Abstract

Clinical History, Laboratory Results, and Gross Findings
A 14-year-old, neutered female miniature dachshund was presented to an animal hospital for a health checkup. The owner and clinician noticed no signs of urinary diseases, such as stranguria and hematuria. Elective abdominal ultrasonography revealed a nodular lesion (5 × 10 × 10 mm in diameter) in the bladder mucosa. A urine sample was collected by aspiration using a urethral catheter and centrifuged to collect the sediment for cytology. Cytological examination of the urinary sediment revealed clusters of epithelial cells with minimal nuclear atypia. A BRAF mutation resulting in an V595E (BRAFV595E) amino acid substitution was detected in the urinary sample using digital PCR (9.07% mutated gene in 30,707 total copies). 6 Based on these results, urothelial carcinoma of the bladder was clinically suspected. Total cystectomy was performed for treatment and histopathological examination. On gross examination, a white nodule measuring 5–10 mm in diameter, corresponding to the lesion depicted on ultrasonography, was found on the mucosa of the left wall of the bladder (Fig. 1). In addition, white to tan nodules measuring 2–3 mm in diameter were found on the bladder mucosa, and dark-red hemorrhagic lesions were found on the bladder and urethral mucosas.
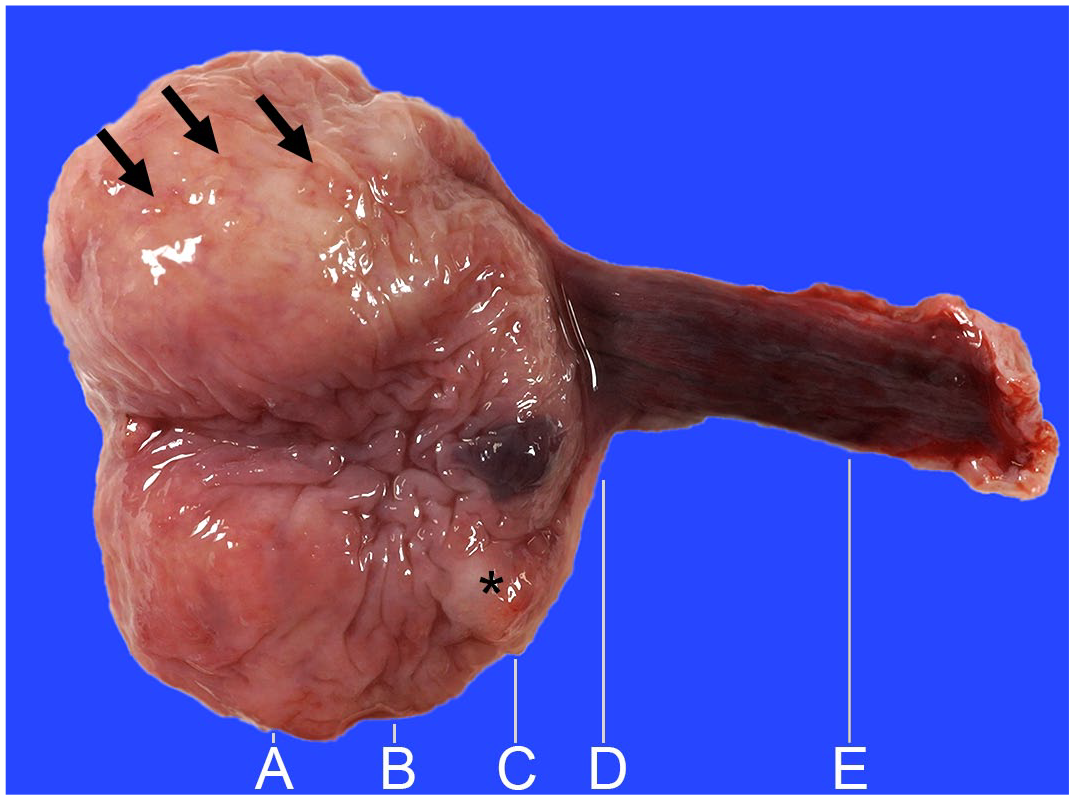
Urinary bladder and urethra, dog. A white nodule is on the mucosa of the left bladder wall (asterisk), which was apparent on ultrasonography. Dark red hemorrhagic lesions are on the mucosa of the bladder and urethra. White to tan nodules are on the bladder mucosa (arrows). Five transverse sections (A–E) were prepared for histological examination.
Microscopic Findings
The bladder was fixed in 10% neutral-buffered formalin, processed routinely for histology, and stained with hematoxylin and eosin. Five transverse sections (A–E) were embedded in two paraffin blocks (Figs. 1, 2a). Block 1 included sections A and B; block 2 included sections C, D, and E. Histologically, the mucosa of the bladder neck (section D) and urethra (section E) was dysplastic with irregularly arranged atypical epithelial cells (Fig. 2b). Mucosal dysplasia was absent in sections A, B, and C. Atypical epithelial cells had slightly enlarged nuclei with one or two distinct nucleoli (Fig. 2c). Mitotic figures in 2.37 mm2 were 3 and 0 in the dysplastic mucosa and non-dysplastic mucosa, respectively (10 FN22/40X fields). Lymphoid follicles were observed in the lamina propria of the bladder, leading to a diagnosis of follicular cystitis (Fig. 2a). Hemorrhagic foci were observed in the lamina propria and the submucosa. The nodule measuring 5–10 mm in diameter in section C was comprised of multiple lymphoid follicles and fibrous tissue. No evidence of neoplasia was observed in any of the sections examined.
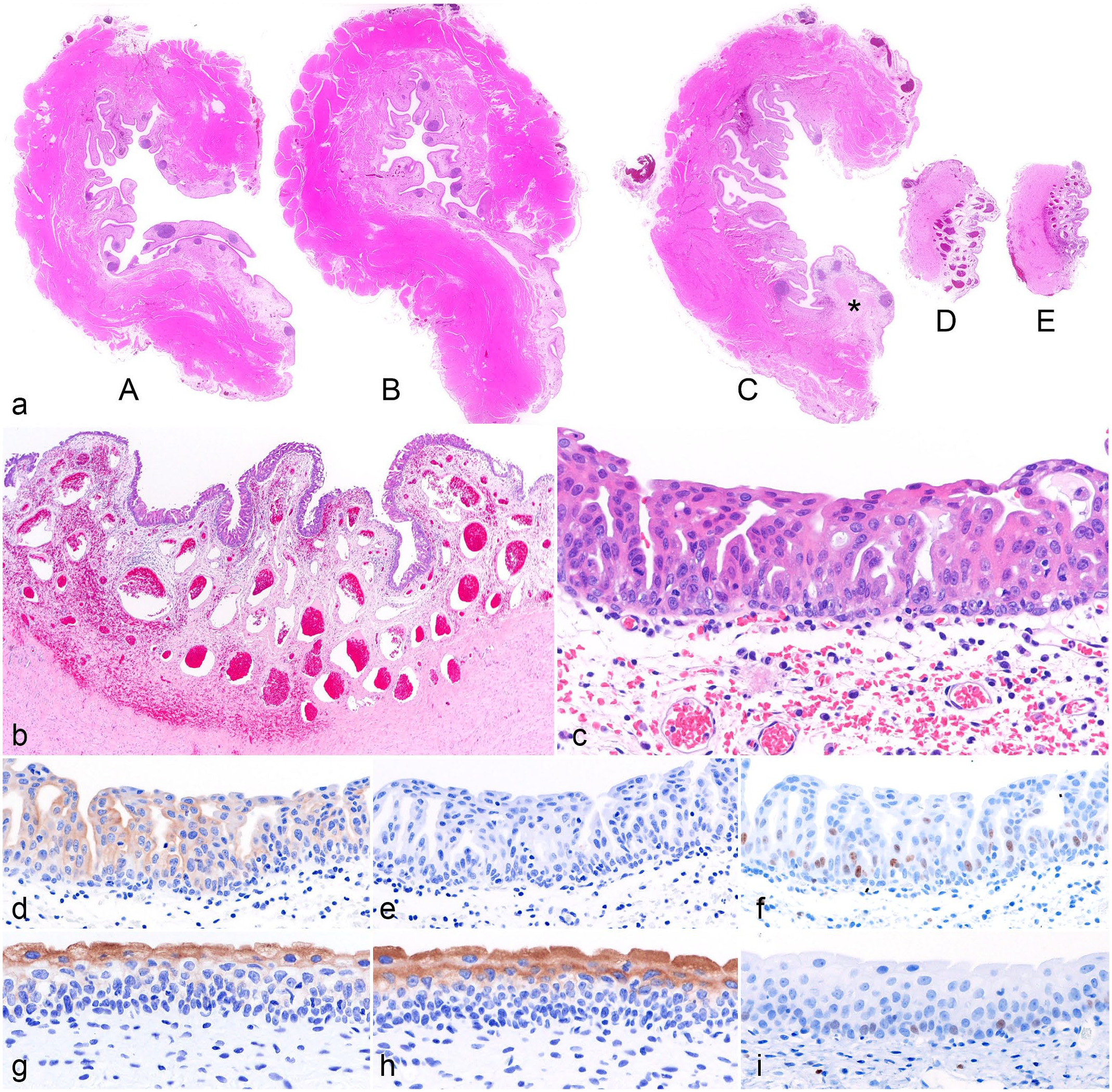
Urinary bladder, dog. (a) Lymphoid follicles in the lamina propria of sections A, B, and C. A nodule in section C comprises lymphoid follicles and fibrous tissue (asterisk). Hematoxylin and eosin (HE). (b) Section E. Polypoid hyperplasia of mucosal epithelium, and congestion and hemorrhage in lamina propria and submucosa. HE. (c) Higher magnification of section E. The dysplastic mucosa has irregularly arranged epithelial cells with enlarged nuclei. HE. (d-f) Mucosa, section E, (d) cytokeratin 7, (e) uroplakin 3, and (f) Ki-67 immunohistochemistry (IHC). The dysplastic epithelium is diffusely immunolabeled for (d) cytokeratin 7 but not for (e) uroplakin 3. (f) Dysplastic epithelial cells have nuclear Ki-67 immunolabeling. (g-i) Mucosa, section B, (g) cytokeratin 7, (h) uroplakin 3, and (i) Ki-67 IHC. Superficial cells and the upper layer of intermediate cells in the mucosa are immunolabeled for (g) cytokeratin 7 and (h) uroplakin 3. (i) Basal cells of the mucosa are immunolabeled for Ki-67 in the nucleus.
Further Investigations and Diagnosis
Formalin-fixed paraffin-embedded tissues of block 1 (without mucosal dysplasia) and block 2 (with mucosal dysplasia) were subjected to BRAF mutation analysis by digital PCR. 6 The BRAFV595E mutation was detected in the DNA extracted from block 2 (0.49% mutated gene in 81,138 total copies) but not from block 1 (0% mutated gene in 68,430 total copies). Immunohistochemistry for cytokeratin 7 (CK7), uroplakin 3, and Ki-67 was performed on both blocks of the present case and a block from a different canine case of invasive urothelial carcinoma as a control. As previously reported, invasive urothelial carcinoma cells had diffuse, cytoplasmic immunolabeling for CK7 and uroplakin 3. 8 In the present case, the dysplastic mucosal epithelium on sections D and E were diffusely immunolabeled for CK7 in the cytoplasm, but not for uroplakin 3 (Fig. 2d, e). The mucosal epithelium of sections A, B, and C was strongly immunolabeled for CK7 and uroplakin 3 in the cytoplasm of superficial cells and upper layer intermediate cells (Fig. 1g, h), which is the normal labeling pattern. 8 Nuclear Ki-67 immunolabelling was observed in the dysplastic epithelium in sections D and E, while the basal cells were immunolabeled in sections A, B, and C (Fig. 2f, i).
Discussion
In the present case, urothelial carcinoma was clinically suspected based on the detection of BRAFV595E mutation in a urinary sample. However, there were no invasive tumors on gross and histopathological examinations, and mucosal dysplasia was found in the bladder neck and urethra. Digital PCR using DNA samples extracted from these lesions revealed the BRAFV595E mutation, suggesting that dysplastic change in the mucosa is associated with an activating BRAF mutation frequently found in invasive urothelial carcinomas of dogs. 1 The nodular lesion depicted on ultrasonography was found to be associated with follicular cystitis. Follicular cystitis is considered a localized immune response to infection, cancer, and other disorders. 3 Previous episodes of urolithiasis or bacterial infection could have predisposed to follicular cystitis and dysplastic changes of the urothelium, although urinary stone or bacterial infection was not found at the time of surgery.
In human pathology, flat urothelial lesions with atypia are categorized into reactive urothelial atypia, urothelial dysplasia, and urothelial carcinoma in situ. 5 Reactive urothelial atypia is often associated with inflammation of various causes. In contrast to reactive urothelial atypia, epithelial cells in carcinoma in situ are disorganized and lose their polarity. Also, nuclear atypia, such as nuclear enlargement, irregular nuclear membrane, and coarse chromatin, is more evident in carcinoma in situ. 9 Cytological features of urothelial dysplasia fall between reactive atypia and carcinoma in situ. However, its morphological distinction has poor reproducibility between observers, and differentiation from carcinoma in situ can be challenging.
In veterinary diagnostic practice, the distinction between urothelial dysplasia and carcinoma in situ is not always clear. In both invasive and noninvasive canine urothelial carcinomas, neoplastic cells are commonly positive for CK7 and uroplakin 3. 8 Immunohistochemistry for these molecules has not been described in urothelial dysplasia. In the present case, dysplastic urothelial cells were diffusely immunolabeled for CK7, but negative for uroplakin 3. In addition, an increased mitotic count, compared to non-dysplastic mucosa, and Ki-67-positivity in the suprabasal layer were observed in dysplastic mucosa. These results suggest aberrant proliferation of suprabasal cells and lack of terminal differentiation of the surface urothelium. Molecular changes such as BRAF mutations have not been demonstrated in flat urothelial lesions with atypia in dogs. In the present case, the BRAFV595E mutation was detected in a formalin-fixed paraffin-embedded tissue sample of urothelial dysplasia, suggesting that BRAF mutation could be associated with urothelial dysplasia in dogs, which is potentially a precancerous lesion that may progress to urothelial carcinoma. The dog received no additional treatment after surgery; however, no evidence of neoplastic lesions was found on abdominal ultrasonography and radiography performed 4 months after surgery. Accumulation of information on BRAF mutation status and clinical outcomes in dogs with flat urothelial lesions is necessary to evaluate the involvement of BRAF mutation in malignant transformation.
Pathological similarities between canine and human invasive urothelial carcinoma suggest that bladder cancer in dogs is a good natural model for cancer biology and intervention research. 4 Genomic analyses of human bladder carcinoma revealed two distinct molecular pathways of tumor progression in noninvasive papillary carcinoma and invasive carcinoma, respectively. 7 Noninvasive papillary carcinoma, which accounts for approximately 75% of human cases, is often associated with FGFR3 tyrosine kinase receptor gene amplification and activating mutations in the RAS and PIK3CA genes, resulting in activation of the phosphoinositide-3 kinase/Akt/mTOR pathway. Invasive carcinoma develops by progression from carcinoma in situ, and mutations in the TP53 and RB genes are associated with both lesions. Carcinoma in situ progresses to invasive carcinoma within 5 years in approximately 50% of untreated cases. 9 In almost 50% of human patients with invasive carcinoma, the tumor has already invaded the bladder wall at the time of presentation. Once the tumor invades the muscle layer, the 5-year mortality rate is 30%. 7
In dogs, canine urothelial carcinomas are mostly of the invasive type, and flat carcinomas in situ account for 3% of cases. 10 Invasive urothelial carcinoma is invariably fatal with current treatment with chemotherapy and cyclooxygenase inhibitors. 4 The BRAFV595E mutation, an activating mutation of BRAF leading to constitutive activation of the mitogen-activated protein kinase (MAPK) pathway, has been detected in 87.0% of canine invasive urothelial carcinoma cases. 1 Detection of BRAF mutations at an early stage of urothelial tumorigenesis, before the development of an invasive tumor, using urinary samples may provide better treatment outcomes in canine patients. However, although not reported in dogs, false-positive BRAFv600E mutations, corresponding to canine BRAFV595E mutation, in cytological samples by highly sensitive BRAF analysis assay have been reported in human cases without any evidence of cancer on histopathology. 2 Detection of mutated genes per se does not confirm a diagnosis, and results of molecular examinations should be interpreted together with morphological assessments.
The present case report shows that BRAF mutations can be detected in dysplastic lesions of the canine bladder, which may be a preneoplastic change of urothelial carcinoma. Further studies with larger numbers of cases are necessary to confirm the prognostic significance of BRAF mutations in dogs with flat urothelial lesions with atypia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
