Abstract
Aim and Objectives:
To compare and evaluate the antibacterial efficacy of various nanoparticles incorporated in orthodontic primer with that of conventional antimicrobial agents at different concentrations on Streptococcus mutans (S. mutans) strain.
Materials and Methods:
Transbond XT Primer was mixed with 2.5% and 5% benzalkonium chloride (BAC), 0.2% and 2.5% chlorhexidine, 1% and 3% titanium dioxide (TiO2) nanoparticles, 0.2% and 0.5% nanohydroxyapatite, and 0.2% and 0.5% silica-doped nanohydroxyapatite powders. Antibacterial activity against S. mutans for all the materials was evaluated by the disk diffusion method for periods of 48 (T1) and 72 (T2) hours.
Results:
There was a significant increase in the antimicrobial activity of the orthodontic primer modified by the addition of antibacterial agents. The highest zone of inhibition against S. mutans was observed for silica-doped nanohydroxyapatite of 0.5% (11.03 mm) among all the nanoparticles, which was similar to the conventional antibacterial agents used in our study.
Conclusions:
Among all the groups, BAC at 5% concentration showed the highest antimicrobial activity, and the least activity was exhibited by 1% TiO2 nanoparticles. Silica-doped nanohydroxyapatite at 0.5% expressed the greatest antibacterial activity among all the nanoparticles. All the materials showed sustained antibacterial activity even after 72 hours.
Introduction
Preserving good oral hygiene with fixed orthodontic appliances is a challenge to both the patient and the clinician, because many of these components act as plaque-retentive areas. Thus, the success of orthodontic treatment lies in correcting occlusion in the best possible manner without affecting the preexisting health of teeth and supporting tissues.
White spot lesions after orthodontic treatment are the most common problem for both the orthodontist and the patient. Despite the best efforts towards the prevention of white spot lesions, they still occur. Thus, to prevent these, novel strategies or therapies were developed, such as incorporation of various antimicrobial agents into resins, glass ionomer cement, orthodontic adhesives, etc. Their use in primers may be beneficial because of their close contact with the tooth surface.1-3
The above facts have prompted a renewed interest in the use of alternative antibacterial agents, such as nanoparticles. Due to their smaller particle size, they can interact more closely with the biological molecules such as lipids, proteins, enzymes, and nucleic acids, damage the biological cell structures, and provide a large surface area to exhibit antimicrobial activity at minimal concentrations. The various types of nanoparticles available are as follows: gold, silver, copper, zinc oxide, titanium dioxide (TiO2), silicon dioxide (SiO2), hydroxyapatite, fluorapatite, and fluorohydroxyapatite.4-6 Due to these nanoparticles’ biocidal and anti-adhesive capabilities, this in vitro study was designed to evaluate and compare the antibacterial effects of an orthodontic primer modified through the addition of various nanoparticles, like TiO2 nanoparticle powder, nanohydroxyapatite powder, and the newly introduced silica-doped nanohydroxyapatite powder against Streptococcus mutans (S. mutans), which was determined by the disk diffusion method.
Materials and Methods
Grouping of Materials Analyzed in This Study
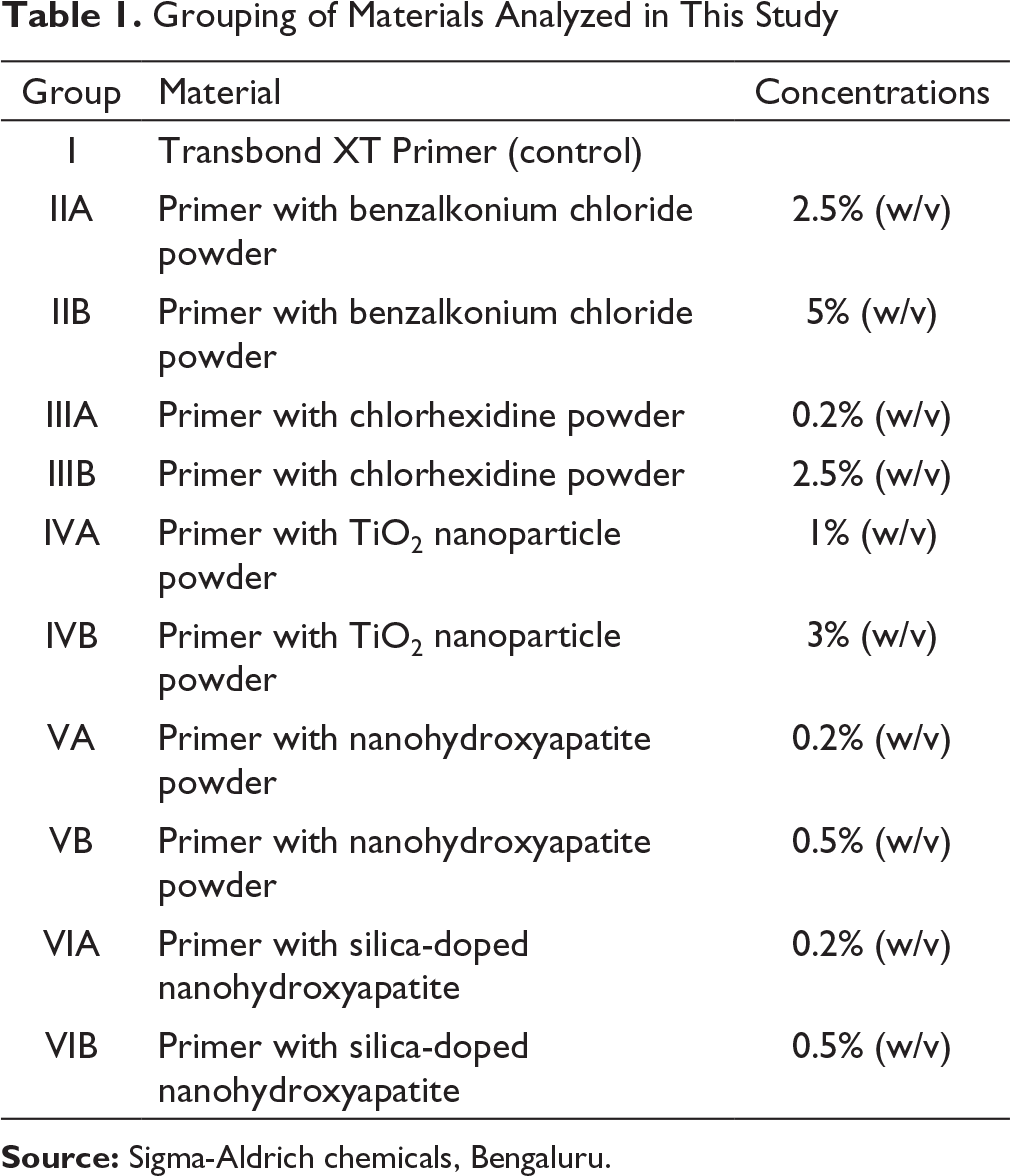
Source: Sigma-Aldrich chemicals, Bengaluru.
All the above concentrations were weighed using an electronic weight balancer and calculated for “1 ml” of bonding agent, mixed with an ultrasonic probe sonicator of 20KHz/1,200,000 rpm frequency to achieve a uniform mix (Figure 4).
Disk Diffusion Assay Method
Media Preparation
98.1 g of mutans sanguis agar was dissolved completely in 1,000 mL of warm distilled water in a beaker and was autoclaved at 121°C and 15 lb pressure for 15 minutes and then allowed to cool. To this media, 4 to 5 drops of antifungal solution clotrimoxazole was added and poured in Petri plates of 2 to 4 mm thickness.
Disk Preparation
A total of 176 disk specimens of 6 mm diameter were prepared with 125 mm qualitative circled Whatman filter paper. The disks were sterilized in an autoclave and later placed in a hot air oven at 60°C for 1 hour to remove moisture. The sterilized disks were impregnated with 10 µl of primer and primer containing various antimicrobial agents.
Procedure
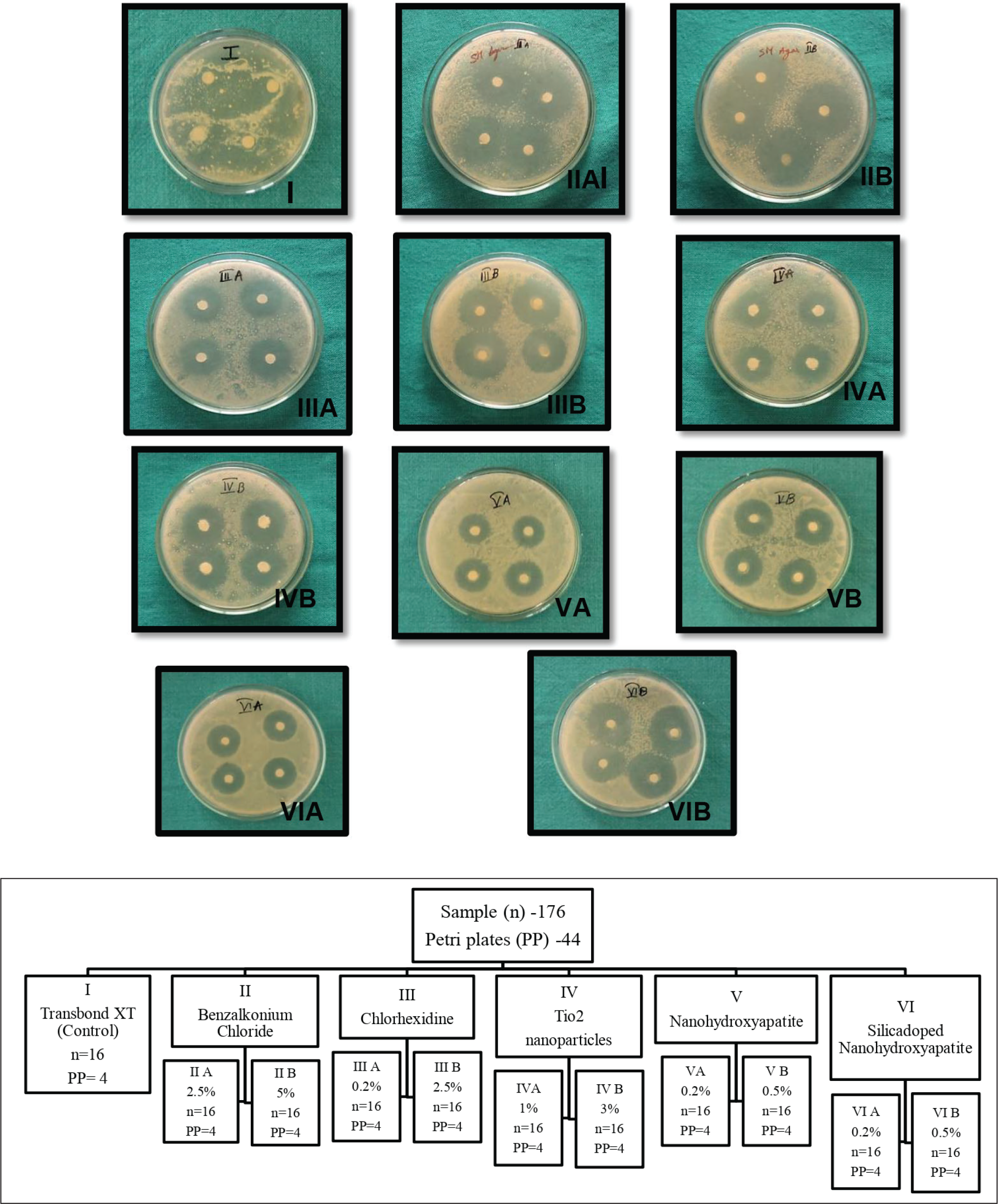
The mutans strain obtained from lawn culture was stored in a phosphate buffer solution (PBS) and was transferred to the solidified agar Petri plates through the pour plate method. Later, 16 sterile disks were taken, and then each disk was impregnated with 10 µL of Transbond XT Primer with the help of a micropipette. In each Petri plate, 4 disks were placed at 4 corners with forceps and labeled as group I. Similarly, labeling was done for all the other groups. For each concentration, 4 Petri plates were utilized for placing 16 disks in each group. Thus, a total of 44 Petri plates were used (Figure 1). Later, all the plates were incubated at 37°C for 48 hours and examined for the zone of inhibition around the disks after 48 (T1) and 72 (T2) hours. These zones were measured with the help of digital Vernier calipers (Mitutoyo, absolute company) of 0.001 accuracy. 7
Source: Sigma-Aldrich chemicals, Bengaluru.
Results
The antimicrobial activity of plain Transbond XT Primer, BAC powder, chlorhexidine powder, TiO2 nanoparticle powder, nanohydroxyapatite powder, and silica-doped nanohydroxyapatite powder mixed with the orthodontic primer were tested in vitro for 48 (T1) and 72 (T2) hours against S. mutans. The disk diffusion assay method was used to demonstrate the ability of the modified orthodontic primer to release antimicrobial agents into the surrounding agar media and thereby inhibit growth of S. mutans. The diameter of the zone of inhibition was observed around all the disks, except the disks containing plain Transbond XT Primer (Figure 2).

Descriptive Statistics of All the Groups
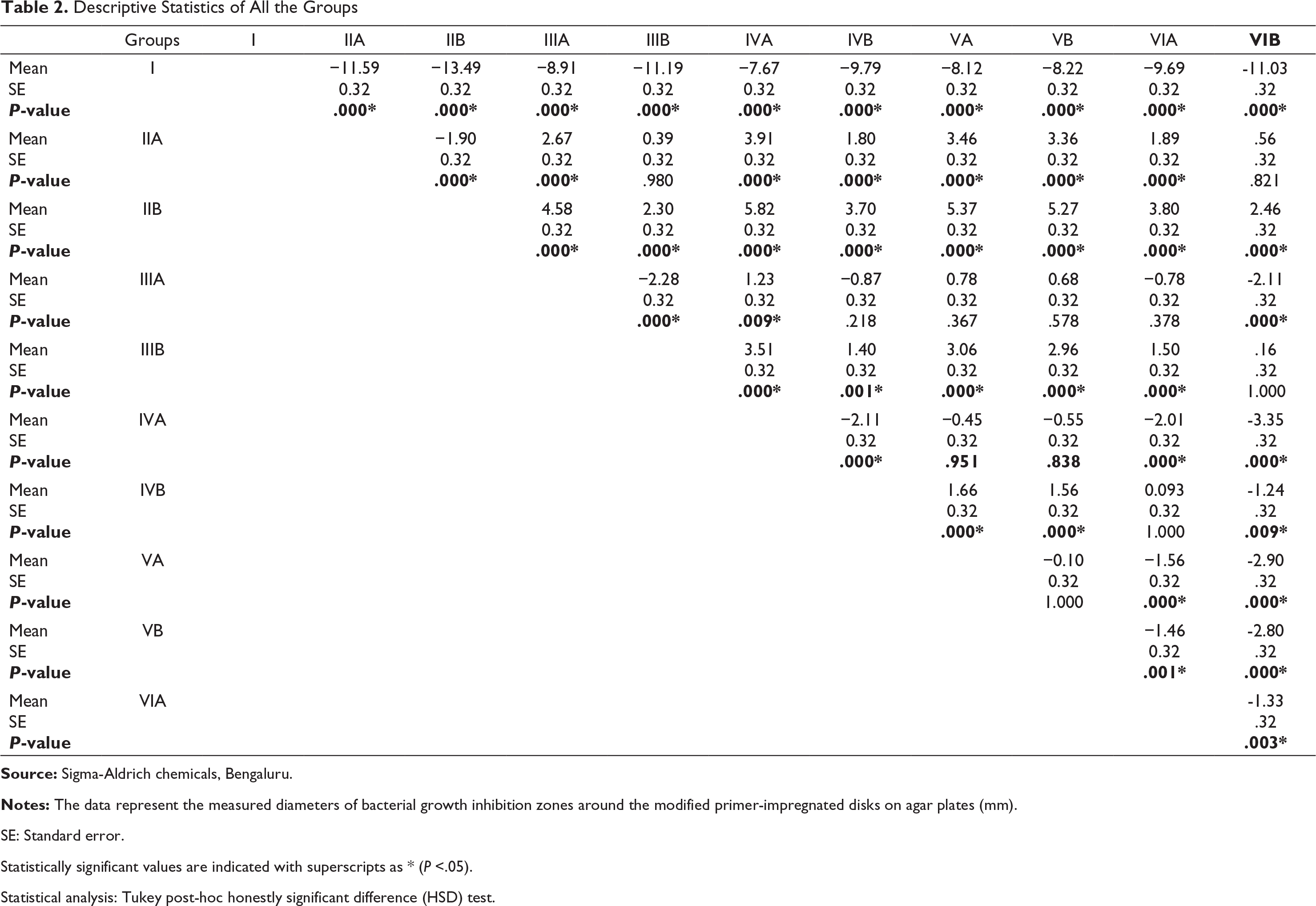
SE: Standard error.
Statistically significant values are indicated with superscripts as * (P <.05).
Statistical analysis: Tukey post-hoc honestly significant difference (HSD) test.
Mean, Standard Deviation, Test of Significance of Zones of Inhibition (in mm) Among Different Groups at 48 (T1) and 72 (T2) Hours
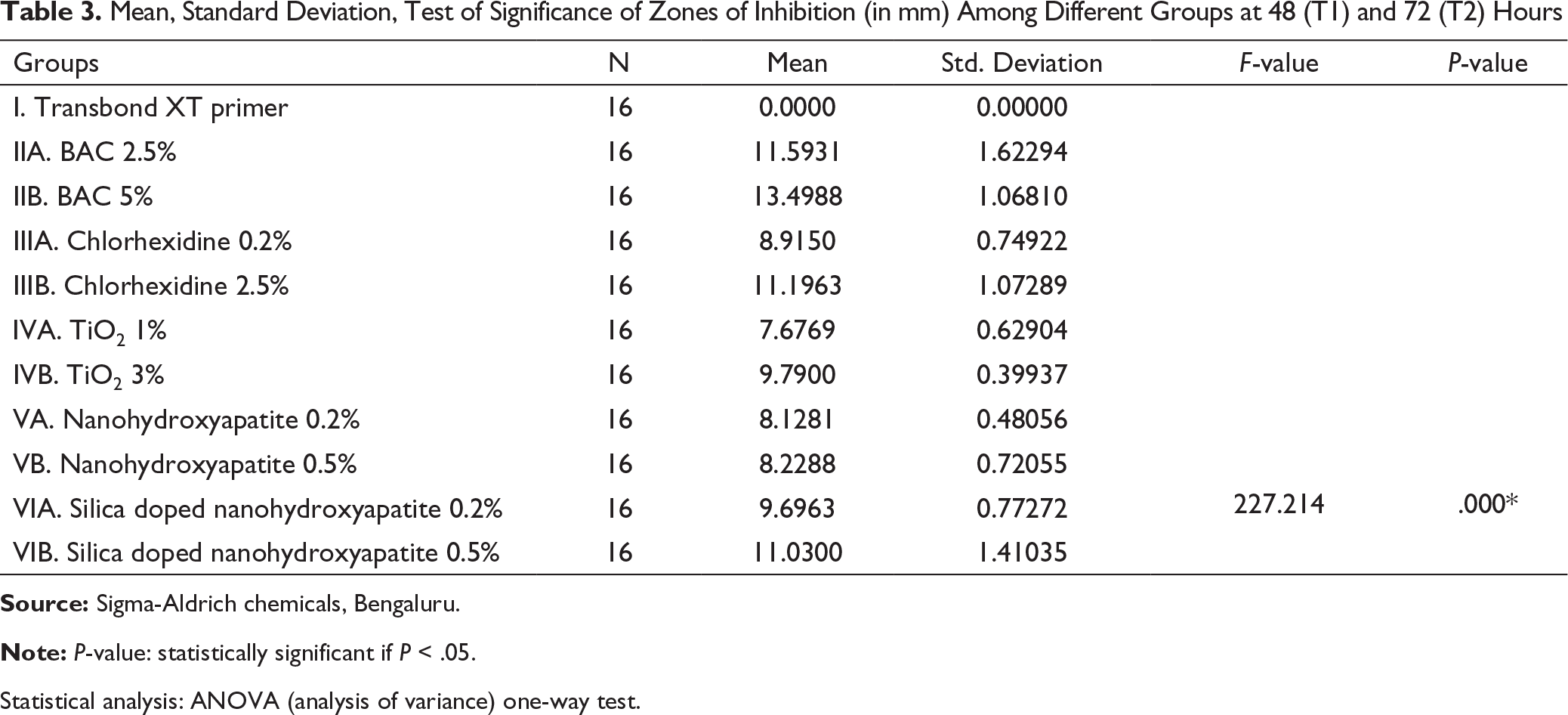
Statistical analysis: ANOVA (analysis of variance) one-way test.
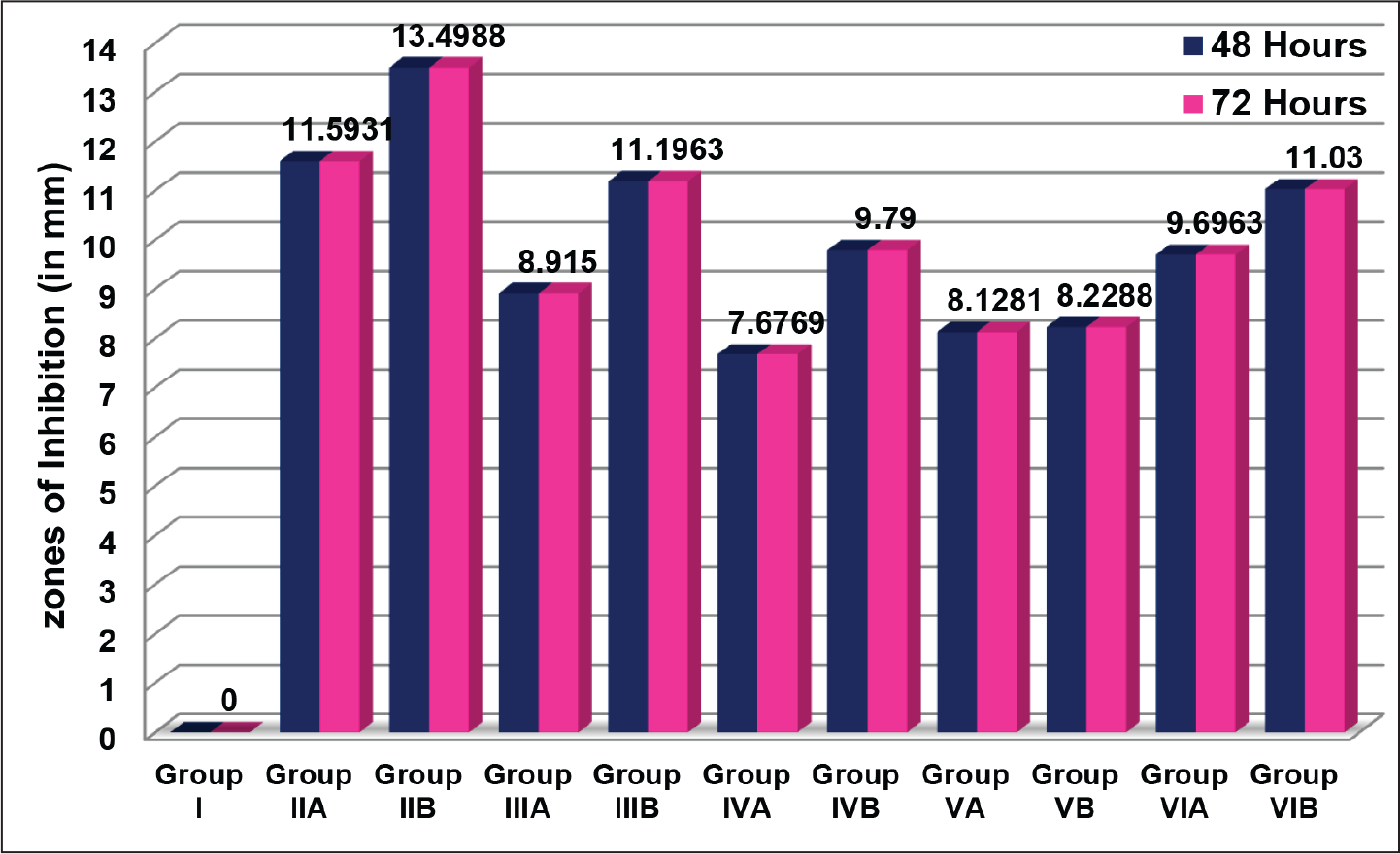
The zone of inhibition for TiO2 3% (group IVB—9.79 mm) and that for silica-doped nanohydroxyapatite 0.2% (group VIA—9.69 mm) were almost equal when compared to that for the other groups, followed by chlorhexidine 0.2% (group IIIA—8.91 mm), nanohydroxyapatite 0.5% (group VB—8.22 mm), and nanohydroxyapatite 0.2% (nanohydroxyapatite VA—8.12 mm). The least zone of inhibition was observed with TiO2 at the concentration of 1% (group IVA—7.67 mm) (Table 3). The results for all the groups after 48 (T1) and 72 (T2) hours were similar. It is inferred that all the agents in this study retained their antimicrobial activity even after 72 (T2) hours (Figure 3).

Discussion
Under normal conditions, the diversity of oral flora is well balanced and the microorganisms work together as a natural defense system. However, long-term treatment with fixed orthodontic appliances causes changes in the normal oral environment and favors increased risk of colonization of pathogenic bacteria, like S. mutans and lactobacilli, initiating a series of events that may lead to decalcification and caries development. Thus, antimicrobial targeting of these organisms has been studied extensively by various microbiological clinical studies. 8
Numerous studies have been performed to improve the properties of orthodontic adhesives through incorporating various antibacterial agents, such as vancomycin, metronidazole, methacryloyloxydo-decylpyridinium bromide (MDPB), chlorhexidine, triclosan, and BAC.9-12 Similarly, in the present study, nanoparticles were incorporated in the primer for antibacterial testing. They were found to be effective against bacteria in a comparable manner to that of conventional antibacterial agents.
No zone of inhibition was observed in our study for Transbond XT Primer due to lack of antimicrobial ingredients, which was in accordance with the results obtained by Al Musallam et al 13 and Sehgal et al, 14 who also observed a 0-mm inhibition zone when antimicrobial agents were added to chemically cured 3M Unite Bonding Adhesive. The zone of inhibition for primer containing BAC 2.5% was 11.59 ± 1.62 mm and that for BAC 5% was 13.49 ± 1.06 mm, which was the highest value obtained in the present study. This inhibition zone was comparable to values obtained by Saito et al 12 in their study, who noticed 12 mm and 14.29 mm inhibition zones when 2.5% and 5% concentrations were used, respectively. Othman et al 15 added BAC 0.25%, 0.75%, 1.25%, 1.75%, and 2.5% (wt/wt) to Reliance Phase II chemical cure composite to evaluate its antimicrobial benefits and physical properties. They concluded that incorporation of BAC into a composite adhesive material added antimicrobial properties to the compound without affecting its mechanical properties.
The mean zone of inhibition observed with respect to chlorhexidine at the concentration of 0.2% was 8.91 ± 0.74 mm, and that with 2.5% chlorhexidine was 11.19 ± 1.07 mm. Similarly, Shanmugaavel et al 16 also added 1% chlorhexidine liquid to a conventional glass ionomer and resin sealants and observed 10.3 mm and 14.8 mm inhibition zones, respectively. Marti et al 17 in their study noted 6.1 mm, 6.6 mm, and 10 mm inhibition zones on addition of 0.5%, 1%, and 2% chlorhexidine to glass ionomer cement. These figures were lower than our results, due to the addition of chlorhexidine to glass ionomer cement, which does not dissolve completely in primer. Transbond XT Primer, a photoactivated, light-cured composite resin, was modified by the addition of chlorhexidine, at a concentration of 2.5 wt%, by Singh et al 18 They found that the addition of chlorhexidine to the orthodontic composite resin enhanced its antimicrobial properties and that there was no significant difference between the bond strengths of the control and the experimental resins tested after 24 hours and 25 days.
The mean zone of inhibition observed for primer containing 1% TiO2 nanoparticle was 7.67 ± 0.62 mm, and that for 3% TiO2 nanoparticle was 9.79 ± 0.39 mm. These inhibition zone values are similar to values obtained by Poosti et al, 19 who noticed an inhibition zone of 8.2 mm when 1% TiO2 was used. They stated that when particles of size less than 50 nm were mixed with the composite paste, it made a homogenous distribution of nanoparticles in the composite structure and did not affect its consistency, and hence its shear bond strength (SBS) and adhesive remnant index (ARI) scores.
The zone of inhibition observed in the current study with respect to nanohydroxyapatite at a concentration of 0.2% was 8.12 ± 0.48 mm, and that for 0.5% nanohydroxyapatite was 8.22 ± 0.72 mm, almost similar values. Similarly, Sodagar et al 20 incorporated 5% silver/hydroxyapatite into a Transbond XT composite and observed a 6.3 mm inhibition zone. This shows that hydroxyapatite has the added advantage of antibacterial efficacy along with its remineralizing property.
Akhavan et al 21 stated that incorporation of 5% silver/hydroxyapatite nanoparticles maintains the shear bond strength of orthodontic adhesives. An increase in bond strength is seen when 1% silver/hydroxyapatite nanoparticles are used, because the nanofillers improve adhesion at the interface between the restorative material and the tooth structure, thereby increasing the mechanical strength of the adhesive layer. On the other hand, increasing the concentration to 10% has an undesirable effect on the shear bond strength when compared to the control group due to an agglomeration of particles, creating defect points and interfering with the curing process of the adhesive.
The mean zone of inhibition for the primer containing 0.2% silica-doped nanohydroxyapatite was 9.69 ± 0.77 mm, and that for 0.5% silica-doped nanohydroxyapatite was 11.03 ± 1.41 mm. Likewise, Camporotondi et al 22 and Capeletti et al 23 tested antibacterial activity of silica and concluded that silica had antibacterial efficacy against gram-positive and gram-negative bacteria. In the present study, due to doping of silica particles with nanohydroxyapatite, the antibacterial efficacy was doubled, and the material showed a similar amount of antibacterial activity at the higher concentration.
Thus, nanoparticles in micro-size aggregates have been shown to elicit better antimicrobial properties, almost at par with conventional materials. However, while this study was performed in vitro for a limited period of time, in a clinical situation, orthodontic treatment would last for about 18 to 24 months. These findings suggest that further investigation is needed to test the clinical performance, safety and possible clinical applications of the nanoparticles studied.
The present study was conducted to evaluate the antibacterial properties of the newer silica-doped nanohydroxyapatite in comparison to other nanoparticles when they were added to a primer instead of to a composite. Previous studies15, 18-20 have shown clinically acceptable bond strength values when these antibacterial agents were added to a composite. Further studies have to be done to evaluate the bond strength of these materials when they are added to a primer.
Conclusions
Within the limitations of the present study, the following conclusions were drawn:
There was a significant increase in the antimicrobial activity of the orthodontic primer modified by the addition of antibacterial agents. BAC at 5% concentration showed the highest antimicrobial efficacy among all the groups. Among all the nanoparticles, silica-doped nanohydroxyapatite at 0.5% concentration expressed the greatest antibacterial activity. The least antibacterial activity was exhibited by 1% TiO2 among all the groups in reducing the growth of S. mutans. All the materials showed sustained antibacterial activity even after 72 hours.
Footnotes
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
Declaration of Conflicting Interests
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
