Abstract
Trypanosomosis of the West African Dwarf (WAD) sheep is often neglected due to emphasis on trypanotolerance. Nevertheless, significant pathological changes may occur in tissues of infected WAD sheep. The purpose of this study was to evaluate the brain, pituitary, and adrenal lesions of Trypanosoma brucei brucei (Tbb) and Trypanosoma congolense (Tc) infections in WAD rams. Fifteen WAD rams were infected intraperitoneally with Tbb or Tc (106 trypanosomes/animal) or were uninfected controls (5 rams per group). Adrenocorticotrophic hormone (ACTH) and cortisol were assayed in serum by enzyme immunoassay technique. The brain, pituitary, and adrenal glands were processed for histopathology. Serum ACTH levels of infected rams were significantly (P < .05) higher than that of controls on days 14 and 70 post infection (PI). Serum cortisol levels of infected rams were significantly (P < .05) higher than that of controls only on day 14 PI. Mortality was 60% in Tbb- and 40% in Tc-infected rams. The brain of the infected groups showed chromatolysis of cortical neurons and Purkinje cells with severe encephalitis. Degenerative, necrotic, and inflammatory changes were seen in the pituitary and adrenal glands of the infected rams. Adrenal corticomedullary ratio was significantly (P < .05) higher in Tc-infected rams than controls. Based on the high mortality levels, likely due to severe encephalitis, the WAD sheep may not be regarded as trypanotolerant.
Keywords
Trypanosomes are single-celled protozoa, some of which cause severe disease known as trypanosomosis in both humans and livestock.2,27 African animal trypanosomosis (AAT) poses a major challenge to livestock farming in sub-Saharan Africa and limits the full potential of livestock production in the 38 countries where it is endemic, 20 with annual economic losses worth 5 billion USD.13,45
In spite of the enormous challenges posed by AAT, it has been reported by meta-analysis, covering from 1960 to 2017, that the disease has been poorly studied in most parts of Nigeria. 33 The reason for this study gap may be due to the frequent emphasis on trypanotolerance. Trypanotolerance of the West African Dwarf (WAD) breeds is defined as the ability of these breeds to survive, reproduce, and remain productive under trypanosomosis risk without the need for the use of chemicals to control the vector or drugs to control the parasite.8,11 This, however, may not preclude the occurrence of severe pathological changes in the tissues of infected WAD breeds.
In the WAD sheep, the brain, pituitary, and adrenal glands are among the organs in which the lesions and the dynamics of trypanosome infections have either been scantly reported or poorly understood. Studies have demonstrated brain lesions in both natural and experimental infections with Trypanosoma evansi in horses, 39 T. vivax in sheep, 10 T. congolense in cattle, 1 T. brucei brucei in cattle, 17 and T. brucei rhodesiense in vervet monkeys. 31 In cattle, T. congolense was reported to accumulate in the microvasculature of the pituitary gland. 1
The presence of trypanosomes in the brain and pituitary gland may also affect the adrenal gland by altering the hypothalamic-pituitary-adrenal (HPA) axis even in trypanotolerant breeds. T. brucei group has been shown to cause several psychoneuroendocrine disturbances.4,9,22 Similar interference with some endocrine functions also occurs in T. congolense infection.15,30 Elevations of plasma cortisol levels were reported in sheep together with adrenal cortical hyperplasia. 28 However, the cause of the rise in plasma cortisol levels was not known but was speculatively attributed to the presence of large number of T. congolense in the microvasculature of the adrenal glands.3,28 This study comparatively evaluated the effects of T. brucei brucei and T. congolense infections on the brain and pituitary-adrenal axis of WAD rams, which are regarded as trypanotolerant.
Materials and Methods
Animals and Parasites
Fifteen WAD rams, aged 12 to 18 months and average weight for the 3 study groups of 10.0 ± 1.41 to 10.75 ± 1.47 kg (mean ± SEM), were used for the study. They were procured from local breeders and allowed to acclimatize for 3 weeks, during which period they were dewormed with albendazole at 10 mg/kg body weight (b.w.) per os and ivermectin at 1 mL/50 kg b.w., subcutaneously. All the rams were screened for trypanosomes and/or other haemoparasites by examination of the buffy coat and Leishman’s-stained blood smear. The rams were fed on fresh forage and drinking water ad libitum. The study was approved by the Institutional Animal Care and Use Committee (IACUC) of the Faculty of Veterinary Medicine, University of Nigeria, Nsukka (approval reference no: FVM-UNN-IACU C-2020-0345).
T. brucei brucei and T. congolense (Savannah group) (Federer strains) were sourced from the Nigerian Institute for Trypanosomiasis Research (NITR), Kaduna State, Nigeria and maintained by passage in rats.
Study Design and Sampling
The rams used for the study were randomly assigned into 3 groups of 5 rams each. Baseline values of the mean body weight, serum cortisol, and adrenocorticotrophic hormone (ACTH) were determined prior to infection. Rams in each study group were infected with Trypanosoma brucei brucei (Tbb) or Trypanosoma congolense (Tc), or were uninfected controls. Rams in the infection groups were inoculated by intraperitoneal (IP) injection of 1 mL of trypanosome-infected donor blood diluted in physiological saline to obtain a concentration of 106 trypanosomes/mL of blood.
About 3 mL of blood was collected from each of the rams in all groups every 14 days for 70 days by jugular venipuncture. Sampling was consistently done between 08:00 and 09:00 hours, which was reported to be the period of peak cortisol concentration in circulation. 29 Blood samples were collected into clean plain glass tubes containing no anticoagulant, allowed to clot for 45 minutes, then centrifuged at 3000 r/min for 10 minutes, and serum samples were aliquoted for hormone assays. Analyses were completed within 48 hours of sample collection. Serum levels of ACTH (MBS017780) and cortisol (MBS7 78300) were measured by enzyme immunoassay using sheep-specific ELISA test kits (MyBiosource, California, USA), following manufacturer’s instructions.
At the end of the 70-day study period, 3 rams each from the Tc-infected and control groups and 2 surviving rams from the Tbb-infected group were euthanized by intravenous injection of 100 mg/kg b.w. of sodium pentobarbital. The brain, pituitary, and adrenal glands were dissected and weighed using sensitive digital balance (B. BRAN, UK). The brain weights included the cerebrum, cerebellum, and brainstem including medulla and pons. Organ weights were expressed as a percentage of body weight. The cerebrospinal fluid was harvested from the ventricles with syringes, avoiding contact with blood, and was examined using phase-contrast microscope (Motic, USA).
Determination of Brain Sodium and Oxidative Stress Markers
Fresh brain tissues (parietal, temporal, and occipital lobes of the cerebrum) were homogenized in phosphate buffer (10 mM, pH 7.4; 0.1 mM EDTA) and centrifuged at 5000g for 20 minutes. The resulting supernatant was used to assay for sodium levels and the oxidative stress indicators-superoxide dismutase (SOD) and malondialdehyde (MDA). The analysis of brain sodium concentration was performed using a commercially available sodium test kit (Agappe Diagnostics, Cham, Switzerland), following the manufacturer’s instructions. The evaluation of brain SOD activity followed a published method 25 based on the ability of SOD to inhibit the autoxidation of epinephrine to adrenochrome at pH 10.2. Brain MDA level was measured by quantifying the formation of thiobarbituric acid reactive substances (TBARS). 42 Test kits for SOD and MDA assays were sourced from Sigma Chemicals (St. Louis, MO, USA).
Histopathology and Histochemistry
The brain was fixed in Bouin’s fixative for 12 hours before it was transferred to 70% alcohol. The pituitary and adrenal glands were fixed in 10% neutral-buffered formalin for 72 hours. After fixation, the organs were routinely processed and sectioned at 5 µm thickness and stained with hematoxylin and eosin. Parallel samples of the brain were stained with cresyl fast violet to demonstrate the Nissl substance in neurons. 40
Histomorphometry of the adrenal gland was evaluated with the aid of a stage micrometer and eyepiece graticle. The average sizes of the different zones of the adrenal cortex (zona glomerulosa, zona fasciculata, and zona reticularis) as well as the adrenal medulla were measured and recorded based on the average of several independent measurements by 2 evaluators. The average corticomedullary ratio of each group in the experiment was calculated.
Statistical Analysis
Data generated from the study were subjected to 1-way analysis of variance (ANOVA) using SPSS, version 21. Data on parasitaemia were analyzed by Student’s t-test. Variant means were separated post-hoc using the least significant difference (LSD) method. The level of significance was P ≤ .05.
Results
Parasitaemia
Parasitaemia was first detected on Day 5 post infection (PI) in both infected groups (Tbb and Tc), and by Day 6 PI, all infected rams had become parasitaemic. Three peaks of parasitaemia were observed in Tbb-infected rams on days 14 (4.0 × 107 trypanosomes/mL blood), 35 (1.0 × 107 trypanosomes/mL blood), and 56 (1.6 × 107 trypanosomes/mL blood), PI. The parasitaemia of Tc-infected rams peaked on days 6 (1.26 × 106 trypanosomes/mL blood) and 14 (1.3 × 106 trypanosomes/mL blood) PI, after which no sharp fluctuations were observed until the end of the study. Motile trypanosomes were also seen in the cerebrospinal fluid of rams in both infected groups at necropsy.
Hormonal Changes
Serum ACTH levels of both infected groups were significantly (P < .05) higher than that of the control on day 14 PI. On day 70 PI, the serum ACTH levels of the Tbb-infected group was significantly (P < .05) higher than that of the control, whereas the mean serum ACTH level of the Tc-infected group was significantly (P < .05) higher than those of Tbb-infected group and the control (Fig. 1).
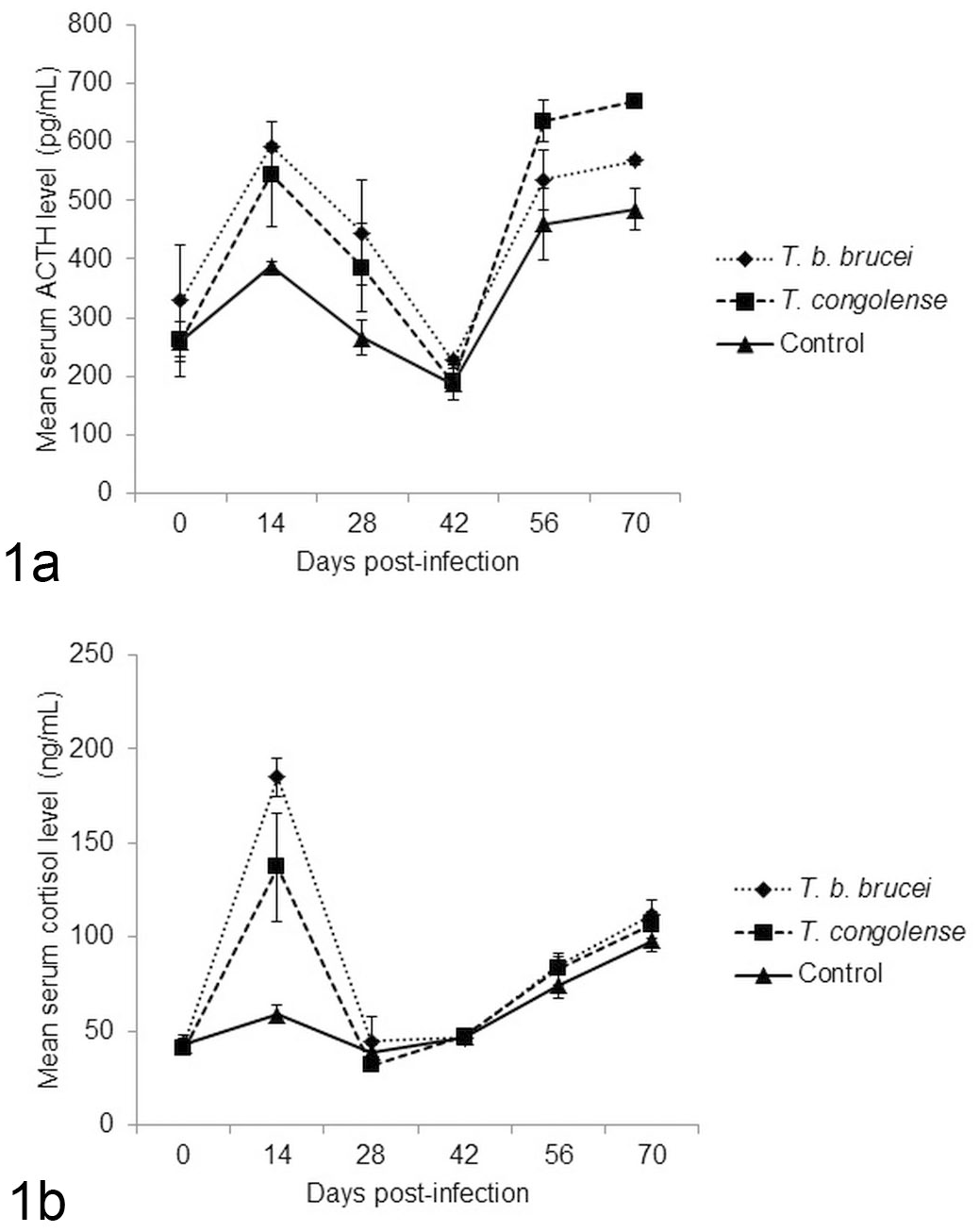
Mean serum ACTH and cortisol levels in West African Dwarf sheep infected with T. brucei brucei or T.congolense, or uninfected controls. (1a) Mean serum ACTH levels. The infected rams had significantly higher values compared to the controls on days 14 and 70 post infection. (1b) Serum cortisol levels. The infected rams had significantly higher values compared to the controls on Day 14 post infection.
Serum cortisol levels of the infected groups (Tbb and Tc) were significantly (P < .05) higher than that of the control group on Day 14 PI. Beyond Day 14 PI, there were no significant differences between groups in the mean serum cortisol levels (Fig. 1).
Clinical Signs and Postmortem Findings
In the Tbb-infected group, 3 of 5 rams died on days 14, 50, and 70 PI. The ram that died on day 50 PI was paralyzed and had a head tilt by day 49 PI. In the Tc-infected group, 2 of 5 rams died on days 20 and 54 PI. There were no mortalities in the uninfected controls.
The carcasses (including the brains) of the Tbb-infected rams were paler than those of Tc-infected and uninfected control rams. No gross lesions were observed in the pituitary glands in any of the groups. The adrenal glands of the infected rams were markedly enlarged compared to those of control rams. The prescapular lymph nodes of the Tbb-infected rams were moderately enlarged while those of Tc-infected rams were larger than those of Tbb-infected rams and were hyperaemic. Similarly, the spleens of the Tbb- and Tc-infected rams were larger than those of the control rams and the cut surfaces were gelatinous. The lungs of the infected rams had similar lesions characterized by moderate emphysema with rib imprints on the pleural surface, hyperaemia, and patchy consolidation that were more pronounced in the caudal lobes. Other notable lesions observed in the carcasses of the infected rams were hydrothorax, hydropericardium, and hydroperitoneum, and these were more pronounced in Tbb-infected than Tc-infected rams. The liver of the infected rams were slightly enlarged. The kidneys, especially those of Tc-infected rams, were markedly enlarged. Renal and pericardial fat was gelatinous in both infected groups of rams.
The mean brain and adrenal weights (relative to body weight) of the infected groups (Tbb and Tc) were significantly (P < .05) higher than that of the controls. There were no significant (P > .05) differences in the mean relative pituitary weights of the rams across the groups (Table 1).
Relative organ weights of West African Dwarf rams infected for 70 days with T. brucei brucei, and T. congolense and the uninfected control.
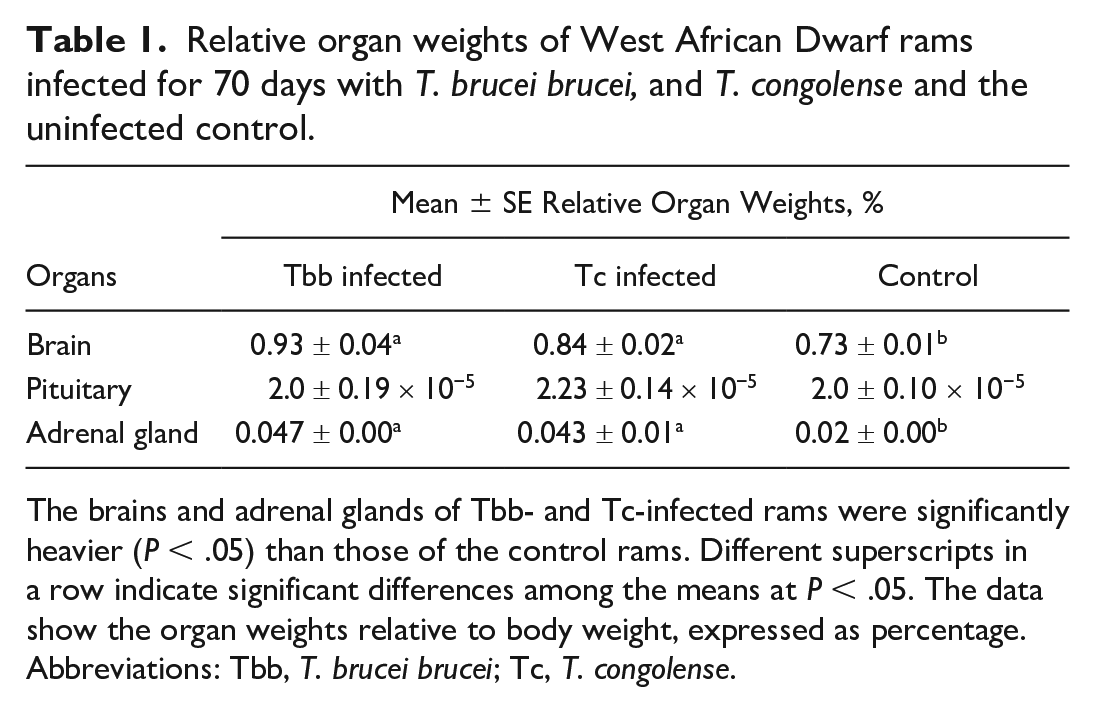
The brains and adrenal glands of Tbb- and Tc-infected rams were significantly heavier (P < .05) than those of the control rams. Different superscripts in a row indicate significant differences among the means at P < .05. The data show the organ weights relative to body weight, expressed as percentage.
Abbreviations: Tbb, T. brucei brucei; Tc, T. congolense.
Brain Sodium, SOD, and MDA Levels
Brain tissue sodium concentrations of the Tbb-infected group were significantly (P < .05) higher than those of the control. Although the mean brain tissue sodium concentration of the Tc-infected group was also higher than that of the control group, this was not statistically significant (Fig. 2).
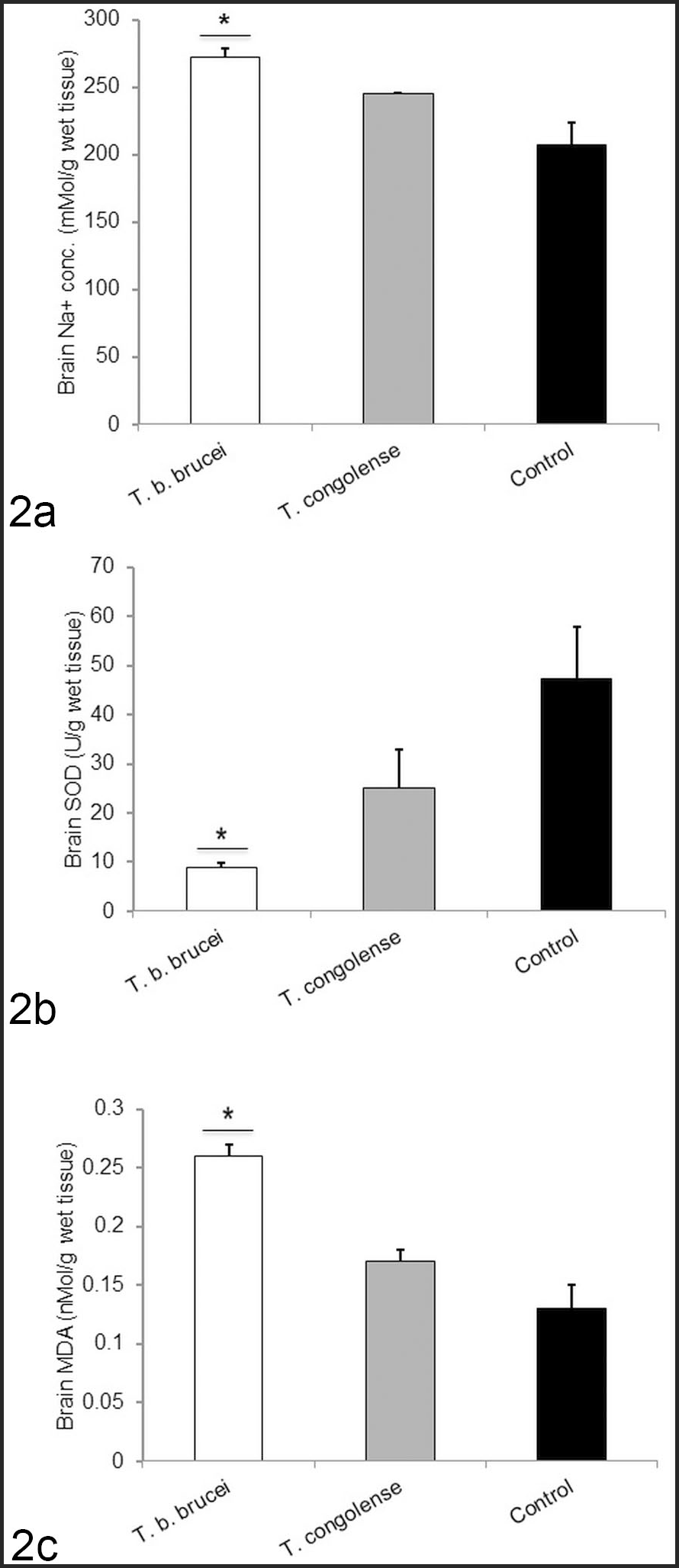
Mean brain sodium concentration, superoxide dismutase (SOD) activity, and malondialdehyde (MDA) levels in West African Dwarf sheep infected with T. brucei brucei (Tbb) or T. congolense (Tc), or uninfected controls. (2a) Sodium concentration. Tbb-infected rams had significantly higher value compared to the controls. (2b) SOD activity. Tbb-infected rams had significantly lower value compared to the controls. (2c) MDA levels. Tbb-infected rams had significantly higher value compared to the controls.
The brain tissue SOD activity of the Tbb-infected group was significantly (P < .05) lower than that of the control. Similarly, the mean brain tissue SOD activity of the Tc-infected group was also lower than that of the control, but this was not statistically significant (Fig. 2).
Brain tissue MDA levels of the infected groups (Tbb and Tc) were higher than that of the control, but this difference was only significant (P < .05) for the Tbb-infected group (Fig. 2).
Histomorphometry of the Adrenal Gland
The average radial sizes of the zona glomerulosa, zona reticularis, and adrenal medulla were significantly (P < .05) higher for the Tbb-infected group compared to the Tc-infected and uninfected control groups. The average sizes of the zona fasciculata of the Tbb- and Tc-infected groups were significantly (P < .05) higher than that of the control (Table 2). The mean size of the entire adrenal cortex of Tbb-infected rams was significantly (P < .05) higher than that of the control; the average size of the adrenal cortex of Tc-infected rams was also higher than that of the control, but this was not statistically significant (Table 2).
The radial sizes of the different anatomical zones of the adrenal gland and the adrenal corticomedullary ratios of West African Dwarf rams infected for 70 days with Tbb and Tc and the uninfected control.
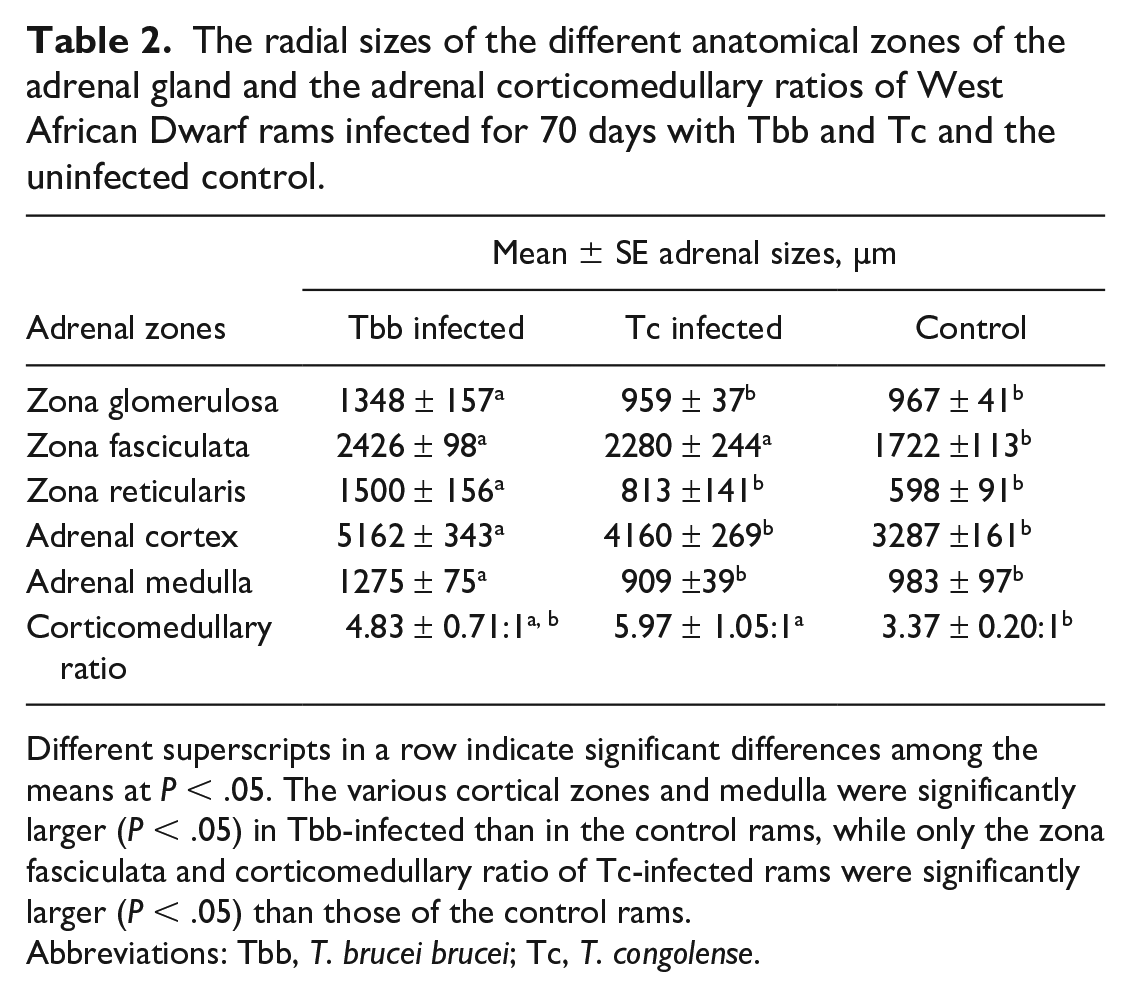
Different superscripts in a row indicate significant differences among the means at P < .05. The various cortical zones and medulla were significantly larger (P < .05) in Tbb-infected than in the control rams, while only the zona fasciculata and corticomedullary ratio of Tc-infected rams were significantly larger (P < .05) than those of the control rams.
Abbreviations: Tbb, T. brucei brucei; Tc, T. congolense.
The corticomedullary ratios of the infected groups were higher than that of the control group, but this was significant only for the Tc-infected group (P < .05; Table 2).
Histopathology
The cerebrum of infected rams had vacuolation in the white matter in addition to gliosis and degeneration of neurons. These lesions were absent in the uninfected rams (Figs. 3–5). Lesions in the brainstem of the Tbb- and Tc-infected rams were characterized by severe perivascular cuffing with macrophages and lymphocytes in addition to glial cell proliferation, and these lesions were absent in the uninfected rams (Figs. 6–8). In the cerebellum, spongiosis occurred around the Purkinje cell layer in Tbb-infected rams (Fig. 9) while loss of Purkinje cells was observed in the Tc-infected rams (Fig. 10), whereas the uninfected rams had histologically normal Purkinje neurons (Fig. 11). Cresyl violet staining showed that neurons in the hippocampal region of the cerebral cortex were poorly basophilic in both infected groups of rams with fragmentation of the Nissl granules in the Tc-infected rams, compared to neurons of uninfected rams with intact and deeply basophilic Nissl substance (Figs. 12–14).
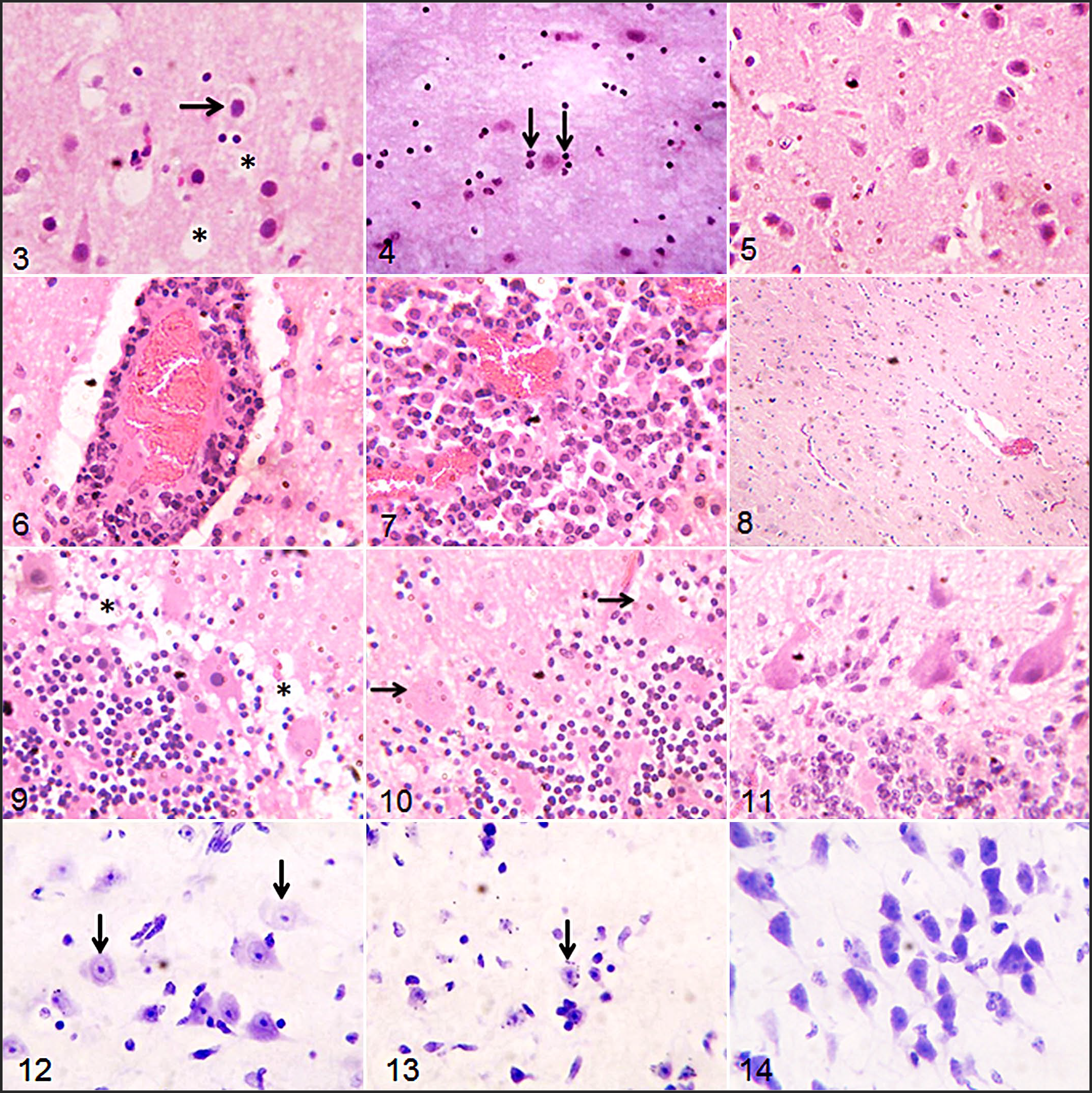
Trypanosomosis, brain, West African Dwarf rams. Rams were infected with T. brucei brucei (Tbb) (Figs. 3, 6, 9, and 12) and T. congolense (Tc) (Figs. 4, 7, 10, and 13), or uninfected controls (Figs. 5, 8, 11, and 14). Figs. 3–10. Hematoxylin and eosin.
The anterior pituitary glands of the Tbb-infected rams had focal necrotic areas in the pars intermedia (Fig. 15). In the Tc-infected rams, severe coagulative necrosis with nuclear pyknosis and necrotic debris were observed in the pars distalis of the pituitary gland (Fig. 16). The pituitary glands of the uninfected rams showed normal basophils, acidophils, and sinusoids (Fig. 17).
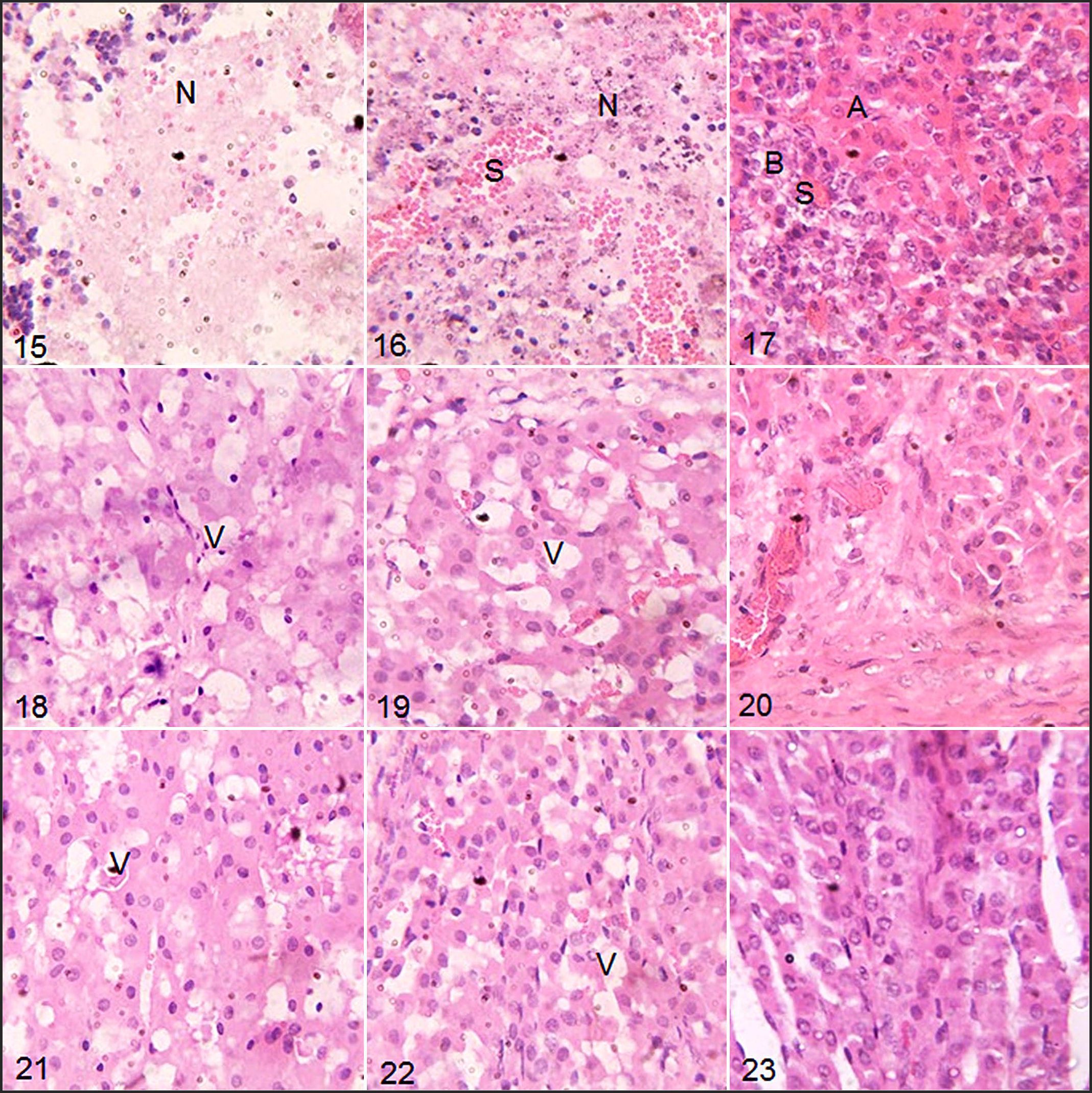
Trypanosomosis, West African Dwarf rams. Rams were infected with T. brucei brucei (Tbb) (Figs. 15, 18, and 21) or T. congolense (Tc) (Figs. 16, 19, and 22) or uninfected control (Figs. 17, 20, and 23). Hematoxylin and eosin.
The adrenal cortices of the infected rams were vacuolated in the zona glomerulosa and zona fasciculata, compared to the adrenal cortex of the uninfected rams which showed normal glandular cells of the zona glomerulosa and zona fasciculata (Figs. 18–23). Compared to Tc-infected rams, Tbb-infected rams, showed more severe vacuolation and mild infiltration of mononuclear inflammatory cells in the zona glomerulosa (Fig. 18).
Discussion
Trypanosome infection of the brain was established in this study by the presence of T. brucei brucei and T. congolense in the CSF, which agrees with previous reports of the presence of trypanosomes in the CSF, including T. brucei spp in cattle 44 and both T. brucei brucei and T. congolense in a mixed infection in cattle. 21 These brain infections may have contributed to the significantly higher relative brain weight and sodium concentration as well as redox imbalance characterized by lower SOD and higher MDA activities observed in the infected groups especially that of Tbb-infected rams. The brain is highly sensitive to oxidative stress because it is rich in iron and polyunsaturated fatty acids and has high affinity for oxygen.7,41,47 Earlier reports in T. congolense-infected rats showed that MDA increased while SOD decreased also in other tissues, especially the liver and heart. 14 These brain infections may be due to the tendency for T. brucei brucei to extravasate from the blood vessels into the brain tissue whereas T. congolense accumulates in the brain microvasculature.
The impacts of the infections in the brain were also revealed microscopically in the cerebral cortex, brainstem, and cerebellum in the form of degenerative, necrotic, and inflammatory changes, which may be responsible for the high levels of mortality in the Tbb- and Tc-infected rams. These CNS changes have been reported in previous studies in different host species including cattle, horse, sheep, pigs, rodents, and man.1,10,36,39 Inflammatory reactions were more prominent in the brainstem compared to the cerebral cortex and cerebellum in this study, probably due to the structures in the brainstem, such as the circumventricular organs, lacking complete blood-brain barrier.23,37
This study identified chromatolysis in the neuronal cell bodies of the cerebral cortex and Purkinje cells of the cerebellum in Tbb- and Tc-infected rams. This lesion of trypanosomosis has not been reported in domestic animals in available literature. However, loss of Purkinje cells was reported in rats infected with Trypanosoma evansi, 6 while central chromatolysis was reported in the spinal cord of horses in the last stage of dourine caused by Trypanosoma equiperdum. 24
It was posited that neurons are not affected in cerebral trypanosomosis, and reports on demyelination were conflicting and inconclusive. 19 However, this study suggests that loss or disaggregation of Nissl substance in many neurons may result following the invasion of the brain by trypanosomes and consequent spread of inflammatory reactions. Nissl granules participate in the synthesis of neurotransmitters,26,46 and their loss or disaggregation may be responsible for the CNS disturbances usually seen in AAT and human African trypanosomosis (HAT), including the paralysis and head tilt observed in the Tbb-infected ram by day 49 PI. We speculate that the loss of Nissl substance might have resulted from activation of ribonucleases, which may have activated intracellular digestion following axonal injury and neuronal demyelination, 26 perhaps initiated by the vasogenic edema observed in the brain parenchyma or by autoantibodies against the myelin components. In the meneingoencephalitic stage in HAT patients and in sheep infected with T. brucei brucei, antibodies directed against myelin proteins and those directed to neurotransmitters were identified in the serum.18,35 Dementia, incoherence, double incontinence, and seizures (which depict CNS demyelination) have also been observed in the terminal stage of the disease in humans. 43 Although neuronal demyelination was not demonstrated in this study, separation of the myelin lamellae due to intramyelinic edema might be responsible for the neuropil vacuolation observed especially in the cerebrum. This intramyelinic edema may have been caused by vasogenic edema occasioned by damage to blood-brain barrier, increased brain Na+ concentration and redox imbalance leading to brain edema and cytotoxic changes in neurons.
In both infected groups, focal degenerative, necrotic, and inflammatory changes in the anterior pituitary were observed, and these agree with previous reports of anterior pituitary lesions in T. congolense-infected Boran cattle 1 and T. brucei brucei-infected goats and sheep. 17 These lesions may have been initiated by the localization of trypanosomes in the pituitary microvasculature in the case of T. congolense 1 or in the pituitary interstitial connective tissue in the case of T. brucei brucei. 17
It appears that the pituitary lesions were not severe enough to depress ACTH secretion in the infected rams, although cortisol secretion was blunted at the later phases of the infections. These findings agree with a previous study in Scottish Blackface sheep in which a biphasic rise in ACTH levels were reported in the early and later phases of the infection with T. congolense. 28 However, the same lone surviving sheep in that study 28 also showed triphasic rise in serum cortisol levels between days 9 and 16 PI, days 51 and 56 PI and days 77 and 79 PI, which differs from the monophasic cortisol rise on Day 14 PI reported in this study. The reason for the lack of significant upsurge in cortisol levels of the infected groups beyond Day 14 PI in this study may be due to breed differences. A blunted cortisol response has been reported in HAT and is mediated by TNF-α and IL-632,38 and may potentially contribute to the survival of trypanosome-infected WAD rams. This blunted cortisol response was more pronounced in the Tc-infected WAD rams that had the highest ACTH values on days 56 and 70 PI than in the Tbb-infected WAD rams and may have contributed to the gross and microscopic changes in the adrenals of the infected rams.
The percentage relative weights also showed that the adrenals of the Tbb- and Tc-infected rams were heavier than those of the controls by 170 and 130%, respectively. These findings agree with previous reports of enlargement of the adrenal glands in sheep infected with T. brucei brucei 9 and T. congolense. 28 The variations in the radial sizes of the adrenal cortex and medulla, as well as in the corticomedullary ratio observed in this study have not been reported previously. However, ACTH, being a hormone of cellular growth and differentiation, is known to cause hyperplasia and hypertrophy of chronically stimulated adrenal cortical cells. 5 The enlargement of the adrenal glands might also be a reflection of the level of stress induced by the trypanosome infections in the WAD rams as the adrenal gland weight and corticomedullary ratio are also used as indicators of stress in animals. 12
Vacuolation of the adrenal cortex was previously reported in T. brucei-infected sheep. 16 Hyperplasia of the zona fasciculata and reticularis were also previously reported in T. congolense infection in Bunaji cattle 34 and Scottish Blackface sheep. 28 In the latter, the adrenal lesion was accompanied by elevated serum cortisol level at the chronic stage of the infection, while in the former, the adrenal lesion progressed to fibrosis and depletion of the adrenal cortical cells with decreased serum cortisol level. These changes differed from the present report in the WAD breed in which the adrenal lesions were not associated with significantly altered serum cortisol levels in both the Tbb- and Tc-infected rams at the long-term phases of the infections.
The major limitation of this study is the lack of identification of the trypanosomes (Tbb and Tc) in the brain, pituitary, and adrenal tissues by in situ hybridization, immunohistochemistry, or electron microscopy due to lack of facility. This would have been helpful in the interpretation of the inflammatory and non-inflammatory changes in the tissues.
In conclusion, the study showed that infection of WAD rams with T. brucei brucei and T. congolense resulted in severe inflammatory, degenerative, and necrotic changes in the brain, pituitary, and adrenal glands. The severity of the encephalitis may be responsible for the high levels of mortality in the Tbb- and Tc-infected rams observed in this study. Based on these findings, the WAD sheep may not be strictly regarded as being trypanotolerant. It may be that introgression of genes from susceptible breeds of sheep had occurred in the WAD breed from poor conservation of the trypanotolerance trait. It is therefore recommended that chemotherapy/chemoprophylaxis should be emphasized in WAD sheep in tsetse endemic areas.
Footnotes
Acknowledgements
The authors are grateful to the Nigerian Institute for Trypanosomiasis Research (NITR) for donating the trypanosomes used for the study and to Mr. O.I. Agbakwuru for helping with tissue processing for histopathology and histochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Institutional Based Research (IBR) grant from Tertiary Education Trust Fund (grant no. TETFund/DESS/UNI/NSUKKA/2018/RP/VOL.1).
