Abstract
Mechanism of cerium-based inclusion formation and influence on impact toughness were researched in industrial Al-killed steel. In the final product, actual inclusions were complex, consisting of Al-O-Ce (Ce-poor), Al-O-Ce (Ce-rich), Al-O-Ce-S(Al-poor) and S-Ca (Mn-rich) from inside to outside. Results of thermodynamic balance calculation were completely inconsistent with the actual inclusions. Considering decomposing or disappearing of formed inclusions in steel difficult, new computing results showed that inclusions were formed by firstly Al2O3 (exist in molten steel before adding Ce), AlCeO3, Ce2O2S, CaS, Ca (Mn)S and MnS in turn, in better agreement with the actual inclusions. Outside of the actual inclusions, Ce2O2S and CaS had a smaller disregistry with ferrite, which could offer effective nucleation sites. And small inclusion was surrounded in a grain and bigger inclusion was surrounded in two grains at least. Microstructures were observed that grains with Ce were finer than grains without Ce. Moreover, V-notch impact toughness of samples was tested at 0 and −60°C, respectively. Compared with samples without Ce, the values of impact toughness were increased by 161% at 0°C and 225% at −60°C, respectively. And fracture surface with Ce had more and finer dimples and finer cleavages than that without Ce.
Introduction
Up to now, many articles have reported on effect of cerium on steel, indicating that suitable content of cerium in steel has an improvement of steel properties1–3. Ce existed mainly in steel in the form of Ce-inclusions. Inclusions have important effects on the properties of steel, such as impact toughness. The positive effects of inclusions were usually achieved by changing micro-structure and modifying inclusions. On changing micro-structure, inclusions could fine grains by offering nucleation site for ferrite,4–7 which was helpful to the properties. And on modifying inclusions, inclusions could make harmful inclusions form beneficial inclusions,4,8 such as Ca-treat in Al-killed steel.9,10 There were two important aspects for inclusions, formation mechanism of inclusions and effect of refining grains.
For Ce-based inclusions, the evolution of the inclusions was different with increasing content of Ce in Al-killed steel. In the lab, the evolution was simple, such as following Al2O3→CeAl11O18→CeAlO3→Ce2O3 in labs.11–13 And in industry, the evolution was complex, such as following CaO-Al2O3 → (Ca-Ce-S-O) + (Al-Ca-O-Ce) and following MnS + Al2O3–MnS → MnS + Ce–Al–O–MnS + Ce–O–MnS → MnS + Ce–S–MnS + Ce–S + Ce–O–S–MnS + Ce–O–S → Ce–S + Ce–O–S.14–15 As known, a key of research value was industrial application. Considering complexity of actual inclusions in steelmaking process, it was meaningful to research the formation of Ce-based complex inclusions. For the effect of Ce on impact toughness, a proper content of cerium (Ce) could markedly improve impact toughness of steel. And mechanism of improving impact toughness was mainly fining grains and modifying inclusions into spheres by Ce-treatment.1,16 However, excess content of rare earth (RE) would be harmful to toughness and effect of content of RE on toughness was closely related to RE/S in steel with the proper Ce/S was about 1.5–1.7.2,17,18 Moreover, element of Ce and S generally could form Ce2O2S, CeS, Ce2S3 and Ce3S4 in steel. CeS was the most effective, then Ce2O2S and finally Ce2O3 as nucleation site for α-Fe.19,20 On the contrary, Ce2O3 was the most effective, then Ce2O2S and finally CeS as nucleation site for γ-Fe. 21 And Ce2O2S was often observed in steel and the Ce2O2S was responsible for the grain refinement of δ-Ferrite.14,15,22
Generally, most studies of steel bearing RE were from labs, with high content of RE. However, the content of RE in industrial products was very low, because of nozzle clogging problem and economic problem. Thus, researching with low content of RE had a theoretical and practical meaningful in industrial products. In this article, industrial Al-killed steel Q355B/D steel was chosen as object of study and formation mechanism of Ce-based complex inclusions and effect of the inclusions on microstructure and impact toughness would be researched.
Materials and methods
Process of experiment
To decrease harmful effect of other factors, industrial-level tests were prepared and Q355 B/D was chosen as test materials. Process route of steelmaking was basic oxygen furnace (BOF) → ladle furnace refining (LF) → continuous casting (CC). Ce-Fe alloy (Ce:30wt%) was added into molten steel during later stag of LF refining, then hold about 5 min, add Ca wire and hold about 5 min before ladle leaving to CC.
The steel was melted at 1843 K in 260t LF. Samples were cut from casting blank and rolled products. The chemical composition, parameters of continuous casting and parameters of rolling were listed Table 1(a), (b) and (c), respectively.
Manufacturing parameters of Q355B/D steel.
Characterization and analysis
The content of Ce was measured by ELAN-9000. Samples were cut from rolled products. Micrographs and chemical composition of cerium-based inclusion were characterised using scanning electron microscopy (SEM, ZEISS Sigma 500) with EDS analysis and TEM. The impact samples were cut off by direction of rolling and width was made of 10 × 10 × 75 mm, tested at 0 and −60°C, respectively. Number and size of inclusions were automatically collected and counted using SEM with EDS on surface of samples from direction of width. Thermodynamic software Factsage 8.2 was used for analysing type and content of inclusions. And FToxid database, FSstel database and CESO database were employed to compute thermodynamic balance in Factsage.
Results and discussion
Observation of cerium-based inclusion
Typical inclusions were observed by SEM in Figure 1 and it showed the inclusions were global and were surrounded by ferrite. And small inclusion was surrounded by a grain (named type I) and big inclusion was surrounded by two grains at least (named type II).

SEM of typical inclusions and schematic diagram of inclusion offering nucleation sites for ferrite.
Figure 2 showed elemental mapping of the inclusions, where distribution of elements of O, Al, Ce, S, Ca and Mn in the inclusion was clearly observed. And the distribution appeared evolution of Al-O-Ce(Ce-poor), Al-O-Ce(Ce-rich), Al-O-Ce-S(Al-poor) and S-Ca (Mn-rich) from inside to outside. It indicated that the inclusions were cerium-based complex inclusion.

Elemental mapping of the cerium-based complex inclusion.
For the cerium-based inclusions, evolution of shape was zone of Al-O-Ce (Ce-poor) with corners, zone of Al-O-Ce (Ce-rich) with unclear corners and zone of Al-O-Ce-S(Al-poor) and S-Ca (Mn-rich) with sphericity from inside to outside. In S-Ca (Mn-rich) zone, distributions of Ca element and Mn element overlapped.
Mechanism of cerium-based inclusion formation
Thermodynamic balance computation
As above mentioned, actual inclusions, in industrial Al-killed samples, consisted of Al2O3, AlCeO3, Ce2O2S and CaS(MnS) from inside to outside. And formation process of the inclusions was calculated using Factsage8.2 with thermodynamic balance. Except dates of Table 1, oxygen content was needed in the calculation. And final total oxygen (T.[O]) was 0.0012% in the steel. The computing results were plotted in Figures 3 and 4.
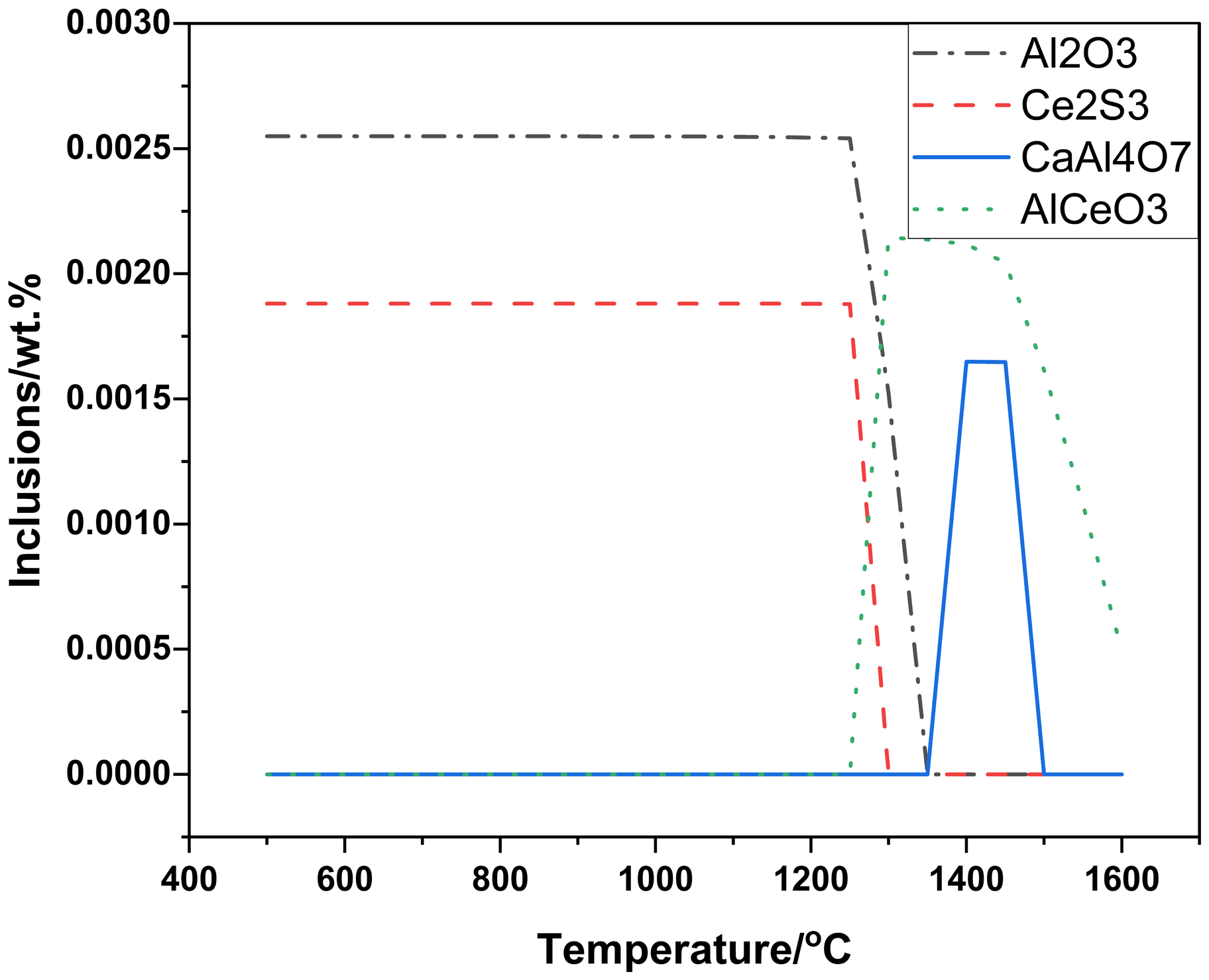
Change of inclusions in process of cooling in balance calculation.
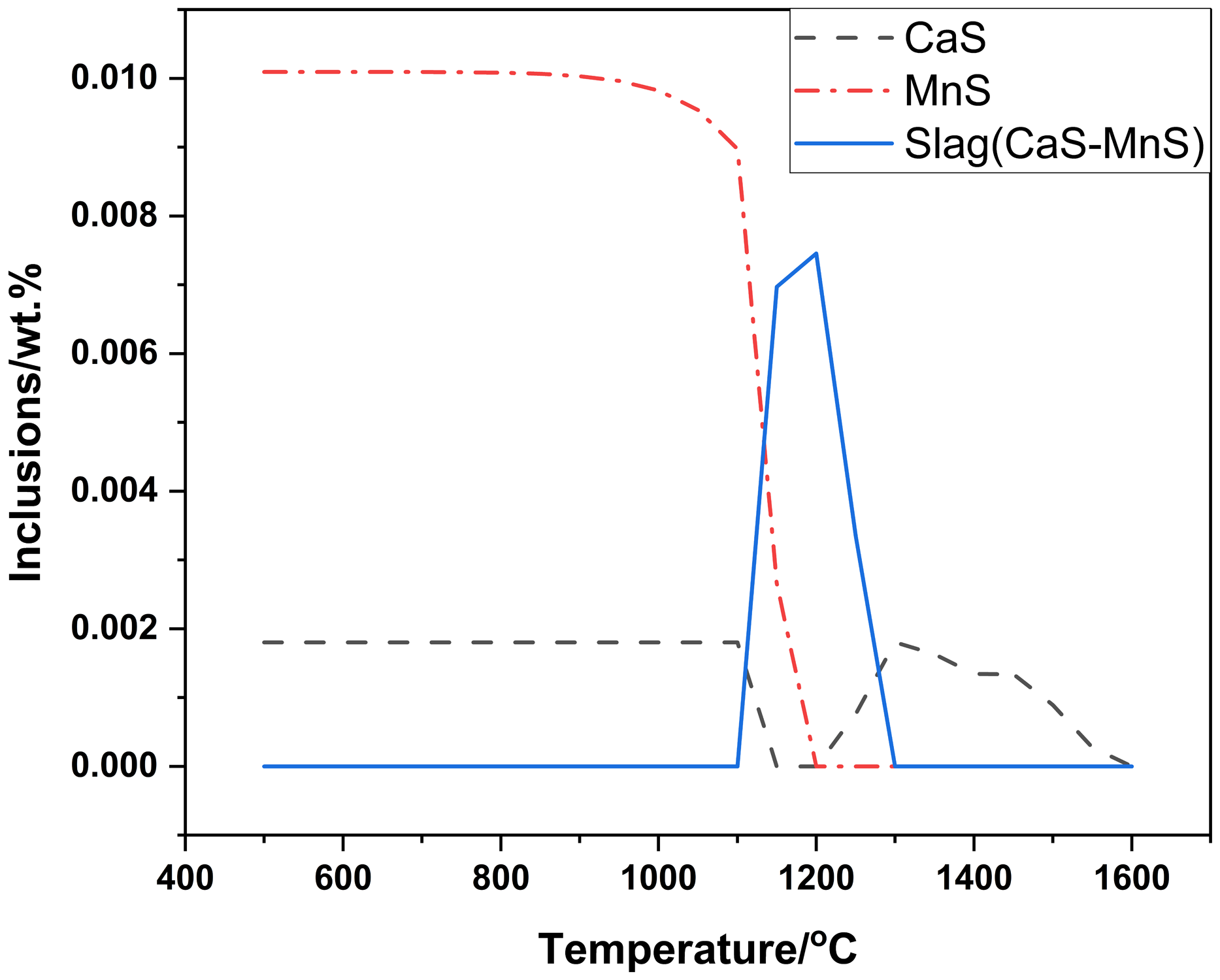
Change of Ca-Mn-S inclusions in process of cooling in balance calculation.
Figures 3 and 4 showed that most Al2O3 and Ce2O2S did not existed before solidification and AlCeO3 and Ce2O2S also did not existed after final solidification with T.[O]:0.0012%. Indeed, only Al2O3, Ce2S3, CaS and MnS existed after final solidification with T.[O]:0.0012%. And actual inclusions in calculation should have appeared at least, but did not do. Above computing results were completely inconsistent with the actual inclusions. A better computing results, for the actual inclusions, Al2O3, AlCeO3, Ce2O2S and CaS(MnS) should appear in turn and finally existed after solidification. Firstly, Al2O3 had existed in molten steel before adding Ce. Secondly, T.[O] included dissolved oxygen ([O]) and undissolved oxygen (Al2O3 for Al-killed steel), meaning that selecting oxygen should be considered again. AlCeO3 should not be decomposed or disappeared after solidification. Thirdly, the [O] should be chosen, which was helpful to form Ce2O2S. Finally, decomposing or disappearing of the formed inclusions should be considered difficult, which was more agree with reality. Park’s research 23 also showed that some phases appear, which should have appeared in thermodynamic balance. It also indicated that decomposing or disappearing of the formed inclusions was difficult.
New balance computation
Decomposing or disappearing of formed inclusions in steel was considered difficult and it was assumed that inclusions once were formed and were not decomposed or disappeared in new balance computation. Before computation, dissolved oxygen ([O]) was needed. Before adding Ce to molten steel, dissolved oxygen ([O]) was 0.0004% and final total oxygen (T.[O]) was 0.0012%. In T.[O], T.[O]-[O] was from Al2O3 in Al-killed molten steel. In computing, Al2O3(T.[O]-[O]) was not chosen and [O] was only chosen, which results were showed in Figures 5 and 6.
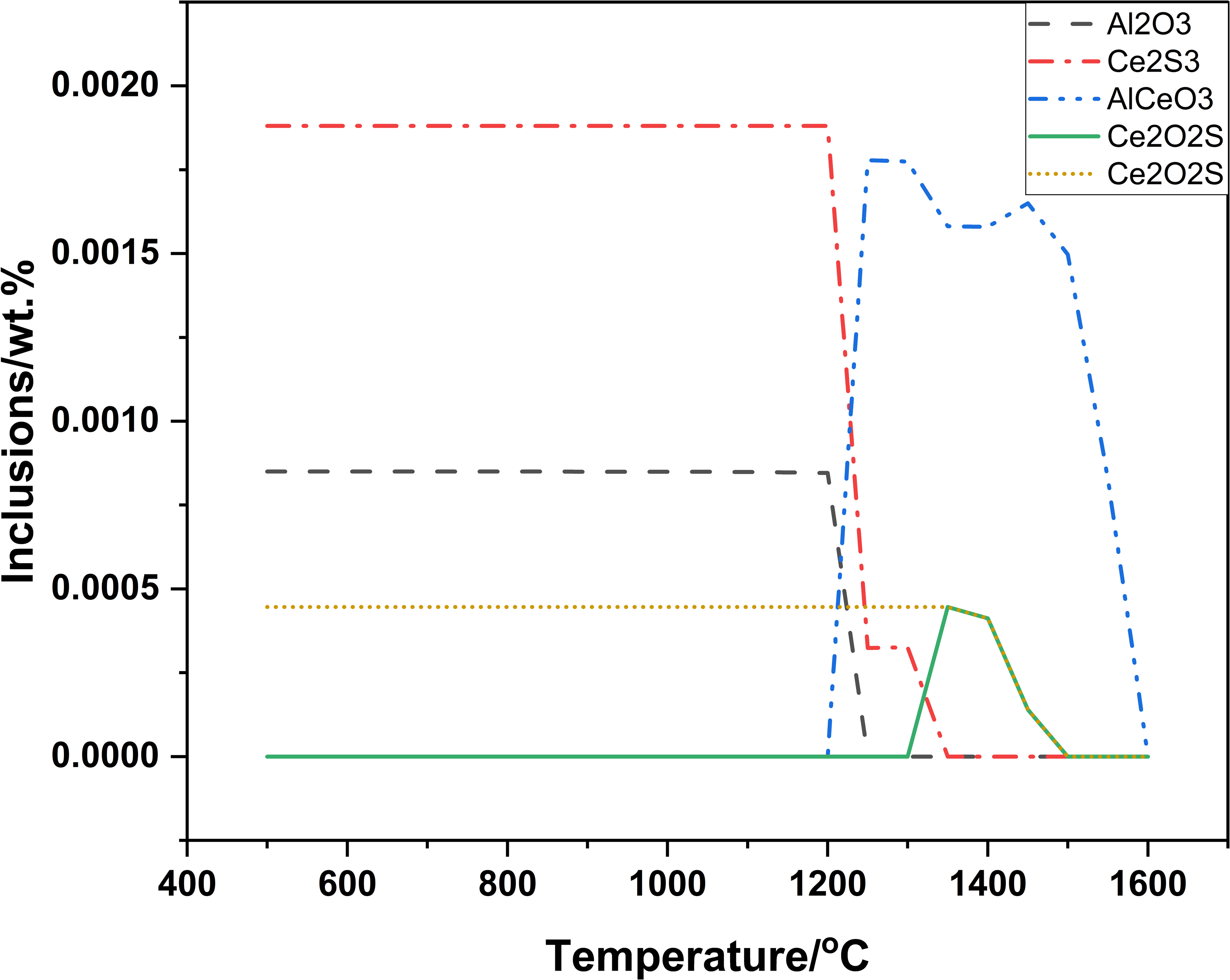
Change of inclusions in process of cooling under [O]:0.004%.
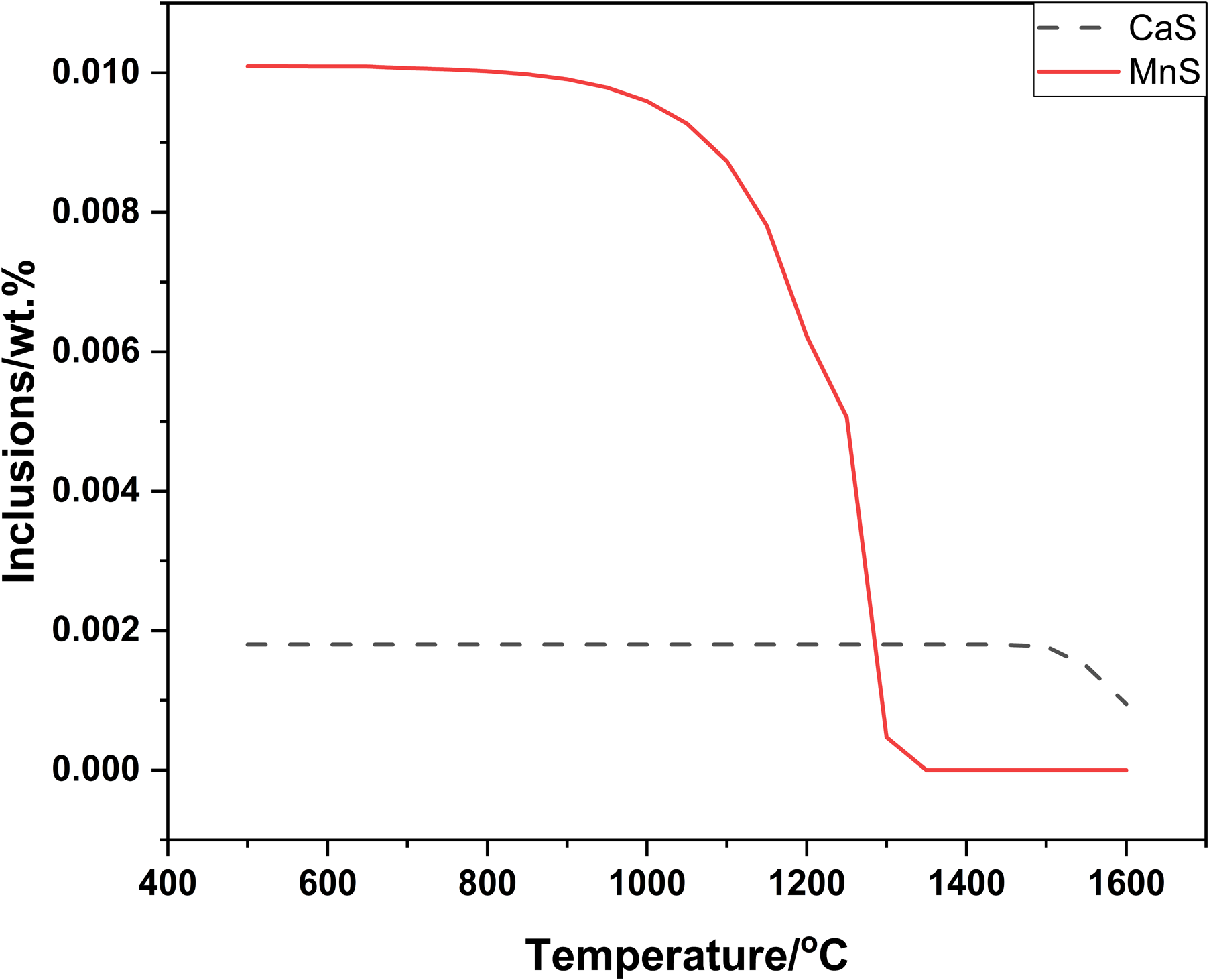
Change of Ca-Mn-S inclusions in process of cooling under [O]:0.004%.
Figure 5 showed that AlCeO3 and Ce2O2S were formed in turn before solidification. The dotted lines were that inclusions were decomposed in cooling process and solid lines were that inclusions were undecomposed in cooling process. Figure 6 showed that CaS, Ca(Mn)S and MnS were formed in turn. Note that decomposing of AlCeO3, Ce2O2S and Ca(Mn)S should be eliminated in Figures 5 and 6. Based on above mentioned, computing results for cerium-based inclusions were firstly Al2O3(exist in molten steel before adding Ce), AlCeO3, Ce2O2S, CaS, Ca(Mn)S and MnS in turn. And it was in better agreement with the actual inclusions.
Fraction of inclusions
Inclusions on smooth broken surface of two samples were automatically collected and counted using SEM with EDS. Figure 7(a) and (b) showed the fraction of the inclusions with average size and number at 16 mm−2. Compared to inclusions without Ce, size of Oxide-Ca(Mn)S and MnS was reduced, number of oxide-Ca(Mn)S was increased, number of MnS was reduced and xCaO yAl2O3 disappeared in inclusions with Ce. According to calculation result in Figure 5, xCaO•yAl2O3 did not existed and the Ce2O2S existed, which would consume partial content of S. And Ca(Mn)S also was formed on surface Ce2O2S, which was the main reason for deducing size and number of MnS. Because Ce has a stronger binding force with O(Al) and S(O), it was easy to fine inclusions. According to the fraction of inclusions, inclusions of steel with Ce were of benefit to improve toughness.
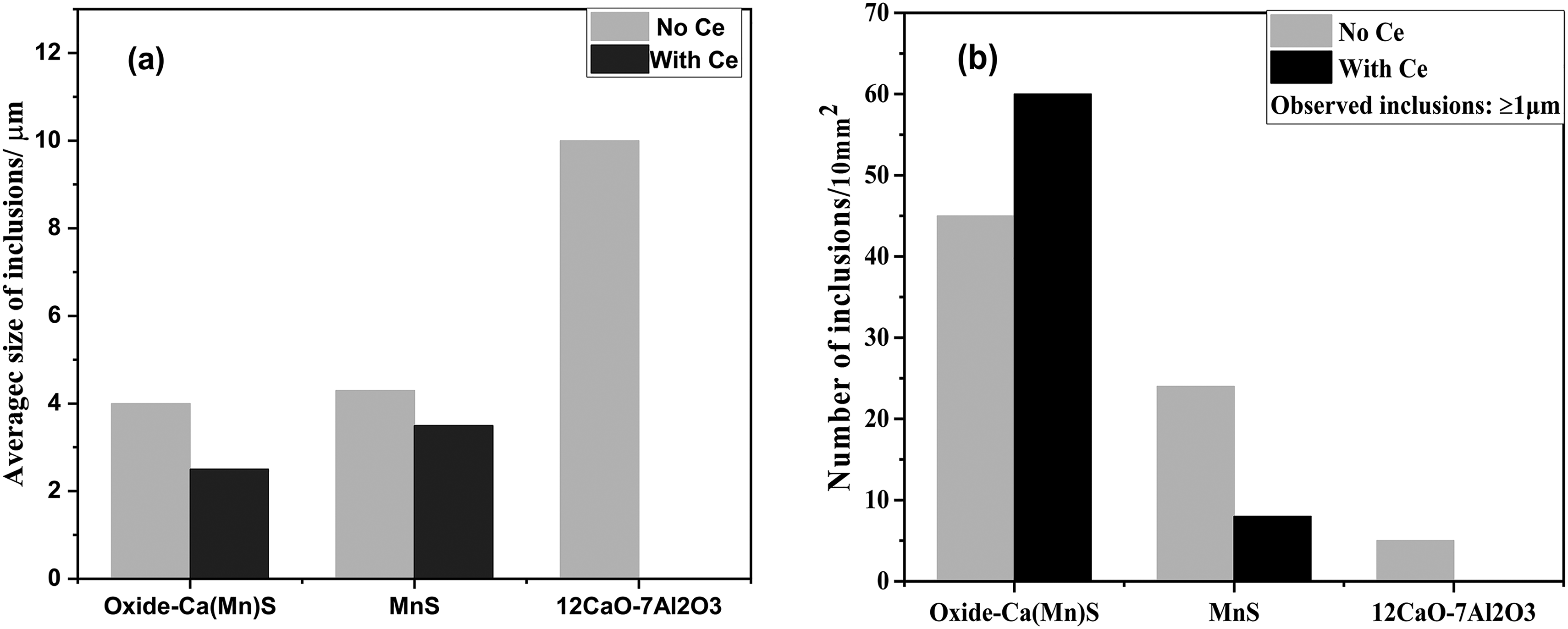
Average size(a) and number(b) of inclusions of steel.
Effect of Ce on microstructure and analysis
Microstructure of casting and rolling state
Figure 8 showed effect of Ce on solidification structure. And the solidification structure with Ce was more refined. And photos of rolling structure were shown in Figure 9, showing that grains with Ce were more refined than grains without Ce in steel.
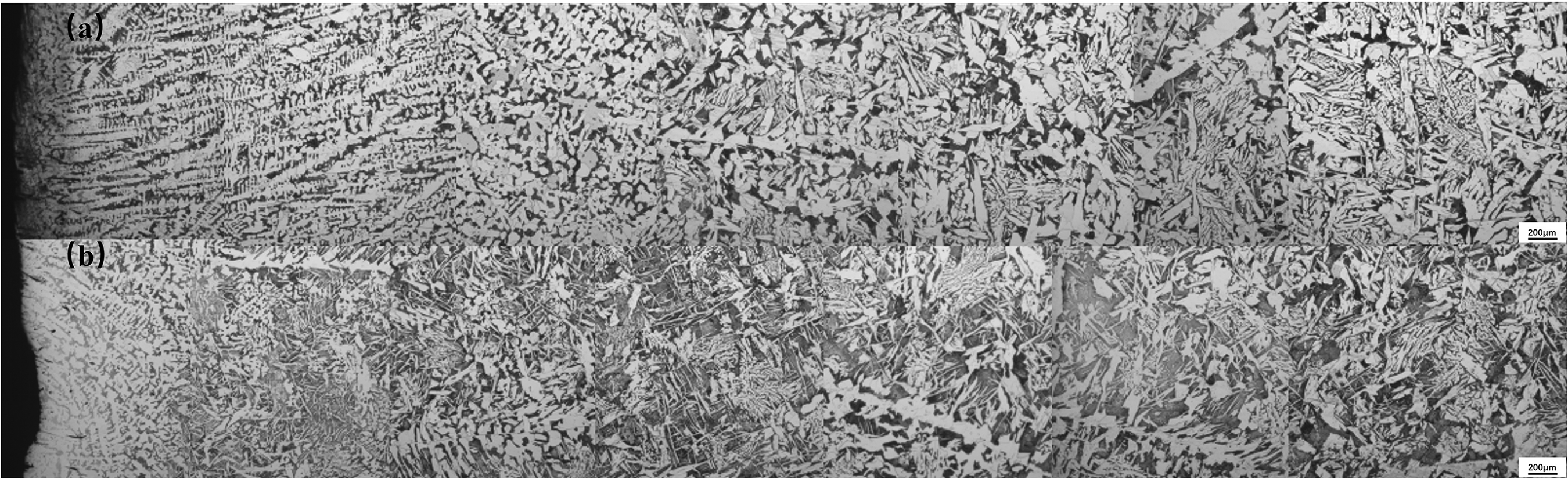
Photos of solidification structure (a)without Ce and (b)with Ce.

Photos of rolling structure (a), (b), (c) without Ce and (d), (e), (f) with Ce.
Disregistry of Ce-based inclusions with ferrite
Two-dimensional lattice misfits were widely used as effective tool to proof effectiveness of inclusions offering nucleation for Ferrite. The boundary energy between the heterogeneous nuclei and the crystallisation phase had an influence for nucleation and it depended on the structure of the two contacting crystalline faces (arrangement condition, atom size and interatomic spacing of the crystal faces).1,3 The value of the two-dimensional lattice misfits usually evaluated where some inclusions may act as the heterogeneous nuclei. The two-dimensional lattice disregistry mathematical model from Bramfitt
24
is: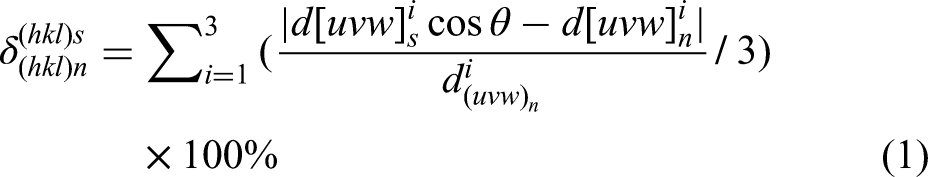
In the process of γ-Fe→α-Fe, the disregistry of high temperature was more effective for heterogeneous nuclei between α-Fe and inclusions. Thus, 800°C was selected as calculated temperature of the disregistry. Outside of the Ce-based complex inclusions was Ce2O2S(main), Ca(Mn)S(main) and CeAlO3(less). And thus, the relevant crystal structure parameters were listed in Table 2. At 800°C, results of calculation of two-dimensional lattice disregistry are shown in Tables 3–5. The lattice disregistry between (0001) Ce2O2S and (111) α-Fe is 1.03% in Table 3. And the lattice disregistry between (100) CeAlO3 and (111) α-Fe is 17.08% in Table 4. And the lattice disregistry between (111) CaS and (111) α-Fe is 0.47% in Table 5. From small to large, the disregistry is CaS, Ce2O2S and CeAlO3, in which CaS and Ce2O2S were the most effective heterogeneous nuclei. Therefore, the Ce-based complex inclusions could act as an effective heterogeneous nucleation in this case. MnS separated out from γ-Fe and γ-Fe → α-Fe in cooling process. MnS could form a solid solution with CaS inclusion, which makes Mn poor areas around the inclusion. And it benefits nucleation of α-Fe. For the type of nucleation for Ferrite, bigger inclusion could offer more sites for nucleation of α-Fe, which was helpful to form type II (inclusion at least in two Ferrite grains).
The relevant crystal structure parameters of inclusions and α-Fe.

Results of calculation of the lattice disregistry of Ce2O2S and α-Fe.

Results of calculation of the lattice disregistry of CeAlO3 and α-Fe.

Results of calculation of the lattice disregistry of CaS and α-Fe.

Effect of Ce on impact toughness
Figure 10 showed experimental values of impact toughness tested at 0 and −60°C. The values induced from 0 to −60°C, where the values with Ce were higher than that without Ce. Compared with samples without Ce, the values of impact toughness were increased by 161% at 0°C and 225% at −60°C, respectively, indicating addition of Ce could markedly improve low-temperature impact toughness.
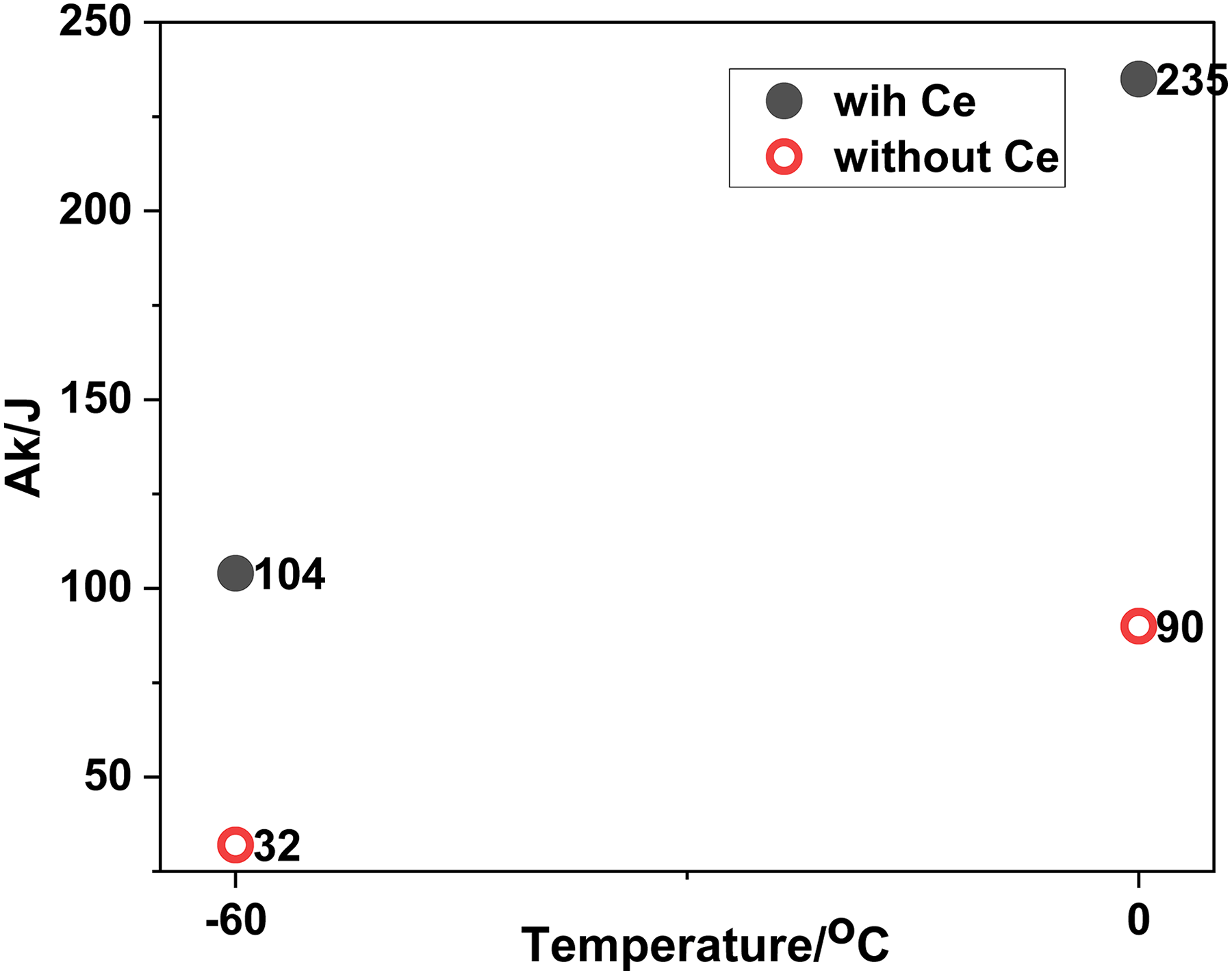
Values of impact toughness tested at 0 and −60°C.
Figure 11 showed the impact fracture morphologies at tested temperature. Beginning from V-notch, the stretch zone with Ce almost occupied all fracture surface and the stretch zone without Ce occupied around half of fracture surface at 0°C. The stretch zone could be seen as a sign of value of impact toughness, which meant that larger zone had higher value of impact toughness under the same conditions. The zone with Ce occupied around 1/3 of fracture surface and without Ce occupied nearly zero at −60°C. Accordingly, the river zone showed opposite result.

Impact fracture morphologies (a), (c) at 0°C and (b), (d) at −60°C.
Figure 12 showed the impact fracture microstructure at 0 and −60 °C, which presented typical dimples cracking, cleavage cracking and mix cracking of dimples and cleavage. The fracture surface with Ce revealed fine dimples and thick dimples away from V-notch at 0°C. The fracture surface without Ce revealed thick dimples and cleavages at 0°C. The fracture surface with Ce revealed fine cleavages and without Ce revealed thick cleavages at −60°C. Overall, the fracture surface with Ce had finer dimples and fine cleavages than that without Ce, which meant that grains with Ce would be finer than that without Ce.

Impact fracture microstructure (a), (c), (e), (g) at 0°C and (b), (d), (f), (h) at −60°C.
As known, fine grains usually had a better value of impact toughness. And in this case, what grains with Ce had much finer than that without Ce had been observed and discussed in the previous section. A suit content of Ce in steel could modify angular Al2O3 inclusions to spherical or ellipsoid inclusions 26 and forming inclusions bearing Ce would be smaller26,27. As above mentioned, inclusions had been modified and were smaller in this case. And it was benefit to low-temperature impact toughness. About fine modified inclusions, morphology of a Ce-based complex inclusion with about 3μm was also observed on fracture surface, showed in Figure 13.
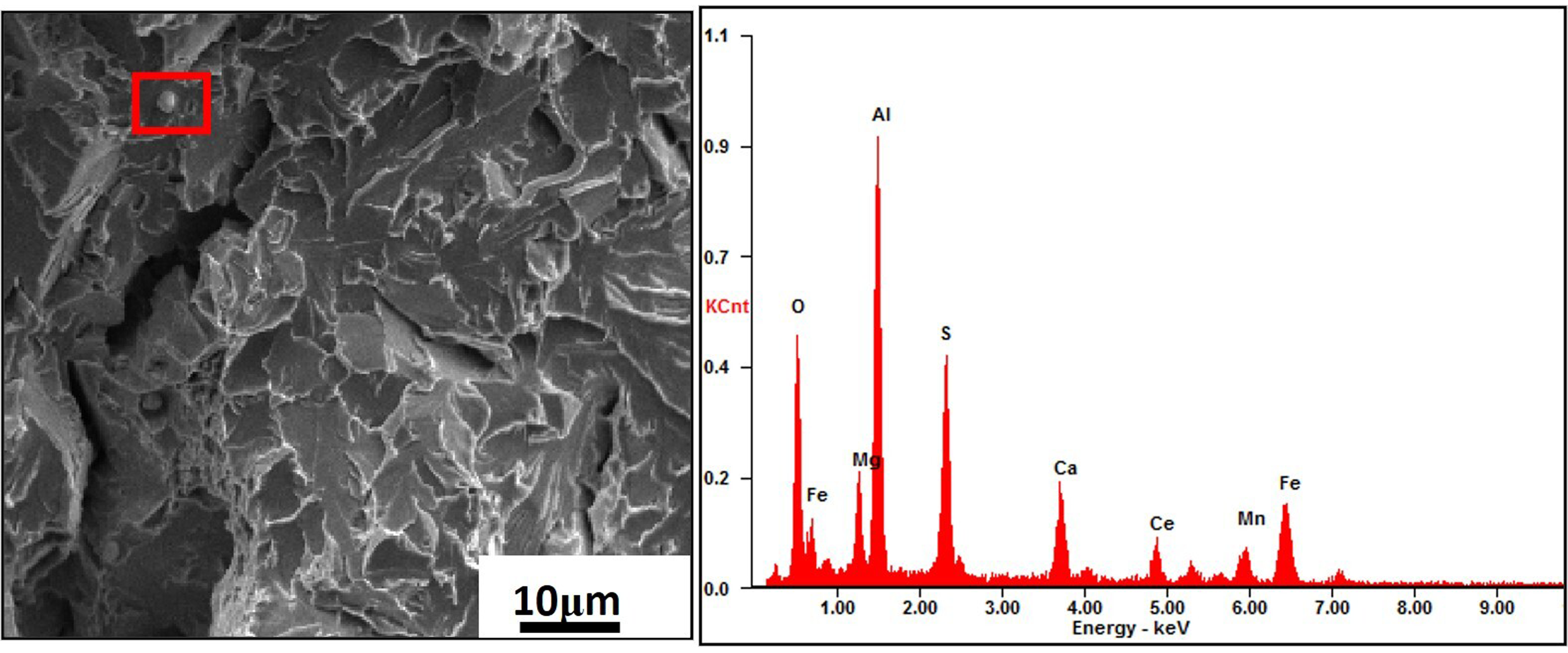
Morphology and EDS of inclusion bearing Ce.
Conclusion
In this article, mechanism of cerium-based inclusion formation and influence on impact toughness were researched in industrial Al-killed steel (Q355B/D). And content of Ce was 0.0014%. The major results are listed below:
The actual inclusions were complex, consisting of Al-O-Ce (Ce-poor), Al-O-Ce (Ce-rich), Al-O-Ce-S(Al-poor) and S-Ca (Mn-rich) from inside to outside in industrial Al-killed steel (Q355B/D). Results of thermodynamic balance calculation were completely inconsistent with the actual inclusions. Considering decomposing or disappearing of formed inclusions in steel difficult, new computing results showed that inclusions were formed by firstly Al2O3 (exist in molten steel before adding Ce), AlCeO3, Ce2O2S, CaS, Ca (Mn)S and MnS in turn, in better agreement with the actual inclusions. Outside of the actual inclusions, Ce2O2S and CaS had a smaller disregistry with ferrite, which could offer effective nucleation sites. And small inclusion was surrounded in a grain and bigger inclusion was surrounded in two grains at least. Grains with Ce were finer than grains without Ce. Compared with samples without Ce, the values of impact toughness were increased by 161% at 0°C and 225% at −60°C, respectively. And fracture surface with Ce had more and finer dimples and finer cleavages than that without Ce. And it indicated addition of Ce with 14 ppm could markedly improve low-temperature impact toughness.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Central Guidance on Local Science and Technology Development Fund of Inner Mongolia Autonomous Region under Grant [2020ZY0034].
