Abstract
The factors that determine the appearance of the different pathologic forms associated with bovine paratuberculosis are not fully understood, but new research suggests a critical role of innate immunity. Toll-like receptors (TLRs) trigger the recognition of invading pathogens by innate immune cells and the onset of specific immune responses. The aim of this work was to assess, immunohistochemically, the expression of TLR1, TLR2, TLR4, and TLR9 in intestinal samples of 20 cows showing different types of paratuberculous lesions: uninfected controls, focal lesions, paucibacillary, and multibacillary diffuse forms. The majority of labeled cells were morphologically consistent with macrophages. A differential cell count was performed in the intestinal lamina propria, gut-associated lymphoid tissue, and mesenteric lymph node. TLR9 immunolabeling between the different types of lesions was compared using a complete H-score. Focal and diffuse paucibacillary forms contained significantly increased TLR2-expressing macrophages outside of the lesions compared with the controls and diffuse multibacillary forms, and moderate TLR9 immunolabeling within granulomas. In the multibacillary granulomatous lesions, the expression of TLR1 and TLR4 was observed as well as increased TLR9 expression compared with the rest of the groups. Differences in the predominance of one type or another of TLR allows us to elucidate the importance of the innate immune response and its possible role in the development of the different types of paratuberculosis lesions.
Paratuberculosis or Johne’s disease, caused by Mycobacterium avium subsp. paratuberculosis (MAP), 34 is a chronic, granulomatous infectious disease of the intestine of domestic and wild ruminants. It has a worldwide distribution and continues to cause important economic loses in the livestock industry. 24
MAP infection occurs primarily via the fecal-oral route, and an early-age susceptibility has been established in calves. 46 Infected animals may show different outcomes of the infection such as resistance, development of lifelong latent stages, or clinical forms appearing in the adulthood.5,19,76 In this sense, intestinal lesions associated with bovine paratuberculosis have been described as focal, diffuse multibacillary, and paucibacillary, 27 and these presentations have been shown to be related to the different phases of the infection. 63 Nevertheless, the pathogenesis of paratuberculosis is poorly understood, and the factors that control the innate immunological response against MAP and determine the outcome of the infection remain unknown. Different lymphocyte11,22,40,82 and macrophages 21 subpopulations have been evaluated in relation to paratuberculosis lesions.
The Toll-like receptors (TLRs) are a family of pattern recognition receptors (PRRs) which play a critical role in the recognition of conserved microbial structures named pathogen-associated molecular patterns (PAMPs). 77 Upon PAMP recognition, TLRs induce the secretion of inflammatory cytokines by innate immune cells, 57 thereby priming lymphocytes to mount an adaptive, antigen-specific immune response. 36 TLRs transduce signals mainly via 2 major pathways dependent on adaptors MyD88 or TRIF, 15 leading to the production of pro-inflammatory cytokines which reinforce immunity at epithelial barriers like the intestine.44,58
Although there are 10 known TLRs in cattle, 47 the most frequently associated TLR genes with susceptibility to MAP infection are TLR1, TLR2, TLR4, and TLR9. 42 TLR2 and TLR4 were the first receptors proven to recognize MAP 23 and to play a critical role in mycobacterial infection. 71 TLR1 serve as co-receptor for TLR2, which is the central receptor involved in the recognition of mycobacteria, expanding the range of its ligands.33,48 TLR2 and TLR4 are both located in the plasma membrane and detect bacterial lipoproteins and lipopolysaccharide, respectively.33,35,62 TLR9 cooperates with TLR2 to mediate optimal resistance to mycobacteria37,48 and is found in intracellular compartments, detecting unmethylated CpG containing single stranded DNA. 51
Most studies on the role of TLRs in bovine paratuberculosis have focused only on the investigation of single nucleotide polymorphisms (SNPs) within TLR genes associated with host resistance or susceptibility to infection,41,50,65,72 with scant information about the expression of these receptors in the tissue. 75 These studies have found significant associations between multiple SNPs within TLR1, TLR2, and TLR4, and host’s resistance or susceptibility to MAP infection, but results for individual SNP significance are inconsistent 42 so that the role of TLRs at the site of MAP infection remains unknown.
The aim of this study was the evaluation of the in situ expression of TLR1, TLR2, TLR4, and TLR9 in relation to the different types of lesions associated with bovine paratuberculosis, using immunohistochemical techniques. Special attention was paid to assessment of the amount of positively immunolabeled cells, immunolabeled cell types, and distribution along different intestinal areas and regional lymph nodes.
Material and Methods
Ethical Approval
Most samples used in this study came from farm animals, slaughtered for production or veterinary reasons, and were procured post-mortem. Some samples were obtained from animals used in old studies for other purposes. Therefore, the animals were not subjected to experimental procedures for the aim of this work and do not require specific ethical approval according to European Directive 2010/63 and Royal Decree 53/2013 (Art 2.5).
Animals and Experimental Design
A total of 20 Holstein cattle were included in the study. Eight were calves used in a previous study of experimental infection with MAP. 53 Briefly, 4, 4-month-old calves were orally challenged with a dose of 6 x 1010 bacilli obtained directly from the intestinal mucosa of a clinically affected cow that was purified and administered in 6 separated doses given every 2 days. Calves were culled at 120 days post-infection (dpi) and 270 dpi. Additional 4 control noninfected cattle were culled at 270 dpi. All these animals were sedated with xylazine (Rompun; Bayer, Mannheim, Germany) and euthanized by an intravenous overdose of embutramide and mebezonium iodide (T61; Intervet, Salamanca, Spain). The remaining 12 animals were adult dry cows ranging from 3 to 6 years of age that belonged to 2 commercial dairy herds not vaccinated against MAP-infection in which a follow-up survey on losses due to paratuberculosis was ongoing. These animals were culled due to production reasons and for consumption in an authorized slaughterhouse according to the standard methods in the current legislation. Infection was confirmed following the methodology described elsewhere.25,27
Tissue Samples and Lesion Classification
Samples from ileocecal valve; distal, middle, or proximal ileum; jejunum (with and without Peyer’s patches) as well as associated cecal and cranial and caudal jejunal mesenteric lymph nodes from each animal were evaluated for the study. After fixation in 10% buffered formalin, samples were routinely embedded in paraffin after dehydration through a graded alcohol series and xylene treatment. Tissue sections of 3-μm thick were obtained from each sample and stained with Harris’s hematoxylin and eosin and Ziehl-Neelsen method for acid-fast bacilli (AFB) detection. A semi-quantitative classification of the samples according to the load of AFB present was made from 1 (scant AFB in the cytoplasm of macrophages, seen with difficulty), 2 (moderate, easily detectable bacilli) to 3 (high load of AFB).
Lesions were then categorized as focal, diffuse multibacillary, and diffuse paucibacillary according to the location of granulomas, inflammatory cell types present in the infiltrate, and the amount of AFB, following the guidelines provided by González et al. 27 . Focal lesions, associated with subclinical stages, were composed of small and well-demarcated granulomas, up to 40–50 epithelioid cells, located exclusively in the interfollicular areas of the intestinal lymphoid tissue and the paracortical regions of the lymph nodes. Diffuse lesions, seen in animals with clinical forms of the disease, were characterized by widespread granulomatous lymphadenitis and enteritis due to the presence of a severe inflammatory infiltrate. Diffuse paucibacillary lesions contained large numbers of lymphocytes in the infiltrate, with some epithelioid and giant cells, but none or very few AFB. Multibacillary lesions were instead characterized by the predominance of epithelioid cells harboring large numbers of AFB.
Immunohistochemistry
Tissue samples were immunolabeled with primary TLR1 (GTX47794, GeneTex), TLR2 (MA1-40080, Invitrogen), TLR4 (ORB11489, Biorbyt), and TLR9 (GTX31295, GeneTex) antibodies with verified customer’s review of reactivity in sections of formalin-fixed samples (Table 1). The specificity of these primary antibodies on bovine tissues was evaluated by western blot analysis of buffy coat cells collected from heparinized cattle blood and stimulated in vitro with concanavalin A (con A) to induce TLR expression. Cultured cell proteins were separated by SDS-PAGE, transferred to a nitrocellulose membrane, and tested for antigen-antibody specificity. The validation of each antibody was confirmed by the presence of specific bands corresponding to the expected molecular weight of the target TLR.
List of primary antibodies and antigen retrieval conditions used in this study for immunohistochemistry.
PTLink (Dako-Agilent Technologies, Santa Clara, USA).
Immunohistochemical analysis was carried out in a total of 20 intestinal tissue sections (with lymphoid tissue) and 20 mesenteric lymph node samples, one from each animal included in the study. Heat-mediated antigen retrieval was performed using PT Link system (Dako-Agilent technologies, Santa Clara, USA) for 20 minutes at 95°C with different target retrieval solutions depending on the primary antibody used. The immunohistochemical procedure was carried out as described elsewhere. 11 Appropriate species- and isotype-matched immunoglobulins were used as control. These included sections with an isotype control for the primary antibody and the omission of the primary antibody.
Immunolabeling Evaluation
Due to the heterogeneous distribution of the immunolabeled cells, a differential cell count was carried out on the lamina propria (LP), gut-associated lymphoid tissue (GALT), and mesenteric lymph node (LN) from each sample included in the study. Immunolabeled cells were classified according to the morphological features.
Thus, 30 randomly chosen fields from each area were photographed at 400× (0.0576 mm2) (Nikon Eclipse Ci microscope with Digital MD-E3-6-3 digital camera) and differential counting of positively immunolabeled cells was performed using the Cell Counting add-on of Image J program (US National Institutes of Health, Bethesda, Maryland, USA). Additionally, the distribution of the labeled cells was evaluated in relation to the presence of granulomas.
For the TLR9 antibody, the immunolabeling intensity between the different types of granulomatous lesions (focal, diffuse paucibacillary, diffuse multibacillary) was further compared using the quantitative H-score. 16 Briefly, a total of 5 different slides for each type of lesion were selected and 5 randomly chosen fields containing granulomas were evaluated at 630×. H-score was calculated by adding the percentage (0–100) of macrophages labeled at a certain staining intensity (weak, moderate, or intense) multiplied by the staining intensity score (1, weak; 2, moderate; and 3, intense) present in each microscopic field. The final H-score between 0 and 300 was obtained for each lesion category considering the mean value for the 25 areas counted.
Evaluation of the differential cell immunolabeling was assessed independently by duplicate (D.Z. and V.P.) and discordant results were reviewed in a multi-headed microscope to reach consensus within a third analysis.
Antibody Validation
Primary antibodies used for immunohistochemistry were validated by immunoblot analysis. Specifically, bovine peripheral blood leukocytes (PBLs) were stimulated in vitro with con A for TLR expression. 73 Afterward, PBL proteins were electrophoretically separated, transferred to nitrocellulose membrane and used for testing the specificity in antigen-antibody recognition.
The isolation protocol of PBLs used herein was modified from that proposed by Criado et al. 12 Briefly, 30 mL of heparinized blood from a 2-week-old healthy Holstein calf were diluted with 20 mL of phosphate-buffered saline (PBS) and centrifuged at 650 × g for 30 minutes at 21°C. The buffy coat was collected, and erythrocyte lysis was performed with 0.03% sodium bicarbonate for 10 seconds at 4°C. After that, PBS was added up to 50 mL and PBLs were centrifugated at 300 × g for 10 minutes at 4°C. PBLs were resuspended in RPMI 1640 (11835-063, Gibco, Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 5% heat-inactivated fetal bovine serum (10500064, Gibco, Paisley, UK). The number of viable PBLs was determined by trypan blue (15250061, Gibco, Paisley, UK) exclusion test in a Neubauer chamber using an automatized Corning Cell Counter (6749, Corning, NY, USA). A total of 6 × 107 cells were transferred to a 6-well plate with 3 mL of supplemented RPMI 1640 medium and 10 µg/mL con A (C2272, Sigma-Aldrich, Saint Louis, MO, USA) and incubated at 37°C in a 5% CO2 atmosphere for 24 hours. After incubation, PBLs were collected, washed with PBS, and centrifugated at 300 × g for 10 minutes at 4°C; the supernatant was discarded, and the cell pellet was stored at −80°C until use.
SDS-PAGE and western blotting was carried out as previously described, 20 with few modifications. Briefly, PBLs were thawed and diluted in 600 μL of 50 mM Tris-Cl containing 2% sodium dodecyl sulfate, 10% glycerol, 0.05% bromophenol blue, 100 mM dithiothreitol, disrupted by ultrasound treatment (UP-200H, Hielscher Ultrasonics, Teltow, Germany) in an ice-bath, and then boiled for 5 min. Electrophoresis was performed in 10-well, precast, 12% polyacrylamide gel (4561043, Bio-Rad Laboratories, Hercules, CA, USA) at constant 150 V. Precision Plus Kaleidoscope weight standards (1610375, Bio-Rad Laboratories, Hercules, CA, USA) were used to estimate the molecular weights of the proteins labeled by the primary antibodies. Proteins were electrophoretically transferred to a nitrocellulose membrane (162-0113, Bio-Rad Laboratories, Hercules, CA, USA). Then, the immunoblotting was performed as described elsewhere. 1 Briefly, membranes were washed in Tris-phosphate-buffered with 0.05% Tween-20 (TBS-T), and then incubated overnight in blocking buffer (TBS-T containing 5% nonfat milk for TLR4 and TLR9 antibodies, or 5% bovine serum albumin for TLR1 and TLR2 antibodies). After being washed in TBS-T, the membranes were incubated overnight at 4 ºC with the primary antibodies diluted in TBS-T containing 0.5% milk or 3% bovine serum albumin according to the blocking buffer. Specifically, TLR1, TLR2, and TLR4 were diluted at 1:500 whereas TLR9 was diluted at 1:1000. After 3 washes with TBS-T for 10 minutes each, membranes were exposed for 1 hour to an appropriate goat anti-rabbit (YH381824, Invitrogen, Waltham, MA, USA) or goat anti-mouse (YJ379318, Invitrogen, Waltham, MA, USA) horseradish peroxidase-conjugated secondary antibodies at 1:500 dilution titer in TBS-T. After 3 washes with TBS-T and one with TBS, membranes were developed using 4-chloro-1-naphtol (1706534, Bio-Rad Laboratories, Hercules, CA, USA) as chromogenic substrate. After 15 min, the reaction was stopped using distilled water.
Blots were photographed using Molecular Imager Gel Doc XR+ System (1708195EDU, Bio-Rad Laboratories, Hercules, CA, USA).
Statistical Analysis
Cell counts, according to the type of lesion, were expressed as means, standard deviations, and ranges (minimum and maximum) using conventional statistical descriptive procedures. The Kolmogorov-Smirnov test was used to assess the normality of the data. Since the values obtained did not fit a normal distribution and could not be statistically transformed, nonparametric tests were used to compare between groups. Specifically, to compare the number of cells between the different lesion status categories (no lesion/lesion) as well as between the different lesion types (control, focal, diffuse paucibacillary, and multibacillary) and the different intestinal locations (LP, GALT, and LN), the nonparametric tests U Mann-Whitney and Kruskal-Wallis were used, respectively. In a second step, to find out which pair of groups differences were found, a post-hoc analysis (Pairwise Wilcoxon Rank Sum) was performed with Bonferroni correction for the level of significance. 26 Immunohistochemical data (H-score) for TLR9 immunolabeling was analyzed likewise. Correlation between AFB category and H-score was evaluated using a Spearman test. Values of P < .05 were considered statistically significant.
All statistical analyses were performed with the R software version 3.6.1(R Foundation, Vienna, Austria)
Results
Distribution of Immunolabeled Cells
Positively immunolabeled cells were identified based on their morphologic features. The majority of TLR1+, TLR2+, TLR4+, and TLR9+ cells had abundant amoeboid-shaped cytoplasm, morphologically consistent with macrophages.
In samples of the jejunum from control cows (Fig. 1), immunolabeled macrophages showing uniform membrane and cytoplasmic labeling for TLR1 (Fig. 1a), TLR2 (Fig. 1d), TLR4 (Fig. 1g), and, to lesser extent, TLR9 (Fig. 1j) were scattered throughout the LP. In the Peyer’s patches, small groups of macrophages expressing TLR2 (Fig. 1e) and, to lesser extent, TLR1 (Fig. 1b) were clustered in the dome, below the M cell layer. Few scattered TLR1+, TLR2+, TLR4+ (Fig. 1h), and TLR9+ (Fig. 1k) cells were present in the interfollicular region. In the jejunal lymph nodes, positively immunolabeled macrophages expressing TLR2 (Fig. 1f, insert) and scarce TLR1 (Fig. 1c, insert), TLR4 (Fig. 1j, insert), and TLR9 (Fig. 1l, insert) cells were concentrated in the medullary cords. Few macrophages in the medullary sinuses had weaker, cytoplasmic TLR9 expression. In the cortex, few TLR1+ (Fig. 1c), TLR2+ (Fig. 1f), TLR4+ (Fig.1j), and TLR9+ (Fig. 1l) macrophages were scattered principally in the paracortical region.
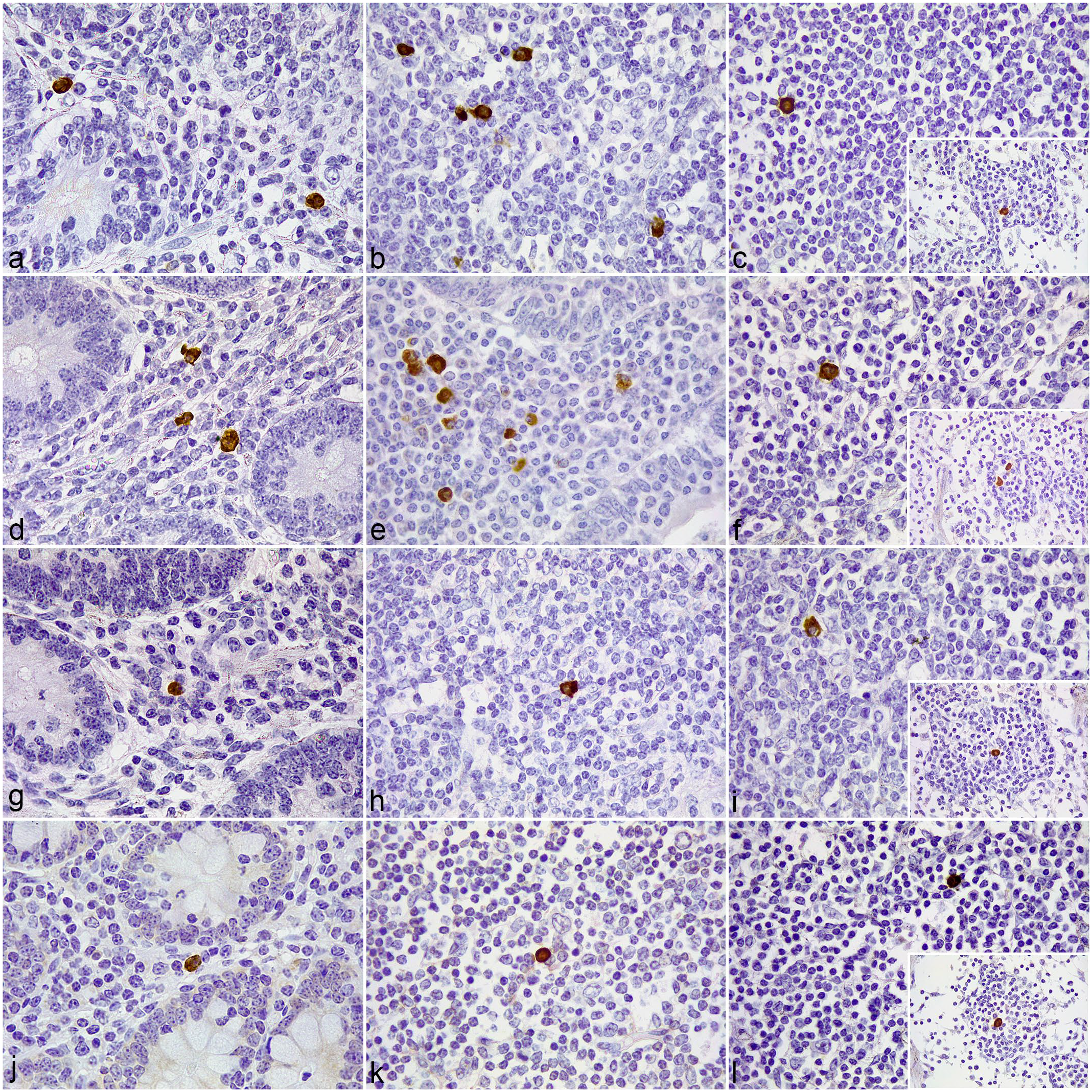
Tissue sections of the jejunum and jejunal lymph node (LN) from an uninfected control cattle.
In jejunal samples with focal lesions (Fig. 2), the distribution of positively immunolabeled cells outside of the granulomas was similar to the immunolabeling pattern observed in the control cows (Fig. 2a, d, g, j). Nevertheless, mild to moderate, uniform to discrete punctate immunolabeling for TLR9 (Fig. 2k-l) (but not for TLR1 (Fig. 2b-c), TLR2 (Fig. 2e-f), or TLR4 (Fig. 2h-i)) was observed in the cytoplasm of the epithelioid cells forming the granulomas in both the jejunal Peyer’s patches and lymph nodes. TLR2-immunolabeled polymorphonuclear (PMN) cells were occasionally present inside the lesions. Occasionally, few TLR1 and TLR2 immunolabeled macrophages gathered around small subcapsular granulomas at the lymph nodes.
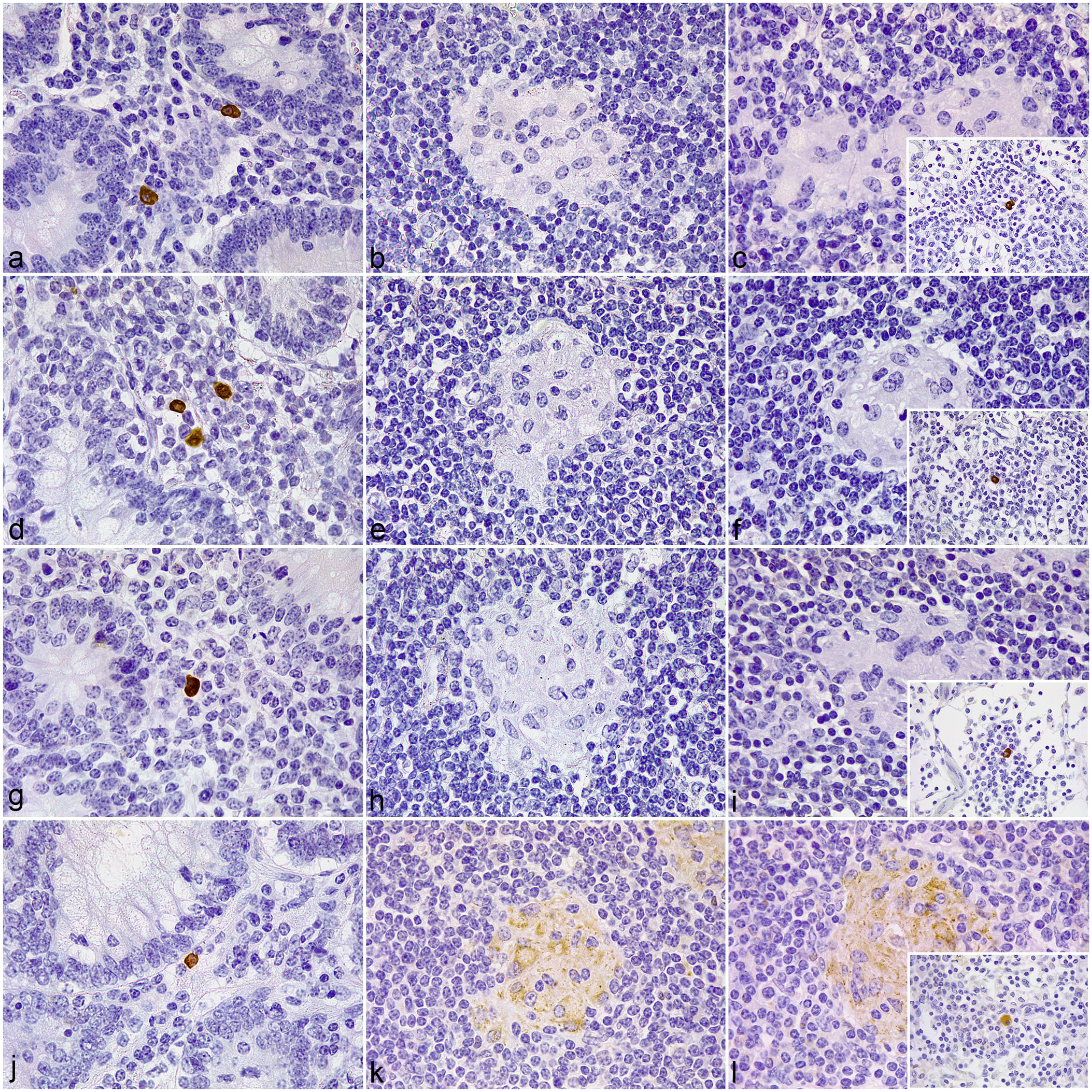
Tissue sections of the jejunum and jejunal lymph node (LN) showing focal lesions associated with Mycobacterium avium subspecies paratuberculosis infection in cattle.
In the diffuse paucibacillary lesions (Fig. 3), immunolabeled macrophages expressing TLR1, TLR2, and scarce TLR4 appeared intermingled between the lymphocytic infiltrate in the ileal lamina propria, lymphoid tissue, and paracortex of the jejunal lymph nodes. Similar to the focal lesions, there was no TLR1 (Fig. 3a-c), TLR2 (Fig. 3d-f), or TLR4 (Fig. 3g-i) immunolabeling detected in the epithelioid and Langhans giant cells forming the granulomas, although these cells expressed TLR9 (Fig. 3j-l). TLR2 immunolabeled PMN cells were also occasionally present within the granulomatous lesions (Figs. 3d-e). However, in animals with intermediate lesions where the presence of relatively large numbers of AFB was confirmed inside the granulomatous lesions, macrophages and Langhans giant cells displayed strong TLR1 and TLR4 immunolabeling.
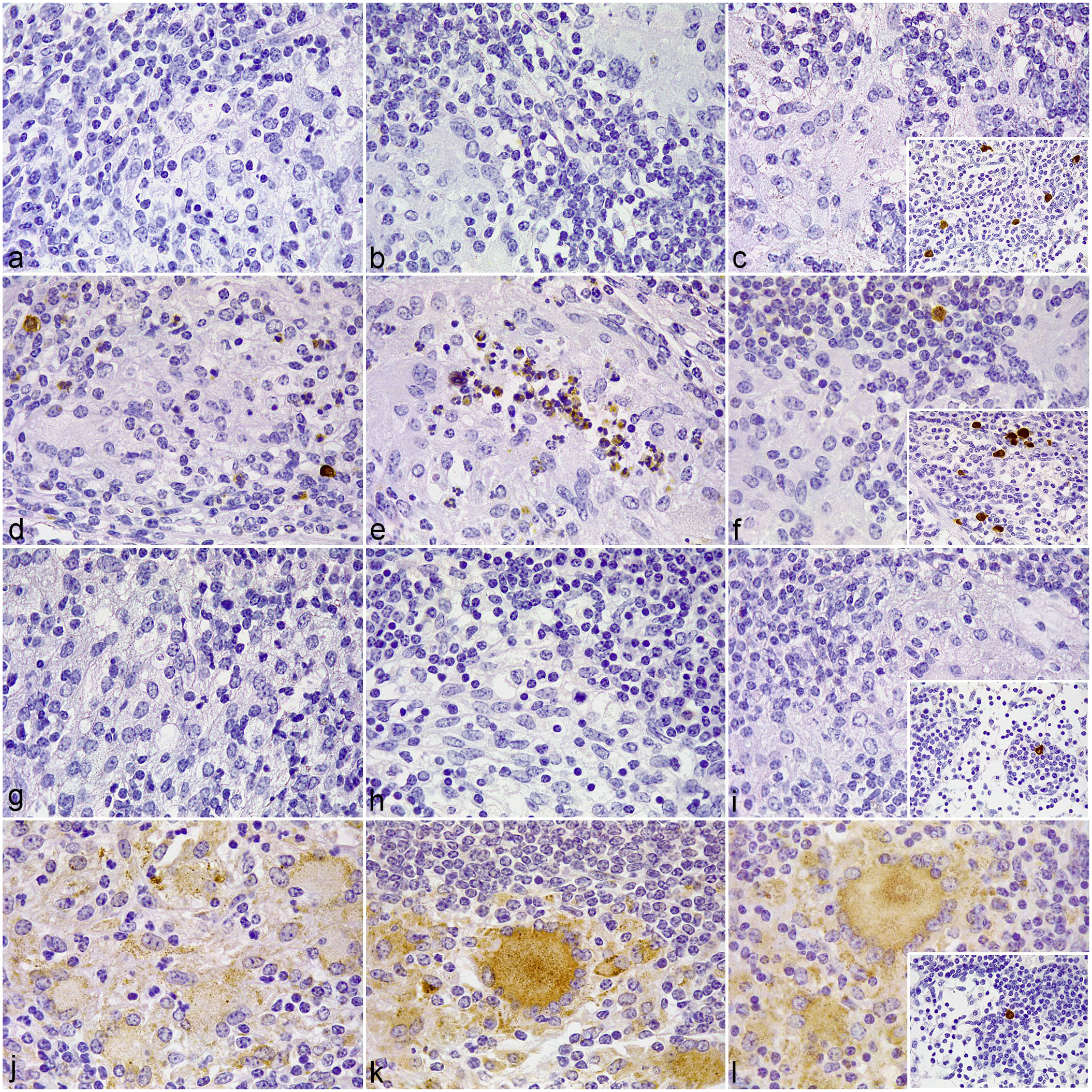
Tissue sections of the ileum and jejunal lymph node (LN) showing diffuse paucibacillary lesions associated with Mycobacterium avium subspecies paratuberculosis infection in cattle.
In diffuse multibacillary lesions (Fig. 4), a high number of positively immunolabeled epithelioid and Langhans giant cells expressing TLR1 (Fig. 4a-c), TLR4 (Fig. 4g-i), and TLR9 (Fig. j-l), but not TLR2 (Fig. 4d-f), were seen in close relationship to large numbers of bacteria inside the granulomatous lesions. Few TLR1+, TLR2+, TLR4+, and TLR9+ macrophages were scattered in unaffected areas of the ileal lamina propria, lymphoid tissue, and paracortical region of the jejunal lymph node. Medullary sinus macrophages had more intense TLR9 immunolabeling compared with the other lesion categories (Fig. 4l, insert). TLR4-labeled cells present in granulomatous lesions from all 3 locations analyzed exhibited a particular point-like type of labeling throughout the cytoplasm (Fig. 4g, insert), in contrast with the coarser granular staining seen for the other antibodies.
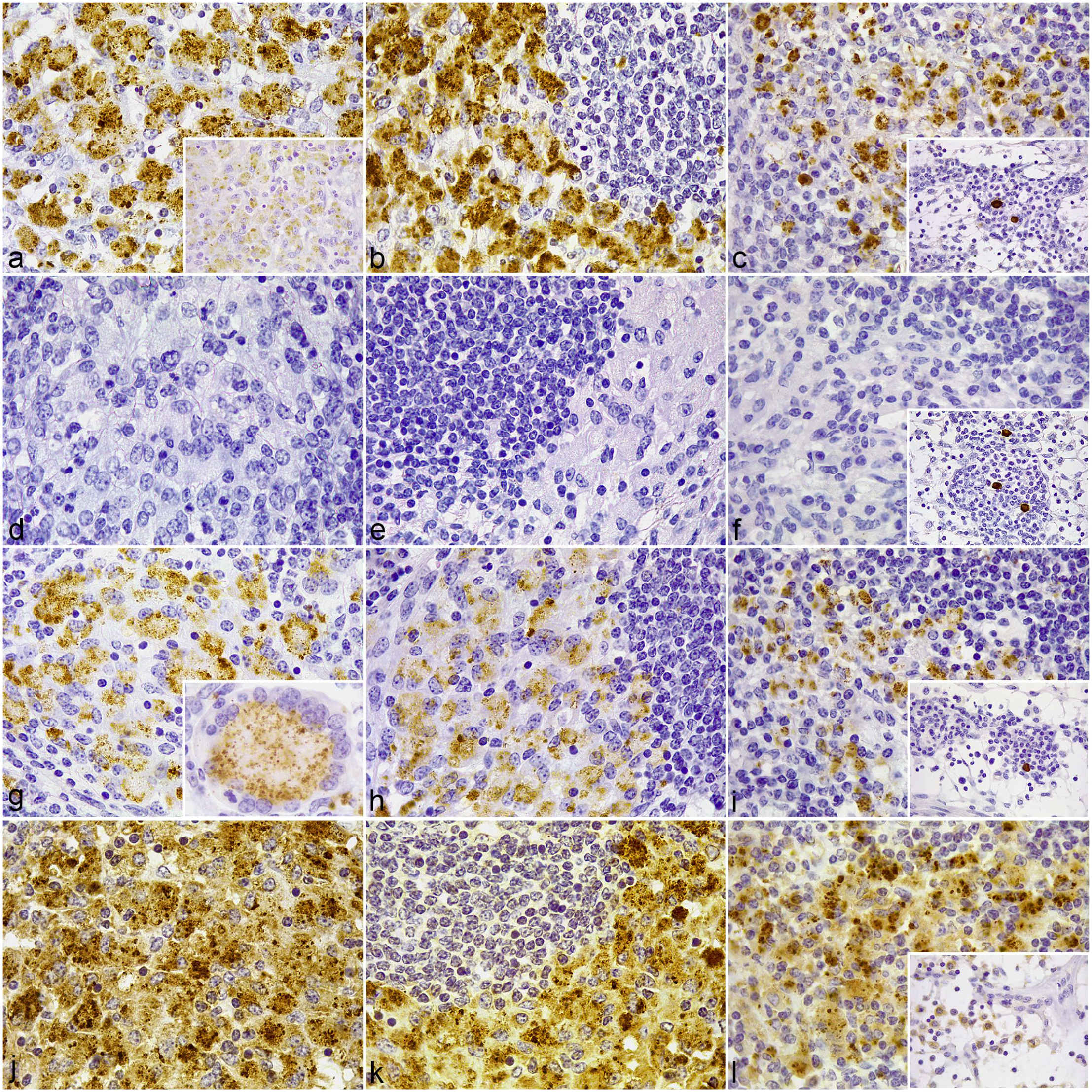
Tissue sections of the ileum and jejunal lymph node (LN) showing diffuse multibacillary lesions associated with Mycobacterium avium subspecies paratuberculosis (MAP) infection in cattle.
Associations Between Immunolabeled Cell Numbers and Lesion Type
Samples of the different types of lesions showed differences in the number of immunolabeled cells. For cell-counting, only those positive cells with a clear macrophage morphology were considered. For all antibodies employed, tissue sections presenting MAP-associated lesions showed significantly higher number of positively immunolabeled cells than those from the control animals (P < .001). The number of TLR1 and TLR4-labeled macrophages was significantly higher in the diffuse multibacillary lesions compared with the rest of the groups (P < .001). The highest number of TLR2-positive macrophages was observed in samples with diffuse paucibacillary lesions and was significantly higher (P < .001) than in the rest of the groups. The number of TLR9-immunolabeled macrophages was significantly higher in the groups with diffuse lesions, both paucibacillary and multibacillary (P < .001) (Table 2).
Mean number of positively immunolabeled macrophages for TLR1, TLR2, TLR4, and TLR9 antibodies per microscopic field according to the type of lesion.
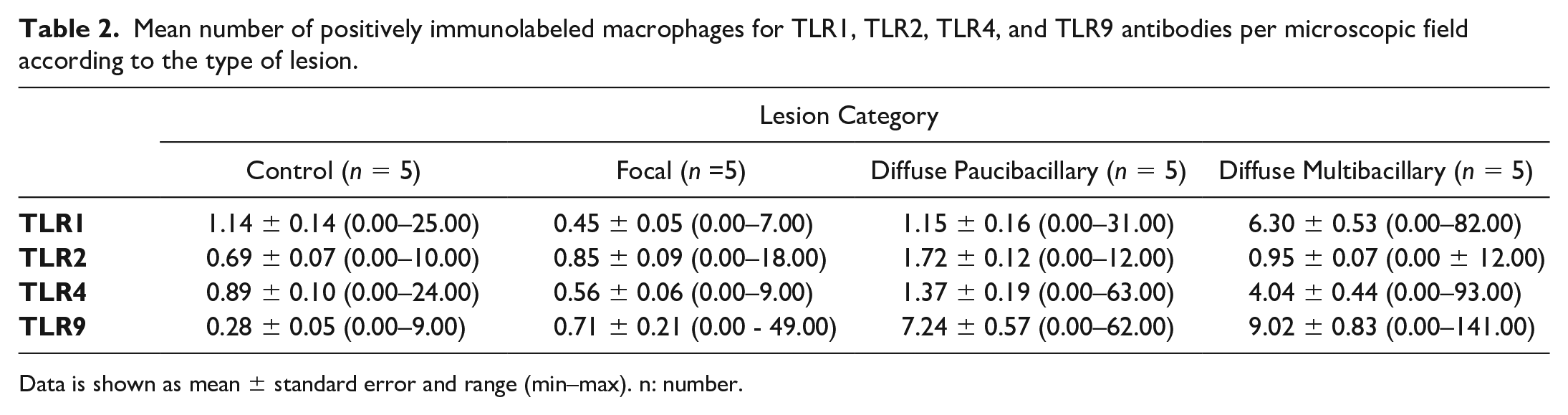
Data is shown as mean ± standard error and range (min–max). n: number.
A significant increase in the number positively TLR1 and TLR4-immunolabeled macrophages was found principally in the LP of cows with multibacillary lesions compared with the other groups (P < .001). These results were similar for TLR9 (P < .001), although in this case the highest number of labeled macrophages was observed at the GALT of animals with paucibacillary lesions (P < .001). Cows with paucibacillary lesions also showed significantly higher levels of TLR2-expressing macrophages in all the locations analyzed (P < .05), except for the LP, which showed no differences with the focal lesion group (P = .25333) (Fig. 5).
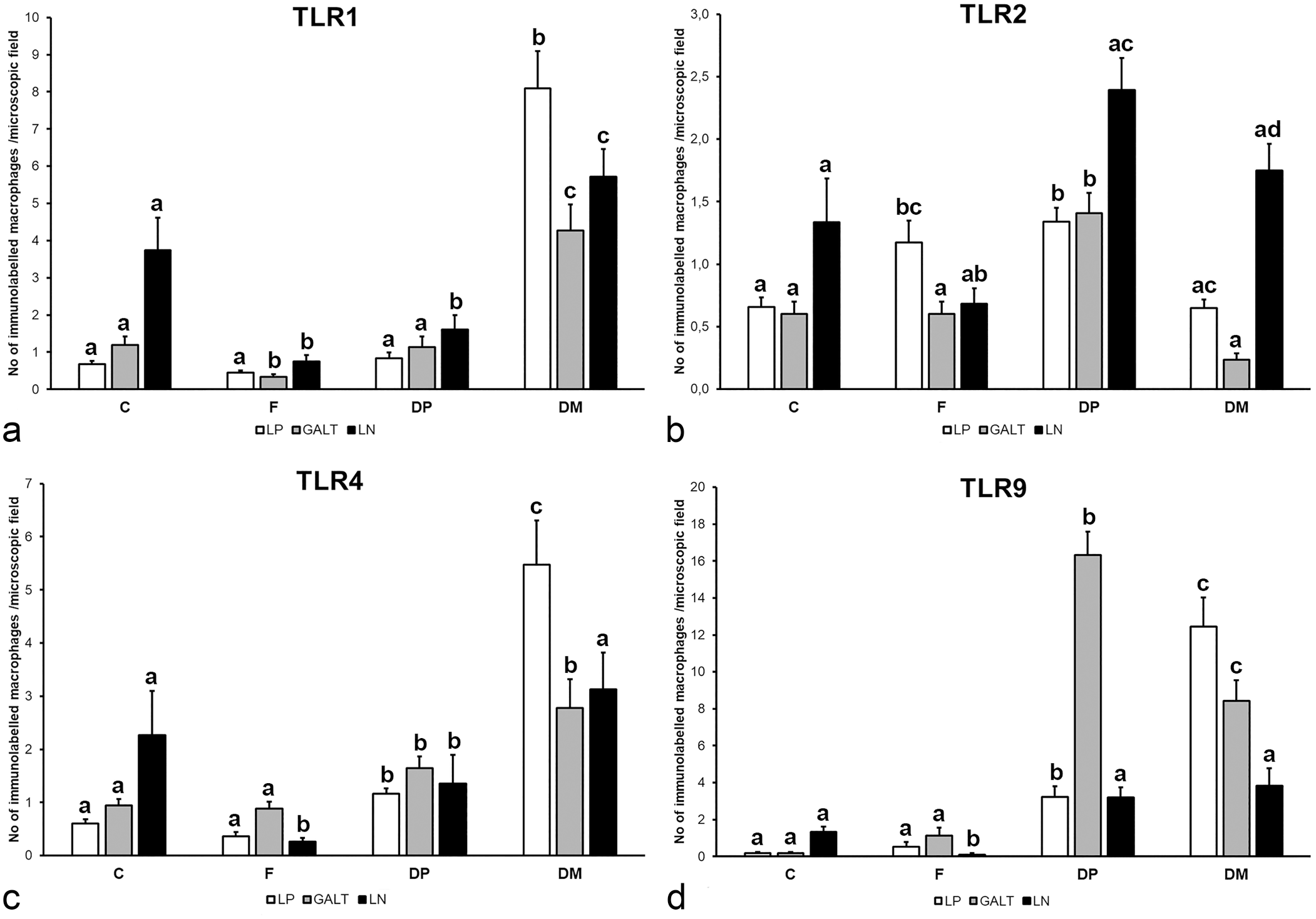
Mean number of immunolabeled macrophages for (a) TLR1, (b) TLR2, (c) TLR4, and (d) TLR9 per microscopic field (0.0576 mm2) in each area according to the type of lesion. C, control (n = 5); F, focal (n = 5); DP, diffuse paucibacillary (n = 5); DM, diffuse multibacillary (n = 5); LP, lamina propria; GALT, gut-associated lymphoid tissue; LN; lymph node. Different superscript letters following the values indicate statistical significance (P < .05). Error bars: standard error.
Histologic Scoring for TLR9 and Correlation with AFB Burden
Epithelioid macrophages forming the different types of granulomatous lesions (focal, diffuse paucibacillary, diffuse multibacillary) showed variable intensity for TLR9 immunolabeling (Figs. 2k-l, 3j-l, 4j-l). Thus, a complete H-score was performed to semiquantitatively evaluate differences between the different lesion categories. 16 The H-score for TLR9 was significantly higher in multibacillary lesions than in focal (P < .001) and diffuse paucibacillary forms (P < .001). The diffuse paucibacillary lesions showed a significant increase in the H-score value over the focal forms (P < .05).
H-score for TLR9 was positively correlated with the burden of AFB (P < .001) (Supplemental Figure S1).
Immunoblot Analysis
The specificity of the primary antibodies used for immunohistochemistry analysis was probed by immunoblotting. Validation of each antibody was achieved through the demonstration of specific band(s) of the appropriate molecular weight for the target TLR protein, with minimal cross-reacting bands (Supplemental Figure S2). A specific band between 75 and 100 kDa was detected for all the primary antibodies analyzed. According to the information provided by Universal Protein Resource (UniProt) database 7 regarding the molecular weights of bovine TLR2 (90 kDa), TLR4 (96 kDa), TLR9 (115 kDa), and human TLR1 (90 kDa), the bands observed were consistent with the detection of TLR1, TLR2, TLR4, and TLR9 proteins. It should be noted that several factors can affect protein migration on SDS-PAGE, including post-translational modifications or detergent binding,64,67 which may explain the slight difference between the expected (115 kDa) and observed (~100 kDa) molecular weight for TLR9 protein. In the case of TLR4, the detection of 2 additional bands of ~37 kDa and ~25 kDa probably indicate the presence of alternative glycosylation states of TLR4/MD-2 complex, as reported in other studies.8,14,59
Discussion
The evidence of the critical role of innate immunity in host resistance to MAP infection has increased in recent years.38,42 In this context, TLRs are an important family of PRRs involved in microbial detection and the onset of innate immune responses against invading pathogens. 61 This work allows the assessment of unexplored features regarding the expression of TLRs in the intestine of cows infected with paratuberculosis, such as the immunolabeled cell types and tissue distribution of positively immunolabeled cells in relation to the different MAP-associated granulomatous lesions. All the TLRs analyzed in this work were principally expressed by the monocyte-derived cells such as macrophages, epithelioid cells, and Langhans giant cells. 31
Interestingly, macrophages and epithelioid cells forming focal and diffuse paucibacillary granulomatous lesions showed no immunoreactivity for cell membrane TLRs including TLR1, TLR2, and TLR4, probably due to insufficient antigenic stimulus because of the lack of extracellular mycobacteria or bacterial components.31,54 The scant bacilli present in these pathological forms are typically located inside macrophages, 27 thereby stimulating expression of intracellular TLR9. However, heavily infected macrophages from multibacillary granulomatous lesions markedly expressed TLR1 and TLR4, along with TLR9, but not TLR2. According to other studies, this strong expression of surface PRRs may be explained by the presence of abundant, free MAP antigens produced during mycobacterial growth 3 and released due to the lysis of highly infected macrophages within granulomas. 39
TLR2 is the main receptor involved in mycobacteria detection.30,79 In the present study, the distribution of positively immunolabeled macrophages showed no relationship to the granulomas in any of the pathological forms analyzed. Nevertheless, the expression of TLR2 by PMN cells located within focal and paucibacillary granulomatous lesions could indicate a potential role of these cells in the clearance of infection. 43 In depth, a significant increase in the number of TLR2+ macrophages was observed in the intestine of cows with focal and paucibacillary forms of the disease, where a Th1-type immune response predominates, compared with multibacillary lesions where the Th2 profile and M2 macrophages abound.21,22 Indeed, the upregulation of TLR2, observed exclusively in the macrophages from unaffected areas, might not be mediated by the presence of the bacteria but rather by the effect of the production of large quantities of interferon (IFN)-γ by T lymphocytes.22,60,74 Therefore, TLR2 may play a role in the surveillance against MAP outside of the granulomas, despite representing a well-recognized target for pathogen-mediated immune subversion. 80 In this sense, it has been demonstrated that both tuberculous and nontuberculous mycobacteria can impair TLR2 expression in human macrophages.68,69 These findings are consistent with the lack of immunoreactivity for TLR2 antibody observed within multibacillary granulomatous lesions, where the release of mycobacterial components by necrotic macrophages stimulated the expression of other surface TLRs.3,39 Nonetheless, it could be also hypothesized that this lack of TLR2 expression may be associated with the anti-inflammatory activity of the macrophages in this type of lesion. 66 However, in vitro studies have shown that MAP can manipulate TLR2 signaling to favor anti-inflammatory IL-10 production and prevent phagosome acidification-maturation in bovine macrophages.5,80 In fact, this TLR2-dependent production of IL-10 is considered one of the most critical immune evasion mechanisms of MAP. 29 Nevertheless, Fernández et al. 21 verified a strong IL-10 immunolabeling in MAP-associated multibacillary lesions despite the lack of reactivity for TLR2. These findings would indicate an alternative source of IL-10 production by infected macrophages in vivo.
TLR1-TLR2 heterodimers trigger the recognition of mycobacterial triacylated lipopeptides where 2 lipid chains interact with TLR2 while the third binds TLR1. 33 In the present study, we demonstrated a significant increase of positively immunolabeled macrophages for TLR1 antibody in the intestine of clinically affected cows presenting diffuse multibacillary forms of the disease, especially within the granulomatous lesions, contrary to TLR2. For this reason, lipoprotein-induced TLR1-TLR2 heterodimerization could be compromised in this type of lesions and, therefore, subsequent signaling.18,33 Thus, the expression of TLR1 observed in multibacillary granulomas cannot be associated to the protection against MAP infection due to the absence of TLR2.
TLR4 participates in the immunological response against mycobacteria in many ways including cytokine production, phagocytosis, autophagy, lysosomal degradation, and regulation of cell death.2,28,45,70,81 The increased expression of TLR4 in cows with diffuse multibacillary forms observed in the present study would be consistent with previous findings. 32 Furthermore, unlike the other TLRs, positive epithelioid and Langhans giant cells located within multibacillary lesions displayed a characteristic point-like type of immunolabeling throughout the cytoplasm. After PAMP-TLR4 binding, 2 signaling pathways are sequentially triggered. 10 The first signaling pathway is induced in the plasma membrane (Myd88-dependent) and the second pathway is induced after receptor endocytosis (TRIF-dependent), which could explain the punctate immunolabeling of TLR4 observed in the cytoplasm of epithelioid and Langhans giant cells. In this sense, TRIF-dependent signaling induces the expression of type I IFN and anti-inflammatory IL-10,9,10 which decrease the Th1 immune response resulting in decreased production of IL-12 and IFN-γ. 17 Therefore, the TLR4-dependent TRIF signaling cascade could contribute to the dampening of the Th1-type response in favor of a Th2-based anti-inflammatory environment in the intestine of clinically affected cows showing diffuse multibacillary forms of the disease, as observed in macrophage profiles in lesions of this type. 21 This TLR4-dependent production of type I IFN may be induced by the mycobacteria as observed in tuberculosis infections 49 or instead be associated to the presence of transforming growth factor-beta 1 in multibacillary granulomas.21,52,55
TLR9 detects unmethylated CpG DNA and cooperates with TLR2 to mediate optimal resistance to mycobacterial infections.6,37,48 In our study, we identified TLR9 immunoreactivity in MAP-associated granulomatous lesions from all pathological forms analyzed, and the H-score for TLR9 was directly correlated with bacterial burden. Thus, the rate of TLR9 expression by infected macrophages may be dependent on the intracellular concentration of mycobacterial DNA. 13 However, TLR9 signaling probably stimulates IL-12 expression by infected macrophages forming the focal and diffuse paucibacillary granulomatous lesions, and thus, contributes to the Th1-associated IFN-γ production by lymphocytes 6 in these pathological forms characterized by strong local Th1 type response. 21 Nevertheless, there is evidence of MAP ability to inhibit classical TLR9-mediated proinflammatory responses in favor of noncanonical Pyk2 signaling pathway for intracellular survival, 4 which could indicate that the overexpression of TLR9 in multibacillary lesions with higher H-score values and a Th2 profile, 21 represent a compensatory rather than protective mechanism.
The patterns of expression of the different TLRs in the pathological forms analyzed in this study differ from those observed in sheep.56,78 This suggests potential differences in the innate responses to paratuberculosis infection of these species and, thus, in the pathogenesis of the disease.
According to the results of this study, TLR1, TLR2, TLR4, and TLR9 could play a role in the development of the different types of lesions associated with bovine paratuberculosis. The role of TLR9 in the innate response against MAP in focal and diffuse paucibacillary lesions and in the support of IL-12 mediated Th1 type immunity may be key, since the scant bacilli present within the granulomas are located inside host mononuclear cells. The liberation of IFN-γ by T lymphocytes, induced by IL-12, probably stimulates the expression of TLR2 by surrounding macrophages for increased surveillance at the mucosa. However, cell membrane PRRs may acquire a more predominant role in the innate response of clinically affected cows presenting heavily infected intestines with diffuse multibacillary forms, where the presence of extracellular mycobacterial antigens released by necrotic macrophages harboring large numbers of AFB stimulate the expression of surface TLR1 and TLR4 by surrounding innate immune cells. Nevertheless, TLR2 expression seem to be totally inhibited in this type of lesion, probably due to the anti-inflammatory nature of the macrophages or as a mechanism of immune evasion of the bacteria, as demonstrated in related mycobacteriosis.68,69 Furthermore, in these pathological forms, TLR4 signaling occurs mainly via TRIF-dependent pathway, which leads to type I IFN and IL-10 production, dampening cell-mediated immunity in favor of adaptive Th2 immune response.9,10,17
This immunohistochemical assessment of the expression of different TLRs within MAP infection–associated lesions pretend to contribute to elucidate the precise role of these receptors during MAP infection for increased knowledge on the pathogenesis of the disease.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241302850 – Supplemental material for Immunohistochemical expression of TLR1, TLR2, TLR4, and TLR9 in the different types of lesions associated with bovine paratuberculosis
Supplemental material, sj-pdf-1-vet-10.1177_03009858241302850 for Immunohistochemical expression of TLR1, TLR2, TLR4, and TLR9 in the different types of lesions associated with bovine paratuberculosis by David Zapico, José Espinosa, Miguel Criado, Daniel Gutiérrez, María del Carmen Ferreras, Julio Benavides, Valentín Pérez and Miguel Fernández in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge the excellent technical assistance of Marta Silva Samos.
Author Contributions
Conceptualization, DZ and MF; methodology, DZ, DG, and MF; results validation, DZ, DG, and MF; Statistical analysis, JE; investigation, DZ and MF; resources, JB and VP; data curation, DZ, MC, and MF; writing—original draft preparation, DZ; writing—review and editing, VP, JE, and MF; visualization, MCF, and JE; supervision, MF All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DZ is a recipient of a predoctoral contract of the University of León. This study has been funded by own resources of the SANPATRUM research group of the University of León.
Data Availability
All data sets generated for this study are included in the manuscript and/or the supplementary files.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
