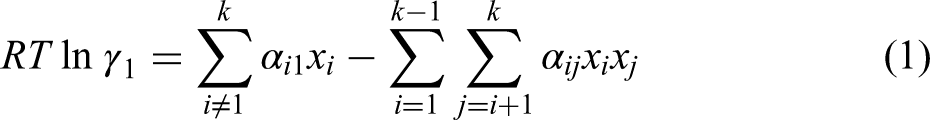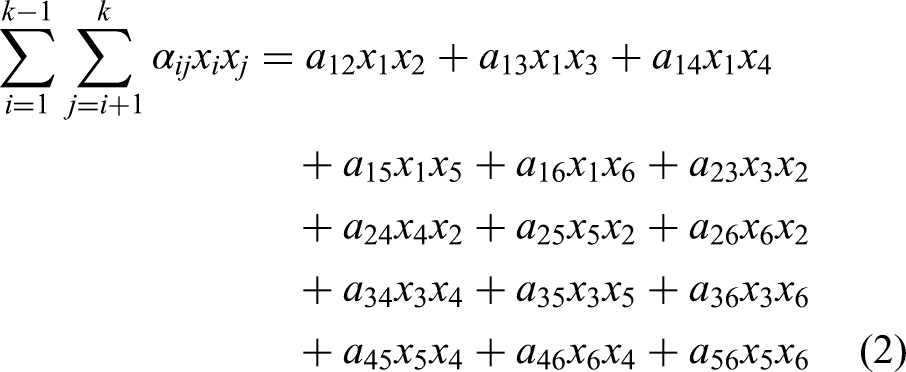Abstract
Phosphorus must be reduced to less than 20ppm for the products subject to hydrogen resistance and other hostile environments. The current dephosphorization process in converter has not reached this level. In this study, the dephosphorization slag with ultra-high Lp was studied, and the kinetic conditions of dephosphorization reaction were optimized too. The industrial production was carried out in a 200t converter in Shougang Jingtang, and the production process was ‘KR-BOF-LF-VD-CC'. By the double slag process in converter, phosphorus contents were reduced to 3.8-11.0ppm in one casting consequence of 10 heats, and the average phosphorus was 7.3ppm. The average tap to tap time was 42min. This technology was successfully applied in the production of 9Ni steel, acid resistant steel and hydrogen resistant steel. 53800 tons of ultra-low phosphorus molten steel have been successfully produced.
Keywords
Introduction
Phosphorus is considered detrimental in most of the steel grades. High content of phosphorus leads to the low plasticity and toughness of materials, especially the impact toughness under low temperature.1–3 In addition, phosphorus seriously affect the tempering brittleness of materials.4–6 The demand on ultra-low phosphorus content was even more profound for the steel grades used in hostile environments, such as low-temperature vessels, low-temperature bridge steels and hydrogen resistance steels, and so on, which all required the phosphorus as low as possible to ensure the better steel quality. According to the production data, the mass fraction of phosphorus decreased from 0.0040% to 0.0010%, the impact energy at −196 °C increased from 188 to 253 J under the same other conditions. So the phosphorus in some critical grades are required less than 20 ppm.7,8,9
There are several methods to remove phosphorus to produce low phosphorus steel. The leading enterprises in Japan, Germany, and South Korea all adopt duplex steelmaking process. The phosphorus was feasible reduced to 20–40 ppm, but the long smelting period (≥65 min) limits the high-efficient production. 9 To this day, the batch production of the steel with phosphorus less than 20 ppm has not been reported.
To produce the steel with phosphorus less than 20 ppm, the phosphorus must be reduced to less than 10 ppm at the end of the converter. Under the condition of same amount of slag, the phosphorus distribution ratio Lp must be above 1500. However, Lp is only about 200 in conventional single slag process, so the amount of slag needs to be increased by 7–10 times to reduce the phosphorus to less than 10 ppm, which is difficult to meet. Lp is up to 400–700 in the LD-ORP process developed by Nippon Steel, which can produce the molten steel with the phosphorus content of 0.0020%–0.0035%. but is difficult to meet the lower phosphorus content.10,11,12 So, it is very necessary to improve the Lp to get the molten steel with ultra-low phosphorus. In this study, the slag composition with higher Lp was calculated firstly, then the process of top and bottom blowing were optimised to promote the dephosphorisation reaction between slag and steel. These technologies have been successfully applied in the industrial production, and realised the stable and efficient production of steel with phosphorus content less than 20 ppm.
Development of the slag for ultra-low phosphorus steel
Both the abilities of super dephosphorisation and phosphorus-fixation are necessary to the slag for ultra-low phosphorus steel. The phosphorus distribution ratio is an important parameter to measure the dephosphorisation ability of slag. Under the same amount of slag, the higher of phosphorus distribution ratio, the lower of phosphorus in liquid steel. To avoid the rephosphorisation, the phosphorus should exist in the slag as the form of solid solution phase.
Calculation method of the phosphorus distribution ratio
The phosphorus distribution ratio is closely related to the slag composition, steel composition and temperature, etc. 13 Many researchers studied the empirical formulas to evaluate the phosphorus distribution ratio, but there were some deviations from the actual results. In order to quantitatively analyse the thermodynamic problems of slag-steel reaction, various ionic solution models have been established based on the ionic structure theory. These models mainly included complete ion solution model, ion and molecular coexisted theory, regular ion solution model. For the complete ion solution model, only the basic characteristics of slag is considered, but the difference in electrostatic potential caused by different types and sizes of ions with the same charge is ignored, which lead to the uneven distribution for cations and anions. So there are differences in the property between complete ion solution and the actual slag, which limits the application of this model. 14 The ion and molecular coexisted theory proposes the structure of slag, which considers that the slag is composed of simple cations, simple anions and molecular compounds with undissociated covalent bond. 15 The regular ion solution model is applied very well in the oxidising slag with high basicity, and is used to analyse the equilibrium distribution of oxygen, phosphorus and sulphur between slag and molten steel. 14 In this paper, the regular ion solution model is used to study the phosphorus distribution ratio of slag to steel.
The activity coefficient of slag component was calculated by the regular ion solution model, which is expressed by Eq. (1).
14
FeO, MnO, CaO, MgO, SiO2, P2O5 are considered as the main components for the slag in this study,
Interaction energy between cations, J/mol.
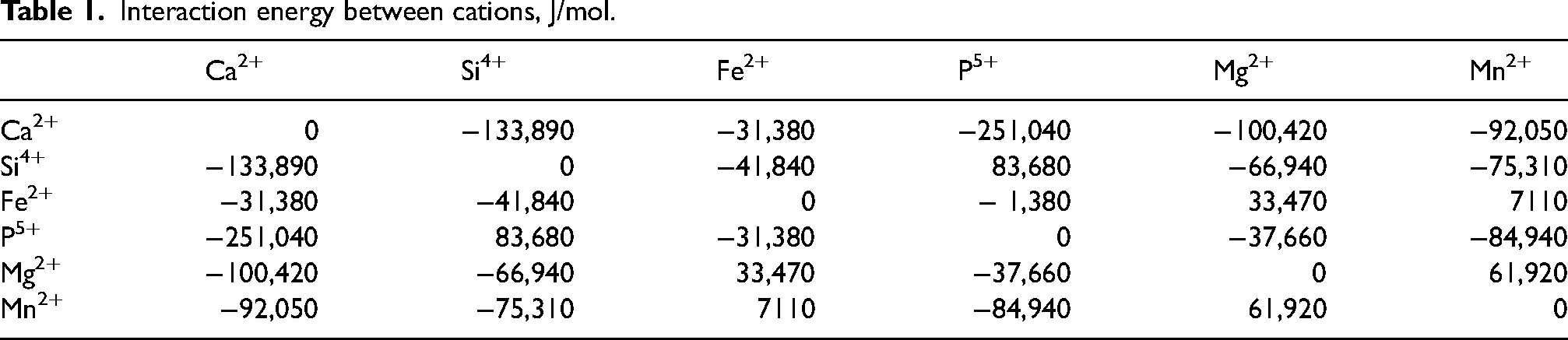
The activity coefficients of FeO (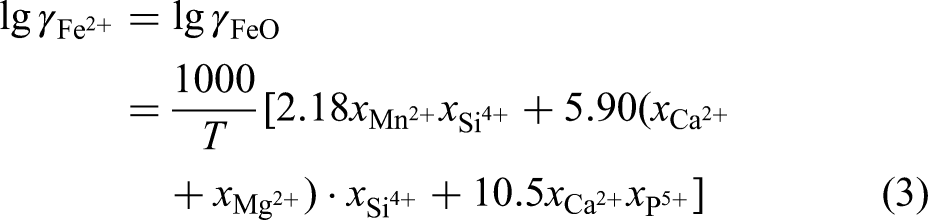
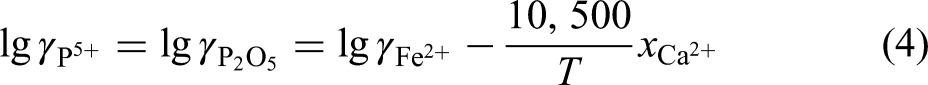
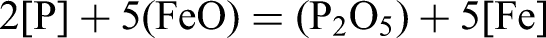
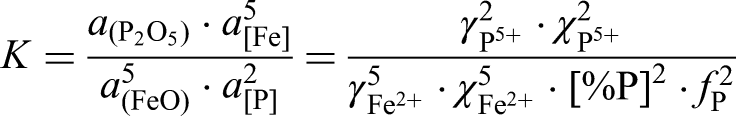
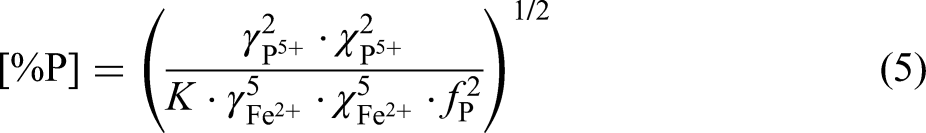
Where,
To verify the reliability of the regular ion solution model, the phosphorus content calculated by the model was compared with the actual data, the result was shown in Figure 1. There is a good consistency between the model and the actual data. But most of the model data are higher than the actual content, that is because the dephosphorus reaction between slag and steel did not reach the equilibrium.
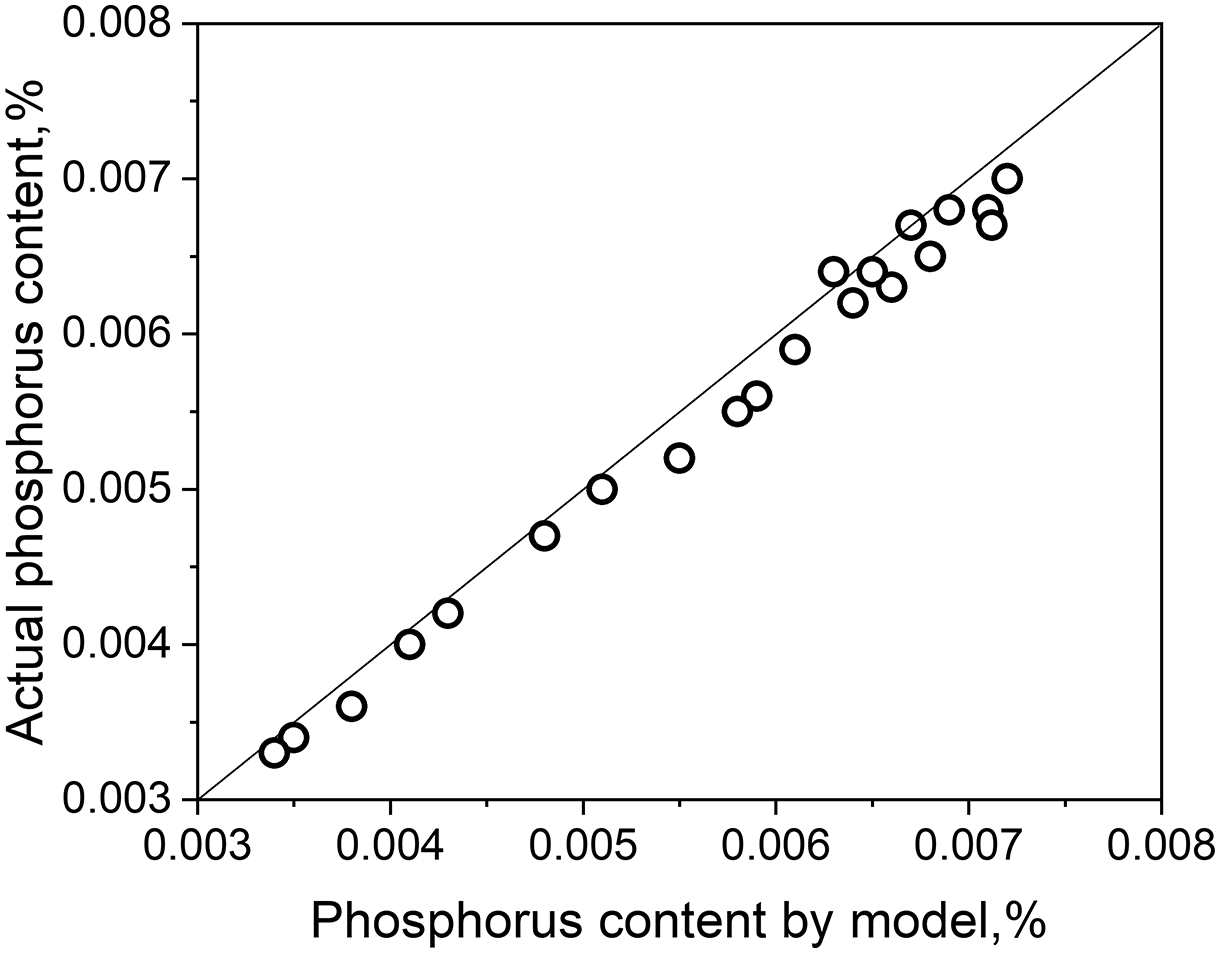
Comparison of phosphorus content from the model and the actual.
So, the equilibrium phosphorus content in liquid steel ([%P]equilibrium) was calculated by Eq. (5), then the equilibrium phosphorus distribution ration of slag to steel (Lpequilibrium) was calculated by Eq. (6). Based on this method, the effect of slag composition and temperature on the equilibrium phosphorus distribution ratio was studied in this paper.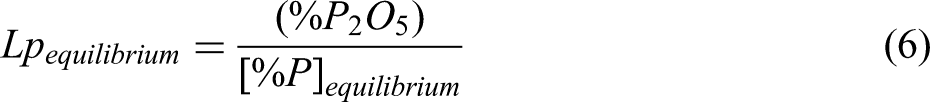
Study of the influencing factors on phosphorus distribution ration
As mentioned above, to produce the steel with phosphorus less than 20 ppm, the end-point phosphorus content in converter must be reduced to less than 10 ppm, and the phosphorus distribution ratio of slag to steel is required to be over than 1500 under the same amount of slag. So the influencing factors on the phosphorus distribution ration were firstly studied, then the optimal composition of slag was given in this paper.
The influence of MgO in slag on the activity coefficients of P2O5 and FeO were calculated by the regular ion solution model. The results are shown in Figure 2(a) and (b). With the increasing of MgO in slag, the activity coefficient of P2O5 increases significantly, but the activity coefficient of FeO reduces, which are not helpful to dephosphorise. So, reducing the content of MgO is beneficial to the dephosphorisation of slag.
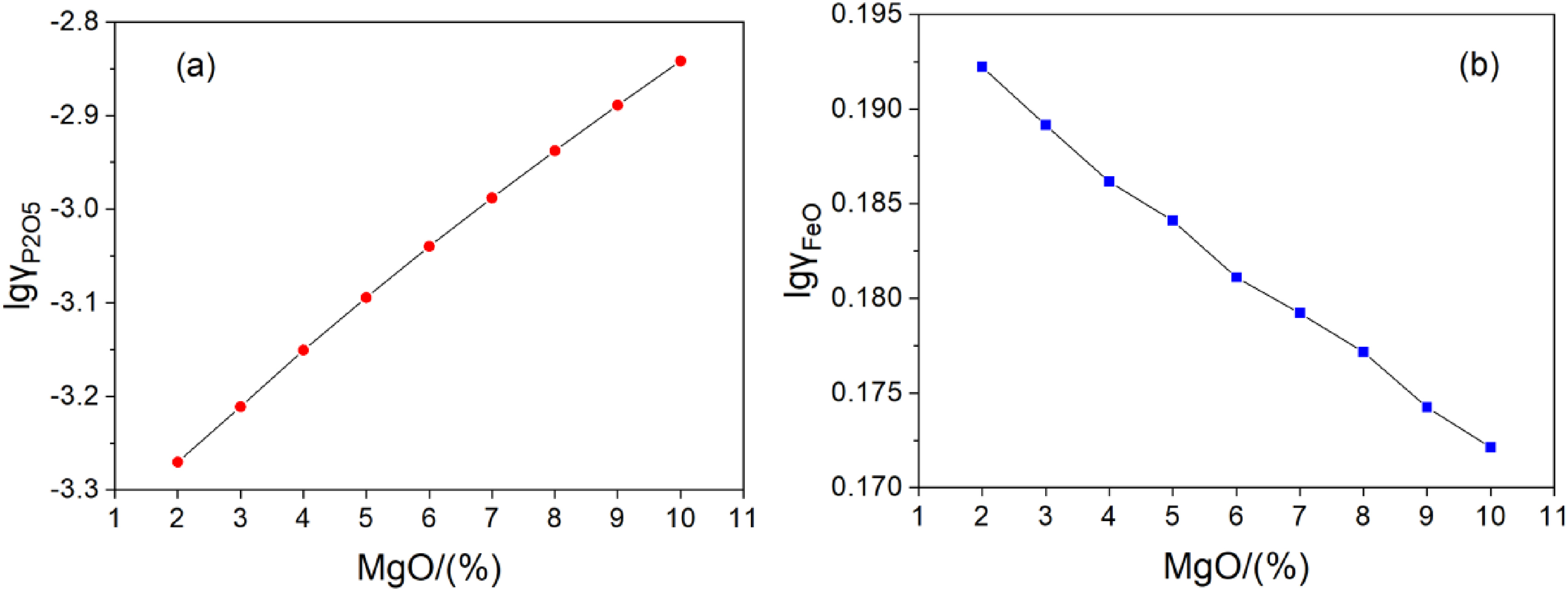
Effect of MgO on the activity coefficients of P2O5 (a) and FeO (b).
Figure 3 shows the effect of the slag basicity R and MgO on the phosphorus distribution ratio Lp at 1610 °C. Under the same content of MgO, Lp increases with the increasing of slag basicity. As basicity increasing, the content of CaO in slag increases relatively. During the process of dephosphorisation, CaO reacts with P2O5 to generate calcium phosphate compounds. So the increasing of CaO promotes the dephosphorisation reaction. In the other hand, CaO provides the amount of free O2− in the slag, which improves the oxidisability of the slag and also promotes the dephosphorisation reaction. Because MgO in the slag increases the activity coefficient of P2O5, so with the increasing of MgO content, Lp decreases accordingly. Figure 3 also shows that Lp would more than 1500 when the slag basicity R > 5 and the MgO content was 3%.
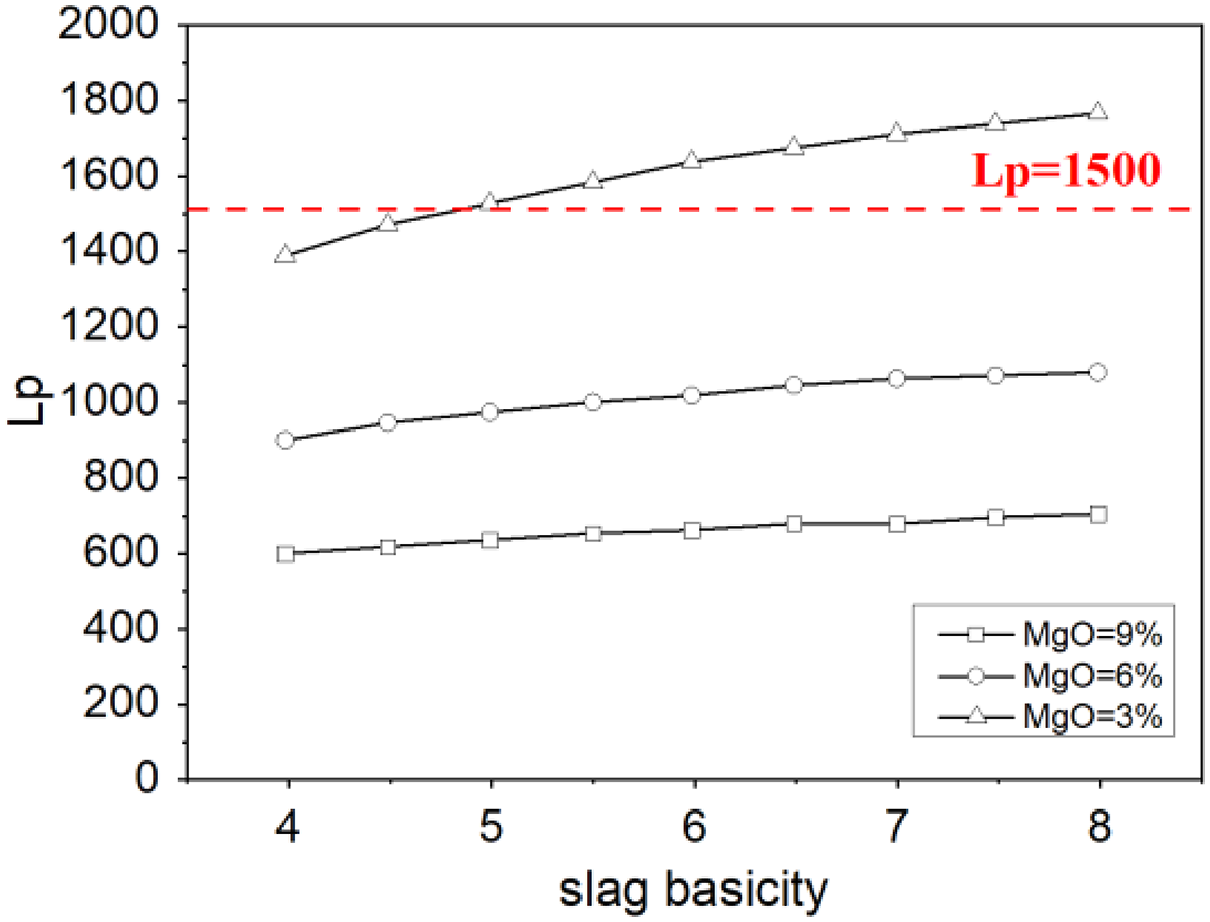
Effect of slag basicity R and MgO on the Lp.
Figure 4 shows the effects of FeO and MgO on the phosphorus distribution ratio Lp at 1610 °C. Under the same content of MgO, Lp increases with the increasing of FeO from 14% to 30%. FeO is a kind of oxide, dephosphorisation reaction is promoted while increasing the activity of FeO. FeO is also a kind of basic oxide and reduces the activity coefficient of P2O5. So FeO is beneficial to remove the phosphorus in molten steel. Figure 4 also shows that there is an optimal content of FeO when the MgO content is 3%, the maximum Lp is obtained at this point. When FeO is lower than this optimal value, Lp increases with the increasing of FeO. When FeO content is higher than this optimal value, Lp decreases with the increasing of FeO. We can also see from the results that the optimal content of FeO increases with the increasing of MgO in the slag. Lp is more than 1500 only the content of MgO is 3%, and the max. Lp is obtained when the FeO is about 23%.
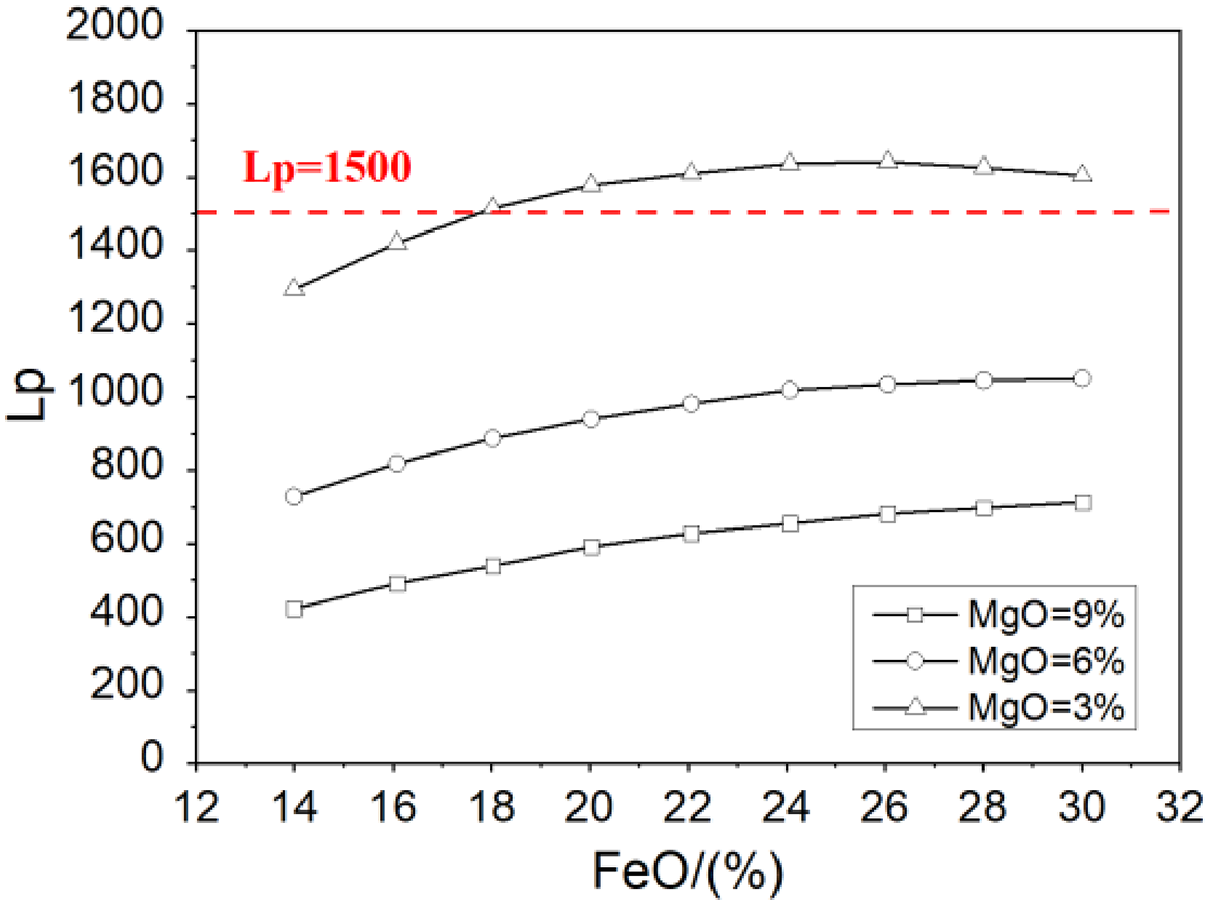
Effect of FeO and MgO on the Lp.
Figure 5 shows the effects of temperature and MgO on the phosphorus distribution ratio Lp. Under the same content of MgO, Lp increases with the increasing of temperature. Lp will be above 1500 when the content of MgO is less than 4%.
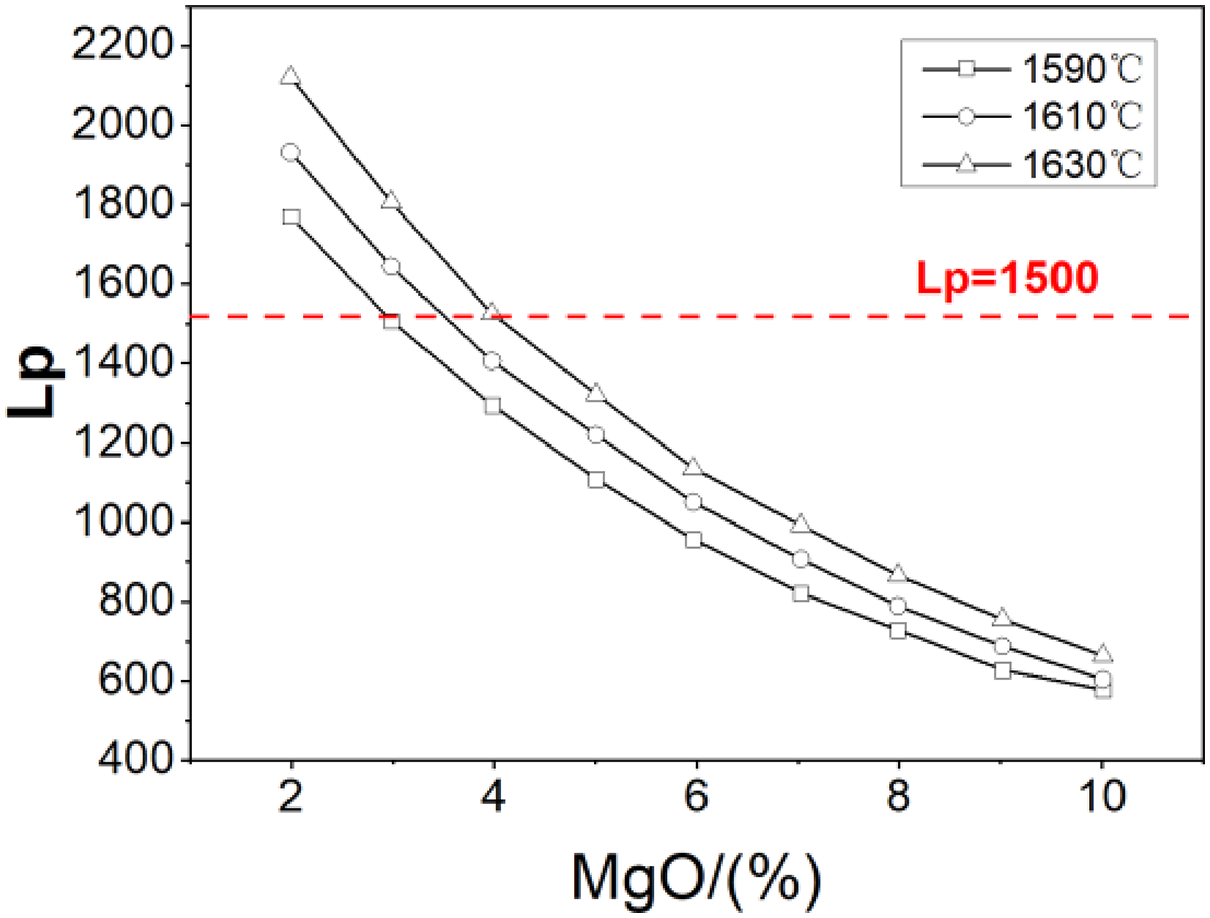
Effects of temperature and MgO content on the Lp.
To summarise, at the temperature of 1590–1630 °C, Lp will be more than 1500 when the composition of dephosphorisation slag is controlled as: high basicity(R > 5), lower content of MgO(MgO% < 4%), higher content of FeO (23% < FeO% < 30%). Then it is possible to reduce the phosphorus to less than 10 ppm at the end of converter smelting.
Control technology of phosphorus fixation phase in the slag
The converter slag is a kind of multi-phase slag. The slag with different components has different phases, and the existing form of P2O5 in slag is also different. Activity of P2O5 is high when it exists in liquid slag. But P2O5 is stable when it exists as the form of solid solution 2CaO·SiO2–3CaO·P2O5 (C2S–C3P). So, to avoid the rephosphorisation, it is very important to control the slag composition to make more P2O5 exist as the form of solid solution C2S–C3P.
Effect of slag basicity on the slag phase
To analyse the existing form of P2O5 in different slag, the semi-steel slag with low basicity and the final slag with high basicity were respectively studied. The slags were cold embedded and polished, then the morphology and the composition of slags were analysed by scanning electron microscope.
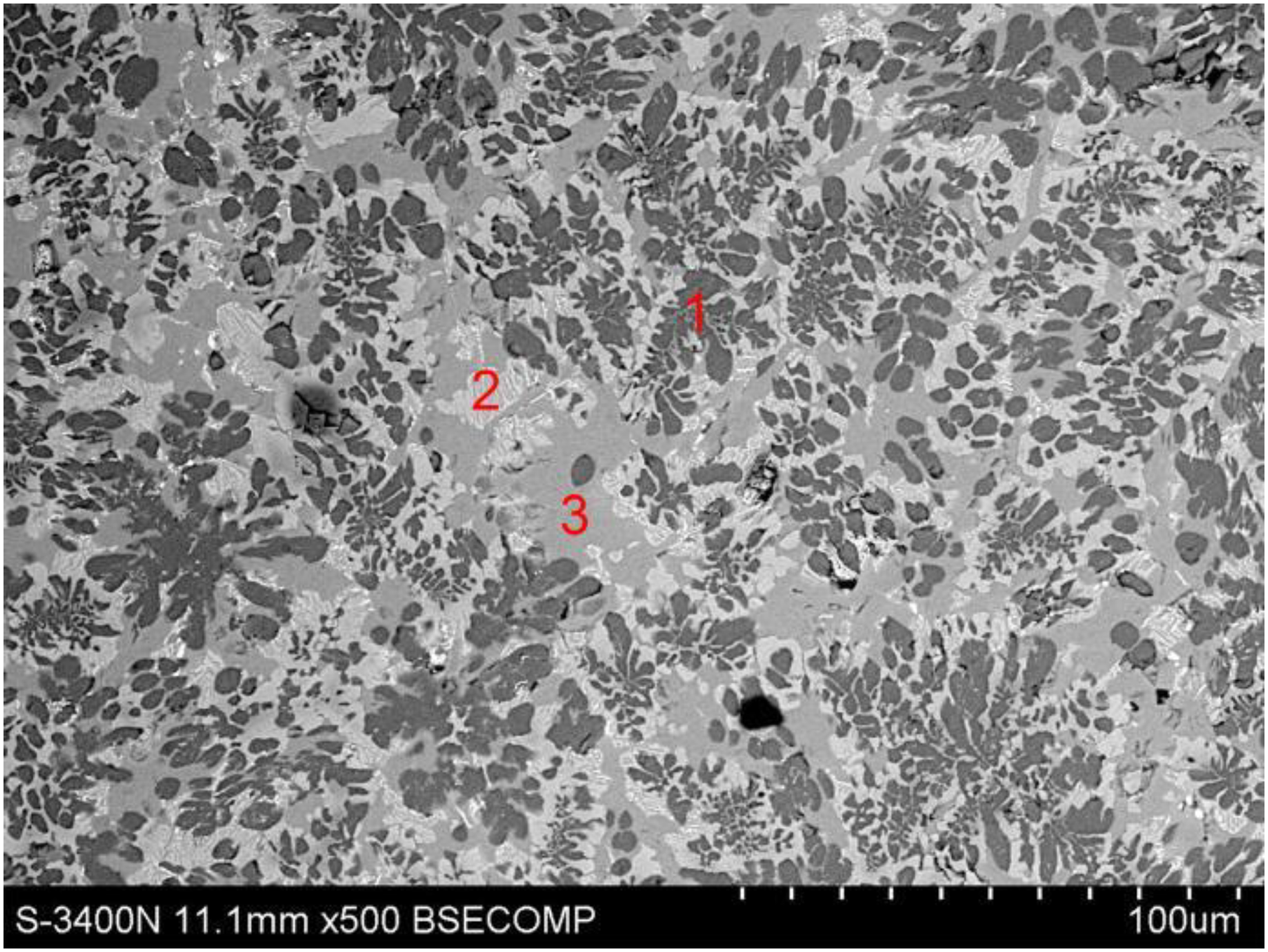
Figure 6 shows the morphology of the semi-steel slag with R = 1.15, TFe% = 25.9%, P2O5% = 2.14%. The slag is liquid at the semi-steel temperature. During the air cooling process, the white dendritic phase (1 and 2 in Figure 6) begin to precipitate, which are composed of higher FeO (above 60%) and lower CaO and SiO2, the contents of P2O5 in the phase 1 and phase 2 are 1.18% and 1.29%, respectively, lower than the average P2O5 content of 2.14% in slag. The black phase (3 and 4 in Figure 6) is super-cooling liquid phase, which is composed of CaO–SiO2–FeO (about 23%), the P2O5 content in the phase 3 and phase 4 is 3.06% and 2.48%, respectively, a little more than the average P2O5 content in the slag. Table 2 shows the results of EDS for positions shown in Figure 6. So, there is no the solid solution phase in semi-steel slag with low basicity. To avoid rephosphorisation, this kind of slag should be poured out as much as possible during the first deslagging.

SEM image of the semi-steel slag with low basicity.
Results of EDS analysis for positions shown in Figure 6.
With the increasing of slag basicity, the slag phase changes too. Figure 7 is the morphology of the final slag in converter with R = 5.93, TFe% = 27.89%, P2O5% = 1.51%. This slag is composed mainly by three kinds of phase. The black dentritic phase is solid solution of 2CaO·SiO2–3CaO·P2O5 (C2S–C3P), which is also the phosphorus-enrichment phase in the slag. The content of P2O5 in this phase is 3.2%, higher than the average content in the slag. The white Gray phase is composed by higher FeO and lower CaO and SiO2. In this phase, a small amount of P2O5 is contained. Gray phase is the low melting temperature phase of calcium ferrite. EDS shows that the content of P2O5 in this phase is zero. Therefore, we can conclude that P2O5 has almost been solid soluted in the black phase, and there is no P2O5 existing in the liquid slag phase. So the activity of P2O5 in the slag is decreased, and the dephosphorisation reaction is promoted. Moreover, 2CaO·SiO2 is easily precipitated in the slag when basicity is high, which is beneficial to combine the 3CaO·P2O5 in the slag to form the stable solid solution of 2CaO·SiO2–3CaO·P2O5 (Table 3).17,18
SEM image of the final slag.
Results of EDS analysis for positions shown in Figure 7.

At the late stage of converter smelting, it is very important to improve the slag basicity to increase the content of solid solution phase of C2S·C3P. But higher basicity also has higher viscosity of slag, the mass transfer coefficient of P2O5 in the slag reduces at the same time, which limits the transfer of P2O5 in the slag,19,20 In addition, the flowability of slag is reduced at high viscosity, which results the difficulty of deslagging. Combined with the results of the previous section, R = 6 is chosen to meet the requirement of Lp ≥ 1500 and the higher content of solid solution C2S·C3P in the slag.
Effect of FeO on solid solution C2S·C3P phase in the slag
At the temperature of 1500 °C, the effect of FeO on the solid solution C2S·C3P phase was calculated by the equilibrium module of Factsage. Results are shown in Figure 8. The increasing of FeO reduces the precipitation of C2S and the content of C2S·C3P phase, then increases the P2O5 in liquid slag phase. Because the solid solution C2S·C3P is the phosphorus-enrichment phase and very stable, which fixes phosphorus in the slag and limits it return to the molten steel. On the other hand, the higher the content of P2O5 in the liquid slag, the easier for phosphorus return to molten steel. So, the content of FeO in the slag should not be too high.
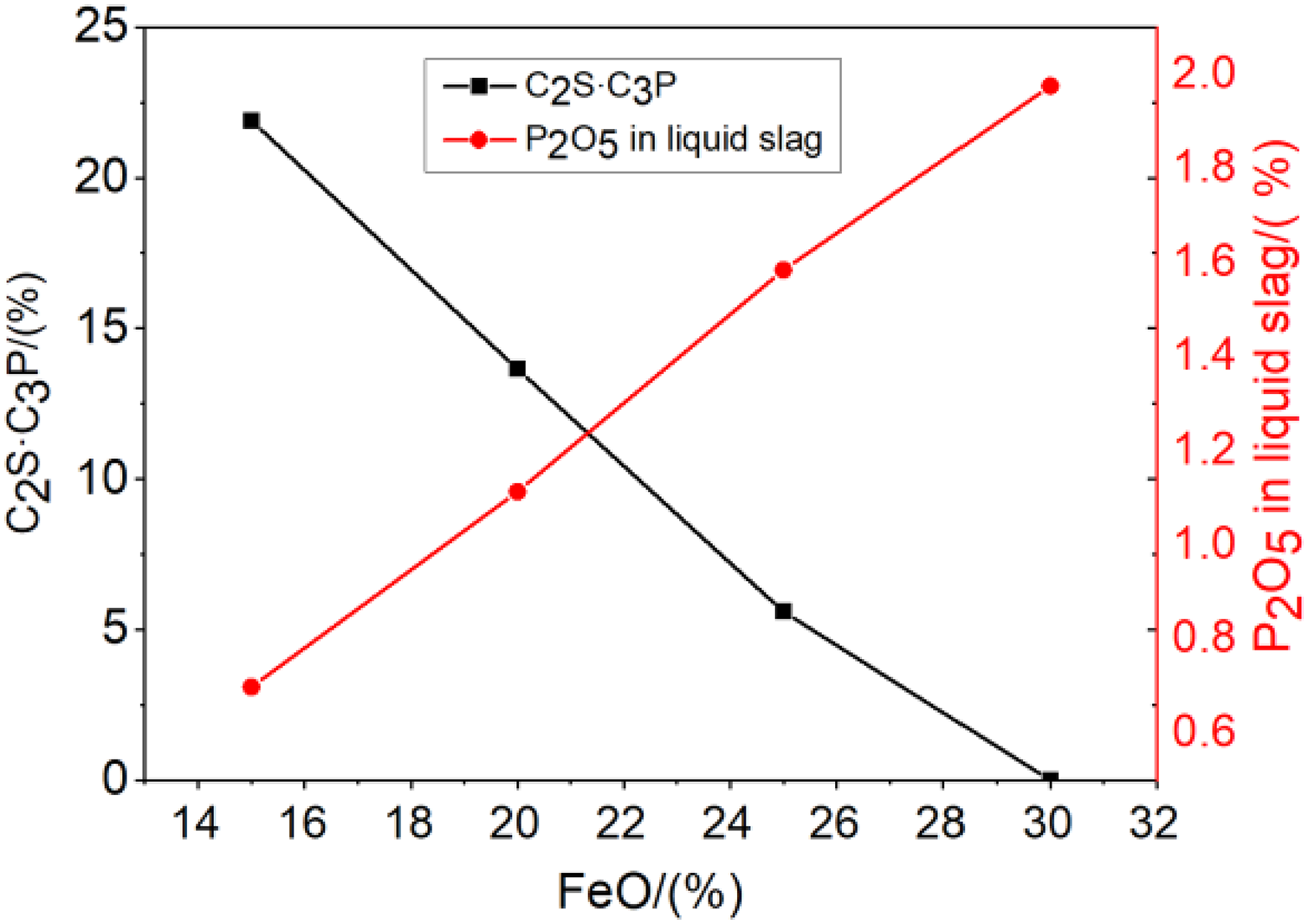
Effect of FeO on the phase of C2S·C3P and the P2O5 in liquid slag.
High efficient blowing technology of converter
The dephosphorisation reaction occurs mainly at the slag–metal interface and in the slag. Accordingly, the stirring plays a very important role during converter smelting, which directly affect the dephosphorisation ratio and the degree of the slag–metal reaction. To give full play to the role of dephosphorisation slag, it is very important to improve the top and the bottom blowing process.
Optimisation of top lance height in converter
The top lance height is a key parameter to dephosphorisation ratio, which can enhance stirring in the bath and promote mass transfer of [P] in molten steel. In this study, the top lance is equipped with a 5-hole nozzle, and the working flow is 44,000–46,000 Nm3/h. To improve the dephosphorisation ratio before the first deslagging, the lance height at the stage B in Figure 9 was lowered to less than 1700 mm to enhance the stirring of molten steel. At the later stage of converter smelting, in order to reduce the slag oxygen potential and accelerate the reaction of [C] with [O], top lance height was kept as less than 2000 mm. 21 During the process of stage C in Figure 9, the top oxygen gas flow rate was increased and the blowing time was extended for 1.5–2.0 min.
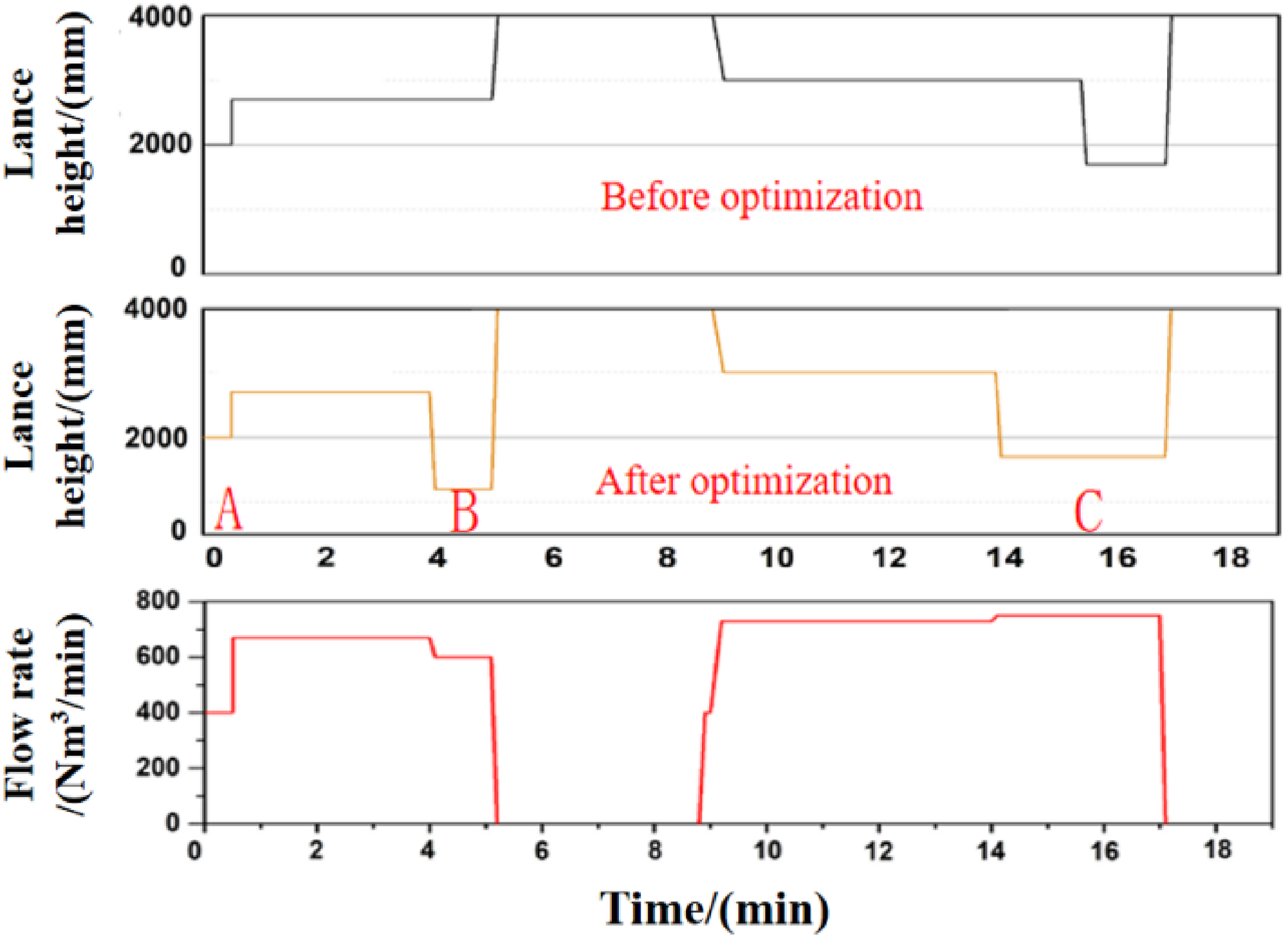
Optimisation of top blowing lance height.
Optimisation of the bottom blowing in converter
The influence of bottom blowing on the mixing of the metal bath is much greater than that of top blowing. The flow rate of bottom blowing has a significant impact on the mixing time of the liquid steel.22,23 The effect of bottom blowing flow rate on turbulent kinetic energy and mixing time of molten steel were calculated by fluent software. During the process of numerical simulation, chemical reactions were ignored, the argon bubbles were assumed as uniform in size, the bottom blowing tuyeres were equivalent to the circular holes, and the flow of liquid steel in converter was regarded as incompressible flow.
The effect of bottom blowing flow rates of 300, 480, 600, 800, 1000 and 1500 Nm3/h on turbulent kinetic energy in the molten steel were calculated in this study. The result was shown in Figure 10, the turbulent kinetic energy in the liquid steel increased with the increasing of bottom blowing flow rate. The mixing time of molten steel under different bottom blowing flow rates were shown in Figure 11, with the increasing of bottom blowing flow rate, the average speed of the molten steel significantly increased, and the mixing time of the molten bath correspondingly decreased. Moreover, the mixing time significantly decreased when the bottom blowing flow rate was above 1000 Nm3/h.
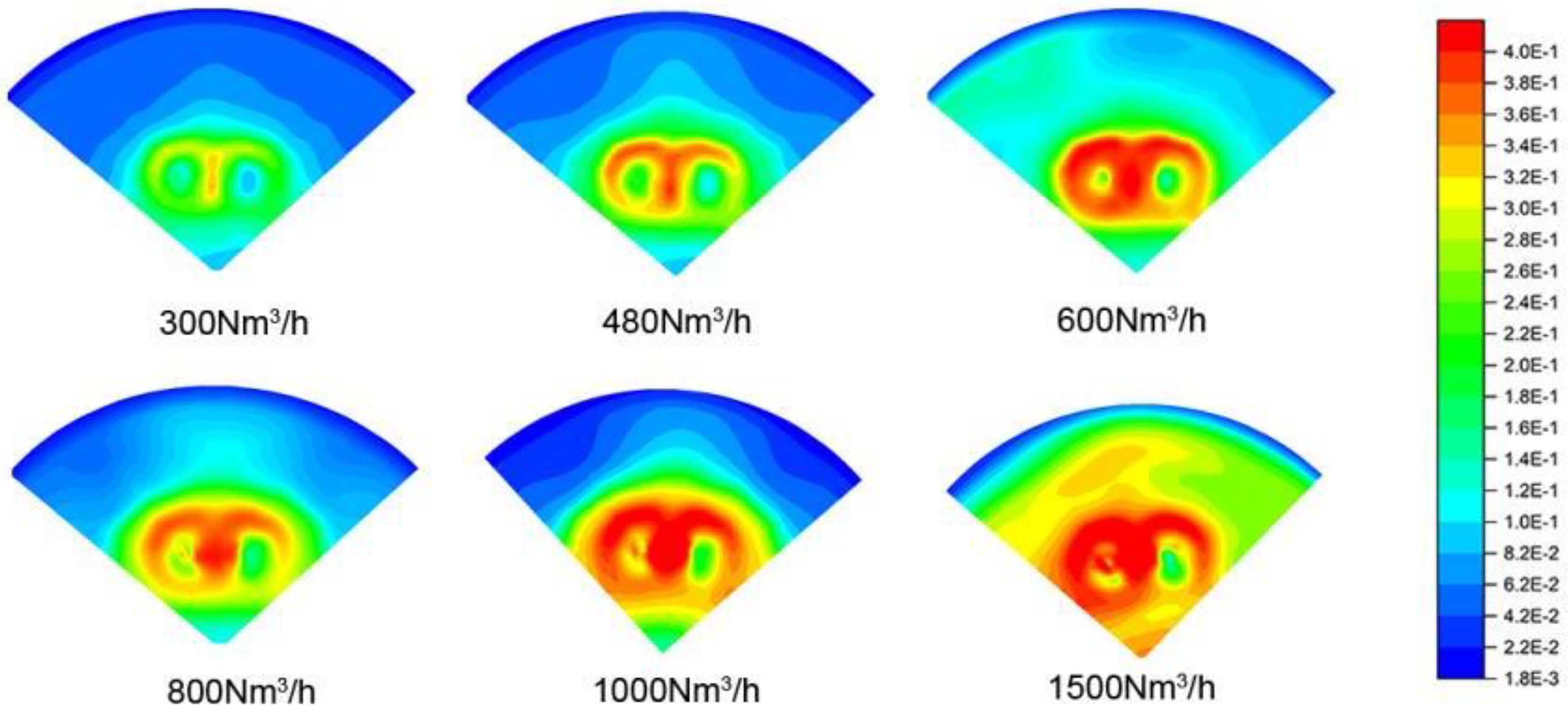
Turbulent kinetic energy under different bottom blowing rate.
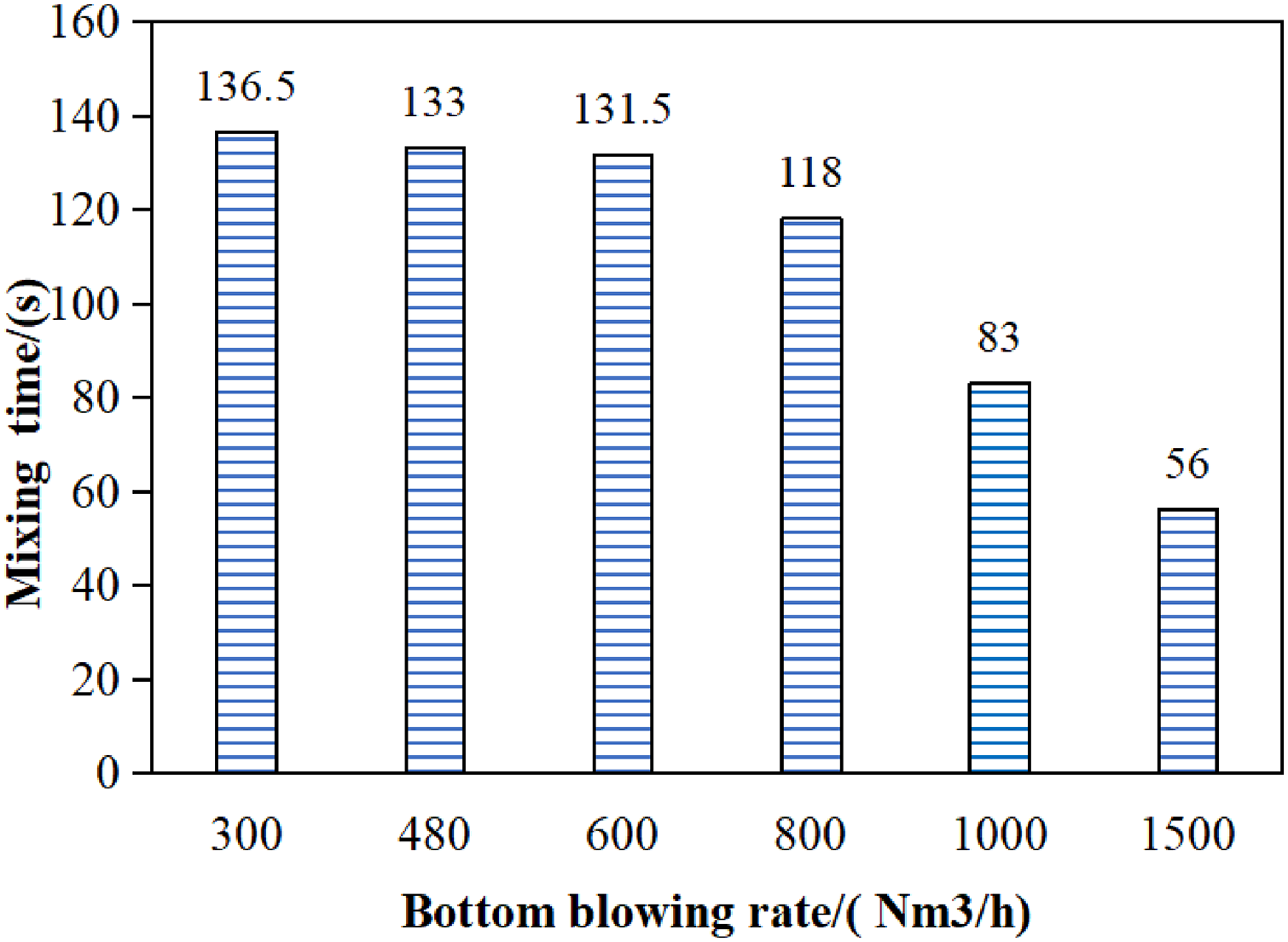
The relationship between bottom flow rate and mixing time.
Based on the above calculation, the bottom blowing rate in the primary period was increased from 600 to 800 Nm3/h. and increased from 800 to 1200 Nm3/h at the stage of TSC–TSO. The corresponding bottom blowing flow control mode was shown in the Table 4.
Bottom blowing rate of different converter process.
Application effect
The above technologies were widely used in a 200-ton converter in Shougang Jingtang, and the production process was ‘KR-BOF-LF-VD-CC’. The ultra-low phosphorus molten steel of 53,800 tons have been produced successfully. Take the production of 9Ni steel as an example. The production was carried out in one casting consequence of 10 heats, and the double slag process was adopted during the BOF smelting of all the heats.
By improving the top and bottom blowing process, the ability of the dephosphorisation slag with high phosphorus distribution ratio was brought into full play. During the production process of 9Ni steel, the average dephosphorisation rate and phosphorus content at different stage were shown in Figures 12 and 13. Results show that the average phosphorus content at TSC was controlled at 9.4 ppm, and reduced to 7.3 ppm at TSO. The average tap to tap time was 42 min.
Dephosphorisation rate at different blowing stage.
Phosphorus content in liquid steel at different blowing stage.
On the other hand, combined with the technologies such as low-temperature tapping, rapid deslagging, adding the alloy with lower phosphorus content and so on, the ultra-low phosphorus steel (P ≤ 20 ppm) was successfully produced. According to the production data of 9Ni steel, Lpequilibrium calculated by the regular ion solution model was 1594–2411, Lpactual at the end of converter smelting was 1511–1946. The comparison of [%P]equilibrium and [%P]actual was shown in Figure 14. The results show the [%P]actual were almost close to the [%P]equilibrium, so the dephosphorisation reactions after optimisation were almost to the equilibrium.
Comparison of [P]equilibrium and [P]actual in one continuous casting.
Conclusion
The end-point phosphorus content in converter was reduced to less than 10 ppm in this study. The technology was successfully applied to the production of 9Ni steel, acid resistant steel and hydrogen resistant steel. After verification by batch production for 53,800 tons of ultra-low phosphorus molten steel, the average converter period was 42 min, this technology was mature and stable.
At the temperature of 1590–1630 °C, when the slag was controlled as high basicity (R > 5), lower content of MgO (MgO% < 4%), higher content of FeO (23% < FeO% < 30%), the phosphorus distribution ratio between slag and steel Lp would be more than 1500. Solid solution phase of C2S·C3P exists in higher basicity slag, the increasing of FeO in slag would reduce the content of C2S·C3P and increase the P2O5 content in liquid slag, which were adverse to dephosphorise. Lower the top lance height less than 1700 mm and increase the bottom blowing flow rate to 800 Nm3/h before the first deslagging. At the later stage of blowing, extend the blowing time for 1.5–2 min while lowering the top lance, and increase the bottom flowing rate to 1200 Nm3/h at the stage of TSC–TSO, which all greatly improve the dynamic conditions of the converter. The converter end-point phosphorus content in one continuous casting (10 heats) was efficiently controlled at 3.8–11 ppm, the average phosphorus content was 7.3 ppm.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.