Abstract
Many psychiatric populations present deficits in the processing of neutral faces. While insomnia is associated with perceptual alterations of facially expressed fear, sadness, and happiness, the perception of neutral faces have not been explored in this population. This study compared normal sleepers and individuals experiencing insomnia symptoms in their expression intensity ratings of neutral faces. A total of 56 normal sleepers scoring <5 on the Insomnia Severity Index and 58 individuals experiencing clinically significant insomnia symptoms scoring ≥15 on the Insomnia Severity Index (19.24 ± 3.53) observed 12 neutral facial photographs from the Karolinska Directed Emotional Faces database. Participants rated the extent to which each face appeared as attractive, sad, happy, trustworthy, approachable, healthy, and sociable. The results revelated a main effect of group, F(1,117) = 4.04, p = .047, and expression, F(7, 819) = 39.08, p = .001, on intensity ratings. While no significant Group × Expression interaction was confirmed, F(7,819)=1.03, p = .41, simple effects analysis determined that those experiencing insomnia symptoms rated neutral faces as significantly more attractive (34.30 ± 14.82; t(117) = −2.73, p = .007; Cohen’s d = 0.50) and happy (34.83 ± 13.87; t(117) = −2.23, p = .028; Cohen’s d = 0.41) compared with normal sleepers (Attractive: 26.89 ± 14.76; Happy: 28.90 ± 12.48). The present outcomes tentatively suggest that individuals experiencing clinically significant insomnia symptoms differentially perceive neutral faces when compared with normal sleepers. These outcomes present potentially negative psychosocial implications for those with insomnia.
Insomnia is the most prevalent sleep disorder, affecting 30% of the general population at symptom level (American Academy of Sleep Medicine, 2005; Espie et al., 2012; Morin et al., 2006) and 10% at disorder level (Espie et al., 2012; Morin, et al., 2006; Ohayon, 2002). Characterized by difficulty with sleep initiation, maintenance and/or early morning awakening, and accompanied by significant impairment to daytime functioning, insomnia is often related to deficits in emotion regulation and socioemotional functioning (Baglioni et al., 2010; Espie et al., 2012). Social perception is vital in shaping the evaluation of other people, particularly when decoding affective and socially relevant information (Singer et al., 2004; Todorov et al., 2008). Individuals use a variety of cues, including facial, vocal, and gestural, to communicate emotions to others; and read these cues to infer another’s internal state (Sauter et al., 2010). For most people, visual processing of the human face remains highly prevalent on a daily basis (Allison et al., 2000). Indeed, facial observations provide a plethora of information including distinguishing characteristics, perceived attractiveness, intended behavior, and emotional state, without any use of language (Haxby et al., 2000). As a result, facial expressions are considered the primary source of information in social perception.
Accurate judgments of facially expressed emotion are vital for effective social interactions and nonverbal social judgments. Therefore, any perceptual deficits accompanying poor sleep may present negative psychosocial consequences for those involved (Axelsson et al., 2010; Sundelin et al., 2013). Perceptual alterations of emotional faces are widely established in psychiatric populations (e.g., anxiety, depression, bipolar disorder, and anorexia nervosa: Bradley et al., 1999; Bourke et al., 2010; Pollatos et al., 2008; Surguladze et al., 2010). In the context of sleep, research examining the emotional processing of facial expressions largely rely on sleep deprivation data, yielding mixed results (Cote et al., 2014; Huck et al., 2008; Killgore et al., 2017; Maccari et al., 2014; Schwarz et al., 2013; van der Helm et al., 2010). Here, a number of studies identify deficits in the ability to categorize emotional faces following total sleep deprivation. Specifically, total sleep deprivation (31.5 hours) has led to performance deficits in the ability to categorize happy and sad faces (Cote et al., 2014). These outcomes were confirmed again in a more recent study (Killgore et al., 2017). In contrast, Huck et al. (2008) evidenced nonspecific deficits in the overall ability to categorize the six basic facial emotions. Reduced intensity ratings of facially expressed anger and happiness at mid-intensity have also been observed following sleep deprivation (van der Helm et al., 2010). When examining valence ratings of positive, negative, and neutral faces in a healthy sample who were subject to total sleep deprivation (24 hours), Maccari et al. (2014) evidenced increased emotional valence ratings for negative and neutral faces when compared with baseline assessment. Partial sleep deprivation (4 hours) is also related to impaired response latency, as measured by electromyography, when asked to facially mimic the emotional valence of positive and negative faces (Schwarz et al., 2013).
Similar, albeit mixed outcomes have been observed in the insomnia population (Akram, Sharman et al., 2018; Brand et al., 2019; Crönlein et al., 2016; Kyle et al., 2014; J. Zhang et al., 2019). In particular, individuals with insomnia often provide blunted responses to emotional faces depicting fear, sadness, and happiness (Crönlein et al., 2016; Kyle et al., 2014) and insomnia salient faces depicting tiredness (Akram, Sharman et al., 2018). Similarly, insomnia and sleep apnea patients have shown impairments in the ability to categorize the expressions of anger, happiness, and sadness (Crönlein et al., 2016; J. Zhang et al., 2019). In contrast, among police officers completing computerized measures of emotional labeling and matching of faces, Brand et al. (2019) failed to evidence any relationships between insomnia symptoms and performance outcomes (accuracy and speed). Likewise, in a series of large-scale studies by Holding et al. (2017), the duration and quality of sleep and experience of total sleep deprivation failed to predict overall speed and accuracy when categorizing facially expressed emotions. While emotional intensity ratings were not gathered, poor sleep was related to deficits in recognizing the expression of disgust (Holding et al., 2017). Despite the variation of outcomes yielded in these studies, the possibility of a relationship between insomnia and socioemotional deficits in the context of facially expressed emotions remains plausible. While the perception of neutral faces has been examined following sleep deprivation in healthy subjects (Maccari et al., 2014), the perception of neutral faces is yet to be examined in the context of disturbed sleep.
Examination of neutral faces have afforded novel insight into alterations of face processing among many psychiatric populations including those with anxiety, depression, and schizophrenia (Arce et al., 2009; Leppänen et al., 2004; Pinkham et al., 2011; Somerville et al., 2004; Wieser & Moscovitch, 2015). Depressed individuals have been found to exhibit slower and poorer performance when asked to categorize neutral faces (Leppänen et al., 2004) and deficits in emotional resilience are associated decreased ratings of happiness in neutral faces (Arce et al., 2009). Similarly, individuals with paranoid schizophrenia are known to over attribute anger to neutral faces (Pinkham et al., 2011). In contrast, healthy subjects are generally accurate in their perception of neutral faces as unambiguous signals of neutrality, even in the presence of subtle emotional signals (Etcoff & Magee, 1992; Leppänen et al., 2004). As such, perceptual deficits of neutral faces in psychiatric patients may not be attributed to task difficulty, that is, that neutral expressions are generally difficult to recognize (Leppänen et al., 2004). To advance the understanding of face perception and insomnia, the present work sought to determine whether individuals presenting insomnia symptoms display abnormalities in the processing of neutral faces. More specifically, this study compared normal-sleeping controls and individuals experiencing insomnia symptoms in their expression intensity ratings of neutral faces. Given that experimental research to date has yielded mixed evidence concerning the perception of emotional faces in insomnia, this work is considered to be an exploratory investigation, with no a priori hypotheses.
Methods
Participants
A cross-sectional online questionnaire-based study was implemented. Both students and members of the population responded to an advertisement for a face rating study via faculty emails, social media platforms (i.e., facebook and twitter), and online research participant forums (e.g., Callforparticipants website). This resulted in complete data from N = 222 people. Following the approach by Courtauld et al. (2017), participants were stratified into low insomnia symptoms (n = 56; 19.69 ± 4.07 years, 73% females) determined as scoring below 5 on the Insomnia Severity Index (ISI: Bastien et al., 2001; 3.73 ± 2.12), and those experiencing clinically significant insomnia symptoms (n = 63; 20.32 ± 4.08 years, 85% females) determined as scoring 15 or more on the ISI (13.14 ± 3.94). A score of 15 or more on the ISI is suggestive of clinically moderate-to-severe insomnia. This resulted in a final sample of 119 individuals who were entered into analysis. The clinically significant insomnia symptoms group scored significantly higher on the ISI (19.24 ± 3.53) relative to low symptom group (2.70 ± 1.69; t(117) = −31.93, p = .001); however, groups did not differ in age, t(117) = −.83, p = .41, or sex, X2(1)> = 3.06, p = .08.
Materials
Insomnia Severity Index
Insomnia symptoms were assessed using the ISI (Bastien et al., 2001). The ISI consists of seven items examining the severity of insomnia symptoms over the past 2 weeks including difficulty initiating and maintaining sleep and awakening too early. Items are scored on a 5-point likert-type scale, with total scores ranging from 0 to 28. Higher scores suggest greater insomnia severity. Total scores between 0 and 7 indicate no clinically significant insomnia, 8 and 14 subthreshold insomnia, 15 and 21 clinical insomnia (moderate severity), and 22 and 28 clinical insomnia (severe). Assessment of internal consistency yielded a Cronbach’s alpha of .89.
Facial Stimuli
Twelve facial photographs (50% females) displaying a neutral expression were taken from the Karolinska Directed Emotional Faces database (Lundqvist et al., 1998). Subsequently, the hair and neckline were cropped from each neutral image, leaving a series of oval-shaped neutral facial images (see Figure 1).
(A) Example of Female Neutral Face. (B) Example of Male Neutral Face.
Procedure
In accordance with the British Psychological Society’s Code of Human Research Ethics and the host institution’s Research Ethics Policy, ethical approval was granted by the (Masked for Review) University Research Ethics committee. This experiment was conducted in accordance with the Declaration of Helsinki, and all participants gave their informed consent before participation. Participants completed an online questionnaire, in which they were presented with the series of 12 images in randomized order. For each image, participants were asked to quantify on a 100 mm visual analog scale the extent to which each face appeared as tired, attractive, sad, happy, trustworthy, approachable, healthy, and sociable. In total, participants provided 96 ratings, and higher scores represented a greater presence of the specified expression. Following facial expression ratings, participants completed the ISI. Once complete, participants were debriefed about the nature of the study. No financial reimbursement was provided.
Statistical Analysis
Pearson’s bivariate correlations examined possible associations between age and intensity ratings, whereas a series of one-way analysis of variance was conducted to explore potential sex differences in ratings. Mean ratings of expression intensity were calculated for both low and clinically significant insomnia symptoms groups. A 2 (Group) × 8 (Rating Type) mixed model analysis of variance was employed with intensity ratings as the dependant variable. This was conducted to assess main effects of group and rating type, as well as the Group × Rating Type interaction. Main effects and interactions were followed up with independent t tests, and the extent of group differences were quantified using Cohen’s d. Significance was considered at the p < .05 level.
Results
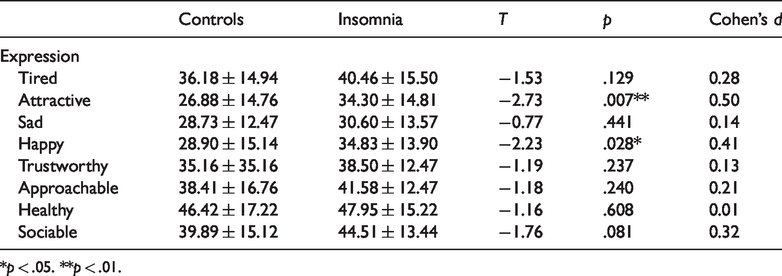
Mean expression intensity ratings for each group are displayed in Table 1. No significant relationships between participant age and each intensity rating were observed (all p’s >.05, see supplemental table for correlation matrix). Examination of sex differences in intensity ratings demonstrated an increased perception of tiredness among females (38.72 ± 15.03) relative to males (32.05 ± 15.03, F(1,217) = 7.31, p = .007). However, no other sex differences were observed for each type of intensity rating (all p’s >.05).
Means and Standard Deviations (±) of Intensity Ratings for Both Low and Clinically Significant (High) Insomnia Symptom Groups.
*p < .05. **p < .01.
The results revealed a main effect of group, F(1,117)=4.04, p = .047, and rating type, F(7, 819) = 39.08, p = .001, on intensity ratings. While no significant Group × Rating Type interaction was confirmed, F(7, 819)=1.03, p = .41, simple effects analysis determined that those experiencing clinically significant insomnia symptoms rated neutral faces as significantly more attractive (34.30 ± 14.82; t(117) = −2.73, p = .007; Cohen’s d = 0.50) and happy (34.83 ± 13.87; t(117) = −2.23, p = .028; Cohen’s d = 0.41) when compared with those low in symptoms (Attractive: 26.89 ± 14.76; Happy: 28.90 ± 12.48: see Figure 2). Interestingly, while not significant, a trend was observed toward increased sociability ratings among those with clinically significant insomnia symptoms (44.51 ± 13.44; t(117) = −1.76, p = .08; Cohen’s d = 0.32) compared with the low symptom group (39.89 ± 15.12). See Table 1 for all group differences and effect sizes, Figure 3 for individual data points.
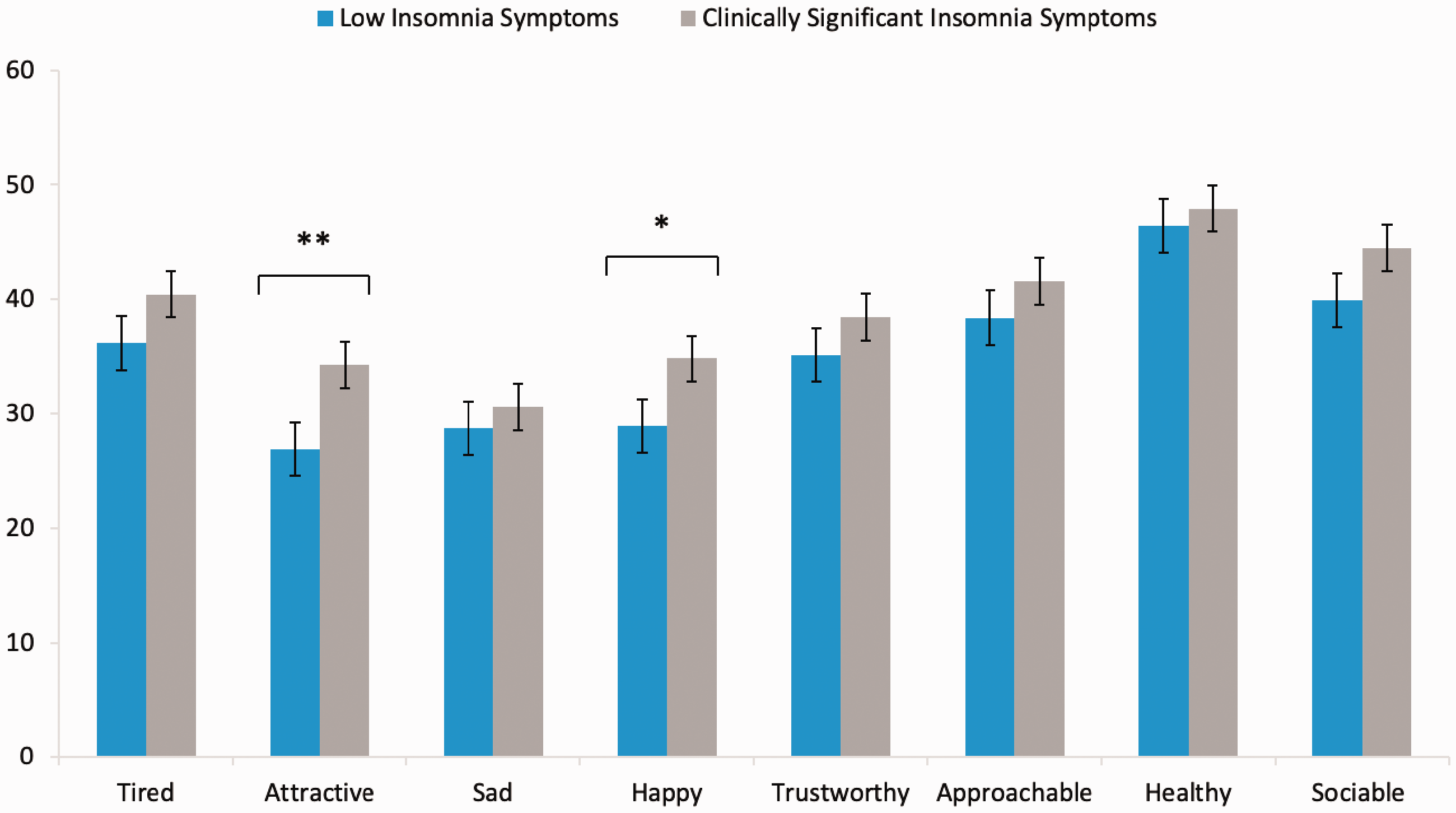
Intensity Ratings for the Control and Clinically Significant Insomnia Symptom Groups Across the Eight Expressions Examined (*<.05, **<.01 for group comparison).
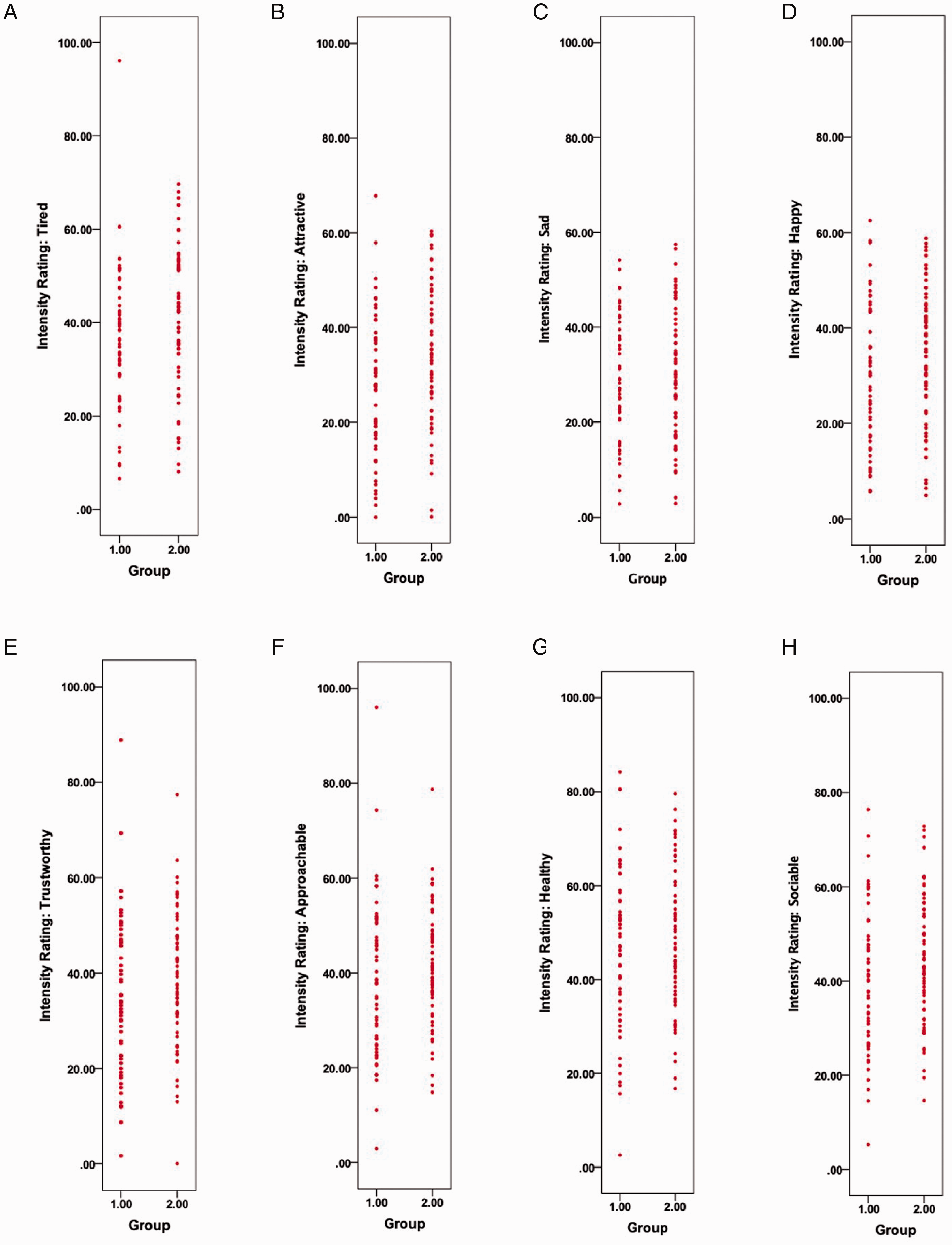
Data Plots of Individual Intensity Ratings for Control (1) and Clinically Significant Insomnia Symptom (2) Groups Across the Eight Expressions Examined. A, Tired; B, Attractive; C, Sad; D, Happy; E, Trustworthy; F, Approachable; G, Healthy; E, Sociable.
Discussion
This study examined whether individuals presenting insomnia symptoms display abnormalities in the processing of neutral faces. More specifically, we compared normal sleepers and individuals experiencing insomnia symptoms in their ratings of expression intensity when observing neutral faces. The current outcomes found that those experiencing insomnia symptoms rated neutral faces as significantly more attractive and happier compared with normal sleepers. No group differences in the extent to which neutral faces appear as tired, sad, trustworthy, approachable, healthy, and sociable. However, a trend was observed toward increased sociability ratings among those insomnia symptoms. The current outcomes cautiously suggest that, in line with other psychiatric disorders (Arce et al., 2009; Leppänen et al., 2004; Pinkham et al., 2011; Somerville et al., 2004; Wieser & Moscovitch, 2015), individuals with insomnia display a propensity to misperceive emotionally neutral faces. However, while other psychiatric conditions such as depression and schizophrenia associate with negative interpretations of neutral faces, the converse appears true for those displaying insomnia symptoms. A number of mechanisms underlying insomnia may offer a partial explanation of this outcome.
Insomnia may be associated with a positive bias when making emotional judgments where, similar to anxiety and depression, an accentuated perception of neutral expressions as positive (i.e., happier and more attractive) may reflect cognitive and affective processes which contribute to their adaptation to difficult interpersonal situations (Arce et al., 2009). Indeed, examination of emotional reactivity in response to experimental sleep loss emphasizes the role of adequate sleep in the adaptive maintenance of emotional processes (see Baglioni et al., 2010 for a review). Nevertheless, this notion may be questioned by previous studies which evidence blunting of both positive (i.e., happiness) and negative facially expressed emotions (i.e., fear, sadness, and anger; Crönlein et al., 2016; Kyle et al., 2014; J. Zhang et al., 2019). As highlighted by Kyle et al. (2014), an alternative explanation may be related to alterations in neural activity. It is well known that the amygdala regulates behavioral responses to socially ambiguous signals (Blasi et al., 2009). Indeed, when examining social judgments in neutral and emotional faces, healthy subjects present greater amygdala activity in neutral faces that were viewed in a positive way (Blasi et al., 2009). In insomnia, imaging studies evidence reduced amygdala connectivity with brain regions (striatum, insula, and thalamus) associated with the processing of emotion, compared with controls (Huang et al., 2012). Whereas increased amygdala reactivity is evidenced in those with insomnia in response to disorder-congruent stimuli (Baglioni et al., 2014). Considering these outcomes, alterations in face processing in insomnia (Akram, Sharman et al., 2018; Crönlein et al., 2016; Kyle et al., 2014; J. Zhang et al., 2019) may perhaps be explained by deficits in amygdala reactivity (Yoo et al., 2007). Certainly, this notion cannot be determined from this study. However, this line of enquiry is certainly warranted in future work.
The ability to accurately gauge another person’s facial expression is important for maintaining healthy and appropriate social interactions (Axelsson et al., 2010). The misperception of neutral faces observed in this study may have negative social consequences for those with insomnia, which are specifically related to evaluating others sates before proceeding with social interaction. Depending on the context, misattributing neutral faces as happier may impair social behavior in sombre work (e.g., important meetings) and health-related (e.g., medical consultations) situations and may inadvertently lead to increased risk of confrontation. Moreover, attractiveness is a known motivator of sexual behavior, collaboration, and superior treatment (Axelsson et al., 2010; Rhodes, 2006). From an evolutionary perspective, misperceived attractiveness related to insomnia may compromise the biological quality of partner choice and gene transmission (Rhodes, 2006).
Neutral faces are used comparatively when examining the perception of emotional faces among individuals with psychiatric difficulties (Fox et al., 2005). Therefore, when presented with the corresponding expressive face, individuals with insomnia may provide an accentuated or attenuated response in comparison (Akram, 2019). Indeed, it is well noted that the neutral expression may be appraised as threatening due to the structural features of the face rather than the emotional content (Oosterhof & Todorov, 2008). To that end, the inclusion of a neutral expression and assessment of emotional valance would be beneficial when clarifying differences in the perception of neutral faces and identifying how emotionally expressive faces are perceived in terms of valence rather than just intensity (Akram, 2019). The latter inclusion would determine whether particular expressions are considered to be threatening in nature among those with insomnia (Akram, Robson et al., 2018).
In this study, all intensity ratings were numerically stronger for those experiencing insomnia symptoms relative to controls. These outcomes directly contrast previous research using emotional faces whereby all ratings were evidenced to be lower among individuals with insomnia relative to controls (Crönlein et al., 2016; Kyle et al., 2014). This may be explained by methodological differences pertaining to the use of 100 point visual analog scales compared with the previously used 7-point likert-type scales (Crönlein et al., 2016; Kyle et al., 2014). In addition, the current work explored the perception of neutral rather than emotional faces which yielded generally lower intensity ratings, irrespective of group, when compared with previous work using emotional faces (Crönlein et al., 2016; Kyle et al., 2014). Finally, while greater intensity ratings for attractiveness and happiness were observed in those experiencing insomnia symptoms, no group differences in related ratings (i.e., approachable and sociable) were observed. As a result, the current findings may be considered spurious in nature. A possibility which is strengthened by outcome variation between studies examining emotional faces in relation to insomnia.
Several strengths and limitations of this study should be noted. The currently used neutral facial stimuli were naturally occurring, gathered from a standardized stimulus set comprised of well-validated facial expressions displayed by trained actors (Lundqvist et al., 1998). As the current sample consisted primarily of female participants, the present findings may not be fully generalizable to males. That said, women are more likely than men to be diagnosed with insomnia (B. Zhang & Wing, 2006). While the role of participant age and sex were accounted for, the role of other potential confounding factors were not explored (e.g., comorbid symptoms of anxiety and depression). That said, while group differences in anxiety and depression would generally be expected, experimental studies do not always control for these symptoms which are often considered a characteristic of the insomnia experience given (e.g., Akram, 2019; Woods et al., 2013). The current outcomes cannot be extrapolated to individuals meeting diagnostic criteria for insomnia. However, the use of large-scale population data could be considered the practical first step in highlighting perceptual deficits of neutral faces in the context of insomnia. Indeed, insomnia symptoms exist along a continuum with the same processes expected in population and clinical samples, albeit diverging in severity (Ree et al., 2006). Therefore, the current outcomes may possibly be accentuated in those meeting the diagnostic criteria for insomnia. It is relevant to note that some effects (i.e., main effect of group) were marginally significant (p = .047) and therefore may not be replicated. That said, key group differences remained somewhat statistically stronger (between p = .007–.028). Nevertheless, future research should examine the present research questions among a more sex balanced and well-screened clinical sample accounting for comorbid sleep-disorder symptoms and psychiatric difficulty.
Deficits in facial emotion processing are an important impairment in insomnia with major consequences for social functioning and subjective well-being. This study compared normal sleepers and individuals experiencing insomnia symptoms in their ratings of neutral faces. The current outcomes suggest that those displaying insomnia symptoms rate neutral faces as significantly more attractive and happier compared with normal sleepers. Further research exploring both neutral and emotional faces is now required to facilitate our understanding of perceptual differences in face processing among individuals with insomnia when compared with normal sleepers.
Supplemental Material
sj-pdf-1-pec-10.1177_0301006620954113 - Supplemental material for Perceptual Differences in Emotionally Ambiguous Neutral Faces Among Individuals Displaying Clinically Significant Insomnia Symptoms
Supplemental material, sj-pdf-1-pec-10.1177_0301006620954113 for Perceptual Differences in Emotionally Ambiguous Neutral Faces Among Individuals Displaying Clinically Significant Insomnia Symptoms by Umair Akram in Perception
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
