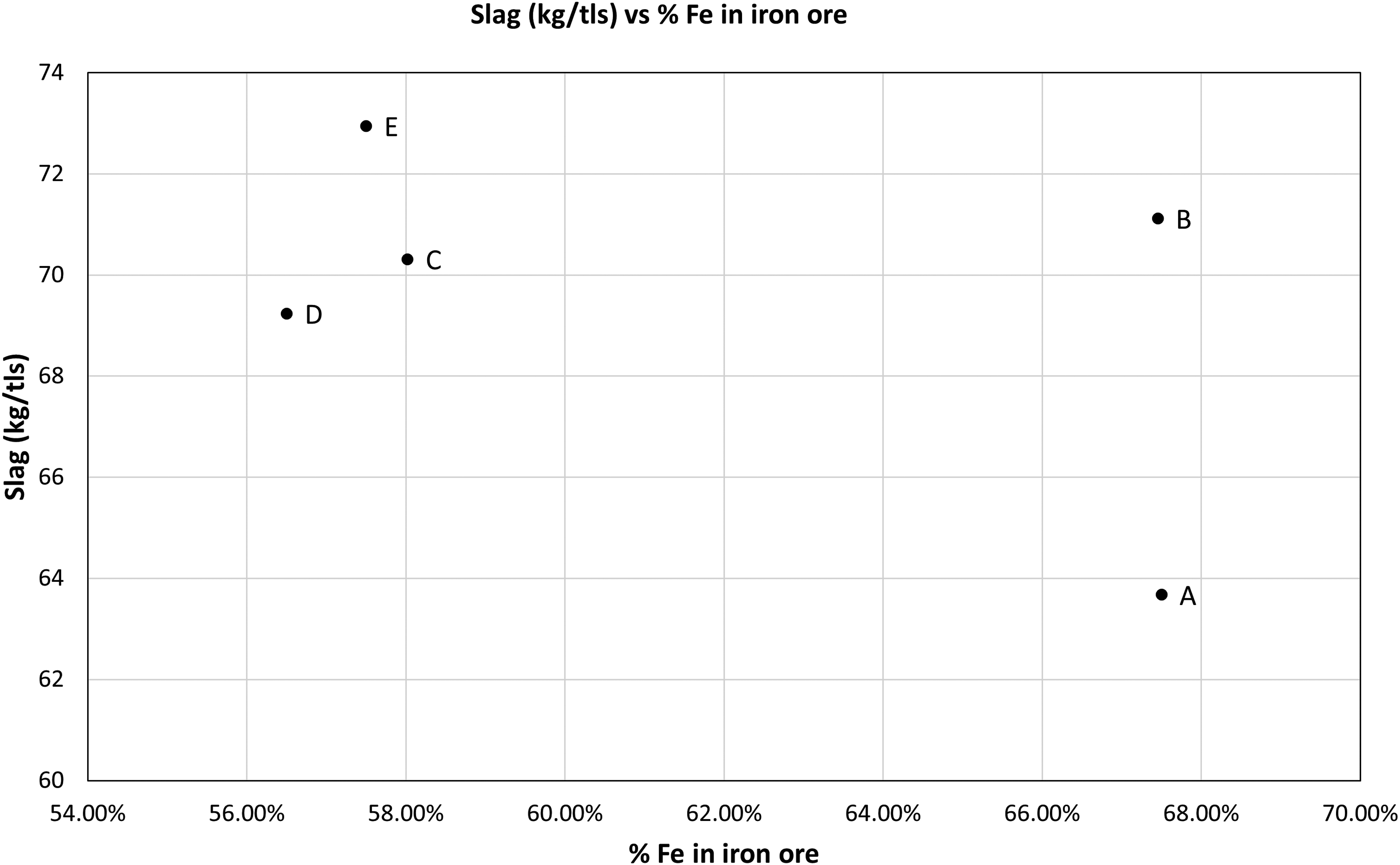
Slag vs % Fe in iron ore.
Amount of flux calculated from the model varied between 38 kg/tls and 40 kg/tls for all type of ores (as shown in Figure 12) whereas flux required in a typical BOF ranges from 13 to 36 kg/tls. The amount of flux did not vary much with the grades of the different ores.
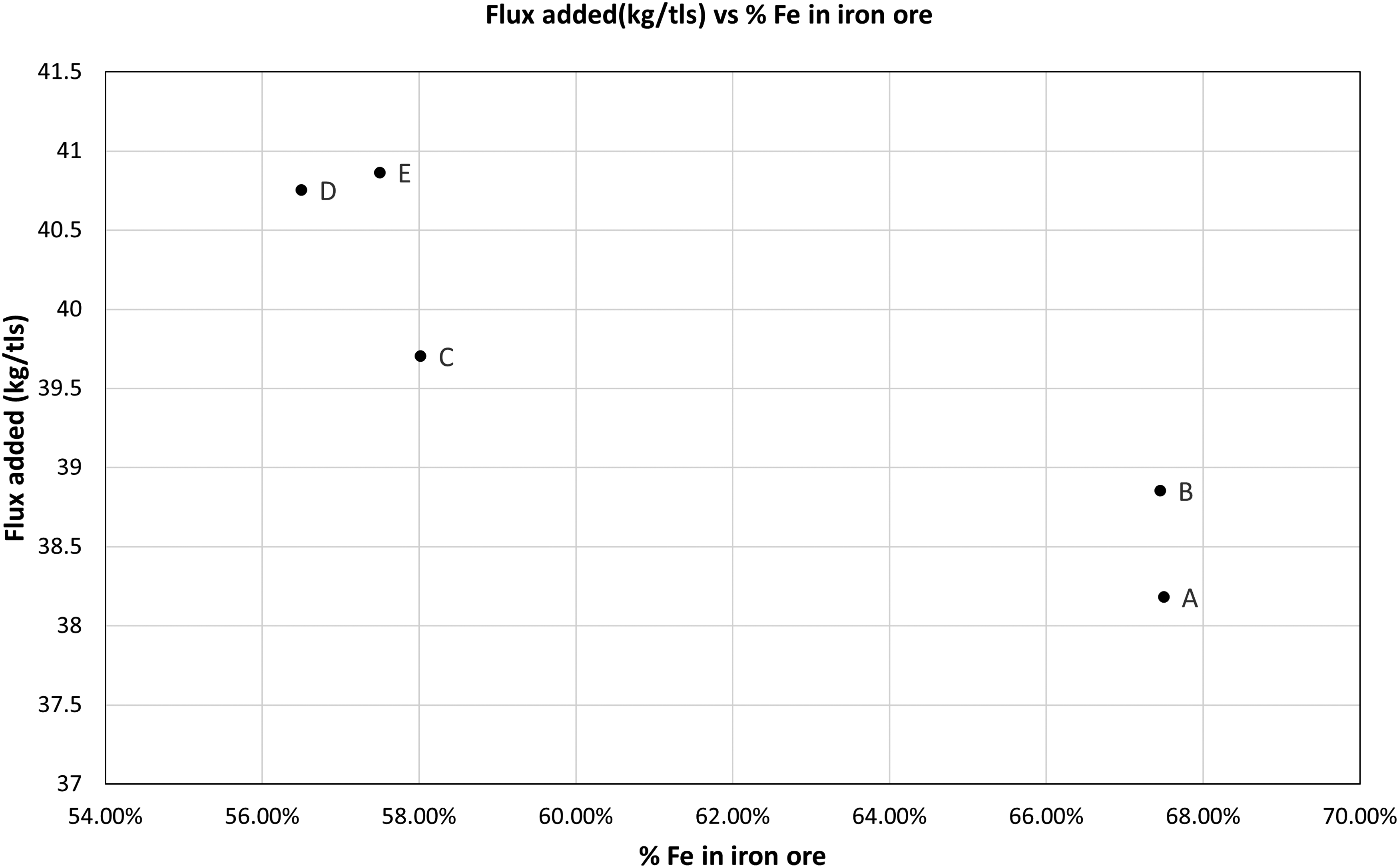
Flux added vs % Fe in iron ore.
CO2 emission from two pathways
CO2 emission from the both pathways – H2DRI-EAF and H2DRI-electric smelter-BOF were calculated from the models and compared with the data available in literature. The following assumptions are considered in the estimation of CO2 emissions:
it is based on direct CO2 emissions from the process as per scope I,
18
CO2 emissions during CaO production is taken into account, electricity requirement for the EAF process is generated from renewable energy resources, CO2 emissions from MgO production or any other upstream mineral processing are neglected.
CO2 emission from H2DRI-EAF pathway for 100% scrap, 100% DRI – higher grade ores and lower grade ores compared in Figure 13. These values are comparable to the value reported in literature.1,19 In general, there is an increase in the CO2 emission with the increasing gangue. As gangue content increases from 10.7 wt-% to 19.1 wt-%, CO2 emission rises from 0.10 t/tls to 0.19 t/tls as more limestone is needed to remove the gangue which also increases the production of CO2.
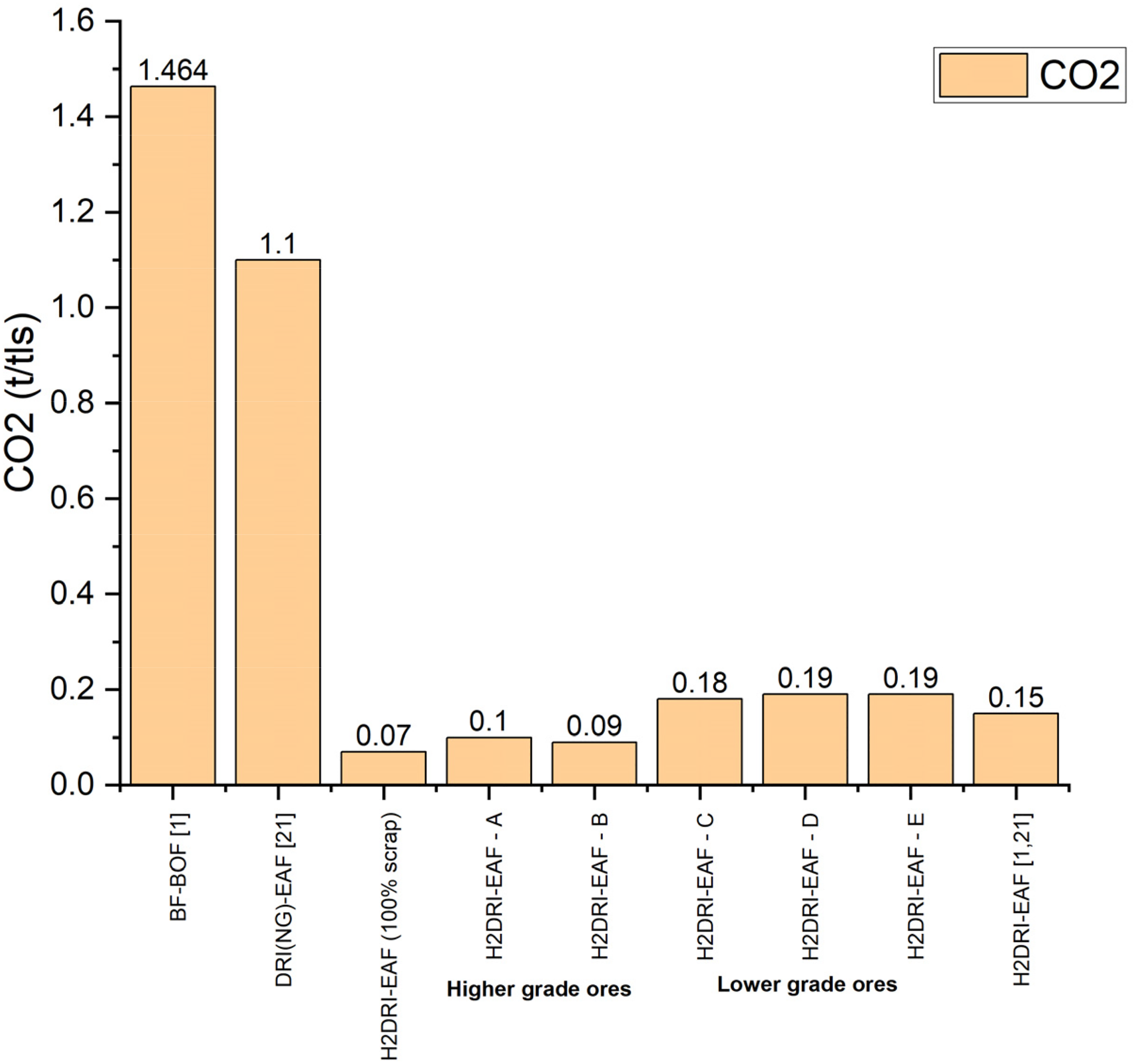
CO2 emission from H2DRI-EAF pathway for different types of ore.
CO2 emission from H2DRI-electric smelter-BOF pathway (about 26 wt-% of the charge was scrap) was found to increase with increase in the gangue content. The values are comparable to the values reported by Wimmer et al. 1 The electric smelter model have been described in detail in a separate publication. 14 From Figures 13 and 14, it can be stated that in comparison to the typical BF-BOF pathway (which has the highest CO2 emission), both pathways showed promising results in terms of reducing CO2 emission in the steelmaking process. There is not much difference in the amount of CO2 emission from both pathways for different types of ores. The difference comes only in terms of loss of yield, slag disposal issue and control issue of the BOF and EAF processes.
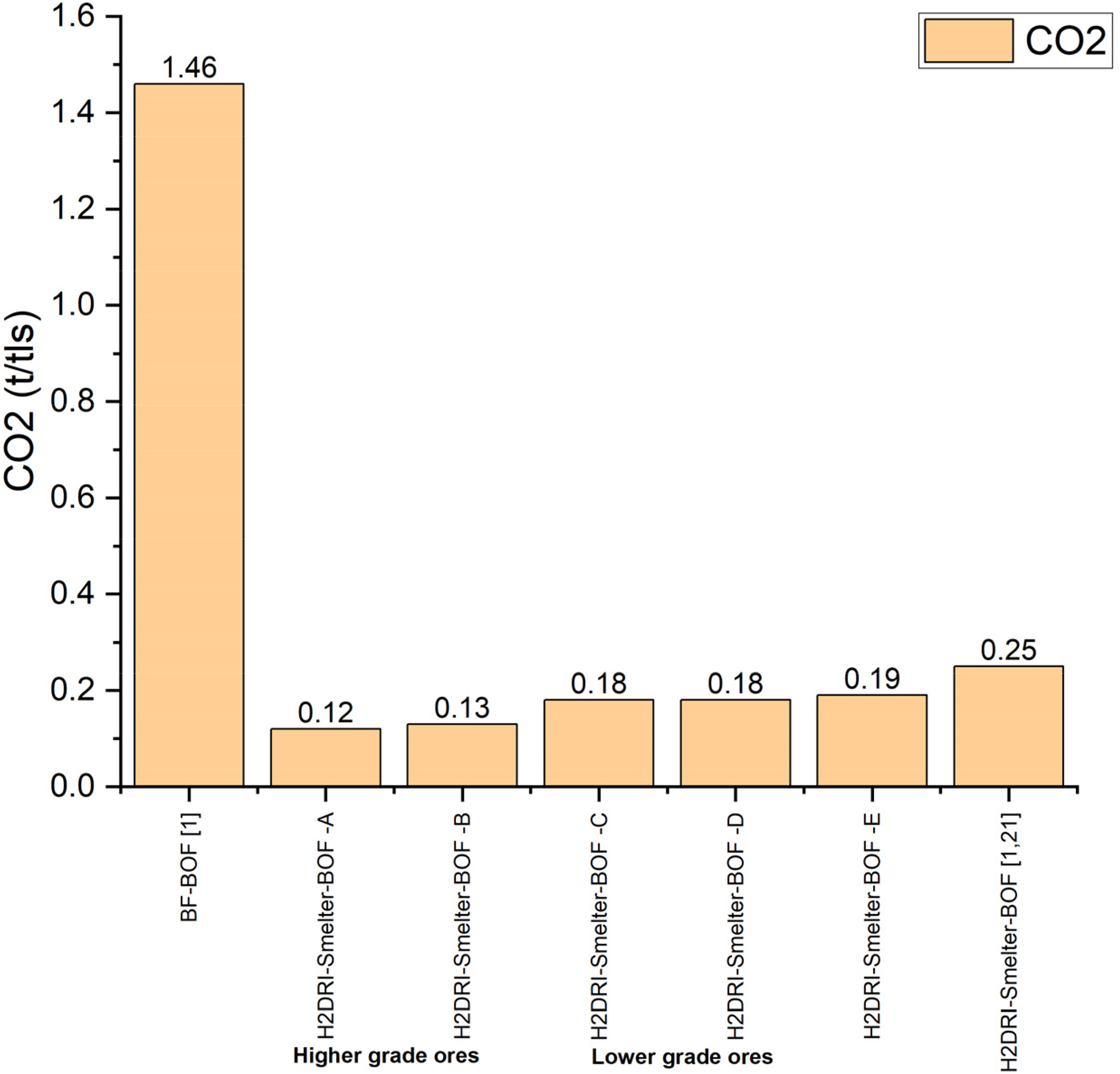
CO2 emission from H2DRI-electric smelter-BOF pathway for different types of ore.
Validation
The results from the present EAF model were validated with the reported value 8 in literature (as shown in Figure 15). The specific electric energy consumption from the model was found to be compatible with the reported values.
Validation of the present EAF model with literature. 8
Conclusions
The current work presents an overall quantitative analysis of the two decarbonisation pathways in terms of slag amount, yield loss and SEC (particularly in an EAF), amount of flux required in a BOF. The aim was to compare how gangue affects outputs and CO2 emissions from both pathways for different grades of ores.
In the EAF model, it was observed that an increase in gangue content in the DRI led to a corresponding rise in the SEC. The SEC for cold-DRI fed into EAF ranged from 514 kWh/tls to 651 kWh/tls, while these values reduced to 399 kWh/tls to 510 kWh/tls for hot-DRI – leading to 21% saving in the SEC. The yield of the EAF process decreased from 88% to 75.5% on the weight basis as a result of increasing the gangue content from 10.7 wt-% to 19.1 wt-%. This was associated with a significant increase in slag weight, ranging from ∼200 kg/tls to 630 kg/tls, which was considerably higher than typical EAF values. In contrast, the amount of slag produced in the BOF process was significantly lower than the EAF process – corresponding to 63 kg/tls to 73 kg/tls depending on the DRI gangue content. CO2 emissions from both the H2DRI-EAF and H2DRI-electric smelter-BOF pathways appear promising in the pursuit of achieving low carbon emissions by 2050 assuming H2 becomes an economically feasible source of energy. The emissions from both pathways were found to be comparable, 0.1 tCO2/tls to 0.19 tCO2/tls, and the trend indicates that CO2 emissions rise as gangue content increases.
In general, the electric smelter-BOF is more appealing to process lower grade DRI than EAF, in which the quantity of slag and loss of yield are considerably higher at higher gangue content. There are greater implications in using EAF for lower grade DRI. Though the costs of electric energy requirement for refining lower grade DRI using can be managed by the application of hot DRI, it will be of great concern and costly endeavour for EAF steelmakers to dispose or use these slags in a profitable way. EAF can be an attractive option for high grade ores whereas the electric smelter-BOF pathway can be economically viable option for processing lower grade ores. The success of the electric smelter-BOF pathway relies heavily on mitigating the technological risks associated with the smelter technology and incorporating more scrap into the BOF steelmaking process. There is also a possibility of beneficiation of the low-grade ores undertaken by the iron ore suppliers to provide greater value to the steelmakers and help them avoid adding an electric smelter into the existing processes, which is the subject of a future publication by these authors.
Footnotes
Acknowledgements
The work has been supported by the Heavy Industry Low-carbon Transition Cooperative Research Centre (HILT CRC) whose activities are funded by its industry, research, and government Partners along with the Australian Government's Cooperative Research Centre Programme with project number HILT.RP1.004.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the HILT CRC,
