Abstract

Clinical History and Gross Findings
A 4-year-old Holstein Friesian dairy cow was presented to the referring veterinarian due to 2 circular areas of alopecia (each approximately 8 cm in diameter) on the black-haired lateral neck. At that time, the second-parity cow was in midlactation. She was held in a freestall barn with cubicles, equipped with a mixture of compost, dried manure solids, straw, and limestone. The farm comprised 140 milking Holstein Friesian dairy cows, fed with a total mixed ration. None of the other cows showed similar skin lesions. Based on the clinical findings and a previous history of Trichophyton infections in the calf barn, the referring veterinarian suspected an infection with Trichophyton verrucosum. Therefore, the cow was treated with enilconazole (Imaverol; Selectavet Dr. Otto Fischer GmbH, Weyarn-Holzolling, Germany). Despite the treatment, the alopecia continued to spread, with the lesions resulting in circular patches to large, multifocal, coalescing areas of hair loss, targeting the black-haired areas of the skin.
In autumn, shortly after dry-off, the cow had lost more than 80% of her black hair (Fig. 1a, b). Further examinations were performed to identify the cause of the hair loss. During the physical examination, the cow had a good general condition and all parameters (ie, behavior, posture, appetite, heart rate, breathing rate, rumen motility, lymph nodes, body temperature, and body condition score) were within the reference ranges. Within the alopecic areas of black pigmented skin, multifocal regrowth of thin, white hairs (leukotrichia) was observed. No signs of scarring or inflammation were present. In addition, the skin along the dorsal midline was multifocally thickened, alopecic, and scaly.

Progression of hair loss of the black pigmented skin. (a) Multiple coalescing foci of alopecia (spring 2023). (b) Generalized alopecia of the black pigmented skin (autumn 2023). (c) Generalized alopecia of the black pigmented skin, with regrowth of thin white hairs (leukotrichia, winter 2023).
At the follow-up examination in winter, the dorsal midline dermatitis had resolved spontaneously. The alopecia had further progressed and almost no black hair remained (Fig. 1c).
Differential Diagnoses
The most common infectious cause of multifocal alopecia in cattle is Trichophyton verrucosum. However, other dermatophytes (Trichophyton mentagrophytes, Trichophyton equinum, Nannizzia gypsea, Microsporum canis, Nannizzia nana), bacterial infections (Staphylococcal spp., Dermatophilus congolensis), or parasitic infections (Stephanofilaria stilesi) can be considered as etiologic differential diagnoses. The primary non-infectious differential diagnosis for acquired loss of black hair in cattle is alopecia areata (AA).10,11,12,13 Further non-infectious reasons for alopecia are ectodermal dysplasia 3 and black hair follicular dysplasia (BHFD); 9 however, both typically occur in younger animals.
For the dorsal midline dermatitis, which resolved in winter, an insect-bite-hypersensitivity due to Culicoides sp. or other insects was suspected.
Microscopic Findings
Biopsy samples were obtained in autumn from alopecic areas of the skin. Biopsies were fixed in 10% neutral-buffered formalin. The specimens were embedded in paraffin wax, sectioned, stained with hematoxylin and eosin, and histologically assessed via light microscopy (Nikon eclipse 80i and Nikon eclipse Ci, Amstelveen, Netherlands). Histologically, a prominent pigmentary incontinence was present around the hair follicle bulbs. In addition, large vacuoles and a subtle aggregation of melanin were seen in several bulbs and the hair shafts present were very thin and sometimes fragmented. A high percentage of hair follicles were in catagen and telogen. In one biopsy taken from the scaly alopecic lesions on the dorsal midline, a moderate, perivascular, eosinophilic, superficial to mid dermal infiltrate was observed. As no final diagnosis could be made based on these biopsies, another set of biopsies were taken in winter. At that time, areas were also biopsied where black hairs were still partially present.
In the biopsy taken in winter, the alopecic skin contained hair follicles in anagen, catagen, and telogen. Similar to the biopsies from autumn, the percentage of hair follicles in the regressing phase was high (Fig. 2a), whereas only a few telogen follicles were seen. Many hair follicles were mildly to minimally distorted and the outer root sheathes were slightly thinner than normal. Of the anagen follicles present, only few bulbs had organized zones of upper bulb melanin transfer. Most of bulbs were malformed, particularly in the central pigment zone and upper region, whereas the outer bulb’s internal root sheath zone was well-formed. Pigment in the central pigment zone was often aggregated into clumps (Fig. 2a, b). In addition, some anagen hair bulbs contained clusters of round pockets that were empty or contained aggregated pigment, presumably from cell loss due to apoptosis. Rare lymphocytes were present in a few of these hair bulbs and clustered in the pigment zone (Fig. 2c). Immunohistochemistry for CD3+ lymphocytes was performed to prove the lymphocytic infiltrate but due to the strong nuclear signal, CD3 immunohistochemistry was unfortunately inconclusive. In addition, a few giant cells were observed around some clusters of released pigment and/or cellular material, which contributed to the distortion of the bulb. There was also excess pigmentation with pigment aggregates in the outer root sheath. The early-stage anagen bulbs were the least affected, and the pigmentation of the hair shafts derived from these bulbs was normal in the suprabulbar areas. Many hair shafts were very thin, had irregular contours, and were often fragmented. They rarely extended beyond the skin surface. Prominent pigment dispersal was seen in peribulbar areas. The epidermis displayed robust pigmentation, which was regularly distributed. A moderate mixed perivascular infiltrate in the superficial and mid dermis was present in all biopsies.
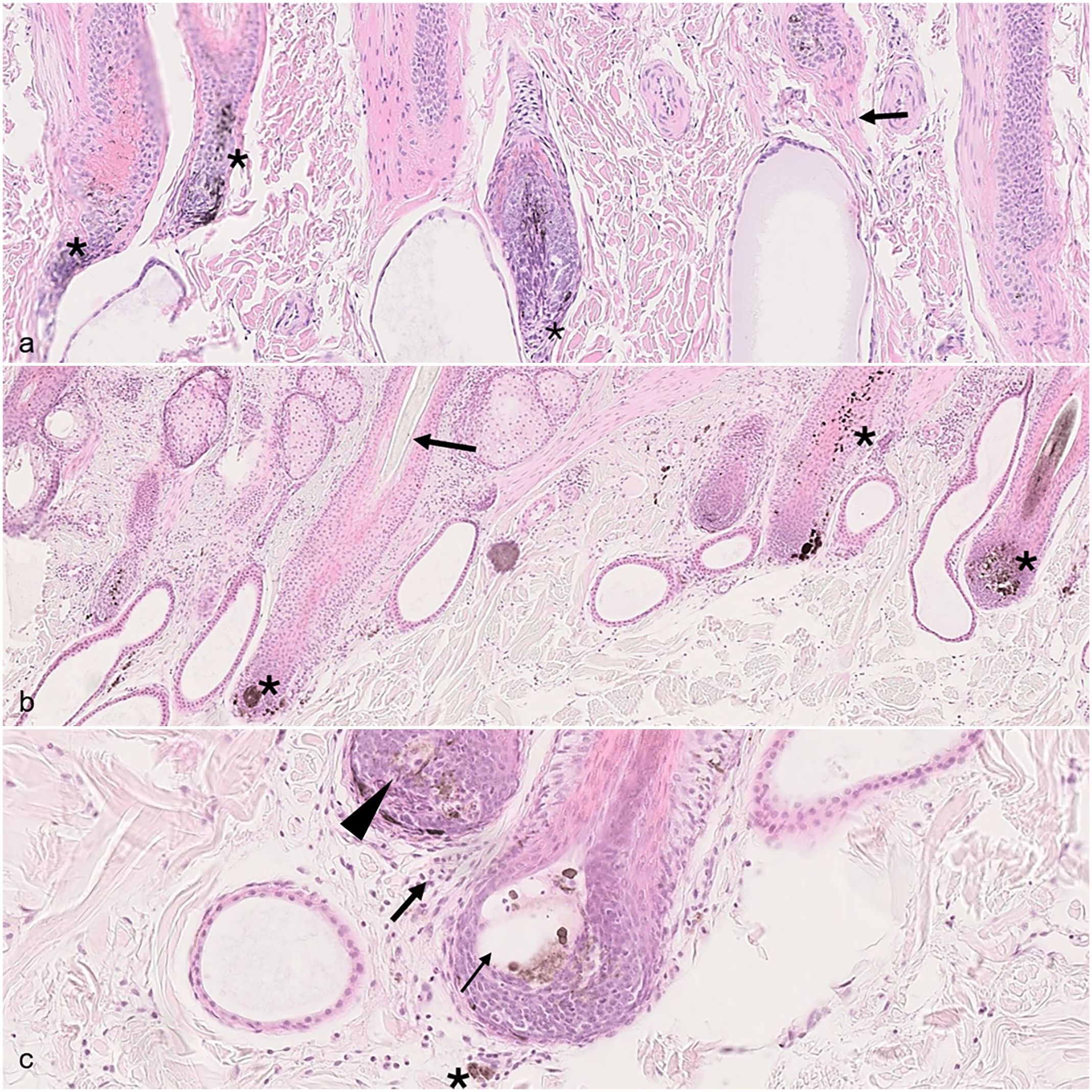
Skin of the cow. Hematoxylin and eosin. (a) Abundance of catagen hair follicles (asterisks), characterized by a thin inferior portion and a trailing connective tissue sheath (arrow). (b) Irregularly dispersed and aggregated melanin in the hair follicle bulbs and the outer root sheath (asterisks) and a thin unpigmented hair shaft with an irregular outer contour (arrow). (c) Peribulbar (arrow) and rare bulbar lymphocytes (arrowhead) and peribulbar pigment incontinence (asterisk). Note the large, clear “pocket” with some aggregated pigment (thin arrow).
Further Investigation and Diagnosis
Cytological examination of the skin revealed occasional coccoid bacteria and Malassezia sp. Deep skin scrapings were negative for Demodex spp. (tauris, bovis, or ghanesis). Trichograms of black and white hair showed a dominance of telogen hair follicles, thinning of the hair shafts, and frayed hair bulbs. Samples of hair and skin tested negative for dermatophytes.
Discussion
In the present case, no infectious cause for the hair loss was identified. This is consistent with the case history, as no other cow of the milking herd was affected by alopecia. Non-infectious causes for loss of black hair are AA, ectodermal dysplasia, 3 and BHFD.6,9 While ectodermal dysplasia is a congenital alopecia, 3 BHFD occurs early after birth or in young adults. In dogs, BHFD develops within the first year of life. 14 There is little information on BHFD in cattle. In the 2 case reports suggesting BHFD as cause of alopecia, the onset of hair loss was at young age and the histological findings were compatible with BHFD seen in dogs.6,9 In one of these case reports, a color-linked alopecia had been mentioned to occur in adult cows; 6 however, for these cases, biopsy findings are, according to the case report, not available and the original case report could not be retrieved by us.
AA is an autoimmune disease targeting the anagen hair bulb and is reported in humans, mice, rats, rabbits, cats, dogs, pigs, horses, cattle, and nonhuman primates.4,7,8 In humans, AA is reported as multifactorial disease. Genetics, infections, stress, drugs, diets, vaccination, and hormones seem to play a role in the development of the disease. This results in a disturbance of a variety of factors resulting in a loss of the immune privilege of the hair follicle and activation of auto-reactive cytotoxic T-lymphocytes. 2 Autoantigens that are most commonly suspected with AA include melanocyte-related proteins and trichohyalin peptides. 2 In cattle, AA has been reported in Ehringer, Black Angus, and Holstein cows of any age.10 –13 Clinically, AA in cattle is reported to be a multifocal, non-inflammatory, patchy to diffuse alopecia affecting primarily dark-haired areas. In humans, also primarily dark hair is lost and hairs often regrow white. 5 Leukotrichia has also been described in cattle.12,13 Common localizations for hair loss are the neck, brisket, shoulder, trunk, and rump, but also the face, limbs, and tail.10 –13 In one dog, nail changes with trachyonychia (brittle claws with longitudinal ridging) were observed, as well as onychodystrophy in a mare.1,8 In cattle, however, claw or horn lesions are not reported.
Diagnosing AA is challenging, as it requires multiple biopsies from early skin lesions to detect the primary lymphocytic infiltration in and around the hair bulb (“swarm of bees”). 10 In acute lesions, the lymphocytic inflammation is often marked, resulting in pigmentary incontinence in the peribulbar region, regression, and miniaturization of the hair follicles or peribulbar fibrosis (“onion skinning”). In cattle, immunohistochemistry of Eringer cows with AA revealed that the majority of lymphocytes infiltrating the hair bulb were CD3+ T cells with only occasional CD20+ lymphocytes. 12 According to Valentine et al, a lack of obvious lymphocytic infiltration is consistent with late-stage AA, in which the characteristic lymphocytic infiltration within and around hair bulbs can be markedly reduced. 13 In this case, further sampling and sectioning may be needed to achieve a definitive diagnosis.
If the lymphocytes attack the melanocyte-related proteins, AA may be restricted to black-haired areas and regrowing hair shafts are often white. This explains the commonly reported leukotrichia in AA cases. Based on the observed leukotrichia (Fig. 1c) and the late age of onset in our case, AA was favored over BHFD as the clinically most likely differential. Histologically, similarities between AA and BHFD, such as aggregated melanin in the hair follicle and the outer root sheath, distorted hair follicles, and fragmented hair shafts, exist. However, no large pigment aggregates, disrupting the cuticle of the hair shaft, as are typical for BHFD, are seen in our case, whereas rare lymphocytes within the hair bulbs, the large vacuoles caused by apoptosis in the bulbs, and the prominent pigmentary incontinence around the bulbs supports damage of the hair bulb, as seen in AA. Thus, despite the lack of final proof of CD3-positive lymphocytes in the hair bulbs, we conclude, based on the clinical and histological findings, that the cow was suffering from AA.
In both sets of biopsies, the percentage of catagen hair follicles was rather high. This may be a result of the damage of the hair bulb since the matrix, cells which are important to maintain the anagen growth phase, are attacked by lymphocytes. However, since the biopsies were taken in autumn and winter, it cannot be ruled out that the hair follicles were physiologically in the regression phase to replace the summer coat by the winter coat, and the winter coat by the summer coat, respectively. However, in the latter case we would expect a more synchronized regression of hair follicles.
During the farm visit in autumn, the skin along the dorsal midline was multifocally thickened and scaly, which resolved by winter. Furthermore, the biopsy from autumn showed an eosinophilic dermatitis that was no longer present in the biopsies taken in winter. An additional eosinophilic dermatitis was also described in the other case reports in cattle with AA. As in our case, authors suspected a reaction to biting insects.11 –13
In conclusion, although rare, AA is a valid differential diagnosis in cows with presumably non-infectious hair loss. For a reliable diagnosis, areas with suspected AA should be biopsied at an early stage of the disease. To obtain biopsies displaying diagnostic findings (lymphocytic infiltration in and around the hair bulbs), biopsies should be taken in the alopecic areas but close to the haired skin. In addition, presumably healthy black-haired areas of the skin should be sampled.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
