Abstract
Introduction:
Fluoride agents to prevent white spot lesions are used often during orthodontic treatment. The beneficial effects of fluoride, when consumed within permissible limits on dental structures, are well known. Their implications on underlying biological tissues, however, are unknown. Mouthwashes and dentifrices with fluorides are associated with metal ion release into the mouth with possible cell genotoxicity. Since these cariostatic agents are frequently used during orthodontic therapy, a deeper understanding of the effects of fluoride on oral tissues was considered necessary.
Methodology:
Three groups of patients (30 each)—group 1 (untreated controls), group 2 (non-fluoridated), and group 3 (Fluoridated) were analyzed. Patients in groups 2 and 3 were bonded with the same bracket prescription and treated with similar archwire sequences. Buccal mucosal cells at 4 specific time periods (before treatment, 1 week, 30 days, and 6 months) were collected, using a wooden tongue depressor, and assessed for any nuclear abnormalities. Comparisons of changes were made with an untreated control group and also between the non-fluoridated and fluoridated groups. Relevant conclusions were drawn after analysis of the results.
Results:
Greater number of nuclei were observed at the 30-day time interval in the fluoridated group, which was statistically significant at P < .001.
Conclusion:
Use of fluoridated oral hygiene products in patients undergoing fixed orthodontic treatment with NiTi archwires could increase the risk of micronuclei formation in buccal mucosal cells.
Introduction
According to a consensus report by International Agency for Research on Cancer (IARC) in 1992, the definition of genotoxicity is broad and includes both direct and indirect DNA effects: (a) induction of molecular-related mutations (gene, chromosomal); (b) indirect surrogate mutagenesis-related events (eg, sister chromatid exchange); or (c) DNA damage, leading to mutations.
Genotoxic factors cause disruption to the genetic material in cells in the form of single- and double-strand breaks, cross-linking, structural and numerical chromosomal aberrations, etc. 1 To assess genotoxicity, micronuclei are used to assess and bio-monitor carcinogen genotoxicity poisoning due to heavy metals, toxins, etc. 2 The benefit of the micronucleus test is due to simple and effective analysis of chromosome mutations in cytological samples relative to the determination of chromosome aberrations and sister chromatid exchanges. 3
Fluoride levels, if within limits in water, have beneficial effects on teeth and reduce dental caries, while prolonged fluoride toxicity above this limit induces both skeletal and dental fluorosis and organotoxicity.4–6
In order to avoid white spot lesions (WSL), orthodontists usually recommend fluoridated toothpastes and mouthwashes to patients. While there are many positive benefits of fluorine, its use is toxic to both teeth and the human body above acceptable limits. It is extremely important that we analyze not just its dental effects but also effects on the surrounding tissues and buccal mucosa. Brackets and archwires used in fixed orthodontic therapy are primarily made of stainless steel, nickel, chromium, and titanium as constituents. Genotoxicity and cell arrest of buccal mucosal cells could result due to leaching of metal ions from fixed appliances on exposure to fluorides during treatment.
The current research used a micronucleus assay to examine buccal mucosal cells of patients (with or without fluoride exposure) for any nuclear irregularities. In both groups, comparisons were made with untreated groups of subjects as a means to reach definite conclusions. Cells were collected, using the technique of exfoliative cytology, and were subjected to micronucleus assay testing, to assess the levels of fluoridated agent–induced toxicity.
This form of in vivo investigation is new and has not been carried out till date. The current research aims to shed light on the biological effects of fluorides, at a cellular level, in groups of human subjects, using oral hygiene care products that are non-fluoridated and fluoridated. The null hypothesis tested was that there would be no differences in buccal mucosal cells in both fluoridated and non-fluoridated groups at all tested time intervals. The results, if significant, could pave the way for determination of safe levels of fluorides in oral hygiene products in future well-planned human studies.
Materials and Methods
A total of 90 subjects (68 females and 22 males) were divided into 3 groups of 30 each. The sample size was finalized using G Power software version 3.1.9.2 after performing a power analysis. Considering the effect size to be measured (f) at 40%, power of the study at 80%, and the margin of error at 5%, the total sample size needed was 66. The final sample size was rounded off to 75. So, each study group comprised 25 samples. An additional 5 samples per group were included to consider dropouts if any.
Group 1 comprised untreated controls; groups 2 (non-fluoridated) and 3 (fluoridated) were patients requiring fixed orthodontic treatment. Inclusion criteria were patients requiring non-extraction fixed orthodontic treatment between the ages of 12 years and 35 years, all permanent teeth till second molars to be present, nonsmokers, teetotalers, and no medications like antibiotics used in the preceding 6 months. Ethical approval was granted prior to beginning and written informed consent obtained from all participating subjects. A single investigator treated all patients in the study. A total of 90 subjects comprised the sample, which was further subdivided into 3 subgroups. The orthodontically treated patients had similar appliances during the 6-month evaluation period with similar archwire sequencing so that comparisons could be made. An equal number of untreated controls were included for assessment and making comparisons. Patients had orthodontic appliances bonded in both arches at the start of the treatment. Stainless steel brackets (Mini Twin 0.022 slot, Ormco Corporation, Glendora, CA) were bonded using Enlight light-cured adhesive (Ormco Corporation). Only nickel titanium archwires were used in the study. The archwires used were 0.014″ NiTi, 0.016″ NiTi and 16 × 22″ NiTi till 6 months of treatment.
All participants were taught oral hygiene methods prior to commencing treatment. The untreated control group and group 2 subjects used only non-fluoridated toothpaste (Dabur Red, Dabur India Ltd, India) for maintaining oral hygiene twice daily. Group 3 participants used fluoridated mouthwash and toothpaste twice daily (Colgate Plax, 225 ppm fluoride, Colgate Strong Teeth, 1000 ppm fluoride Colgate Palmolive Co, India).
Method of Collection of Samples
After obtaining written consent from patients, sample collection was done at 4 time periods (before treatment,1 week, 30 days, and 6 months) for all 3 groups of subjects in the study. Samples collected before treatment would allow the researchers to determine if any cellular changes were already present in any of the samples leading to inherent bias. Leaching of metal ions is known to occur as early as 7 days after appliance placement with maximum leaching occurring at the 1-month time interval. Since orthodontic treatment takes a relatively long time, the 6-month evaluation period was also considered in order to make definitive conclusions. 7
Sampling Procedure
Before obtaining samples, subjects were asked to rinse with water in order to remove debris. Oral mucosa scrapings were taken with a sterilized wooden tongue depressor, which was made moist prior to use. The buccal mucosa on both right and left sides were used to obtain scrapings.
The mucosal scrapings were smeared onto microscopic slides, which were cleaned previously. All smears were air-dried and fixed using 80% methanol within 24 h.
Staining Procedure
The Feulgen technique of staining was utilized in this study, with modifications, as suggested by Tates and Neuteboom et al. 8 Slides were rinsed using distilled water for 10 min at room temperature and then subjected to hydrolysis in 1 N HCl at room temperature for 1 min followed by 5 N HCl for 1 h and then again in 1 N HCl at room temperature for 1 min. The slides were then rinsed 3 times with distilled water. Schiff reagent was used for an hour for staining. Slides were then rinsed in bisulfite solution three times (2 min each time) and rinsed well in water. Slides were counterstained with 1% light green in 1% acetic acid for 40 s. Finally, slides were dehydrated, cleared, and mounted with DNA–protein cross-link (DPX).
Scoring
All slides were randomized, coded, and scored blind at 400× magnification. A total of 200 cells were scored per slide for micronuclei by moving systematically in a zigzag fashion from one field to the next, avoiding the previous field. Micronuclei were scored only in cells with intact cellular and nuclear membranes. Overlapped cells, cell clumps, and cells showing karyorrhexis and karyolysis were excluded. The Countryman and Heddle
9
criteria for identification of micronuclei were followed as under:
micronuclei diameter less than one-third the main nucleus. non-refractility (to exclude small stain particles); color same as or lighter than the nucleus (to exclude large stain particles); micronucleus location within 3 or 4 nuclear diameters of a nucleus and not touching the nucleus (to make frequency measurements meaningful); and a maximum of 2 micronuclei associated with one nucleus.
The untreated control group showed no cellular changes or micronuclei formation at all tested time intervals. Statistical comparisons were thus not deemed necessary for this group of subjects.
Results
Formation of micronuclei in buccal mucosal cells is generally observed in adults with long-term smoking and tobacco chewing habits, alcohol consumption, or exposure to toxic mutagens at the workplace (welders, painters, factory workers, etc). The subjects in the study range in age between 12 years and 35 years, are healthy, and do not have any tobacco or alcohol habits. Micronuclei formation in this group is generally not observed unlike in adults of older age where cellular changes in buccal mucosa are more evident.
The results of the study have been tabulated as follows:
intragroup comparison of mean micronuclei values at different time intervals in group 2: non-fluoridated group; intragroup comparison of mean micronuclei values at different time intervals in group 3: fluoridated group; and intergroup comparison of group 1 versus group 2 versus group 3 in relation to the mean difference in micronuclei values at different time intervals.
Intragroup Comparison of Mean Micronuclei Values at Different Time Intervals in Group 2: Non-fluoridated Group
Comparison of Mean Micronuclei Between Different Time Intervals in Group 2 Using Friedman’s Test.
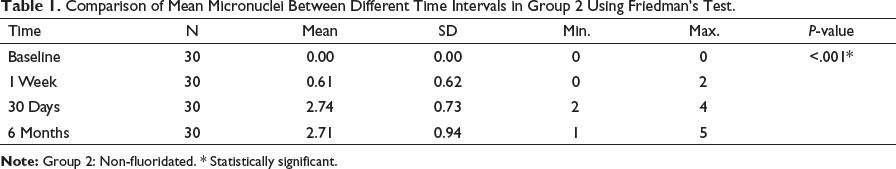
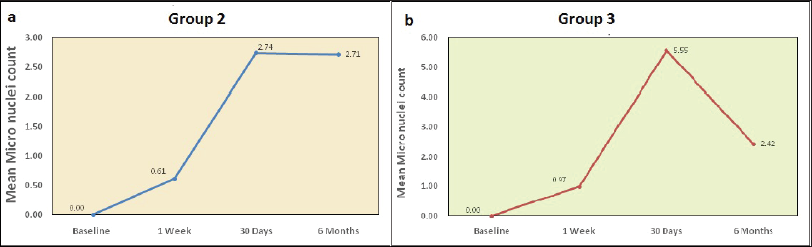
Mean Micronuclei Between Different Time Intervals in Group 2 (Non-fluoridated), and Group 3 (Fluoridated).
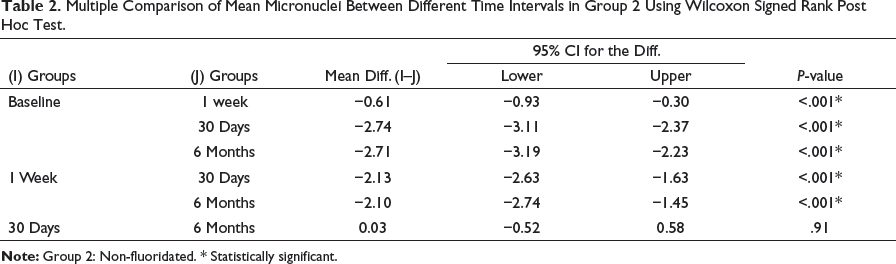
Multiple Comparison of Mean Micronuclei Between Different Time Intervals in Group 2 Using Wilcoxon Signed Rank Post Hoc Test.
Intragroup Comparison of Mean Micronuclei Values at Different Time Intervals in Group 3: Fluoridated Group
Comparison of Mean Micronuclei Between Different Time Intervals in Group 3 Using Friedman’s Test.
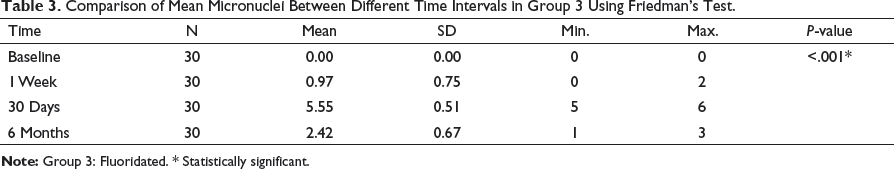
Multiple Comparison of Mean Micronuclei Between Different Time Intervals in Group 3 Using Wilcoxon Signed Rank Post Hoc Test.
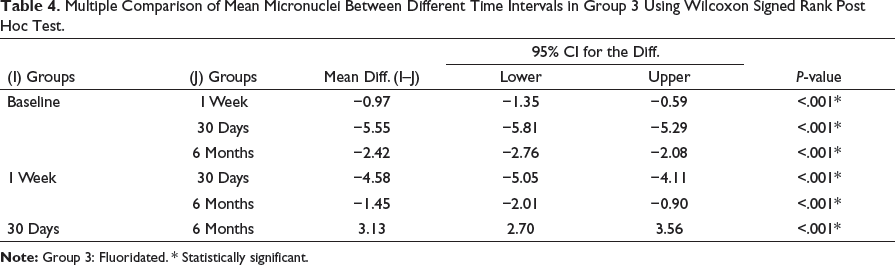
Intergroup Comparison of Group 1 Versus Group 2 Versus Group 3 in Relation to the Mean Difference in Micronuclei Values at Different Time Intervals
The mean micronuclei scores among all three groups were assessed using Kruskal–Wallis test. At 1 week, the difference between group 2 to baseline was 0.61 and group 3 to baseline was 0.97 at P < .001 (Table 5 and Figure 2a). At 30 days, the difference between group 2 to baseline was 2.74 and group 3 to baseline was 5.55 at P < .001 (Table 6 and Figure 2b).
Mean Micronuclei Count in Group 1 (Control), Group 2 (Non-fluoridated), and Group 3 (Fluoridated) at 1 Week, 1 Month, and 6-Month Intervals.
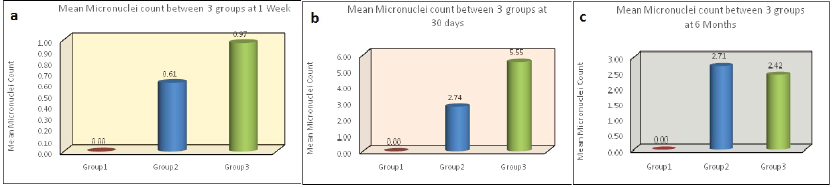
Comparison of Mean Micronuclei Between 3 Groups at 1-Week Period Using Kruskal–Wallis Test.
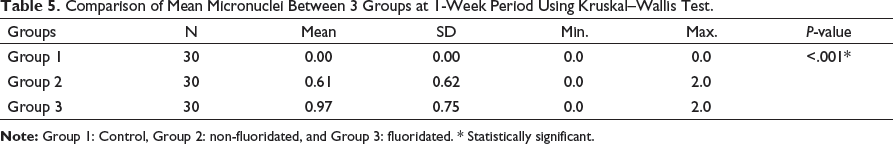
Comparison of Mean Micronuclei Between 3 Groups at 30-Days Period Using Kruskal–Wallis Test.
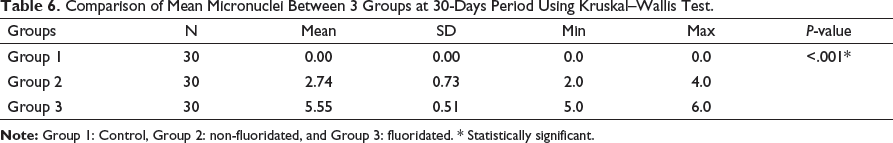
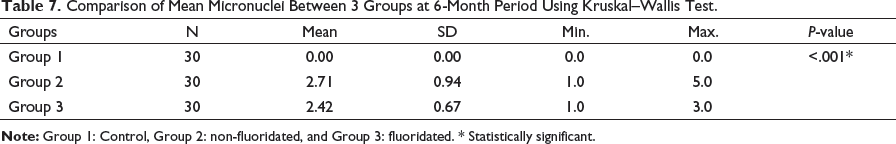
Comparison of Mean Micronuclei Between 3 Groups at 6-Month Period Using Kruskal–Wallis Test.
Discussion
Exfoliated buccal mucosal cells have been used to showcase genotoxic changes due to tobacco and alcohol, as well as exposure to mutagens and carcinogens. 10 The buccal mucosa can be easily accessed, and the technique of sampling is minimally invasive. 11 Buccal mucosal cells are the first in the ingestion route and have been known to metabolize several carcinogens into reactive products. 12 Micronuclei, thus, serve as good biomarkers in order to estimate exposure to potentially mutagenic agents like fluorides. 13
A side effect of orthodontic treatment is formation of WSL and/or enamel demineralization in some cases. This is usually apparent around fixed orthodontic appliances. 14 To decrease WSL, orthodontists consider using cariostatic agents like fluoridated toothpastes and mouthwashes, as fluoride improves enamel remineralization.15–17 The cariostatic influence that fluorides have is attributed largely to the deposition of calcium fluoride. 18 Since these are used intraorally for substantial periods of time, their biosafety is of paramount importance. Early hazard detection is an integral component of exposure prevention and reduction and associated carcinogenic risk. 19
The intraoral environment is constantly subjected to varying pH and temperature fluctuations and also has multiple types of microorganisms. The release of metal ions from orthodontic appliances is mainly due to the behavior of saliva as an electrolyte, predisposing the orthodontic apparatus to corrosion. It is difficult to understand the complex in vivo setting as well as the effects on specific material types. For that reason, the majority of experiments till date were in vitro or animal-based.
It has been observed that many dental materials used for prolonged periods intraorally undergo progressive degradation with release of metal ions, which, in turn, affects their mechanical properties. 20 Several studies have been carried out to assess the impact of metal ion release from brackets and archwires.21, 22 However, there are very few reported on fluoridated agents in vivo. Fluorides have a corrosive effect on metals when exposed for sufficient time periods like during orthodontic therapy, which generally is of long duration. This period is sufficient for the protective surface oxide layer on brackets and archwires to be damaged, exposing the metal to the corrosive effects of fluorides. This, in turn, causes leaching of nickel and chromium ions into the oral cavity. The buccal mucosal cells are in direct first contact with released metal ions and can be easily examined to identify cellular changes, if any. The study, thus, aimed to evaluate fluoride effects at a cellular level in humans and determine if any damage in buccal mucosal cells could be discerned.
Fluoride is a biologically active element that has the ability to accumulate in the body as renal clearance is reached by just 50%. 23 Fluoride interferes with particular enzymes that are involved in the process of DNA repair and causes substantial chromosomal damage due to the formation of covalent bonds with DNA due to which it is considered a true mutagen. 24 This may be due to its oxidative stress–causing potential. It leads to formation of reactive oxygen species (ROS). 25 The insult caused by ROS is not only confined to the cell membrane alone but also can affect protein and nucleic acids. Therefore, due to oxidative stress, DNA damage becomes apparent. 23
This study also demonstrated a significant increase in the number of micronuclei in both fluoridated and non-fluoridated-treated groups over a period of 6 months, which was statistically significant.
The results of the study show the mean MN values to be highest in the fluoridated subject group at 1-month time interval. This is probably due to exposure of the orthodontic brackets and wires to the harsh intraoral environment during the initial time period. Exposure to acidic media like fluorides causes an even greater increase in MN formation as was evident in this treated group. The exact mechanism for reduction in MN levels at the 6-month time period has not yet been fully understood. It could be related to the formation of a protective oxide layer or plaque deposits on the brackets and archwires used in the mouth, which prevents further degradation. Due to the in vivo nature of the study, continuous monitoring of intraoral pH as well as buccal mucosal cells was difficult. The MN frequencies in group 2 non-fluoridated also showed increases at the 1-month interval. This period has been previously associated with maximum leaching of metal ions from orthodontic appliances. A marked increase in nuclear degradation was found at 1 month of fluoride exposure along with reduction in cell viability, which was similar to the studies conducted by Srilatha et al, 23 Bhowmik et al, 26 Podder et al, 27 and Liu et al, 28 who documented excessive fluoride exposure with DNA damage. The in vivo detrimental effects of fluorides on buccal mucosal cells in the form of increased micronucleus formation were demonstrated in the study.
The intellectual level of children could show disturbances due to exposure to high levels of fluorides. Slightly lower intelligence quotient (IQ) levels were observed in children in fluoride endemic zones (2.47 ± 0.79 ppm) relative to children whose drinking water fluoride levels were below acceptable limits (0.36 ± 0.15 ppm). 29 This indicates that prolonged exposure to fluoride may have detrimental effects on the neurodevelopment of children.
In order to simulate human exposure, Zhao et al exposed Sprague–Dawley rats with NaF (10 ppm, 50 ppm, and 100 ppm) from prepregnancy until 2 months of birth. 30 Impairments to learning and memory due to fluoride exposure were observed. Additionally, variations in the hippocampal mitochondria morphology of rats, rapid apoptosis, and neuronal failure were also found.
Fluoride’s genotoxic effect is a controversial topic. Mice exposed to 1 ppm, 10 ppm, 50 ppm, and 75 ppm of NaF for 21 weeks in a study by Dunipace et al showed no genotoxic effect of fluoride on bone marrow cells. 31 Quadri et al reported, however, that fluoride exposure increased generation of ROS, altered mitochondrial calcium homeostasis, changed membrane permeability, and, thus, promoted damage to mitochondria that finally paved the way for apoptosis. 32
A revolutionary genotoxicity technique in exfoliated cells that aids in the evaluation of epithelial carcinogens is the MN assay. 33 Multiple studies have corroborated the efficacy of MN examination in oral cells for evaluating genetic damage.34, 35 Thus, an MN assay was undertaken to observe changes in buccal mucosal cells in 3 groups of subjects.
Damage to cells on fluoride exposure were observed in several previously published animal studies.5, 6, 28 However, extrapolation of these data onto human subjects could not be carried out. In vivo studies of this type help shed light on the detrimental effects of prolonged fluoride exposure to metals used during orthodontic treatment. Since fluoride agents are commonly prescribed during orthodontic therapy to prevent demineralization and formation of white spots, unintended leaching of metal ions could be a cause for cellular changes in the buccal mucosa. The link between increased leaching of metal ions and micronuclei formation requires further investigation. This becomes important in situations where orthodontic therapy is prolonged due to the severe nature of the malocclusion.
Formation of micronuclei predisposes buccal mucosal cells to DNA damage, and further investigation on the synergistic effects of metal ion leaching from orthodontic archwires and brackets due to the corrosive effects of fluorides is thus advisable.
Conclusion
The study has demonstrated the potential for fluoridated agents to cause higher release of metal ions from orthodontic appliances, which, in turn, could cause damage to buccal mucosal cells seen as increase in MN frequencies. Orthodontic appliance exposure to fluorides during the treatment period caused reduced cell viability. Findings of the MN assay provide confirmation of the same. In vivo conditions may, however, show contrasting results due to the presence of several variables, which could affect results. Type and pH of saliva, type of food, and habits of subjects may also act as confounding factors to results obtained.
It is suggested that patients undergoing orthodontic treatment with fixed orthodontic appliances be prescribed non-fluoridated toothpaste and mouthwashes, if caries risk is low, in order to reduce the risk of fluoride-induced genotoxicity. In some cases with moderate malocclusion and high caries risk, use of aligners may also be considered.
Study Limitations
The fluoridated subject group showed higher micronuclei as compared to non-fluoridated groups. Ingestion of fluoride through dietary sources in such patients was impossible to verify. Compliance with instructions in all subject groups was also difficult to confirm, and the responses given were taken as correct. In general, the effects of diet on alteration of nuclear abnormalities are very difficult to identify. Systemic levels of fluorides were not evaluated to come to definite conclusions. It was not possible to exclude inhabitants, if any, from fluoride endemic areas.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
