Abstract
The European hedgehog (Erinaceus europaeus) is a common wildlife species in European countries. Populations are declining due to anthropogenic factors and natural diseases. Verminous pneumonia has been observed as a frequent infectious disease in hedgehogs submitted for diagnostic postmortem examination. This prompted the present in-depth investigation on the lungs of 27 necropsied hedgehogs with confirmed lungworm infections, with or without antiparasitic treatment prior to death. The histological and/or parasitic (fecal samples) examination identified Capillaria aerophila infection in most animals (82%). The parasites were found free in the airway lumen and/or within the airway epithelium, from the larynx to bronchioles. Embedded worms and eggs were associated with epithelial hyperplasia or metaplasia, and long-term inflammation. More than half of the animals (59%) carried Crenosoma striatum, and 41% had a coinfection. C striatum adults were predominantly found free in the lumen of bronchi and bronchioles, and larvae were occasionally seen in granulomas in the pulmonary interstitium, the liver, and the intestine. Independent of the parasite species, a lymphoplasmacytic peribronchitis and, less frequently, interstitial infiltration of eosinophils, neutrophils, and macrophages as well as pneumocyte type II hyperplasia was seen. Interestingly, the extent of pneumonia was not correlated with age, respiratory clinical signs, antiparasitic treatment, or single or coinfection. Verminous pneumonia appeared to be the cause of death in over 25% of the animals, indicating that these parasites not only coexist with hedgehogs but can also be a primary pathogen in this species.
Keywords
The brown-breasted hedgehog (Erinaceus europaeus; Linneaus 1758), a glacial period relict, inhabits northern Europe, mainly in structured grassland and forest with sparse tree cover often in suburban, agricultural, or riparian areas. The species is an omnivorous but primarily insectivorous, nocturnal, hibernating, solitary, and nonterritorial mammal with variable, but generally small, habitats of 0.5 to around 100 hectares. The lifespan is up to 16 years in the wild and 10 years in captivity; the mating season is from April to September, with 2 litters of 3 to 8 offspring per season.7,33
Wild hedgehog populations are declining dramatically at least in some regions. Recent monitoring data show an alarming population loss in European countries, such as the UK and Switzerland.21,37,42 This decline is believed to be driven mainly by negative effects on their natural territories, such as habitat loss and fragmentation, increased urban densification, road traffic, reduction of insect biomass and pesticide use, as well as parasitoses and other diseases. 37
Hedgehogs are a wildlife species of relatively broad public interest. For decades, private and public rehabilitation and release efforts have been active in several European countries.11,14,17,19 Indeed, hedgehogs are often the wildlife most commonly presented in veterinary practices.1,23
Being a wild animal, the hedgehog is frequently infested with parasites. These are either ectoparasites or endoparasites mainly located in the gastrointestinal or respiratory tract. The helminths Crenosoma striatum (Zeder 1800) and Capillaria aerophila (syn. Eucoleus aerophilus) are described as the hedgehog’s pulmonary endoparasites. For the ease of reading, C aerophila is used as species name throughout. Reported prevalences of these parasites may reach more than 40%.10,16,26,29 Infection can be associated with severe clinical manifestations or death.15,29,43 The hedgehog is known as the only final host of C striatum, while C aerophila also parasitizes in cats, dogs, foxes, martens, and badgers. The life cycle of both parasites includes earthworms or snails, which the hedgehog ingests. These intermediate hosts are mandatory for C striatum and facultative for C aerophila.2,15,39 C striatum are ovo-viviparous. Adult females parasitize the hedgehog airways (trachea and bronchi) and shed larvae within a thin egg capsule. These are coughed up, swallowed, and larvae are excreted in the feces. The larvae are taken up by the intermediate hosts (eg, snails) in which they develop into the infectious third larvae. 15 After ingestion of the infected intermediate host, the larval migration starts. The larval migration pattern of C striatum in the hedgehog is not entirely clear. Experimental studies in dogs infected with Crenosoma vulpis have shown that the larvae penetrate the intestinal wall and reach the liver via the portal circulation, then migrate within the liver parenchyma to access the hepatic veins, pass the heart, and then reach the lung parenchyma via the pulmonary circulation. 36 However, a similar experimental study in hedgehogs did not find evidence of migration in liver or other organs (ie, spleen, mesenteric lymph nodes). 15 In the lungs, the parasites mature with the final molt; C vulpis reaches this stage around 8 days after infection. 36
For C aerophila, in contrast, the first larval stage is infectious. The hedgehog ingests eggs containing larvae, either directly or with the intermediate host.6,31,39 The larvae hatch in the intestines and reach the lung. Larvae then migrate in the lungs, where they molt, mature, and populate the airways (trachea, bronchi, and bronchioles). 6 The literature is not clear as to whether the larvae travel to the lungs via the bloodstream or, less likely, the lymphatics.6,38,39 Adult C aerophila are typically located in the airway epithelium 28 and shed nonembryonated eggs into the airway lumen or epithelium.2,6,38
Due to the high prevalence of verminous pneumonia, hedgehogs in rehabilitation centers are frequently medicated with various antiparasitic drugs, 14 with treatment regimens based on years of experience of hedgehog caretakers rather than scientific data on the treatment success, since hedgehogs are generally released into the wild relatively shortly after the treatment. However, both rehabilitators and veterinarians frequently express their frustration about the limited success of their often substantial efforts to rehabilitate injured or diseased hedgehogs (personal observation), an observation that we shared through the collaboration with wildlife rescue centers and private caretakers who submitted wild hedgehogs for diagnostic postmortem examinations. Indeed, we found lungworm infection in the hedgehogs that underwent a pathological examination in recent years, regardless of whether they had received antiparasitic treatment or not. Also, the release rate of hedgehogs at the wildlife station in Landshut, Switzerland is lower than that of other species, and the most common reason for the euthanasia of adult hedgehogs has been therapy-resistant respiratory disease (personal communication).
Given the species’ population decline, the obviously high prevalence of clinical respiratory disease in combination with the often unsuccessful rescue attempts, and the limited literature on the pathological processes associated with lungworm infections in the hedgehog, 12 we undertook an in-depth investigation on verminous pneumonia in European hedgehogs, with the aim to characterize the associated pathological processes and collect any morphological evidence of the potential effect of routine antiparasitic treatment, as a basis for future systematic clinical approaches.
Materials and Methods
Animals and Tissue Sampling
The study was performed on 27 juvenile and adult European hedgehogs that had been subjected to a diagnostic postmortem examination between February 2020 and November 2021 either at the wildlife station in Landshut, Switzerland (Stiftung Wildstation Landshut), a foundation that treats a variety of injured, diseased, or orphaned wild animal species with the aim to reintroduce the animals back into nature (n = 17), or at the Institute of Veterinary Pathology, Vetsuisse Faculty, University of Zurich (n = 10). Animals had been submitted to the latter by private hedgehog caretakers or care centers in Switzerland. During this time period, a total of 36 animals had been examined. These all exhibited histological changes consistent with verminous pneumonia; 12 however, 6 were excluded from the study due to severe postmortem autolytic changes, and an additional 3 were excluded as neither the histological nor the parasitological examination could identify lungworms.
The study animals had died or been euthanized for various reasons. For all animals, anamnestic and clinical data were provided, and in all cases, verminous pneumonia with intralesional parasites was diagnosed. Animals were grouped based on whether or not they had received antiparasitic treatment prior to death, comprising 18 untreated and 9 treated hedgehogs (Supplemental Table S1). Treatment protocols showed a wide variation in the choice of drug, number of applications, and time points prior to the animal’s death (Supplemental Table S2). The most common antiparasitic preparation included levamisole, followed by fenbendazole, imidacloprid, and moxidectin. Supportive medications contained mainly bromhexine, as well as meloxicam, dexamethasone, and/or antibiotics.
Two hoglets that had been euthanized at the age of 7 days due to persistent anorexia were also examined. Neither exhibited any pathological changes in the lungs and therefore served as controls for the histological, immunohistochemical, and ultrastructural examinations of the diseased animals.
All hedgehogs underwent a full postmortem examination within a maximum of 48 hours after death. Samples from all major organs and tissues were collected according to routine diagnostic protocols. Two samples were obtained from the lungs; one from the cranial lobes, including trachea or large bronchi and the other from the caudal lobes. In 17 cases, the head was also collected. Tissue samples were fixed in 10% neutral-buffered formalin.
Histology and Immunohistochemistry
After formalin fixation for approximately 48 hours, tissues were trimmed and routinely embedded in paraffin. Heads were sawn (cross-sections) using a diamond saw (Exakt 300; Exakt Advanced Technologies GmbH, Norderstedt, Germany) and a slice comprising the conchae (mid-nose) was prepared and gently decalcified in RDF (Biosystems Switzerland AG, Muttenz, Switzerland) for 3 days at room temperature on a shaker, then was paraffin embedded. Sections (2–4 µm thick) were prepared and routinely stained with hematoxylin and eosin (HE). In selected cases, consecutive sections from the lungs were subjected to a Giemsa stain to visualize mast cells (n = 6) or a Van Gieson stain to highlight collagen deposition (n = 6).
Based on the overall extent of the verminous pneumonia in the sections (area covered by inflammatory processes and amount of parasites observed in the airways) a simple semiquantitative grading was established, ranging from mild (1) to moderate (2) to severe (3) (Supplemental Table S1). To specify, the grading considered representative sections of the cranial, caudal, and accessory lung lobes at low magnification, where the extent of consolidation (ie, nonaerated, inflamed areas) and the amount of discernible parasites were taken into account. Roughly, tissues with a maximum one quarter of the area affected was graded as mild, up to half as moderate, and anything above as severe.
Extrapulmonary changes relevant for the animal’s death as well as histological changes that were not relevant for the course of the disease are listed in Supplemental Table S1. Changes relevant for the parasitic infection/migration route are further commented on in the result section.
Immunohistochemistry was applied to the lungs of the hoglets and selected diseased animals. This served to identify leukocytes and parenchymal cells in the lungs (Table 1). The horseradish peroxidase method was applied using a Dako autostainer (Agilent/Dako, Glostrup, Denmark). To establish suitable staining protocols for the hedgehog, a range of antibodies and protocols used in other species were tested on sections of spleen, lymph node, and lung of a hoglet for their suitability to highlight T cells, B cells, macrophages, epithelial cells, and type II pneumocytes. Hereafter, the lungs of 2 adult hedgehogs with moderate to severe verminous pneumonia (cases 5 and 22) were immunolabeled for the expression of CD3 (T cells), CD20 (B cells), cytokeratin (pan-cytokeratin, epithelial cells), IBA1 (macrophages), and surfactant protein (SP)-C (type II pneumocytes) using cross-reactive antibodies (Table 1). Briefly, after deparaffinization, antigen retrieval was performed for all antigens, by incubation of the slides with citrate buffer (pH 6) at 98°C for 10 minutes or ethylenediaminetetraacetic acid (EDTA) buffer (pH 9) at 98°C for 20 minutes. Endogenous peroxidase was blocked by incubation with hydrogen peroxide solution for 10 minutes. Slides were incubated with a dilution buffer (Agilent/Dako) for 30 minutes at room temperature followed by the primary antibodies and the matching detection kits (Table 1). Sections were washed with phosphate buffered saline (pH 8) between each incubation step. Finally, sections were counterstained with hematoxylin for 40 seconds and coverslipped. Spleen and lymph node sections from the same hedgehogs served as positive controls for lymphocyte and macrophage identification, and the lungs for cytokeratin, IBA1 and SP-C expression. Consecutive sections incubated with an isotype-matched irrelevant antibody or without the primary antibody served as negative controls.
Antibodies and immunohistochemical methods. Immunohistochemical protocols established in other species (eg, dog, cat, hamster, sheep, goat, and horse) in our laboratory and applied to tissue sections from the European hedgehog. For all antibodies, the matching Envision (Dako, Glostrup, Denmark) kit was employed. The spleen and lung of a 7-day-old hoglet served as positive control tissues.

Abbreviations: CD, cluster of differentiation; citrate, citrate buffer (pH 6) pretreatment; EDTA, ethylenediaminetetraacetic acid (pH 9) pretreatment; h, human; IBA1, ionized calcium-binding adaptor molecule 1; mAb, monoclonal antibody; ON, overnight; pAb, polyclonal antibody; PCK, pan-cytokeratin; RT, room temperature; SP-C, surfactant protein C.
Agilent Technologies, Inc.
Transmission Electron Microscopy
From 2 adult hedgehogs with verminous pneumonia (cases 1 and 5) and one of the hoglets without histological changes in the lungs, lung samples (1 mm3) were collected within 5 to 10 minutes after death and fixed for 24 hours in 2.5% glutaraldehyde, buffered in 0.2 M cacodylic acid buffer, pH 7.3, then trimmed, and routinely embedded in epoxy resin. Toluidine blue-stained semithin sections (1.5 μm) were prepared to select areas of interest for the preparation of ultrathin sections (75 nm) that were contrasted with lead citrate and uranyl acetate and viewed with a Philips CM10, operating with a Gatan Orius Sc1000 digital camera (Gatan Microscopical Suite, Digital Micrograph, Gatan Inc., Pleasanton, California, United States).
Parasitologic Analysis
Histological sections: Since C striatum and C aerophila are the only parasites described in the lungs of the hedgehog,10,16,26,29,34,37 we generally referred to these 2 species only throughout the manuscript. C aerophila infection was confirmed in histological sections based on the characteristic morphology of this species’ eggs and the presence of adult worms embedded in the bronchial epithelium.2,28,39 When adult worms with larvae in their abdominal cavity were found free in the lumen of the airways, these were referred to as C striatum, as this species, unlike C aerophila, has an ovo-viviparous life cycle.1,15 Single larvae found in the lung parenchyma were also referred to as C striatum since previous histologic studies stated that this parasite may be found in lower parts of the respiratory tract, whereas C aerophila is predominantly located within the bronchi.22,28,36 Some individual worms free in the airway lumen and larvae in the bronchi could not be definitely allocated to 1 worm species, as relevant criteria were not fulfilled, these remained unclassified. However, in each case, some parasites could be assigned to a species, thus allowing a diagnosis of C striatum, C aerophila, or dual infection.
Fecal samples: Feces were collected from 14 animals, either intravitam at the wildlife station in Landshut or during the postmortem examination (Supplemental Table S1). These were examined according to standard sedimentation and flotation protocols to identify C aerophila. When enough fecal material was available, a standard Baermann funnel protocol was also applied to identify C striatum.
Statistical Analysis
To reveal any potential association of the pneumonia grade with age and sex of the animals, and whether they had received treatment, a statistical analysis was performed using a commercially available software package (R version 4.1.0 [2021-05-18]—“Camp Pontanezen”; GGPLOT2, VCD). The raw data were used without modifications, and the analysis made creating contingency tables and checking the independence of the variables using the chi-square test. P-values higher than .05 rejected the “null hypothesis” (the data have an association with the pneumonia grades) with a 95% confidence level.
Results
Study Population
Among the 27 animals included into the study, 12 were male and 9 were female; the sex had not been documented for the remaining 6. Animals weighed from less than 500 g to more than 1000 g, thereby representing both juveniles and adults. Twelve hedgehogs had shown clinical respiratory disease, others exhibited traumatic lesions, gastrointestinal signs, severe ectoparasite infestation, persistent anorexia, or poor general condition (Supplemental Table S1). In addition, the lungs of 2, 7-day-old E europaeus hoglets were examined. These did not exhibit any pathological changes and served to determine the morphological features of the hedgehog lung.
Morphological Features of the Unaltered Hedgehog Lung
The gross and histological examination of the lungs revealed an architecture comparable with other mammals as described in Richard Parent’s “Comparative Biology of the Normal Lung.” 30 Grossly, both the left and right lungs were divided into a cranial and caudal lobe, with an accessory lobe branching from the right bronchus (Figs. 1a, b). The histological features were determined in the hoglet lungs without evidence of respiratory disease and in unaffected lung lobes of adult hedgehogs with mild verminous pneumonia. Bronchi and bronchioles were covered by a ciliated pseudostratified respiratory epithelium with a thin underlying lamina propria containing a few leukocytes, surrounded by the tunica muscularis and, in the case of the bronchi, a tunica adventitia with a few glands and embedded cartilage (Supplemental Figs. S1a–d). Besides the ciliated epithelial cells, goblet cells were observed; these were present in moderate numbers and immature in the hoglet (Supplemental Fig. S1e) and found in large numbers, and both fully differentiated and developing in the unaltered bronchi of adult hedgehogs (Supplemental Fig. S1f). Small bronchioles opened into alveoli, and the alveolar walls were lined by the cytokeratin positive pneumocytes, including the flat type I pneumocytes and the more cuboidal type II pneumocytes that showed punctate cytoplasmic SP-C expression. Interestingly, the latter generally appeared to bulge toward the alveolar interstitium rather than into the alveolar lumen. Macrophages (IBA1+) were occasionally found within alveoli (alveolar macrophages), but were otherwise present as individual cells in the interstitium. In the hoglets, alveoli were variably aerated. The described features are illustrated in Supplemental Figs. S2 (hoglet) and S3 (adult hedgehog). Macrophages (IBA1+) were also present in low numbers in the bronchial tunica adventitia, around bronchioles and around larger vessels; a few individual T cells (CD3+) and B cells (CD20+) were found in alveolar walls, likely within capillaries (Supplemental Fig. S4). Lymphoid structures were not observed.
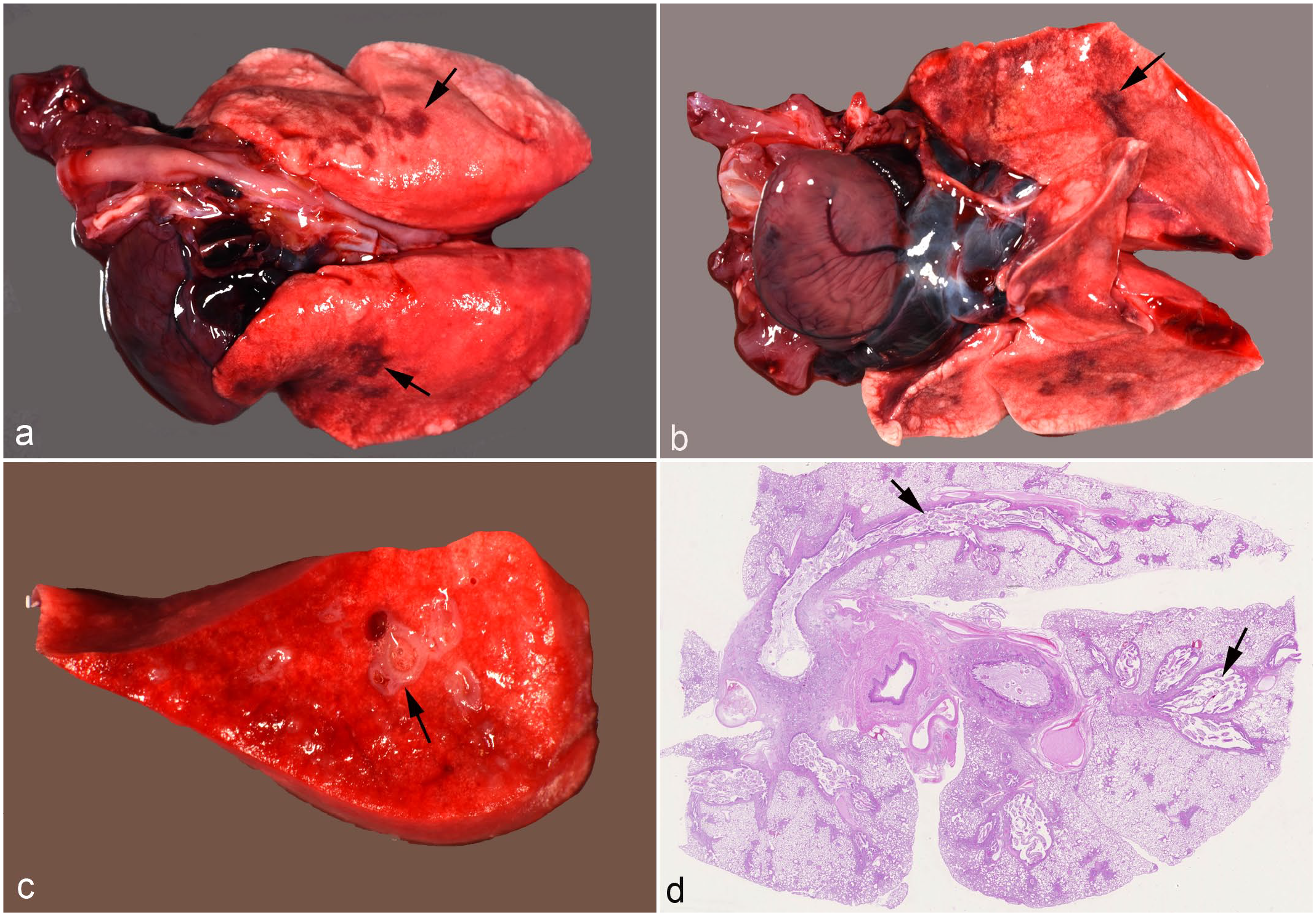
Lung, hedgehogs with verminous pneumonia.
Confirmation of Lungworm Infection
The 2 lungworm species known to occur in the European hedgehogs, C striatum and C aerophila,10,16,26,29,34,37 were identified based on the combination of histological examination and parasitological examination of the fecal samples. Coproscopy identified C aerophila in 12 of the 14 examined samples and C striatum in 3 of the 8 examined samples (Supplemental Table S1). Considering also the results of the histological examination, C aerophila infection was diagnosed in 22 of the 27 examined hedgehogs (82%), and C striatum infection was diagnosed in 16 animals (59%); the latter was the only parasite in 2 cases, whereas coinfection was confirmed in 11 cases (41%). Detailed information for individual cases is provided in Supplemental Table S1.
Lungworm Infection: Gross Changes
Gross changes of the lungs ranged from a mildly increased consistency of the lobes to slightly depressed, consolidated, ill-defined, multifocal to coalescing, and gray to red areas that were predominantly found in dorsal portions of the lung lobes (Fig. 1a, b). In some animals, bronchi(oles) were prominent and thickened at the cut surface (Fig. 1c), and the lumen of larynx, trachea, tracheal bifurcation, and main bronchi was filled with mucus admixed with a few filamentous worms.
Lungworm Infection: Histological Differentiation of the Parasites
The histological examination confirmed the presence of lung worms in the lumen (Fig. 1d) and/or epithelium (Fig. 2a–e) of the respiratory tract in all but 2 animals (cases 4 and 24; in both cases, parasites were identified in the feces), from the larynx to the alveoli, but sparing the nasal cavity. The morphological features of the parasites (see the “Materials and Methods” section: Parasitologic analysis; Supplemental Fig. S5) did not only allow their identification but also revealed that their location in the respiratory tract varied slightly. This was particularly obvious in confirmed coinfections. In 19 cases (70%), eggs and/or adult female nematodes with intrauterine eggs with the typical morphology of C aerophila (thick-capsuled brown eggs with an operculum evident at both ends; Supplemental Fig. S5a) were found both free in the lumen and embedded in the pseudostratified ciliated epithelium of the larynx, bronchi, and, less frequently, bronchioles (Fig. 2c–e). In one case (no. 18), C. aerophila were also within alveoli. In contrast, adult C striatum nematodes, identified based on the presence of a pseudocoelom with prominent digestive tract and a reproductive tract that contained larvae (Fig. S5b), were found free in the lumen of bronchi and bronchioles in 11 cases. Larvae (10–15 μm in diameter with a thin eosinophilic cuticle and a basophilic granular interior) in the pulmonary parenchyma were generally also assigned to C striatum, since the first larvae of this parasite are known to move down into the lung parenchyma, whereas C aerophila is known to remain in the upper airways.22,28,36 The larvae were found free in the lumen of alveoli (Supplemental Fig. S5b, inset) or, more often, encapsulated in well-demarcated, randomly distributed small granulomas. The latter were found in an additional 3 cases, where adults were not present in the sections, leading to a total of 14 cases with histologically identified C striatum infection.
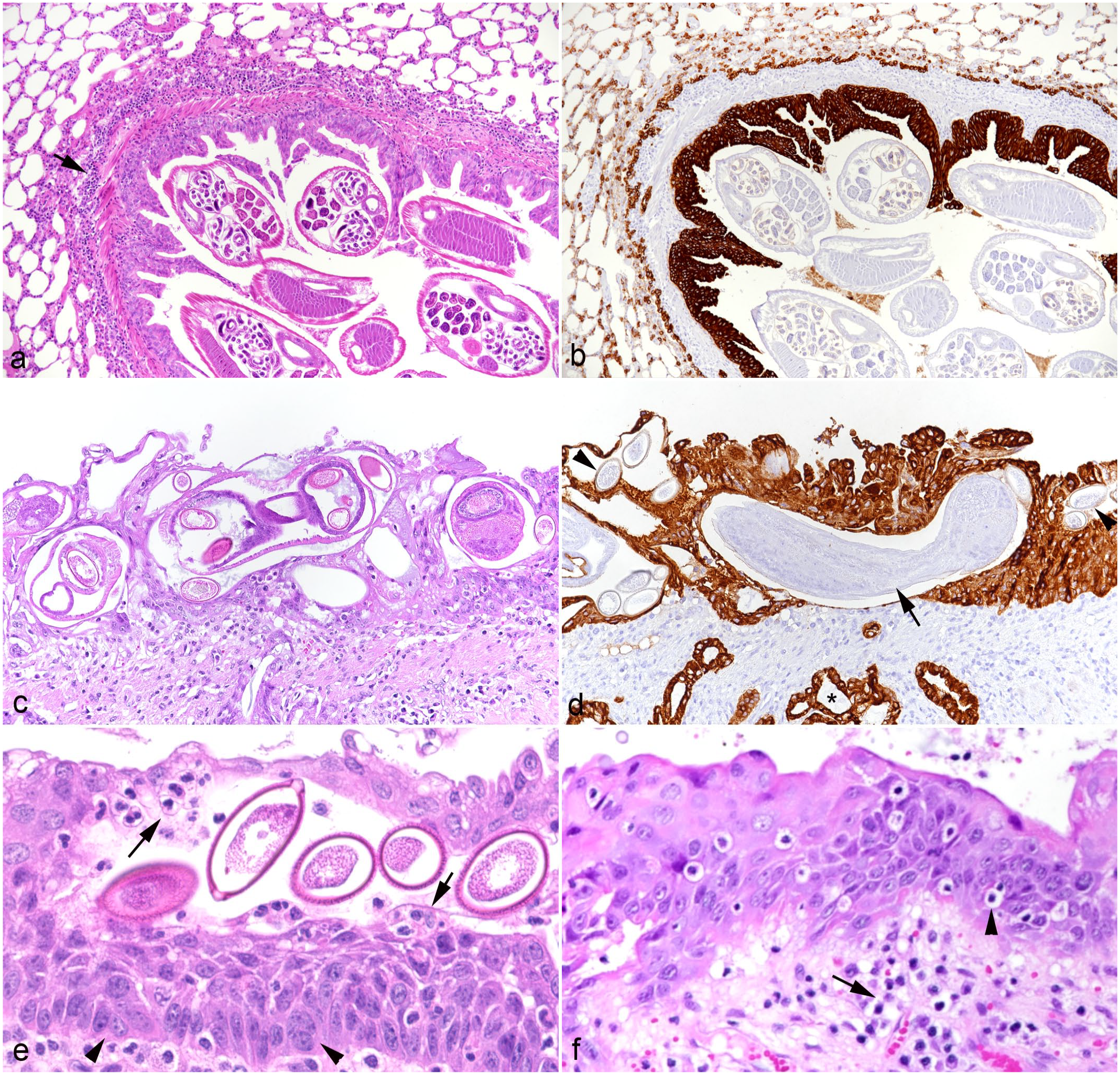
Lung, hedgehogs with verminous pneumonia. Changes in lower airways.
Morphological Features of Verminous Pneumonia in Hedgehogs
Changes in the airways
Larynx, trachea, bronchi, and bronchioli were the sites that most commonly showed changes associated with the presence of the parasites. Adult lung worms were in some cases noted in the lumen of the larynx, bronchi, and bronchioles without epithelial changes or very mild epithelial hyperplasia and peribronchial mononuclear infiltration (Fig. 2a, b). The presence of lungworm stages within the epithelium (identified as C aerophila worms, larvae, and/or eggs), however, was always accompanied by alterations in the respiratory epithelium. There were variable degrees of epithelial hyperplasia. Severe cases had more than 10 rows of cells, prominent nuclear crowding along the basement membrane, and strong mitotic activity (Fig. 2a–d). Occasionally, the hyperplastic epithelium formed finger-like intraluminal projections and showed squamous metaplasia and/or exocytosis of leukocytes (eg, neutrophils, lymphocytes) (Fig. 2e, f). In such severe cases, extensive, predominantly mononuclear infiltration of the lamina propria was seen, consistent with a long-term proliferative laryngitis, (peri)bronchitis, and (peri)bronchiolitis. Within the lung parenchyma, the peribronchial infiltrates stretched further and were also found around vessels (Fig. 3a). Infiltrates were composed of macrophages (IBA1+; Fig. 3b) with some T cells (CD3+; Fig. 3c), B cells (CD20+; Fig. 3d), mast cells (Giemsa positive; Fig. 3e), and plasma cells. Affected airways were often filled with cellular debris, proteinaceous exudate (mucus), sloughed (and degenerate) epithelial cells, and/or degenerate neutrophils (Fig. 3b). The bronchial submucosal glands were hyperplastic, and the smooth muscle bundles were hypertrophic (Fig. 3a). In rare cases, parasite larvae/eggs were not restricted to the lumen of bronchioles but were also present within adjacent alveoli; this was occasionally accompanied by evidence of bronchiolar destruction, with apparent discontinuity of the epithelium and partial replacement by sloughed epithelial cells, necrotic debris, and inflammatory infiltrates (Fig. 4a). The described focal lesions were more prevalent in cases with advanced long-term bronchiolar/peribronchiolar inflammatory infiltrates and epithelial hyperplasia.
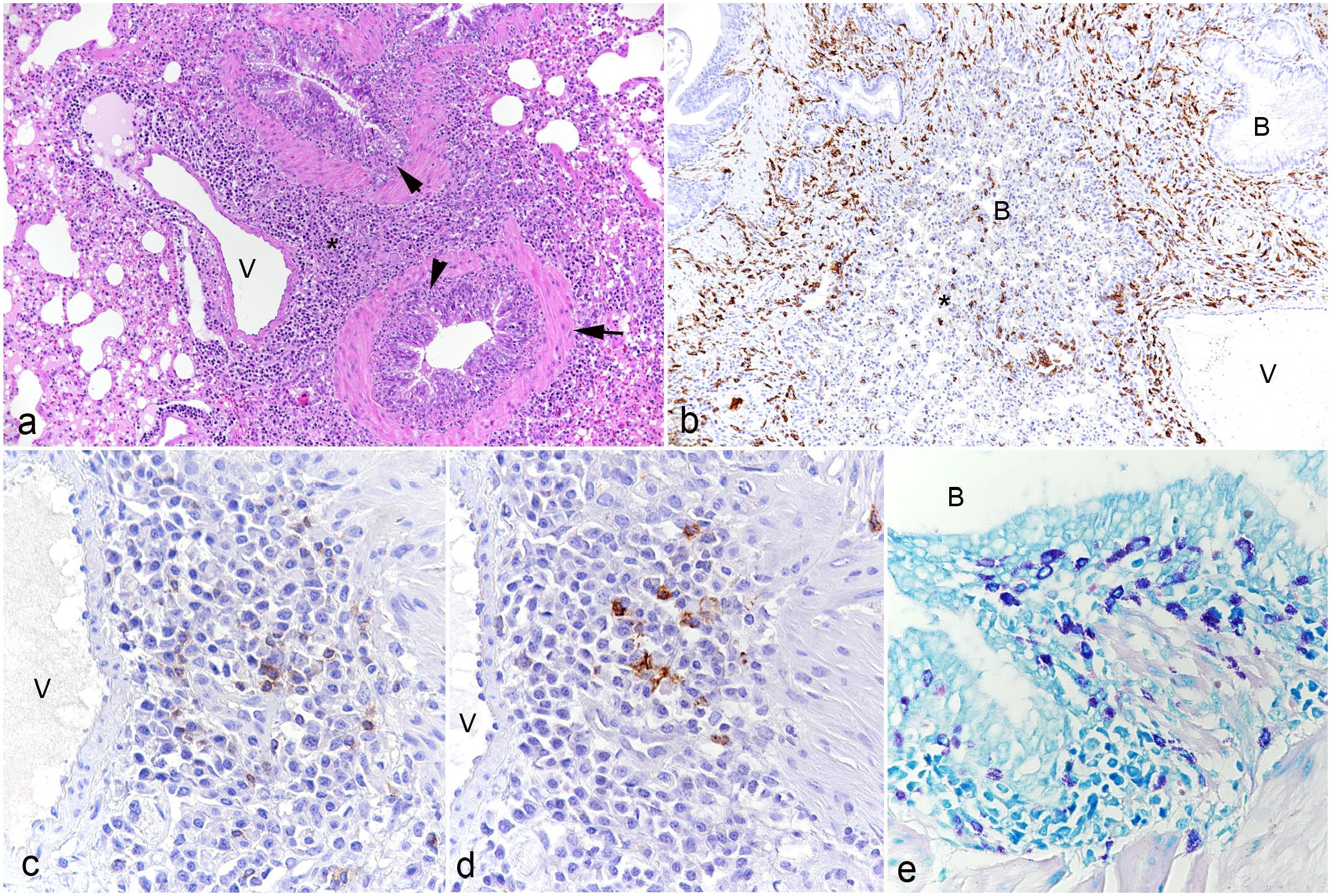
Lung, hedgehogs with verminous pneumonia. Long-term bronchiolitis and peribronchiolitis.
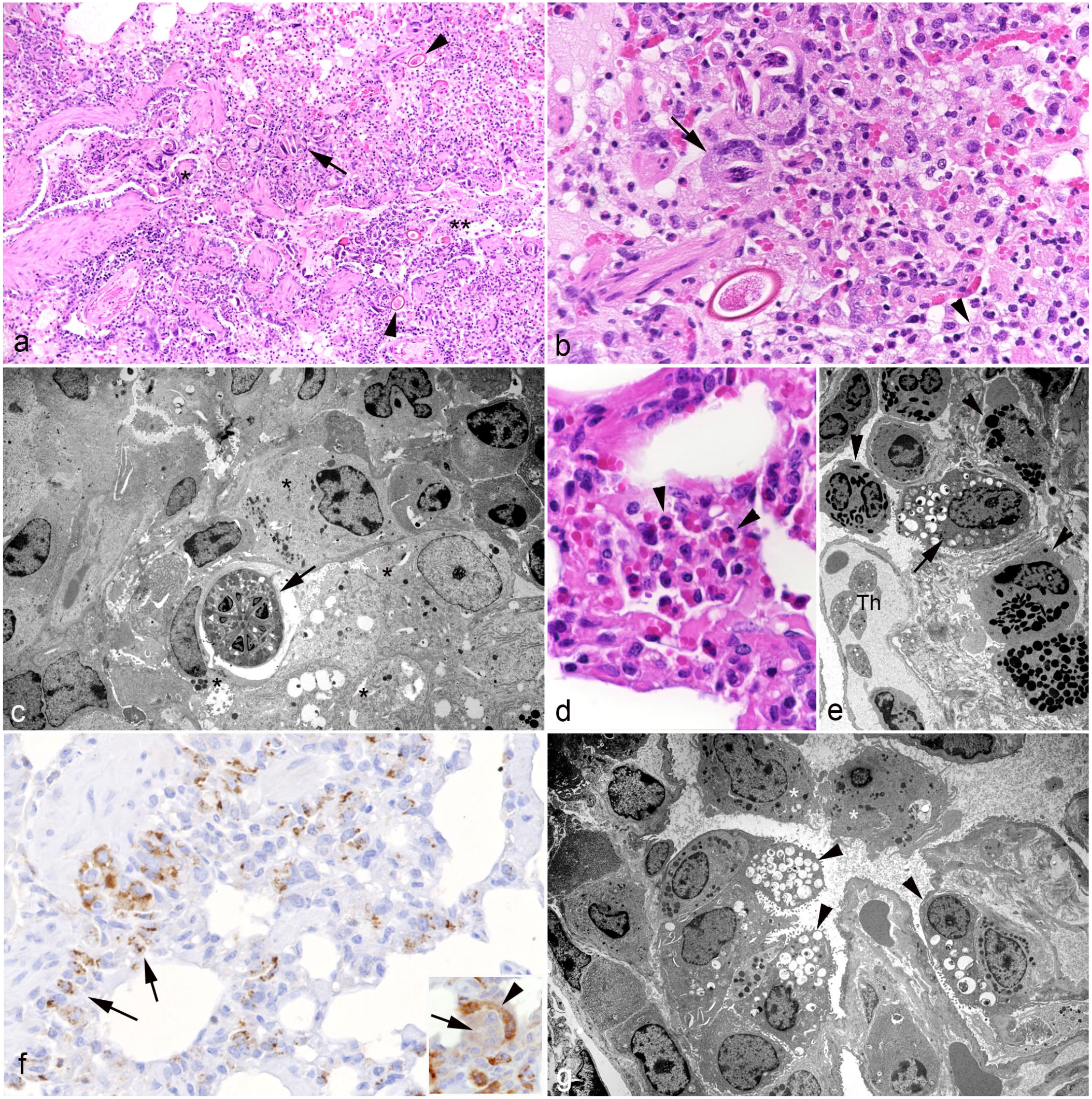
Lung, hedgehogs with verminous pneumonia. Parenchymal changes.
Involvement of the pulmonary parenchyma
In addition to the above-described focal lesions, some cases contained random foci where larvae, and rarely C aerophila eggs, were found free in alveolar lumina (Fig. 4b). In those cases, the parasitic infection was accompanied by a strong inflammatory response, characterized by degenerated and viable eosinophils, neutrophils, and often foamy macrophages (IBA1+) (Fig. 4b–e). A few foreign body-type multinucleated giant cells, which occasionally contained larval structures (Fig. 4b), were also observed. Interestingly, we recorded a distinct SP-C reaction on larval structures and, weakly, in the cytoplasm of multinucleated giant cells (Fig. 4f). Those cases also exhibited further changes in the pulmonary parenchyma, all with a variable extent. There was type II pneumocyte hyperplasia (SP-C+) at the bronchoalveolar transition and multifocally aligning the alveolar walls (Fig. 4f, g), alveolar smooth muscle hypertrophy, and lymphocytic perivascular cuffing. The interstitium exhibited mild multifocal fibrosis and infiltration by T cells (CD3+), B cells (CD20+), and plasma cells.
An additional finding that appeared to be independent of the overall extent of parasite infection and/or inflammation was granuloma formation in the pulmonary parenchyma; this was seen in 11 cases. These granulomas were randomly distributed throughout the parenchyma. They comprised macrophages (IBA1+), often with an epithelioid appearance that surrounded central larval structures (assigned to C striatum) and were arranged in an onion peel-like pattern (Fig. 5). There were also granulomas that only harbored macrophages in the center, some of which were multinucleated, and exhibited a dense fibrous capsule (Fig. 5c). The larvae again showed a strong reaction for SP-C (Fig. 5e). The ultrastructural examination did not reveal any evidence of degeneration of the embedded parasite (Fig. 5f).
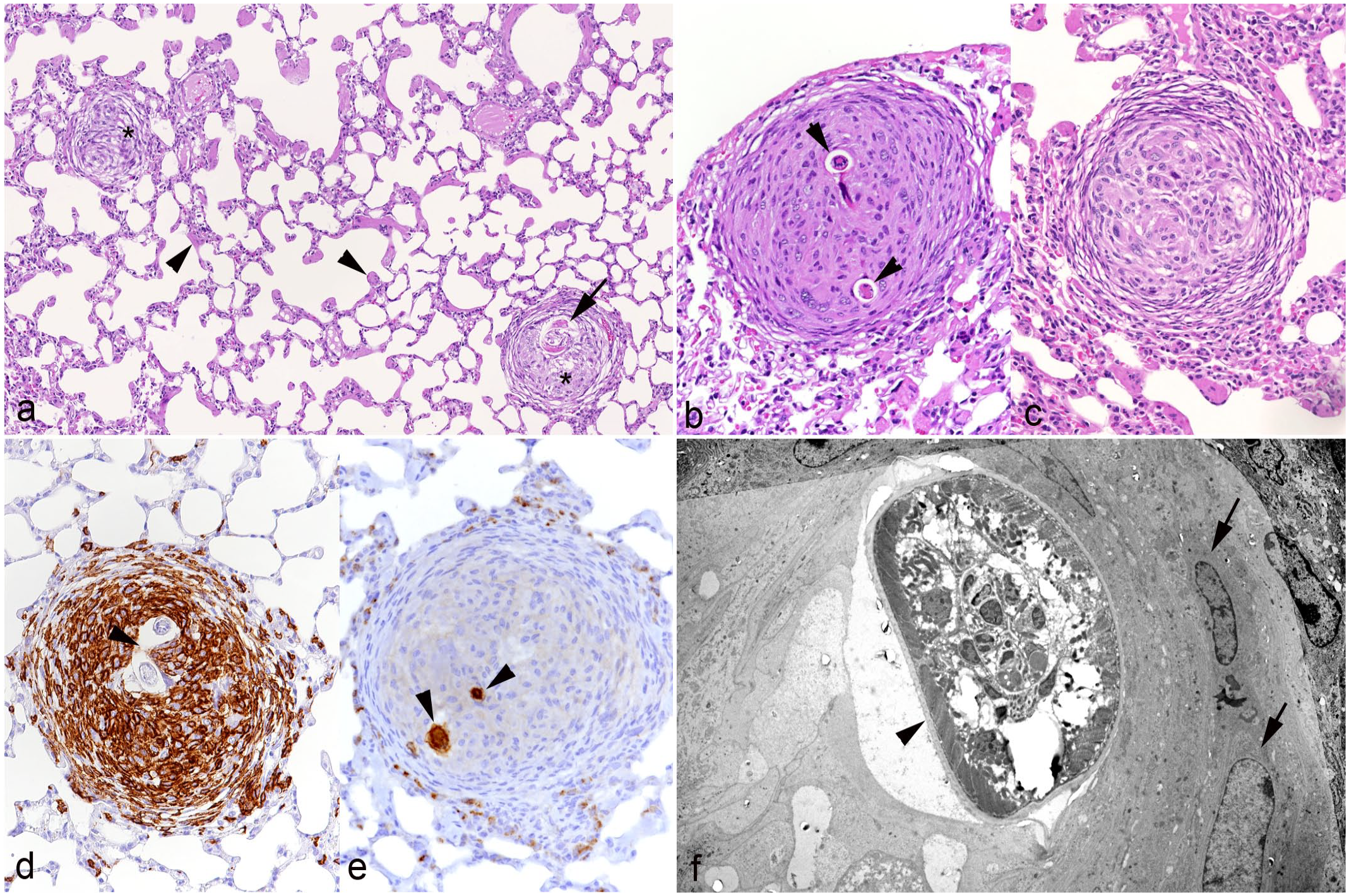
Lung, hedgehog with verminous pneumonia, parenchymal granuloma formation. Case 5.
Evidence of Parasitic Migration
In 4 individuals (cases 8–11), a low to moderate number of small, well-demarcated granulomas were randomly distributed in the hepatic parenchyma (Supplemental Table S1). Similar to the pulmonary granulomas, in one individual, they were centered around larvae and could be attributed to the migration of C striatum, as supported by a former experimental study on C vulpis in dogs. 36
Pneumonia Severity, Lungworm Prevalences, and Cause of Death
In 6 cases (5 adults and 1 of unknown weight/age), the pneumonia was graded as mild, in 13 cases (4 juveniles and 9 adults), it was considered as moderate, and in the remaining 8 cases, it was graded as severe (6 adults and 2 unknown weight/age) (Supplemental Table S1). There was no correlation between the degree of pneumonia and the sex and/or the age group of the hedgehogs (P = .5047 and P = .1153, respectively). Animals with respiratory signs showed a slightly higher average score (mean of 2.17 vs. 2.00 in animals without respiratory signs), but the difference was not significant (P = .3472).
Considering the overall clinical information and postmortem findings, we could identify an obvious cause of death unrelated to verminous pneumonia in 14 animals (52%), these were classified as traumatic injuries (33%; cases 3, 9, 11, 13–17, and 24) or disorders of the digestive tract (19%; cases 4, 6, 7, 20, and 21). In another 6 cases (22%; cases 2, 8, 10, 18, 19, and 22), death was likely due to a combination of verminous pneumonia and other pathological processes of similar severity. In 7 animals (26%; cases 1, 5, 12, 23, and 25–27), all with clinical respiratory signs, the verminous pneumonia was the main finding and hence considered as the cause of euthanasia.
Antiparasitic Treatment
Nine of the 27 hedgehogs (37%) had been medicated with antiparasitics (Supplemental Table S1). Three animals had been treated for less than 5 days before death or euthanasia, 5 had received treatment for a period of 5 to 10 days, and 1 hedgehog (case 27) had been treated for more than 3 months, but showed recurrent respiratory signs (Supplemental Table S2). Most treated hedgehogs had received levamisole twice (n = 6). Interestingly, the degree of pneumonia appeared higher in the treated animals (mean of 2.33 vs. 1.94 in untreated animals), but the difference was not significant (P = .1337). Also, the histological changes did generally not differ between treated and untreated animals; however, the amount of cellular debris, proteinaceous exudate, and neutrophils in the lumen of the airways appeared overall higher in the treated animals.
Discussion
The European hedgehog is a common wild animal in countries, such as Switzerland, where the current study took place, with a generally positive reputation that is also reflected by the abundance of rescue initiatives.18,24,43 However, its population is under pressure due to several risk factors, including life-threatening diseases like pneumonia and many anthropogenic changes, such as habitat fragmentation.8,10,20,37 Indeed, both in the present study and a retrospective study undertaken in Portugal on European hedgehogs between 2002 and 2019, trauma was a frequent cause of death (33.3% and 37.5%, respectively). The latter study identified infectious (including parasitic) diseases as the cause of death in a relatively small proportion of animals (8.1%). 9 In contrast, in the present study, verminous pneumonia appeared to be the single cause for euthanasia in more than a quarter of the animals (26%). People seem to feel a rather unique responsibility toward hedgehogs, and there is a great willingness to help. There are numerous hedgehog care centers where injured or diseased patients are treated mainly based on acquired personal experience due to the lack of empirical data on hedgehog anatomy, diseases, and treatment.4,7,8
There has been anecdotal and empirical evidence (eg, loud coughing, “you hear the hedgehog before you see it”) that hedgehogs often suffer from verminous pneumonia,8,10,20 and there are publications highlighting it as a relevant disease in this species.1,8,20 Parasitic pneumonia was diagnosed in 91% of the cases in a recent study on 35 wild hedgehogs in France that had died naturally or had been euthanized due to medical reasons. 44 Indeed, in our diagnostic material, all routinely necropsied juvenile and adult hedgehogs examined during the last few years were found to harbor lungworms and exhibit various associated changes, regardless of antiparasitic treatment. This prompted us to undertake the current detailed study, making use of 27 cases where the lung tissue was sufficiently preserved and lungworm infection was confirmed by histology or parasitology (feces).
As a first step and since we could not refer to literature on the topic, we studied the normal pulmonary architecture in hedgehogs. This was pursued on the lungs of 1-week-old hoglets that had been euthanized due to persistent anorexia and progressive weakness, but without any respiratory changes. To highlight potential differences between the lung architecture of newborn and adult animals, we comparatively assessed unaltered lung lobes of 2 hedgehogs with mild verminous pneumonia. The results confirmed that the morphology of the hedgehog lung is very similar to that of other mammals. 30 However, it lacked any distinct lymphoid structures, which suggests that hedgehogs do not possess bronchus-associated lymphoid tissue, similar to pigs, mice, and humans.13,32 However, in some of these species, bronchus-associated lymphoid tissue is inducible after infections or long-term inflammation. 32 This seems not to be the case in the hedgehog. We did not observe any lymphoid structures in any verminous pneumonia case, although it would be expected at least with Crenosoma spp. infection, considering that foxes and dogs with C vulpis infection exhibit substantial bronchus-associated lymphoid tissue hyperplasia. 28
European hedgehogs have been shown to harbor 2 lungworms, C striatum, of which the hedgehog is the only known final host, 31 and C aerophila, which is also found in carnivores, such as cats and dogs. 38 The literature reports variable prevalences (C striatum: 45%–79%; C aerophila: 9%–41%), based on postmortem and/or fecal examinations.3,20,26,35 Interestingly, the present study found C aerophila more frequently (81.5%) than C striatum (59.3%). This may have several reasons. First, the study had a focus on the detailed histological examination and hence detected the parasites solely via this approach in a large proportion of cases (n = 13). For C striatum, this required definite identification of adult worms with larvae in their pseudocoelom, whereas C aerophila could be more readily identified by its eggs and intraepithelial localization,1,2,15,28,39 leading to a potential underdiagnosis of the former. Second, when available, the amount of feces that could be collected from the animals was often only sufficient to undertake a sedimentation-flotation protocol and identify C aerophila, not allowing a funnel protocol for the identification of C striatum, likely resulting in further underrepresentation of the latter. Finally, coproscopy might not be sufficiently sensitive to identify all infested animals, due to intermittent excretion of both parasite species and because the Baermann tests rapidly loses sensitivity postsampling.1,31 However, the current study is the first that thoroughly considers coinfections of the 2 parasites in hedgehogs, which we found in more than 40% of the cases. So far, the literature has provided inconclusive data on coinfections. While a study undertaken on fecal samples of hedgehogs across Europe concluded that double infections are common, 29 a study on 40 hedgehogs collected in Italy could merely detect C striatum by combined organ and fecal examination. 20 However, a comparative study in foxes in Canada detected coinfections of C vulpis and C aerophila in as much as 75% of the animals. 28 We are aware that the proportion of coinfection in our material might be even higher as we did not perform parasite specific polymerase chain reaction assays on lung tissue; an approach that would provide more solid epidemiological data.
The present investigation is also the first to fully determine the pathological changes associated with the 2 lungworm species in hedgehogs. We observed C aerophila adults and eggs in the lumen, and embedded within the respiratory epithelium, mainly in larynx, trachea, bronchi, and sometimes in bronchioles. This is largely consistent with its airway distribution reported in red foxes. 28 The presence of this parasite species was always accompanied by pathological changes, such as epithelial hyperplasia and/or metaplasia and an often chronic (peri)bronchial/(peri)bronchiolar inflammatory response, indicating a reaction to long-term irritation. 28 In one animal, we detected a few C aerophila eggs within alveoli, without associated changes, possibly representing agonal aspiration.
In contrast, C striatum adult worms always resided free in the lumen of bronchi, bronchioles, and alveoli. Their presence also had an apparent effect on the respiratory epithelium, but this was restricted to often mild hyperplasia and inflammation. We also observed granulomas in the lung parenchyma that often harbored a parasite larva in the center. Although the morphological features did not always allow definite species allocation, we decided to assign these to C striatum, since C aerophila larvae are known to remain in trachea and bronchi.22,28 The recent study on hedgehogs infested with C striatum alone reported occasional granulomatous foci but did not mention these to be directly associated with larvae. 20 However, an older detailed morphological investigation of the course of experimental C vulpis infection of dogs reports early granuloma formation by macrophages and granulocytes arranged around larvae at 4 days postinfection, 2 days after larvae were first observed within alveoli and respiratory bronchioles and before they appeared to gradually move up the bronchial tree. 36 The authors reported an inflammatory response to the empty cuticles of the third-stage larvae after molting. 36 The presence of the cuticle around the larva in the ultrastructurally examined granuloma in the present study would therefore indicate that the granulomatous response is indeed evoked by the cuticle and restricted to larvae shortly after their emigration from the vasculature. 36 Neither this nor the mentioned experimental study detected larvae of either parasite in vessels or found vascular changes, indicating that the emigration itself does not induce any damage. 36 However, we sometimes found evidence of direct parenchymal damage in response to the larvae, such as focal type II pneumocyte hyperplasia in alveolar walls and at the bronchioloalveolar transition, and bronchiolar destruction. This provides further evidence of direct mechanical irritation as well as alveolar epithelial cell damage and loss due to the larvae, with subsequent regenerative attempts. 36 Also, parasite larvae and C aerophila eggs occasionally elicited a mixed inflammatory reaction (eosinophils, neutrophils, and macrophages) with foreign body giant cells. Interestingly, while we used the immunohistochemical labeling for SP-C to identify type II pneumocytes, we also observed distinct SP-C labeling on larval structures and in the cytoplasm of some multinucleated giant cells that were also occasionally found to carry larval structures. SP-C is one of the proteins in the surfactant.5,25 Inflammatory signals generally influence SP-C expression; as an example, SP-C can interact with CD14 on phagocytes. 25 To our knowledge, there is no evidence of an association of SP-C with pulmonary metazoan parasite infection. However, its deposition on the nematodes cannot be excluded, and further studies might be required to elucidate whether this might initiate or support a host response.
In line with previous studies in hedgehogs and foxes,12,27,28,36 we generally observed a disseminated granulocyte (ie, eosinophil and neutrophil) infiltration with occasional regional clustering that could not be assigned to 1 of the 2 parasite species. The latter also applies to the bronchial/bronchiolar and peribronchial/peribronchiolar infiltration by lymphocytes and plasma cells seen alongside the respiratory epithelial hyperplasia in association with the presence of adult worms in the airway lumen. However, what could be clearly assigned to C aerophila was the substantial epithelial hyperplasia and metaplasia together with the mixed inflammatory response of the airways in association with the presence of intraepithelial parasite stages. To our knowledge, this has so far not been described in any previous study dealing with Crenosoma and Capillaria spp. coinfections. 28
Clinical respiratory signs were reported in 12 of the 27 animals, independent of the parasite species they harbored. The literature suggests that C striatum is more often associated with dyspnea in hedgehogs, 31 possibly due to mucous airway obstruction and emphysema6,31 or due to nematodes bundling in lower airways. 20 In the present cohort, we also found evidence that adult C striatum may have completely obstructed the airways in some cases; however, we observed more pronounced epithelial hyperplasia with intraepithelial C aerophila infection, which may have contributed to airway obstruction and thereby dyspnea by narrowing of the airway lumen. This would be particularly severe in dual infections. However, an absence of respiratory signs excludes neither verminous pneumonia nor its impact on the animals’ health. Hedgehogs with cough and dyspnea may simply represent the “tip of the iceberg,” that is, the seriously ill proportion of the population with other underlying diseases/alterations, possibly also as a consequence of increased stress levels in the suburban environment due to the overall reduction of natural habitats.
Among the hedgehogs with respiratory signs, verminous pneumonia was considered the reason for death or euthanasia in 7 cases (58%). This consideration is relevant since snails and earthworms, the intermediate hosts of both parasites, are part of the hedgehog’s normal diet, which would explain the high prevalences and raise the question of whether hedgehogs are more likely to die “with” or “from” verminous pneumonia. Since merely one quarter of our infested hedgehogs might have died of it, verminous pneumonia might also represent a sentinel for other factors that influence the hedgehogs’ survival.
Previous studies found evidence of a significant correlation between lungworm burden and some population parameters, such as C striatum was more prevalent in male hedgehogs in Ireland, 10 in female hedgehogs in the UK, 35 and in adults than in juveniles in Italy. 20 The present study did not determine parasite burdens but rather focused on the severity of the verminous pneumonia for which it did not find any correlation with population parameters. This also applied to the antiparasitic treatment prior to death. Currently, rescued hedgehogs are frequently treated with antiparasitic drugs, among which levamisole, considered the first choice for C striatum, and flubendazole, reported to be successful against C aerophila, are the most common.14,43 Levamisole, as an acetylcholine mimetic agent, causes depolarization in the ganglia and neuromuscular end plates, ultimately leading to the parasite’s persistent spastic paralysis, 41 whereas flubendazole functions as an inhibitor of tubuline polymerization, disrupting the parasite’s cytoskeleton formation and essential nutrient transport. 40 Ultimately, both drugs should lead to local death of the parasites within hours or few days.41,40 However, C aerophila eggs have stable envelopes that make them highly persistent 31 and therefore more difficult to clear from the airways, leading to a longer recovery period after treatment. 1 In the present study, one third (9/27) of the cohort had been treated with several different antiparasitic drugs and for varying lengths of time. Considering that the effect of levamisole is dose dependent, 41 and that of flubendazole more dependent on the persistence of the drug level, 40 it is not unlikely that at least a proportion of treatment approaches was ineffective. We found no significant differences between treated and untreated animals regarding the histopathological features; in particular, there was no clear morphological evidence of any degenerative changes in the parasites. However, there appeared to be more cell degeneration and more exudate and neutrophils in the airway lumen of the treated animals. The low number of cases and the lack of any empirical significance do not allow any further conclusions on this, but hints toward an area that warrants further investigation.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231193103 – Supplemental material for Verminous pneumonia in European hedgehogs (Erinaceus europaeus)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231193103 for Verminous pneumonia in European hedgehogs (Erinaceus europaeus) by Simone Lehmann, Eva Dervas, Andres Ruiz Subira, Ulrike Eulenberger, Angela Gimmel, Felix Grimm, Udo Hetzel and Anja Kipar in Veterinary Pathology
Footnotes
Acknowledgements
The authors are grateful to the technical staff of the Histology Laboratory and the Electron Microscopy Unit, Institute of Veterinary Pathology, and the Institute of Parasitology, Vetsuisse Faculty, University of Zurich, for excellent technical support. They also wish to express their gratitude to the hedgehog caretakers who supported and enabled this study with great interest and commitment, especially at the Stiftung Wildstation Landshut.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
