Abstract
Objective
To evaluate effect of growth hormone (GH) therapy in craniofacial development in children with or without growth hormone deficiency.
Method
The systematic review was conducted by searching the electronic databases—PubMed, Cochrane clinical trials register, Google scholar, and EBSCO host. Randomized controlled trials and observational studies published till January 2022 were included. The risk of bias was assessed by Risk-Of-Bias VISualization (ROBVIS) tool. The risk of bias of the included studies was presented as a “traffic light” plot of individual studies and a summary diagram. Data was studied by two authors and any dispute was resolved by another reviewer.
Result
A total of 964 articles were obtained through electronic database search. After eliminating the duplicates, 13 articles met inclusion criteria. Of the 13 studies, 8 studies had moderate risk of bias, 2 had serious risk of bias, and 1 had critical risk of bias as per the ROBINS-I assessment. According to the RoB 2, two studies were having high risk of bias.
Conclusion
The maxillary length, the mandibular length, and the cranial base length increased post-GH therapy, causing an overall increase in the facial height. The limitation of GH therapy was in the transverse direction as maxillary and mandibular arch dimensions remained almost equal to those in the untreated subjects. Dentition had minimal influence of GH therapy.
Registration
The systematic review was registered in the International Prospective Register of Systematic Reviews database under CRD42021257044.
Introduction
Understanding the growth and development of craniofacial complex is an integral part of orthodontics. Since the beginning of dentofacial orthopedics, efforts have been made to fabricate appliances for modifying/redirecting the facial growth in a favorable way. Genetics, hormones, nutrition, and environment are some of the multiple factors that impact craniofacial growth. 1
Growth hormone is one such hormone which has a significant role in craniofacial growth. An excess or lack thereof causes jaw deformities which may hamper the aesthetic harmony of face due to disproportionate facial features. GH, which is secreted by the pituitary gland, plays an important role in longitudinal bone growth. 2 Growth hormone deficiency (GHD) is a disease that leads to growth disturbances, mainly short stature, because of inhibited pituitary gland hormones. The etiology can be classified as idiopathic, organic, or genetic. 3
Since the first patient was treated with pituitary-derived GH in 1958, 4 numerous studies have documented beneficial effects of GH treatment on linear growth in both GH-deficient and non-GH deficient subjects (idiopathic short stature).5, 6 Study by Segal et al 7 focused on effects of GH on other GH-dependent areas such as hand and foot size apart from head circumference.
A case report by Newman and Newman 8 included a longitudinal study of six years of an 11-year-old male with hypopituitarism and retrognathic mandible. GH along with orthodontic therapy enhanced the condylar growth and thus increased the mandibular length post treatment. Wang et al 9 conducted an experiment on adolescent rat model to analyze the role of GH in supplementing orthodontics/functional appliance therapy and found enhanced condylar growth at the end of experiment. Another case of a girl with idiopathic short stature reported by Hwang and Cha 10 showed positive effects of GH therapy with orthodontic treatment on facial profile improvement. The lack of randomized controlled trials (RCTs) in this subject could be attributed to ethical issues of leaving a group of patients untreated as the effect of GH is dependent on the age at which therapy is started other than the length of administration.
The primary question was—“Does growth hormone therapy play a role in enhancing craniofacial growth in growth hormone-deficient children or those who may not have growth hormone deficiency (such as idiopathic short stature)?” This systematic review aims to shed light on the existing studies on GH therapy and its effects on craniofacial development in children. Secondary aim was to assess whether GH therapy supplements orthodontic treatment in improving the profile of children with or without GHD.
Material and Methods
Protocol and Registration
This systematic review was registered in the International Prospective Register of Systematic Reviews database under CRD42021257044 (PROSPERO database (
It followed the PICO criteria 11 where the population (P) was children with or without GH deficiency, intervention (I) was GH administration, control was children who did not receive GH therapy, and outcome (O) was changes in the craniofacial development.
Selection Criteria
Inclusion criteria:
RCTs, controlled clinical trials, and prospective and retrospective longitudinal studies. No restrictions were set for sample size. Only human studies were included. Articles written in English were included. Studies published till January 2022 were included.
Exclusion criteria:
Abstracts, lab investigations, descriptive studies, case reports, case series, reviews, and papers with an opinion were disregarded. In order to eliminate the effects of genetics and other interventions as additional confounding factors, studies done only on syndromic patients (such as Down syndrome, Silver Russell syndrome, etc.) and involving other interventions such as therapy with other hormones along with GH therapy were excluded.
Search Strategies
Two writers (SK and US) each conducted the primary research for this systematic review, while the third author (SB) dealt with any conflicts. From their launch date until January 2022, the following electronic databases were searched: PubMed, Google Scholar, Cochrane Library, and EBSCO host. On the basis of the lists of references from the articles that were obtained from the main databases, a number of manual searches were also carried out. There were just English-language articles present.
Medical Subject Headings (MeSH) terms were mixed with keywords, free-text phrases, and the proper Boolean operators for PubMed in the search process. Each database required a different search approach. A similar collection of MeSH terms and phrases was used for an advanced search on each database. Table 1 lists the specific MeSH phrases and keywords, as well as the combinations of MeSH terms and keywords or free-text words, that were employed in various databases.
MeSH Terms and Keywords Used for Research.
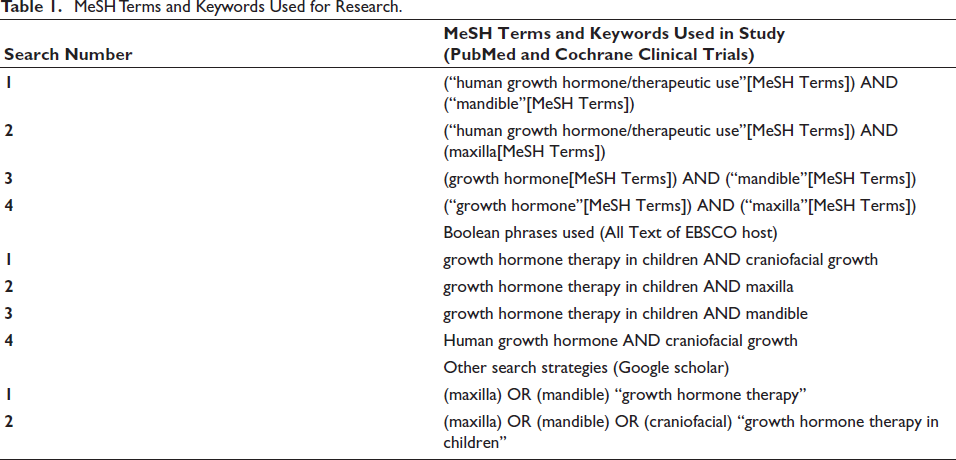
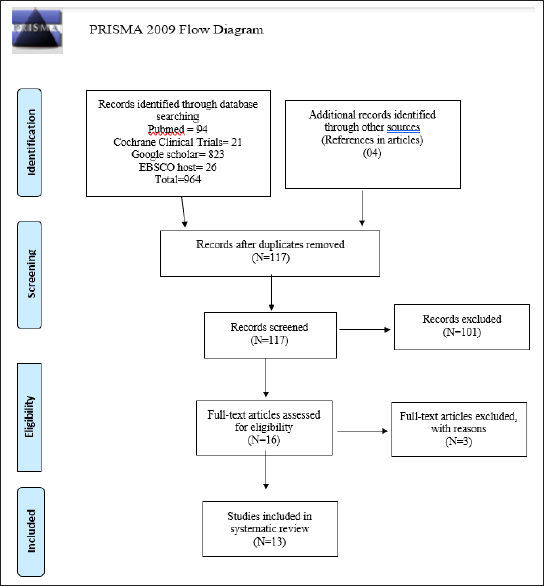
The list of search engines used and the results obtained are presented in Table 2. Table 3 presents the articles that were excluded after reading along with the reason for exclusion. The studies’ identification and selection process is displayed as a PRISMA flow diagram, as seen in Figure 1.
List of Search Engines and the Results Obtained.

List of Excluded Studies.
Results
A total of 964 articles were identified after the electronic search (PubMed, Cochrane Library, Google Scholar, EBSCO host). Duplicate articles were eliminated and 117 articles were selected for primary screening of titles and abstracts. A total of 16 articles qualified for full-text reading out of which 13 articles finally met the inclusion criteria and were included in the systematic review. Due to heterogeneity of method of assessment, study sample, intervention, and duration of therapy, meta-analysis could not be done.
Data was collected under the following headings: general information (the name of the authors and the year of publication), participants (sample size, age, and gender), intervention, method of assessing changes due to GH therapy, primary aim of the study, and conclusion.
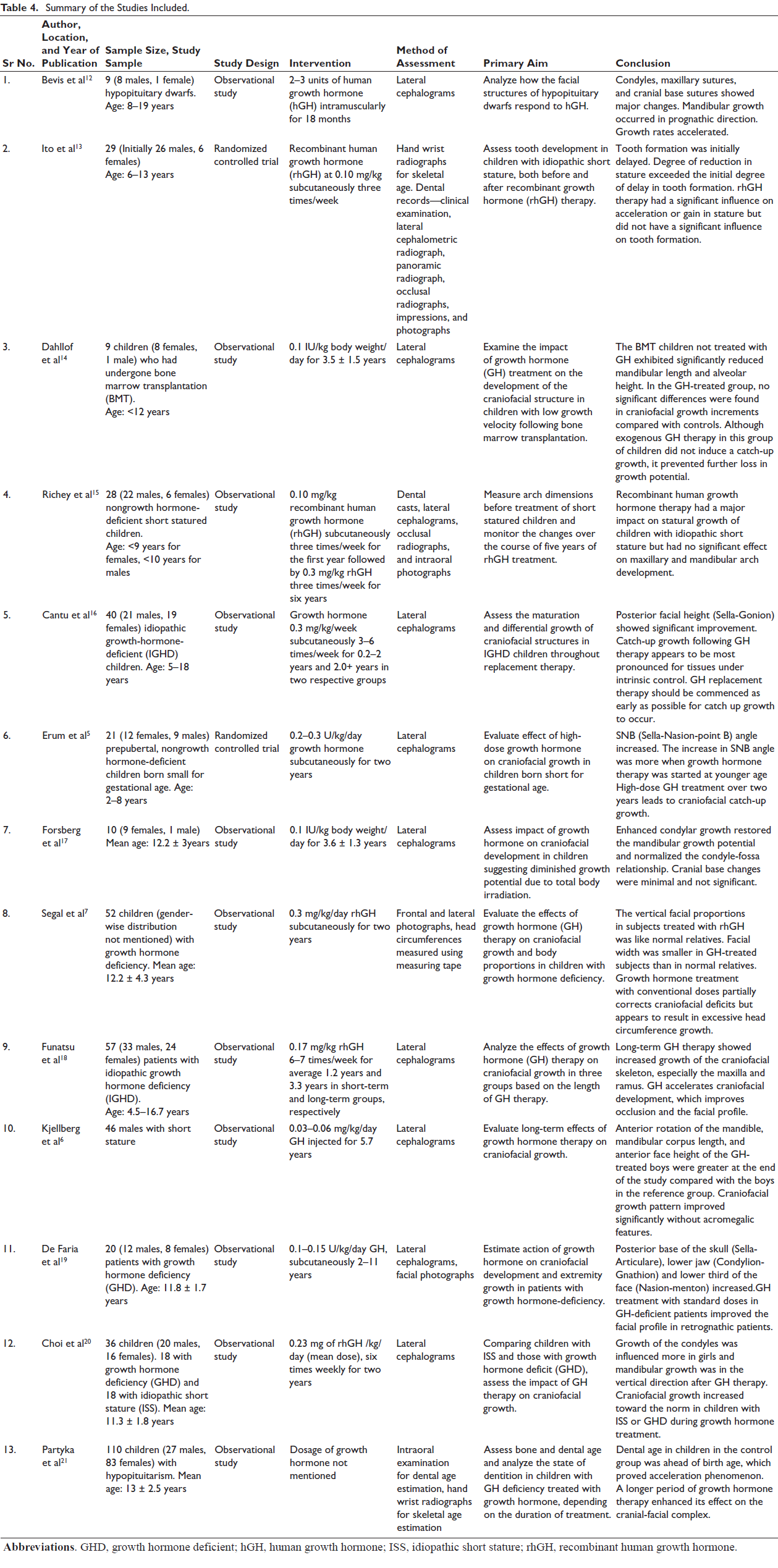
A summary of the included studies has been shown in Table 4.
Summary of the Studies Included.
Risk of Bias Assessment
The risk of bias was assessed by Risk-Of-Bias VISualization tool (ROBVIS) tool. The risk of bias of the included studies is presented as a “traffic light” plot of individual studies and a summary diagram (Figures 2–5). The domains of bias that were assessed are mentioned in Figures 2 and 3. The ROBINS-I tool for nonrandomized studies and RoB 2 tool for randomized controlled studies were used in the ROBVIS. Of the 13 studies, 8 studies had moderate risk of bias, 2 had serious risk of bias, and 1 had critical risk of bias as per the ROBINS-I assessment. According to the RoB 2, two studies were having high risk of bias. The overall summary of risk of bias is presented in the summary graphs.
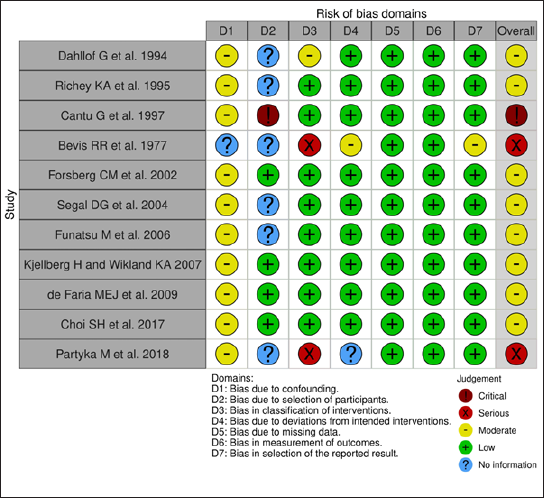
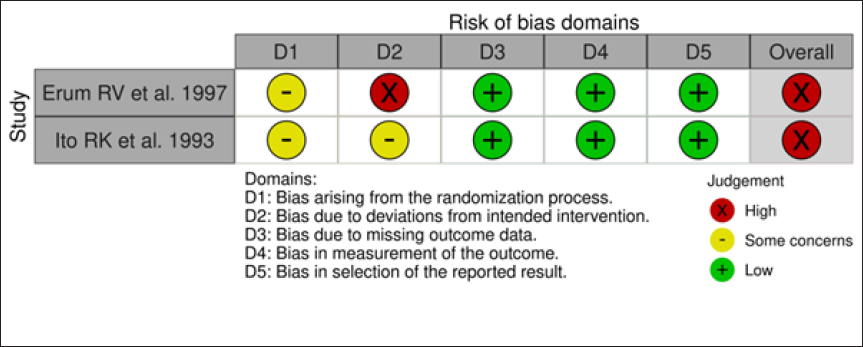
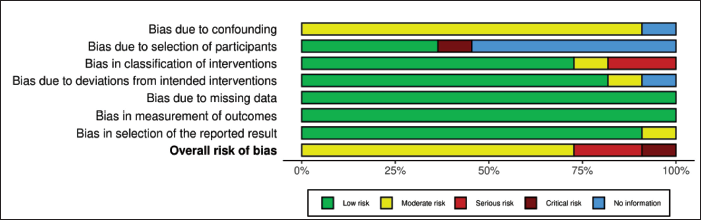
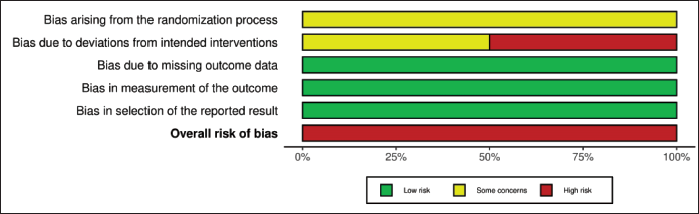
Discussion
The aim of the present systematic review was to assess whether GH therapy enhances the craniofacial development, especially the dimensions of maxilla and mandible in children with or without GH deficiency. A total of 13 studies met the eligibility criteria and were included in this systematic review and the risk of bias assessment was done using ROBVIS tool.
All the studies except the study conducted by Richey et al and Partyka et al used serial lateral cephalograms to analyze and measure the growth changes occurring in the study subjects. Richey et al as well as Bevis et al included dental casts to measure maxillary and mandibular arch width changes after GH therapy.12, 15 Partyka et al used hand wrist radiographs and conducted intraoral examination for skeletal age and dental ages determination, respectively. 21 Photographic and anthropometric measurements were involved in studies done by Segal et al, 7 Ito et al, 13 Richey et al, 15 and Faria et al. 19
Craniofacial components such a mandibular length, ramal length, anterior facial height, and anterior cranial base length showed a positive and clinically significant change in dimensions after GH administration. Transverse dimension of maxillary and mandibular arch did not show significant change. Evaluation of transverse dimension on study models by Richey et al 15 and Bevis et al 12 showed similar results and proved that GH therapy had minimum effect on it and was clinically insignificant. The maxillary and mandibular arch dimensional changes noted by Bevis et al ranged between 0 and 1 mm with majority of the values less than 0.5 mm. 12
One of the most common aesthetic problems observed in patients with GH deficiency is facial convexity. Due to the anterior growth of mandible, the facial angle improved as per the results of three studies. Even though the corpus length was not much affected by GH replacement therapy, the total mandibular length (Condylion-Pogonion) and ramal length (Articulare-Condylion or Condylion-Gonion) were significantly increased and this was attributed to the accelerated growth of condylar cartilage. Condylar growth direction changed from forward-backward direction to backward-upward direction as observed by Forsberg et al. 17 According to Ito’s study, mandibular growth not only improved but exceeded than those in control group. However, disharmony of facial profile was not observed. 13 Increase in the ramal height was observed in three studies. However, the catch-up growth in ramal height was the least as per the results of Cantu et al. 16
A positive effect of GH therapy on maxillary length was found in only two of the six studies that measured maxillary length changes. The improved growth in maxillary length as explained by Kjellberg might be due to stimulative effect on membranous as on endochondral bone formation by growth. 6
Changes in the anterior cranial base were reported in eight studies. All suggested a positive effect of growth on sphenooccipital synchondroses which led to the lengthening of anterior cranial base. It was also noted that the effect of GH on this synchondroses is however age dependent and thus failed to have much impact on the catch-up growth in the subjects above the age of seven years, which is approximately the age of fusion of sphenooccipital synchondroses. 20 Similarly, posterior cranial base length and posterior facial height also increased significantly in all the four studies which included them as a parameter.
Dental effects were specifically paid attention to by the study conducted by Partyka et al, 21 Ito et al 13 where they found that dental age was retarded or delayed in children with GH deficiency and in children with idiopathic short stature, respectively. Tooth formation or dental age was not significantly influenced by GH therapy in either of the studies. Bevis et al also concluded that dental age improved only slightly as compared to other parameters. 12
Finally, only one study by Kjellberg et al included myofunctional appliance therapy in certain subjects and found that it supplemented the effect of GH in improving the mandibular growth in a sagittal direction. 6
Although there was a moderate risk of bias in most of the studies, all studies suggested that the efficacy of GH therapy depends on the timing of administration as growth of different craniofacial components ceases at different times. The earlier the administration of GH, the better is the corrective growth of maxilla and mandible in a sagittal direction. This favorable growth of mandible improved facial profile and made it more harmonious. Maxillary and mandibular arch dimensions did not improve significantly. Dental changes were also not considerable post GH therapy.
Limitation
The limitation of this systematic review was the limited number of RCTs available to know whether there is additive effect of GH therapy in children undergoing myofunctional therapy. A meta-analysis could not be carried out as there was a lot of heterogeneity among studies. All studies displayed heterogeneity regarding the parameters assessed on lateral cephalograms to measure effect of GH.
Future Scope
Future research is required to assess whether GH therapy in conjunction with myofunctional therapy can accelerate profile improvement in children with a retrognathic profile. Also, it is necessary to evaluate whether targeted supplementation of GH in adults undergoing orthodontic treatment can aid in improving sagittal dimensions of maxilla and mandible as most of the present studies were conducted only on children. Targeted supplementation of GH may not only improve a retrognathic, convex profile but also aid in correcting facial asymmetry caused by conditions such as unilateral mandibular hypoplasia.
Conclusion
GH therapy plays a significant role in craniofacial development of children with or without GH deficiency and has an enhancing effect on the maxillary length, the mandibular length, and the cranial base length, causing an overall increase in the facial height.
The limitation of GH therapy is mainly in the transverse direction as arch dimensions remained almost equal to those in the untreated subjects.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not applicable
