Abstract
Swine dysentery (SD) is an enteric disease associated with strongly β-hemolytic Brachyspira spp. that cause mucohemorrhagic diarrhea primarily in grower-finisher pigs. We characterized alteration of colonic mucin composition and local cytokine expression in the colon of pigs with acute SD after B. hyodysenteriae (Bhyo) infection and fed either a diet containing 30% distillers dried grains with solubles (DDGS) or a control diet. Colonic tissue samples from 9 noninoculated pigs (Control, N = 4; DDGS, N = 5) and 10 inoculated pigs experiencing acute SD (Bhyo, N = 4; Bhyo-DDGS, N = 6) were evaluated. At the apex of the spiral colon, histochemical staining with high-iron diamine–Alcian blue revealed increased sialomucin (P = .008) and decreased sulfomucin (P = .027) in Bhyo pigs relative to controls, with a dietary effect for sulfomucin. Noninoculated pigs fed DDGS had greater expression of sulfomucin (P = .002) compared to pigs fed the control diet. Immunohistochemically, there was de novo expression of mucin 5AC (MUC5AC) in the Bhyo group while mucin 2 (MUC2) expression was not significantly different between groups. RNA in situ hybridization to detect the pro-inflammatory cytokine IL-1β often showed increased expression in the Bhyo group although without statistical significance, and this was not correlated with MUC5AC or MUC2 expression, suggesting IL-1β is not a major regulator of their secretion in acute SD. Expression of the anti-inflammatory cytokine TGF-β1 was significantly suppressed in the Bhyo group compared to controls (P = .005). This study reveals mucin and cytokine alterations in the colon of pigs with experimentally induced SD and related dietary effects of DDGS.
Keywords
Swine dysentery (SD) is a production-limiting disease associated with strongly β-hemolytic Brachyspira spp. infection that manifests as profuse mucohemorrhagic diarrhea and typhlocolitis. 28 Disease is most often observed in grower-finisher pigs and results in poor body condition and significant economic losses. 7,28 Three unique Brachyspira species have been recovered from pigs with clinical SD and have been shown to reproduce disease in pigs experimentally. These include Brachyspira hyodysenteriae, 29,60 Brachyspira hampsonii, 15,53,81 and Brachyspira suanatina 58,66 and typical isolates of all 3 induce strong β-hemolysis when cultivated on blood agar. 28 Ingestion of infected feces is the main route of natural transmission and infection is limited to the cecum and colon where the spirochetes colonize the mucus layer and within crypts; however, during clinical disease spirochetes may be observed within goblet cells and intracellularly. 7 Infected pigs frequently develop a markedly thickened colonic mucosa with mixed leukocytic infiltration and increased mucus production with altered mucin composition. 62,81
The mucus layer of the intestines serves as an immune barrier against pathogens. Increased mucus production is usually observed when encountering pathogens such as bacteria or intestinal parasites. 48,61 Different from the mucus layer in the small intestine that is loose and unattached with large pores to allow penetration of nutrients, the colon has a 2-layered mucus system composed of the inner anchoring layer that is devoid of bacteria, and a thick outer layer that is rich in microbiota. 4,32,33 These mucus layers are mainly composed of mucins, which are large glycoproteins that can be simply classified as neutral and acidic. Acidic mucins are the main component of the colonic mucus layer and can be further categorized as sialomucin and sulfomucin based on the terminal group of either sialic acid or sulfate groups. 16 These 2 mucin types can be identified using the histochemical staining of high-iron diamine–Alcian blue (HID-AB) in which sulfated mucins are black and sialylated mucins are blue. Besides these chemotypes, mucins can also be classified by their different protein components depending on the expression of associated genes. 11,13,33,42,48 There are 3 subfamilies of mucins: secreted gel-forming mucins, secreted non-gel-forming mucins, and cell-surface mucins. 13,48 In pigs with SD, an increased expression of mucin 2 (MUC2, secreted gel-forming), de novo expression of mucin 5 AC (MUC5AC, secreted gel-forming), and a decrease in mucin 4 (MUC4, cell-surface) has been reported. 62,81 As pathogenic Brachyspira spp. exhibit a strong chemotaxis to mucin, 36,52 the dynamic changes of mucin during SD and the interaction of mucins with these pathogens may play a role in disease pathogenesis. However, there is limited information regarding the underlying mechanisms that drive these changes.
Host cytokine homeostasis during enteric diseases can contribute to regulation of mucin production. 4,48 Nonetheless, information regarding the host immunopathogenesis and cytokine expression in pigs with SD is scarce. Previous studies of SD revealed increased IL-1β at the beginning of the disease accompanied with increased serum amyloid A. 43 Upregulation of neutrophil elastase, IL-17A, IL-1β, IL-6, and IL-8 mRNA expression has also been reported in the colons of infected pigs with SD, and neutrophil elastase and IL-17A were found to induce mucin production in vitro. 63 IL-1β has been shown to contribute to the expression of MUC2, MUC5AC, or both in human studies. 24,40,41,71 Besides the pro-inflammatory cytokines mentioned above, systemically increased IL-10, an immunosuppressive cytokine, has been detected in pigs with SD during the recovery phase. 43 The anti-inflammatory cytokine TGF-β has not been reported in pigs with SD but is reported to play an important role in development of ulcerative colitis and Crohn’s disease in humans. 17 The dysregulation of the TGF-β signaling pathway contributes to inflammatory bowel disease. 2,17,56 In our study, we hypothesize that there is an increased expression of local IL-1β in pigs with SD, which may contribute to mucin alteration, and a lower expression level of TGF-β1 in infected pigs causing diminished anti-inflammatory and repair responses.
Alterations in dietary insoluble fiber have been shown to affect the intestinal microbiome in pigs and may predispose to the development of SD in previous studies. 8,30,80 In the past few decades, commercial swine diets have increased the use of distiller’s dried grains with solubles (DDGS), which is a coproduct of biofuel production. 73 DDGS have relatively more insoluble dietary fiber than whole corn and this fiber passes through to the colon where it is fermented. 74 Dietary fiber can affect goblet cell differentiation and mucin production and composition. 31,54,55,69 For instance, feeding DDGS increased ileal MUC2 expression in pigs 22 and dietary fiber ingestion altered the amount of sulfomucin and sialomucin in intestines of rats. 31,55 However, information regarding the relationship between diet, SD, and the resultant mucin alteration in pigs is lacking.
Accordingly, the objectives of this study were to (1) characterize and quantify sulfomucin, sialomucin, MUC2, and MUC5AC expression in the colonic mucosa of pigs with acute SD relative to uninfected controls; (2) determine if these responses differ in pigs fed a control diet versus one containing 30% DDGS; and (3) assess expression of the pro-inflammatory cytokine IL-1β and the immunoregulatory cytokine TGF-β1 in serial sections from the same tissues to provide further insights into the pathogenesis of SD and associated mucin production.
Materials and Methods
Source and Selection of Tissue Samples
Paraffin-embedded, formalin-fixed tissue blocks of ileum and spiral colon from a subset of pigs in a previous animal inoculation experiment were included in this study. 80 Animals utilized in this experiment were crossbred gilts and barrows that were obtained at 4 weeks of age and originated from a commercial source with no known history of Brachyspira-associated disease. All pigs had been individually confirmed as culture-negative for all Brachyspira spp. prior to their enrollment in the inoculation experiment. Pigs had been acclimated to their diet for 2 weeks prior to inoculation with Bhyo. Colon tissues from all animals included in this study were negative for Salmonella spp. by direct culture at necropsy. As the previous study included a dietary fiber treatment (0% or 30% corn DDGS), 80 approximately half of each group in the current investigation came from each dietary treatment. There were 9 samples from sham-inoculated controls (Control, N = 4; DDGS, N = 5) and 10 samples from pigs experiencing acute SD due to infection with Brachyspira hyodysenteriae (Bhyo, N = 4; Bhyo-DDGS, N = 6).
Histopathology
The colonic and ileal tissues were sectioned at 4 μm, stained with hematoxylin and eosin, and examined for microscopic lesions. Investigators were blinded to treatment group throughout the experiment. Sections of ileum were evaluated for the presence of lesions indicative of Lawsonia intracellularis or Salmonella spp. infection. Sections from the apex of the spiral colon were reviewed and neutrophils in the lamina propria were counted in ten 400× fields and the mean per section of each individual pig was obtained. Crypt depths in each sample were measured using the ruler function in a commercial software package (cellSens ver. 1.18, Olympus USA). Measurement was made in the crypts from the base of the crypt to the mucosal surface, and only crypts that were oriented perpendicularly to the mucosa with intact epithelium were selected. The mean of 10 measurements from the apex of spiral colon was recorded for each sample.
Histochemistry
Formalin-fixed, paraffin-embedded tissue samples from the apex of the spiral colon were sectioned at 4 µm onto positively charged slides, deparaffinized, and stained with a combination of high-iron-diamine and Alcian blue (pH 2.5) (HID-AB) staining to reveal the presence of sulfomucins (black staining) and sialomucins (blue staining). Sections were placed in high-iron-diamine solution for 18 hours at 57 °C, rinsed with water, placed in Alcian blue solution for 5 minutes, rinsed with water, dehydrated, and mounted.
Immunohistochemistry
MUC5AC
Immunohistochemistry was performed on formalin-fixed, paraffin-embedded colonic samples using a primary monoclonal mouse anti-human antibody for MUC5AC (Zymed Laboratories, Invitrogen Corporation) as previously described. 81 Briefly, paraffin-embedded tissues were sectioned at 4 µm onto positively charged slides, baked at 57 °C for 2 hours, and deparaffinized through graded alcohol. Slides were incubated with 3% hydrogen peroxide to inhibit the endogenous peroxidase for 20 minutes followed by 3 rinses with ultrapure water. Antigen retrieval was attained by microwaving slides in Tris/EDTA (pH 9.0) for 1 minute followed by a steam chamber for 20 minutes. Blocking of nonspecific protein binding sites was performed by application of 1:10 dilution of goat serum and PBS (phosphate-buffered saline) buffer for 20 minutes at 37 °C. Slides were then incubated with the primary antibody MUC5AC (dilution 1:50 in PBS containing 0.1% Tween 20) overnight at 4 °C in a humidified chamber. Sections were washed with PBS and incubated with a commercially available biotinylated secondary antibody (MultiLink, BioGenex; 1:80) at 22 °C for 30 minutes. After rinsing in a bath of PBS solution for 5 minutes, sections were incubated with horseradish peroxidase-streptavidin conjugate (Zymed, Invitrogen) at 22 °C for 15 minutes and then rinsed in a bath of PBS for another 5 minutes. The reaction was visualized using a commercial chromogen (NovaREDTM, Vector laboratories) for 5 minutes at 22 °C and rinsed with ultrapure water. Slides were subjected to a Shandon’s hematoxylin counterstain, placed in Scott’s Tap water for 1 minute, rinsed with ultrapure water, dehydrated through a graded alcohol series, and mounted.
MUC2
Staining was performed on formalin-fixed, paraffin-embedded colonic samples using a monoclonal mouse anti-human antibody MUC2 (Santa Cruz Biotechnology). Tissues were sectioned at 4 µm onto positively charged slides and processed as previously described for MUC5AC but with a few modifications. Antigen retrieval was performed using citrate buffer (pH 6.0) and a pressure cooker (1100-W microwave, 45 seconds at 100% power followed by 5 minutes at 30% power). Slides were then placed in an automated cell staining system (OptiMaxPlus, BioGenex) and incubated with goat serum (1:10 dilution in PBS buffer) for 20 minutes at 22 °C to block nonspecific protein binding sites. The primary antibody for MUC2 which has been proven to show specificity in pig large intestine 26 was diluted to 1:100 in PBS containing 0.1% Tween 20. Slides were then incubated with the primary antibody for 1 hour at 22 °C and processed as described above.
RNA In Situ Hybridization (ISH) for IL-1β and TGF-β1
Chromogenic RNA ISH for IL-1β (Gene ID: 397122, Catalog No. 300031, customized, targeted for IL-1β1, Newark, CA) and TGF-β1 (Gene ID: 397078, Catalog No. 300031, customized, Newark, CA) were performed by using the RNAscope 2.5 HD Assay-Red reagents obtained from Advanced Cell Diagnostics (ACD RNAscope, Catalog No. 322350, Newark, CA). Blocks of formalin-fixed, paraffin-embedded colonic samples were sectioned at 4 μm, mounted onto positively charged slides, and dried overnight at room temperature. Slides were then de-paraffinized by passage through xylene (2 × 5 minutes), and dehydrated by immersing in 100% alcohol (2 × 1 minutes). The slides were air-dried and quenched with RNAscope hydrogen peroxide at room temperature for 10 minutes and rinsed with distilled water. After immersing in the prepared RNAscope 1X Target Retrieval Reagent for 15 minutes at 100 °C, tissue slides were rinsed in distilled water, immersed in 100% alcohol for 3 minutes, and then air-dried at room temperature. Slides were the incubated with RNAscope Protease Plus for 30 minutes at 40 °C in the HybEZTM Oven and rinsed in distilled water 3 times. The slides were incubated with the preheated buffer containing the 2 specific RNA probes and hybridized for 2 hours at 40 °C in the HybEZTM Oven. Following amplification of 6 rounds in hybridization buffer, slides were washed, incubated with red chromogenic detection solution for 10 minutes at room temperature, counterstained with hematoxylin, and finally coverslipped with mounting media. Appropriate controls were present in every run, including the positive control probe-Ss PPIB (ACD RNAscope) and negative control probe-dapB (ACD RNAscope).
Image Analysis
Slides labeled by immunohistochemistry, histochemical staining, or RNA ISH were imaged using an Olympus DP74 camera mounted on an Olympus BX41 microscope with UPlanFLN objectives and operated with cellSens Dimension software (version 1.17, Olympus Corporation). Five representative 400× images of colonic mucosa were captured for each slide. For immunohistochemical and histochemical staining, captured images were analyzed quantitatively using the Area Quantification module v1.0 within the HALO image analysis platform (v2.0.1145.19, Indica Labs). Briefly, representative staining color of nuclear stain (blue hematoxylin, Stain No. 1) and positive signal (red chromogen in IHC or black staining in HID-AB, Stain No. 2; blue staining in HID-AB, Stain No. 3) were selected and set as the referenced color baseline. Base threshold intensity values were manually adjusted and assigned by using the real-time tuning option in the software to reflect accurate staining distribution in the areas of interest. Quantification of the total chromogen, black or blue staining area was then analyzed by the module algorithm as percentage Stain No. 2 or No. 3 positive tissue, which refers to the percentage of positive staining of the region of interest. All staining artifacts or regions of white space were manually excluded from the analyzed area by using the software exclusion tool. For RNA ISH image analysis, the ISH module v2.2 within the HALO image analysis platform (v2.0.1145.19, Indica Labs) was used. The output from this platform reflects a cell-by-cell quantitative result reported as an H-score as previously described. 34 Briefly, optimal density of the nuclear stain and positive signal stain color were specified and set manually until reaching the representative color value. Individual cells in the image were circled and separated by adjusting a series of input data including nuclear size (6.5-127.7 µm for IL-β, 6.875-114.5 µm for TGF-β), nuclear segmentation aggressiveness (1 for IL-β, 0.94 for TGF-β), cell radius from nuclei (2.37 µm for IL-β, 1.65 µm for TGF-β), and so on. Once an ideal output could reflect the corresponding signals and separated individual cells, all pictures were analyzed using the module algorithm and a single H-score was obtained for each image, reflecting the overall target expression on a scale of 0 to 400. 34
Statistical Analysis
Statistical analysis of the histopathology, histochemistry, immunohistochemistry, and RNA ISH data was performed using commercial statistical software packages R (version 4.0.2, R Foundation for Statistical Computing) and GraphPad Prism 6 (GraphPad Software). Results are expressed as median with interquartile ranges (IQR). Linear models considering pathogen, diet, and the interaction effect were used to detect statistical differences in neutrophilic inflammation, crypt depth, area quantification of immunohistochemical/histochemical staining, and the H-score of RNA ISH. In the linear model, the pathogen effect was assessed by comparing the responses between the control group and the Bhyo group, while the diet effect was obtained by comparing the responses between the control group and the DDGS group. The heteroscedasticities of the linear models were examined, and the datasets did not violate the assumption. When the overall F-test showed significance, further effect tests of diet, pathogen, and interaction were examined and presented. Correlation analysis was performed in several data pairs, between IL-1β and neutrophils, IL-1β and MUC5AC, IL-1β and MUC2, and MUC5AC and sialomucin, comparing the values from all of the animals and using the Pearson correlation test. The analysis was built out of the residuals of the models so the fixed effects for diet and treatment have been removed. Results were shown as a correlation coefficient (R) with 95% confidence interval (CI). Statistical significance was set as P < .05.
Results
Histopathology
Neutrophil count and crypt depth both were significantly different between groups (P < .05, overall F-test, linear model). At the apex of the spiral colon there was a significant increase in neutrophilic inflammation between the Bhyo group and the control group (P = .0004, Table 1). There was no statistically significant effect of diet on the neutrophil count (P = .95) and there was no interaction between diet and inoculum (P = .10). Crypt depth data of the Bhyo group were significantly greater than the control group (P < .0001, Table 1). There was no statistically significant effect of diet on the crypt depth (P = .48) and there was no interaction between diet and inoculum (P = .07).
Histopathologic Findings in Pigs With and Without Swine Dysentery After Brachyspira hyodysenteriae Infection and Fed a Diet Containing Either No or 30% Corn Distillers Dried Grains With Solubles (DDGS).a
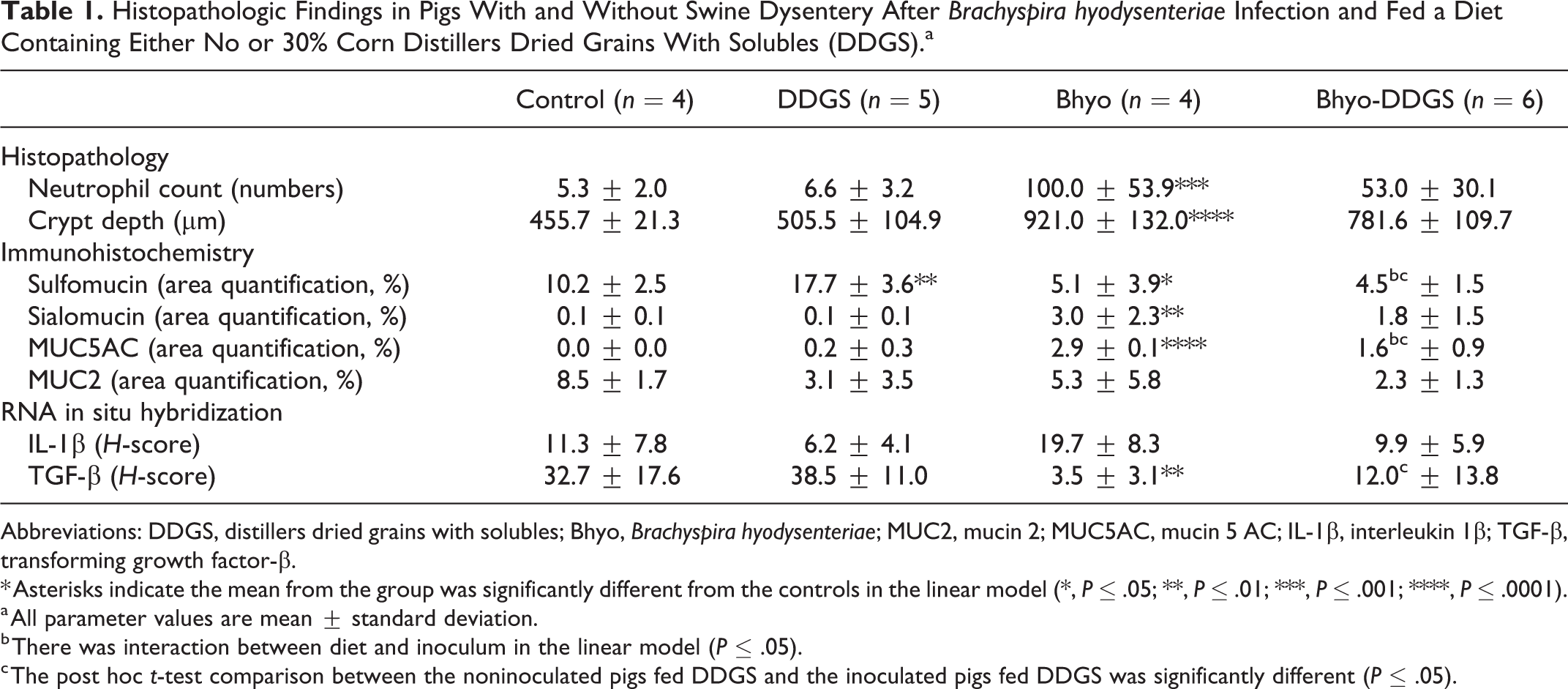
Abbreviations: DDGS, distillers dried grains with solubles; Bhyo, Brachyspira hyodysenteriae; MUC2, mucin 2; MUC5AC, mucin 5 AC; IL-1β, interleukin 1β; TGF-β, transforming growth factor-β.
* Asterisks indicate the mean from the group was significantly different from the controls in the linear model (*, P ≤ .05; **, P ≤ .01; ***, P ≤ .001; ****, P ≤ .0001).
a All parameter values are mean ± standard deviation.
b There was interaction between diet and inoculum in the linear model (P ≤ .05).
c The post hoc t-test comparison between the noninoculated pigs fed DDGS and the inoculated pigs fed DDGS was significantly different (P ≤ .05).
Histochemistry
Histochemical staining of sulfomucin and sialomucin both were significantly different between the 4 groups (P < .05, overall F-test, linear model). The amount of sulfomucin at the apex of the spiral colon was significantly decreased in the Bhyo group with histologic lesions consistent with SD compared to the control group (P = .027), and significantly increased in the pigs fed DDGS (P = .002) compared to pigs fed normal diet (Figs. 1, 3, Table 1). There was significantly lower expression of sulfomucin in the Bhyo-DDGS group than in the DDGS group (P < .0001, post hoc t-test). There was also a diet and inoculum interaction (P = .01). There was a significant increase in the amount of sialomucin (P = .008) in the Bhyo group compared to the control group (Figs. 2, 4, Table 1). There was no statistically significant effect of diet on the expression of sialomucin (P = .98) and there was no interaction between diet and inoculum (P = .35).
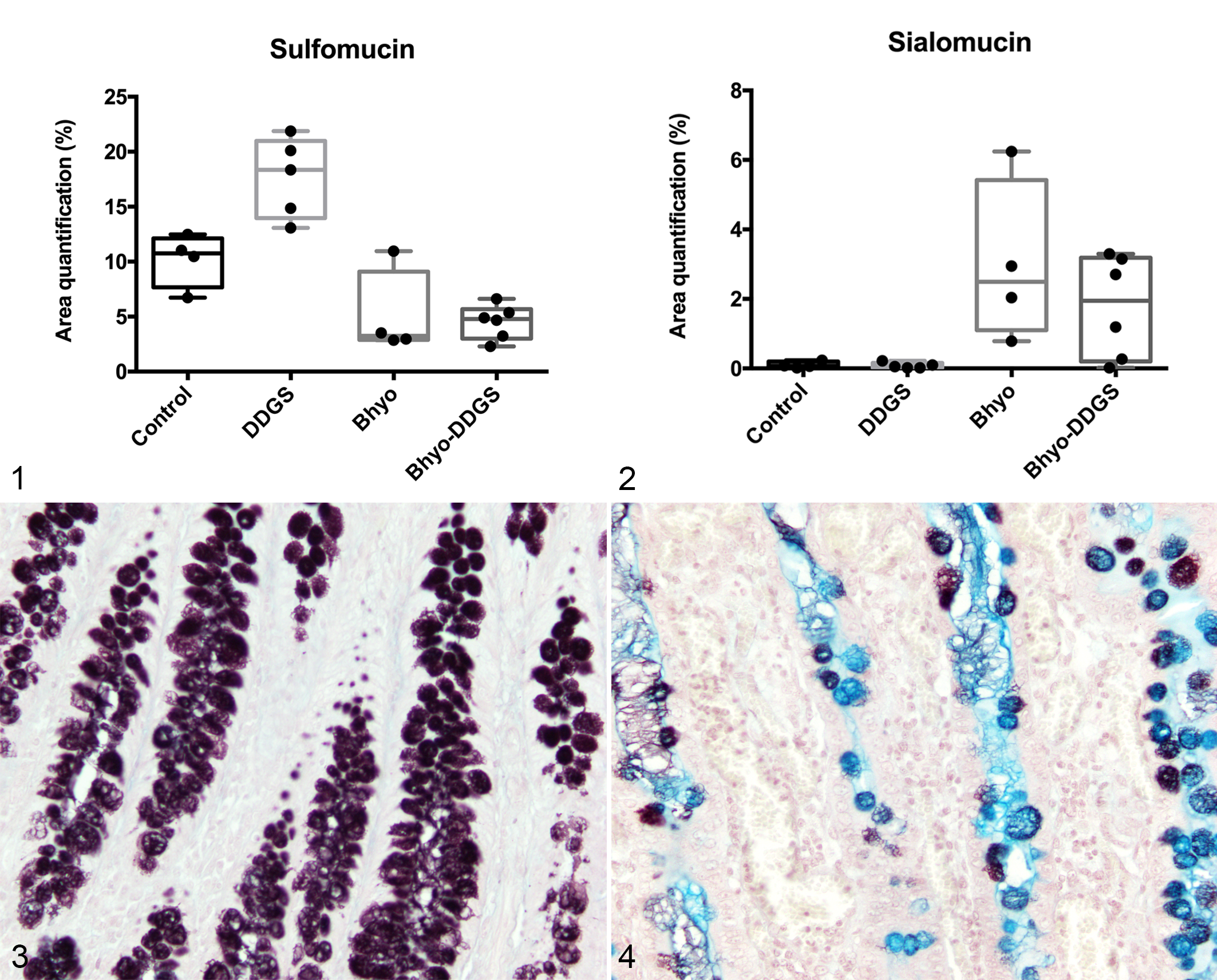
Area quantification of high-iron diamine–Alcian blue (HID-AB) staining in sections of porcine spiral colon. Results reflect the percentage of positive staining in the region of interest for control pigs, pigs fed distiller’s dried grains with solubles (DDGS), pigs inoculated with Brachyspira hyodysenteriae (Bhyo), and pigs with Bhyo and fed DDGS (Bhyo-DDGS). The data are represented as box plots showing all data points. Linear models considering pathogen, diet, and the interaction effect are used. Immunolabeling area of sulfomucin is significantly lower (P = .027) while sialomucin is significantly higher (P = .008) in the Bhyo group, which had histologic lesions consistent with swine dysentery. Significantly increased sulfomucin is also observed in non-inoculated pigs fed DDGS compared to pigs fed the control diet (P = .002) and there is a diet × inoculum interaction (P = .01).
Immunohistochemistry
MUC5AC expression was significantly different between the 4 groups (P < .05, F-test, linear model). MUC5AC expression was higher in the Bhyo group compared to the control group based on area quantification (%; P < .0001, Figs. 5, 6, Table 1). There was no statistically significant effect of diet on MUC5AC expression (P = .569), while there was an interaction between diet and inoculum (P = .007). There was significantly higher expression of MUC5AC in the Bhyo-DDGS compared to the DDGS group (P = .0004, post hoc t-test, Table 1). MUC2 expression was not significantly different between the 4 groups (P = .0556, F-test, linear model, Figs. 8–10). There was no significant correlation between the expression of MUC5AC and sialomucin (R = 0.045; 95% confidence interval [CI] = −0.4178, 0.4892; P = .8549).
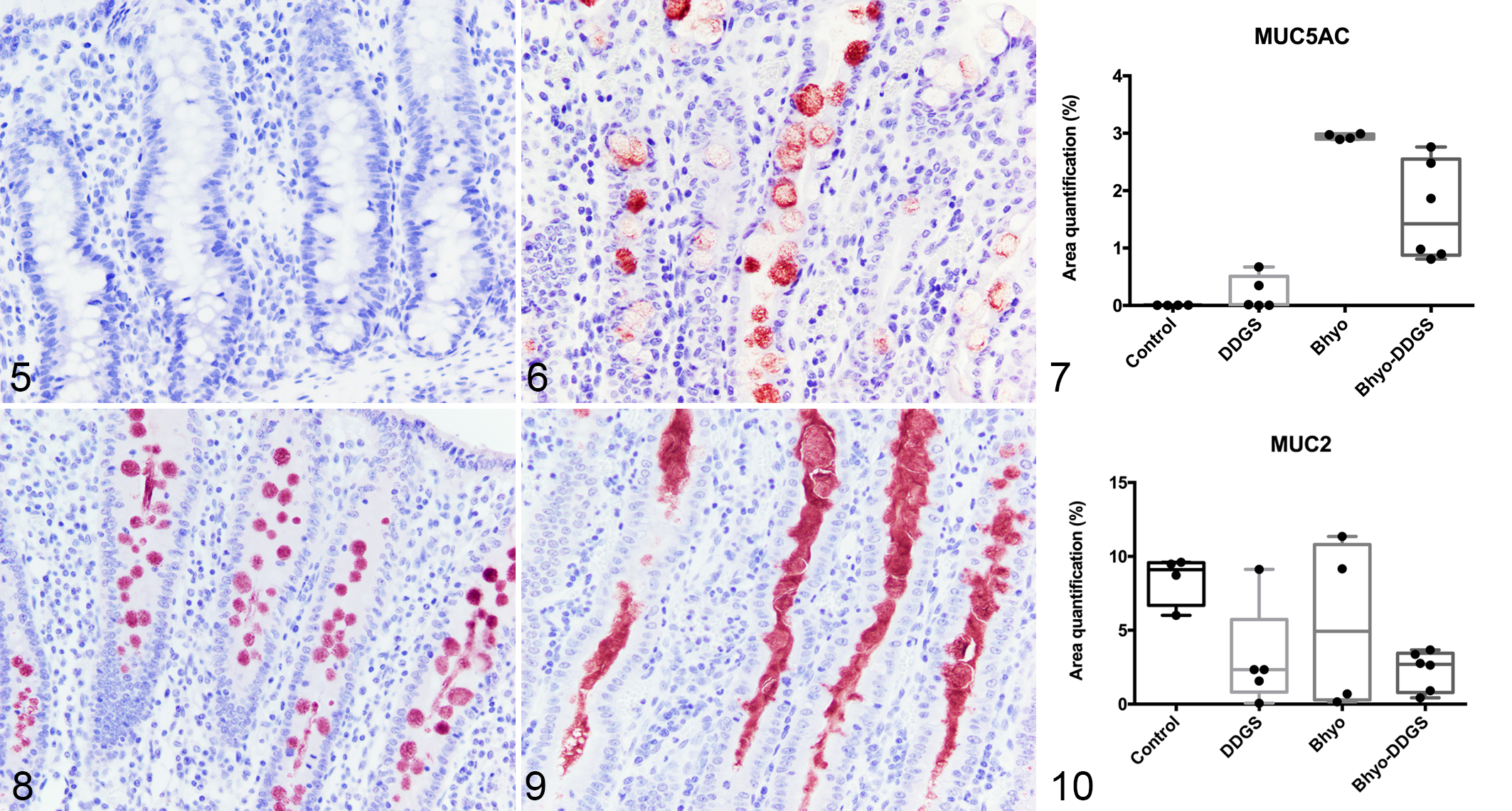
Colon, pig. Immunohistochemistry (IHC) for mucin 5 AC (MUC5AC).
RNA In Situ Hybridization
IL-1β was not significantly different between the Bhyo group and the control group, but had a trend toward higher expression levels in the Bhyo group (P = .086, Figs. 11–13, Table 1). There was no statistically significant effect of diet on the expression of IL-1β (P = .25), and there was no interaction between diet and inoculum (P = .45). Bhyo group showed significantly lower TGF-β expression based on the H-score (P = .005, Figs. 14–16, Table 1). There was no statistically significant effect of diet on TGF-β expression (P = .50) and there was no interaction between diet and inoculum (P = .82). The Bhyo-DDGS group showed significantly lower expression of TGF-β compared to the DDGS group (P = .004, post hoc t-test, Table 1). Neutrophil counts moderately correlated with IL-1β but without statistical significance (R = 0.4268, 95% CI = −0.0341 to 0.7379, P = .07). There were no correlation between the expression of IL-1β and MUC5AC (R = 0.27, 95% CI = −0.21 to 0.65, P = .3), and IL-1β and MUC2 (R = −0.22, 95% CI = −0.61 to 0.26, P = .36).
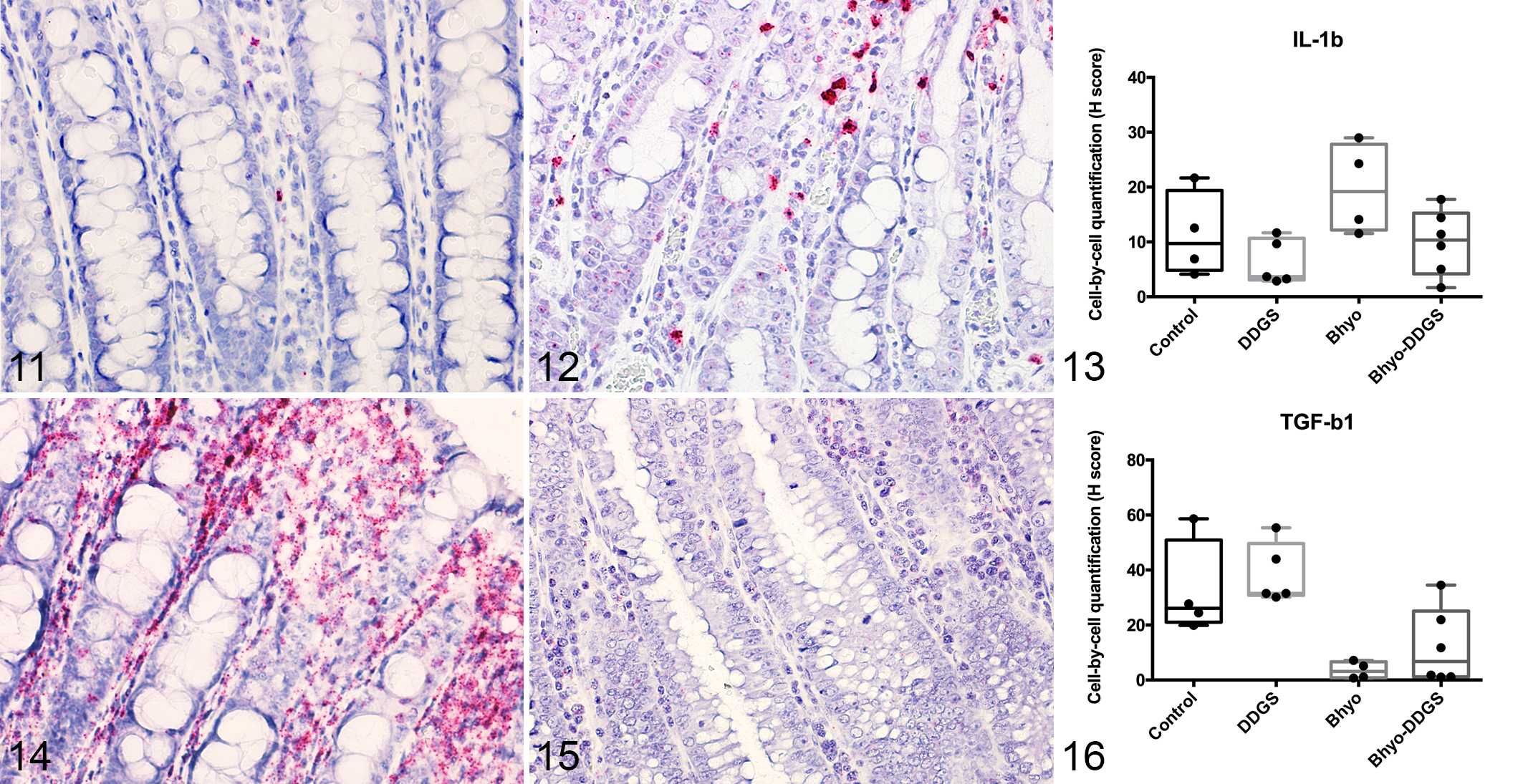
Colon, pig. RNA in situ hybridization (ISH) for IL-1β.
Discussion
The present study characterized alterations of colonic mucin composition and cytokine expression in pigs with experimentally induced acute SD using mucin histochemistry, IHC, and RNA ISH. At the apex of the spiral colon, there was a significant neutrophilic colitis with crypt hyperplasia in pigs inoculated with Bhyo, which are typical morphological lesions of acute SD. The histochemical staining of HID-AB revealed a significant increase of sialomucin accompanied by a significant decrease of sulfomucin in pigs that developed acute SD. Meanwhile, sham-inoculated pigs fed DDGS expressed more sulfomucin in the colon. Immunohistochemically, there was marked de novo expression of MUC5AC in pigs with SD, as expected, 62,81 while MUC2 expression was not significantly different between groups. RNA ISH showed significantly lower expression of TGF-β1 in SD pigs compared with controls and IL-1β expression was often increased but did not reach statistical significance.
SD is characterized by mucohemorrhagic diarrhea and histologically there is extensive mucus production in the colon, accompanied by goblet cell hyperplasia, elongated hyperplastic crypts, mucosal erosions, ulceration, and hemorrhage, as well as locally extensive infiltration of neutrophils and lymphocytes. 7,28 While normally being perceived as a protective barrier in the gastrointestinal tract, the mucus layer has been shown to be a potential niche with increased binding sites for Brachyspira hyodysenteriae. 62 Epithelial mucins are the main component of the mucus layer and are a large family of O-glycosylated glycoprotein macromolecules associated with mucin-encoding genes. 11,48 MUC2 and MUC5AC both belong to the subclass of secreted gel-forming mucins; the former is constitutively expressed throughout the small and large intestine in healthy pigs, 3,38,62 while MUC5AC is constitutively expressed in the respiratory tract, stomach, and gallbladder of pigs but not in the colon of healthy pigs. 37,75 In our study, the expression of MUC5AC in the colonic tissues was significantly greater in pigs with acute SD compared to the uninfected control groups regardless of diet. This result is consistent with previous studies demonstrating increased expression of MUC5AC in pigs with SD in both protein 81 and mRNA levels, 62 depending on the stage of the disease.
To the authors’ knowledge, immunohistochemical quantification of MUC2 expression in pigs with SD has not been previously reported. MUC2 is one of the most abundant mucins throughout the intestine and serves as a barrier between the colonic epithelial cells and bacteria. 33,48 The expression of MUC2 in this study was not statistically different between the 4 groups. Nonetheless, we found the location of MUC2 staining did differ in some sections. MUC2 was commonly present in crypt lumens and on the surface of the epithelium of pigs with SD regardless of diet, while the majority of MUC2 in the control group was present within goblet cells and remained in the epithelium. In a previous study, the authors performed quantitative PCR (polymerase chain reaction) and found substantially increased MUC2 mRNA levels in pigs with SD. 62 They also performed immunofluorescence that showed increased expression of MUC2 in colonic tissues but without quantification. Our analysis is limited as it was based on quantification of expression in the crypt and crypt epithelium, but did not include the mucosal surface area in which there was considerable immunolabeling in the SD group. It is likely that a large amount of MUC2 has been secreted into the intestinal lumen and some secreted mucin may have been lost during tissue processing. Moreover, decreased MUC2 expression has been reported in some other bacterial infections such as Lawsonia intracellularis, which results in altered mucosal integrity and thereby enabling cellular invasion. 3,70 Additional investigation with modifications in sample fixation and processing, as well as the image analysis parameters, may be useful to further explore and compare the expression of MUC2 in SD.
A broad classification of epithelial mucins separates mucins into neutral and acidic chemotypes, and further categorizes them into sialomucins or sulfomucins based on the presence of terminal sialic acid or sulfate groups on the oligosaccharide chain. 16,72 The traditional histochemical technique of HID-AB staining clearly demonstrated the 2 kinds of acidomucin. 72 We found the expression of sialomucin in pigs with SD was significantly higher compared to the control group, while expression of sulfomucin was significantly lower. The latter finding is consistent with previous studies showing a reduction of sulfomucin in pigs with SD using HID staining 81 and decreased levels of sulfated mucin in SD pigs using porous graphitized carbon liquid chromatography tandem mass spectrometry to analyze the mucin component. 78 On the other hand, the sialomucin changes in pigs with SD using HID-AB staining have not been previously reported. Changes from a predominance of sulfated mucin in healthy intestine to increased sialomucin and decreased sulfomucin expressions has been reported in humans with colorectal cancer or inflammatory bowel diseases. 12,14,16,65 The decreased sulfated mucin is associated with mucin degradation and probably influenced by bacterial sulfatases, leading to a disruption of the protective mucin layer. 12,14,16,65 The decrease of sulfated mucin is also likely related to sulfate reducing bacteria, such as Desulfovibrio spp., which have been found to significantly increase in the mucosa-associated microbiota of pigs with SD. 9 Meanwhile, our finding of increased sialomucin expression is consistent with a recent study showing that B. hyodysenteriae might utilize sialic acid for growth and as an epitope for bacterial adhesion. 64 The increased sialomucin in pigs with SD might provide more binding sites for these bacteria. In previous studies, it has been shown that Helicobacter pylori colonizes the gastric mucosa by adhesion to the blood group antigen LeB that is present on gastric mucin MUC5AC. 47,76,77 Increased biosynthesis of those sialylated Lewis glycan structures during H. pylori infection indicates modulation of glycosylation by the pathogen. 49,50 However, there was no direct correlation between the quantitative expression of MUC5AC and sialomucin in our study. Further investigation is needed to explore the mucin binding sites for B. hyodysenteriae, the specific mucin moieties involved, and the modulation mechanism.
Dietary fibers have great impact on gut epithelium and mucus production, which may directly, or indirectly by intestinal microbiota, change the intestinal environment and the susceptibility to infections. 54,55 DDGS, a coproduct of biofuel production, have been increasingly added to commercial swine diets in recent decades and have been found to enhance the disease expression of SD through alteration of the colonic microbial profiles of pigs. 8 In our study, we found that feeding pigs DDGS would increase the expression of sulfomucin, and lower the extent of increased MUC5AC expression in pigs with SD, while there was no statistically significant dietary effect on the expression of sialomucin, IL-1β, and TGFβ. The loss of sulfated residues on mucins has been linked to the first step in degradation of mucin, which provides a nutrient source for growth of niche-specific microbiota and enable bacteria to reach the underlying epithelium. 23,67 It is thus not surprising that pigs with SD showed less sulfated mucin. 8,81 On the other hand, the finding of increased sulfomucin expression in control pigs fed DDGS is unexpected given that pigs fed 30% DDGS develop SD faster than pigs fed a control diet. 80 Feeding pigs DDGS in this study also seemed to lower the extent of increased MUC5AC expression in pigs with SD, and further investigation is needed to clarify the effects of feeding DDGS and mucin expression in the spiral colon of pigs.
We originally hypothesized that pigs with SD may show increased expression of IL-1β in local colonic tissue, which may participate in the local regulation of increased and altered secretion of MUC2 and MUC5AC. Our results showed a mild tendency (but without statistical significance) of increased IL-1β in pigs with SD compared to the uninfected control group. IL-1β is a pro-inflammatory cytokine that is produced by macrophages, epithelial cells, and various other cells during local and systemic response to injury, infection, and inflammation. 57 Its expression is higher in patients with inflammatory bowel diseases, while the regulator IL-1 receptor antagonist (IL-1 ra) is lower in the plasma of the same patients, hence with a decrease of the IL-1 ra: IL-1 ratio. 20,25,46,68 A previous study also showed pigs with SD had an increased level of IL-1β systemically during early disease onset, 43 and there was an upregulation of IL-1β mRNA level in the colonic epithelial cell culture that was incubated with Brachyspira pilosicoli sonicates. 59 However, information regarding local/mucosal expression of IL-1β in pigs with SD is lacking. IL-1β mRNA levels in porcine colonic tissues were increased in 2 out of 5 pigs infected with B. hyodysenteriae; however, these results were not statistically significant. 63 In our study, IL-1β mRNA signals were intense and prominent in the colonic tissues of many pigs with SD, but a few of them showed relatively low expression and several control cases also presented multifocal positivity of IL-1β. It is possible that the local immune status may be slightly different from the systemic cytokine response, or the pigs with SD were at different stages of disease resulting in a wider range of IL-1β expressions. The moderate agreement between IL-1β and neutrophils indicates that some cases showed less of an acute inflammatory response. Furthermore, since a variety of causes may contribute to the upregulation of IL-1β, the higher expression in a limited number of the control pigs may be a result of some other underlying stimulation.
In human airway diseases or colorectal cancers, the upregulation of IL-1β has been reported to increase MUC5AC and MUC2 secretion. Previous studies found increased mRNA and protein abundance of MUC5AC and/or MUC2 in lung or intestinal epithelial cells after activation by IL-1β through various molecular signaling pathways. 10,21,24,39 –41 However, different from our hypothesis that IL-1β may be directly involved in the regulation of MUC2 and MUC5AC, we did not find any correlation between IL-1β and the 2 mucin proteins. Only IL-1β and MUC5AC showed fair agreement but without significance. While most of the previous studies came from human diseases and cell cultures, it is possible that the mechanism of mucin alteration in pigs with SD may be slightly different. Also, our finding is in line with the aforementioned study which demonstrated that the increased MUC2 and de novo expression of MUC5AC are correlated with upregulation of neutrophil elastase and IL-17A, but not with IL-1β. 63 Since mucin alteration is multifactorial and may not only be due to IL-1β, more studies are needed to determine the mechanisms in SD.
Regulation of the immune response in SD is seldom discussed. 7,79 While an increased level of IL-10 was found during the recovery phase of pigs infected with B. hyodysenteriae, 43 information about the other suppressor cytokine, TGF-β1, is lacking. TGF-β1 is a multifunctional cytokine produced by various immune and nonimmune cells that regulates cell growth, differentiation, and has an immunosuppressive activity. 18,19,44 In the intestine, the major sources of TGF-β1 are dendritic cells (DCs) and intestinal epithelial cells. 1,35 As expected, pigs with SD in our study had significantly lower levels of TGF-β1 regardless of diet. On the contrary, the diffuse positivity in the control groups is mostly absent in the pigs with SD. This is consistent with previous studies showing that TGF-β1 serves as a negative regulator of mucosal inflammation both in vitro and in vivo, which plays a significant role in the pathogenesis of inflammatory bowel diseases such as ulcerative colitis and Crohn’s disease. 6,27,45,56 TGF-β1 is also reported to serve as a complex regulator between gut microbiota and immune cell regulation. 1,5,35 Previous reports found that the complex mixture of Clostridium strains from cluster IV and XIVa and the probiotic strain Clostridium butyricum provided a TGF-β1-rich environment that increased induced regulatory T cells (iTreg) in the colon. 1,35 Other microbiota members such as species belonging to the Firmicutes and Fusobacteria phyla are also reported to be the potent inducers of TGF-β1 in vitro. The metabolites derived from commensal bacteria may modulate the expression of TGF-β1. 51 Pigs affected by SD in a previous study showed a significantly higher Firmicutes: Bacteroidetes ratio and higher abundance of Fusobacteriales and Clostridiales in pigs with SD in the luminal contents relative to those without disease, 9 which in theory might induce a higher TGF-β environment. However, despite the changes of the microbiota mentioned above, pigs with experimentally induced SD in this study had lower expression of TGF-β. Further studies are needed to elucidate the role of TGF-β in the pathogenesis of SD and the mechanisms underlying its suppression.
Our findings showed that pigs with experimentally induced SD had a significant neutrophilic colitis with significant crypt hyperplasia, accompanied with altered mucin composition and cytokine expression. The markedly increased expression of sialomucin in pigs with SD suggests that sialylated moieties may provide increased binding sites for Brachyspira spp. While feeding DDGS increased sulfated mucin in control pigs, decreased expression was observed in pigs with SD which suggests that in the presence of microbial sulfatase activity this may provide an additional nutrient source for bacterial growth. The pro-inflammatory cytokine IL-1β was not significantly increased in pigs with SD and thus may not be a major regulator of MUC5AC and MUC2 secretion in acute SD unlike its role in other diseases. We also found that the expression of the immune-regulatory cytokine TGF-β was significantly suppressed in pigs with SD. Taken together this study provides insight into mechanisms of mucin alteration and inflammation in the pathogenesis of SD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
