Abstract
Pneumonia has been reported in both free-ranging and captive koalas and a number of causative agents have been described. Between 2016 and 2019, 16 free-ranging and 1 captive koala (Phascolarctos cinereus) from the Mount Lofty Ranges of South Australia were identified with pyogranulomatous lobar pneumonia, which involved the left caudal lobe in 14/17 (82%) cases. Within lesions, numerous gram-positive or gram-variable, non-acid-fast filamentous bacteria were observed in association with Splendore-Hoeppli phenomenon. Culture yielded growth of anaerobic bacteria, which were unidentifiable by MALDI-TOF-MS (matrix-assisted laser desorption ionization-time of flight mass spectrometry) analysis in 5/5 cases. Sequencing of the bacterial 16S rRNA gene identified a novel Actinomyces species in 4 samples, confirming a diagnosis of pulmonary actinomycosis. Concurrent examination of resin lung casts from healthy koalas suggested greater laminar flow of air to the left caudal lung lobe in koalas. Actinomyces spp. have been reported as commensals of the oral microbiome in other species, and an association with similar pulmonary lesions in other species. Considering the predilection for involvement of the left caudal lung lobe, aspiration is suggested as the likely cause in some cases of pulmonary actinomycosis in koalas. Pulmonary actinomycosis has not been previously described in koalas and further work needs to be undertaken in order to classify this organism within the Actinomyces genus.
Pneumonia has been reported in both free-ranging and captive koalas with varying prevalence between the states of Australia, ranging from 0% (n = 44) in free-ranging koalas of Victoria, 43 4% (3/85) in free-ranging koalas in South Australia, 54 to 21% (n = 28) in captive koalas in New South Wales. 3
Pathogens reported to cause pneumonia in koalas include Bordetella bronchiseptica, 14,39 Chlamydia spp., 9,17,35 and Cryptococcus gattii (previously Cryptococcus neoformans var. gattii). 14,27 B. bronchiseptica has been identified in outbreaks of pneumonia within captive koala colonies in Queensland 39 and New South Wales, 13,14 either as a primary pathogen or secondary to other diseases such as cryptococcosis or chlamydiosis. 14 Association of Chlamydia spp. (C. pecorum, C. pneumoniae) with a rhinitis/pneumonia complex of koalas has been well documented. 11,46 In 1984, when C. pecorum was categorized as C. psittaci, 1 in 12 koalas diagnosed with rhinitis-pneumonia complex had C. psittaci cultured from nasal samples. 8 However, chlamydial pneumonia had not been definitively reported as a primary agent until recently when Mackie et al identified C. pecorum as the cause of pneumonia in a juvenile koala. 35 Cryptococcus gattii can cause an extensive fungal pneumonia, which can disseminate to other tissues, including the central nervous system. 27 Cryptococcal infections have been reported in koalas in Queensland 39 and New South Wales, 13,27 but not in Victoria or South Australia. Other documented infections associated with pneumonia in koalas include Pseudomonas aeruginosa, 13,44 Nocardia asteroides, 57 Staphylococcus epidermidis, 57 Staphylococcus aureus, 8 Mycobacterium ulcerans, 41 α-hemolytic Streptococcus spp., 8 Corynebacterium sp. and other unidentified diptheroids, 8 Pasteurella spp., 8 Acinetobacter lwoffii, 8 Aspergillus spp., 8 and a parasitic pneumonia associated with Marsupostrongylus sp. 38
This study describes the gross and histopathological features and the microbiological investigations of the first reported cases of pulmonary actinomycosis in koalas and proposes its pathogenesis and significance.
Materials and Methods
Routine postmortem examinations were conducted between October 2016 and August 2019 on free-ranging and captive koalas from the Mount Lofty Ranges and surrounding suburbs of Adelaide, South Australia. Koalas were euthanized based on veterinary decision, due to clinical findings indicating a poor prognosis for rehabilitation. Bodies were submitted to the University of Adelaide as part of a broader study into koala pathology (Adelaide University Animal Ethics Committee approval S-2016-169). Sex, tooth wear class (TWC) as an estimate of age, 37 and body condition score (BCS) 9 of koalas were determined. From the broader study, cases with grossly evident lung lesions were identified. Representative tissue samples were placed in 10% buffered formalin for histopathological investigation. When postmortem examinations were performed within 24 hours of death, Amies dry swabs (Copan Italia) were taken from lung lesions for microbiological culture. Blood samples taken during clinical assessment or postmortem spleen samples were collected for koala retrovirus analysis.
Histopathology
Tissues were processed routinely, embedded in paraffin, and sections cut at 4 μm. Slides were stained with hematoxylin and eosin (17/17), Gram (17/17), periodic acid–Schiff (PAS; 17/17), modified Ziehl-Neelsen (15/17), Alcian blue (13/17), and Giemsa (13/17). Formalin-fixed paraffin-embedded (FFPE) tissue from animals that had undergone advanced autolysis (n = 4) were not subjected to all special stains.
Microbiological Investigations
Swabs of lung lesions (5/17) were initially plated on sheep blood agar and MacConkey agar plates (Thermo Scientific) and grown both aerobically and anaerobically. No koalas that had microbiological investigations performed had received antimicrobial therapy prior to euthanasia. Unidentified anaerobic colonies were subcultured for purity and identification on anaerobic agar (Thermo Scientific) in aerobic and anaerobic conditions, as well as on actinomyces agar (Thermo Scientific) in anaerobic conditions. Anaerobic conditions were achieved by placing AnaeroGen 2.5 L pack (Thermo Scientific) and CampyGen 2.5 L pack (Thermo Scientific) in a sealed culture jar with an anaerobic indicator and incubated at 37.0 °C for 48 to 72 hours. The isolates were analyzed by MALDI-TOF-MS (matrix-assisted laser desorption ionization-time of flight mass spectrometry). Isolates that were not identifiable by MALDI-TOF-MS analysis or not speciated to a previously identified commensal of the marsupial oral microbiome or previously known respiratory pathogen were further investigated by PCR (polymerase chain reaction) for 16S rRNA. DNA was extracted from pure cultures using QIAMP DNAEasy Minikit (Qiagen) as per manufacturer’s instructions. Conventional PCR was run on 8 samples with primers to the 16S rRNA sequence. 58 Purified PCR products were sent for Sanger Sequencing at Australian Genome Research Facility, and BLAST (NCBI) was used to align the sequence to any known bacteria.
Resin Study
In order to assist the understanding of lesion distribution, resin studies of bronchial anatomy were undertaken in 4 koala cadavers, acquired following euthanasia due to non-respiratory morbidities. Tracheal and bronchial washes were carried out with heparinized saline (3×) and then water until the wash ran clear. Epoxy resin (Barnes) was syringed into the airways in situ via a transverse incision through the cervical trachea and allowed to cure overnight with the koala in an upright position. The lungs were removed from the body and the tissue digested in a 5% w/v sodium hydroxide bath. A rotary tool (Dremel) was used to isolate the bifurcation of the trachea and the branching of the lobar bronchi from the left and right primary bronchi in the resin cast.
PCR Analyses
DNA was extracted from either whole blood collected antemortem (when available) or spleen using QIAMP DNAEasy Minikit (Qiagen) as per manufacturer’s instructions for koala retrovirus PCR analysis. DNA was extracted from affected FFPE lung tissue using QIAMP DNA FFPE Tissue Kit (Qiagen) as per manufacturer’s instructions for Chlamydia pecorum PCR analysis. All extracted DNA was measured on NanoDrop One spectrophotometer (Thermo Fisher Scientific) and then stored at −20 °C until testing. The 20 µL PCR reaction consisted of 4 µL AllTaq Mastermix (Qiagen), 3 µL of 1/10 dilution DNA template, and 10 µL of PCR-grade water. For detection of koala retrovirus proviral DNA 0.5 μM of forward primer 5′-TCCTGGGAACTGGAAAAGAC-3′ and 0.5 μM of reverse primer 5′-GGGTTCCCCAAGTGATCTG-3′ 56 was added (321 bp amplicon). For detection of C. pecorum OMP gene, 1 μM of forward primer 5′-CCAAGCATAATCGTAACAA-3′ and 1 μM of reverse primer 5′-CGAAGCAAGATTCTTGTC-3′ 24 was added (140 bp amplicon). PCR conditions were the following: initial activation and denaturation of 95 °C for 2 minutes, followed by 40 cycles of denaturation at 95 °C for 5 seconds, annealing at 60 °C for 15 seconds, and extension at 72 °C for 10 seconds. This was followed by a final extension step of 72 °C for 10 seconds. Electrophoresis was used to separate the PCR product on 1.0% agarose gels with samples loaded in combination with gel red, visualized with UV light.
Statistical Analysis
Chi-squared analyses were carried out in Microsoft Excel v. 16.31 to find any interactions of sex, TWC, BCS, koala retrovirus or C. pecorum status with pulmonary actinomycosis.
Results
Gross Pathology Findings
Nine male and 8 female koalas were diagnosed with lobar pneumonia (Table 1); there was no statistical association with sex (P = .81). There were 5 young adult koalas (TWC II/III), 9 adult koalas (TWC IV/V), and 3 aged koalas (TWC VI/VII). No juvenile koalas were represented. Older koalas represented 12/17 (71%) of affected koalas (TWC ≥IV), although this was not statistically significant (P = .09). A low BCS (≤3) was found in 14/17 (82%) affected koalas (P = .01). Comorbidities were present in all koalas: 3 presented with additional lesions of the upper respiratory system including ulcerative stomatitis (n = 1) and fungal (candidiasis) glossitis (n = 2). Lesions consistent with urogenital chlamydiosis were evident in the majority of koalas (n = 14) and included mild-moderate ulcerative cystitis (n = 10), paraovarian cysts (n = 4), urethritis (n = 1), prostatitis (n = 1), conjunctivitis (n = 5), and mild “wet bottom” (pericloacal urine staining) (n = 3).
Case Details of Mount Lofty Ranges Koalas With Pyogranulomatous Lobar Pneumonia.
Abbreviations: TWC, tooth wear class; BCS, body condition score; KoRV, koala retrovirus; PCR, polymerase chain reaction; FFPE, formalin-fixed paraffin-embedded.
a Bold type denotes cases subjected to culture.
b Tooth wear class. 36
c Body Condition Score. 9
d KoRV proviral DNA PCR on whole blood or spleen tissue.
eChlamydia pecorum PCR on FFPE lung tissue.
f LCa = left caudal, LCr = left cranial, RCa = right caudal, RM = right middle, RCr = right cranial.
g Gram stain attributes of filamentous bacteria associated with Splendore-Hoeppli phenomena.
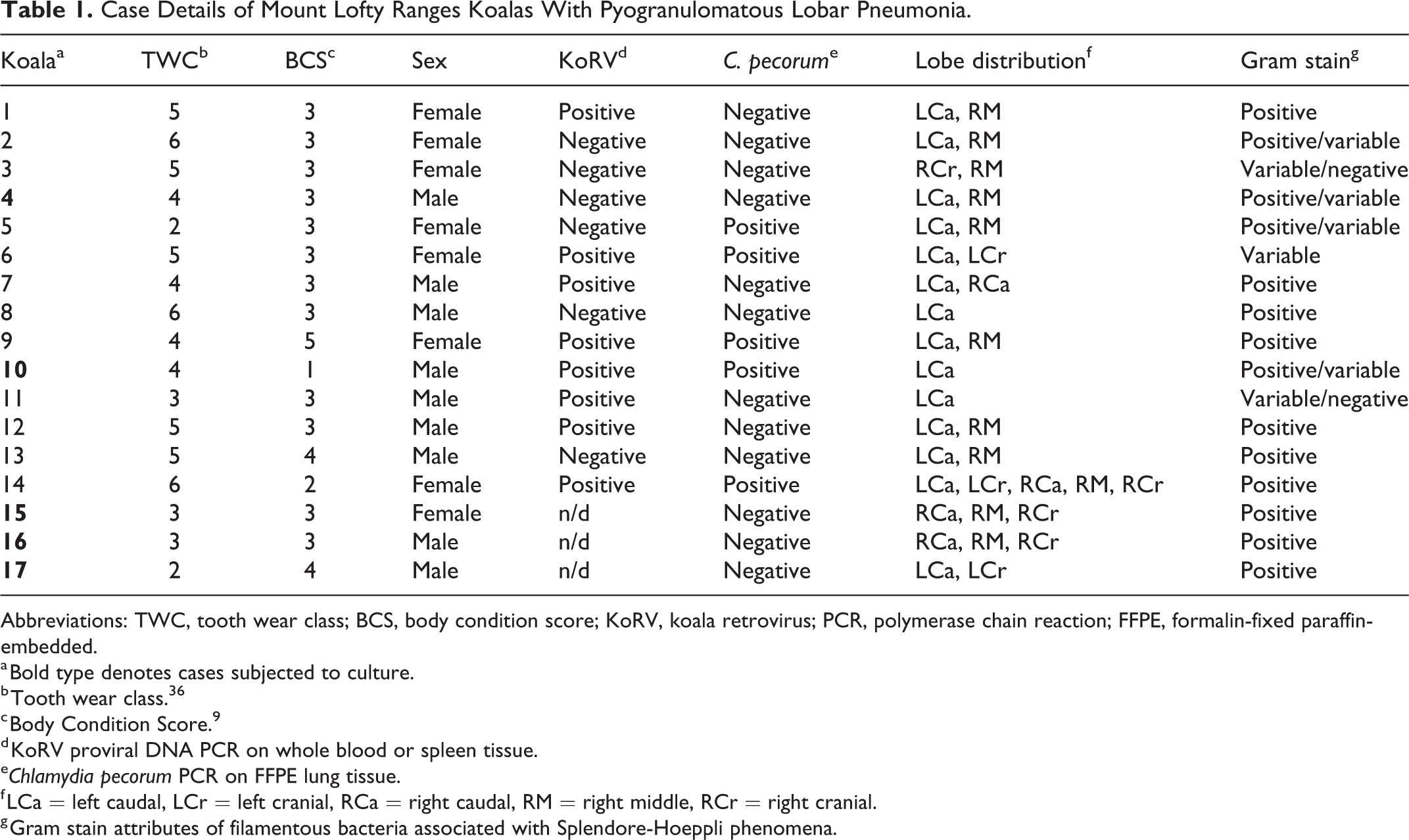
The lobar pneumonia involved the left caudal lung lobe in 14/17 (82%) of the affected koalas. Of these 14 cases, 3/14 had only the left caudal lung lobe affected, 6 had both the left caudal lung lobe and the right middle lung lobe predominantly involved (Fig. 1), and 5 had involvement of 2 or more lung lobes. Only 3/17 (18%) cases had no involvement of the left caudal lung lobe (Table 1).
Pulmonary actinomycosis, lung, koala, case 13. Ventral view, heart reflected cranially. There is lobar pneumonia predominantly affecting the left caudal lobe (white arrow) and right middle lobe (black arrow). The right caudal lobe (arrow head) is normal.
Pulmonary actinomycosis, lung, koala.
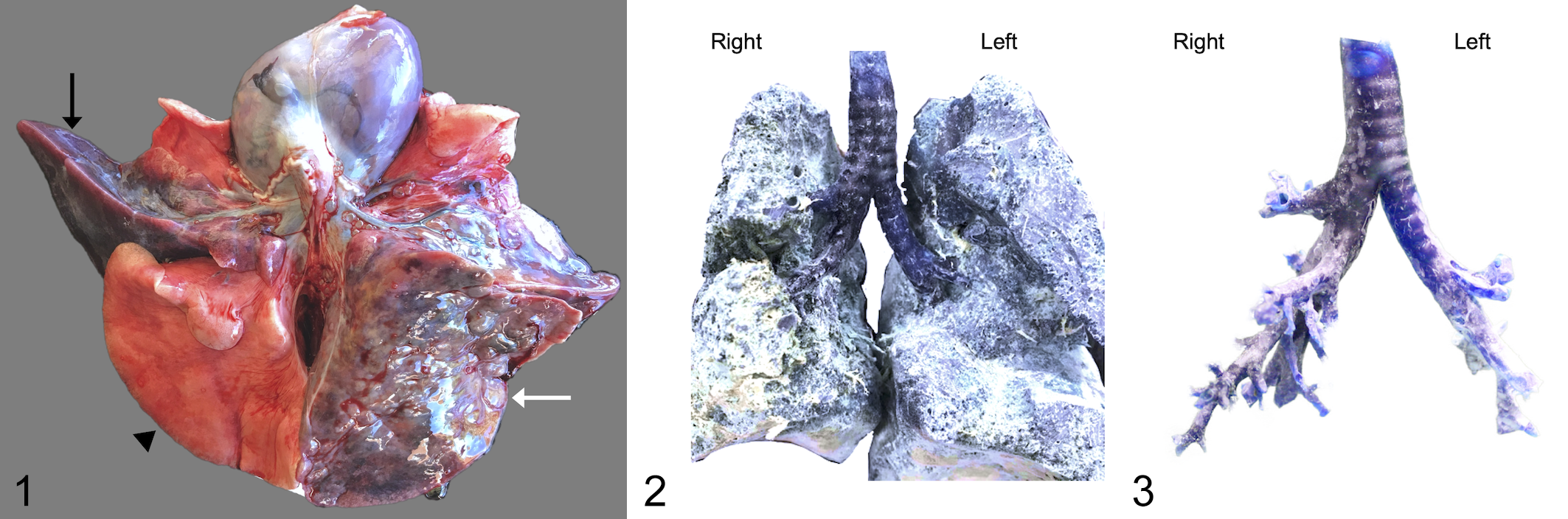
Resin Study
Resin casts demonstrated that the left primary bronchus followed a caudal path and did not branch until deep into the left lung. The right primary bronchus branched much earlier in a more lateral direction from the trachea, into lobar bronchi (Fig. 2 and Fig. 3).
Histopathology
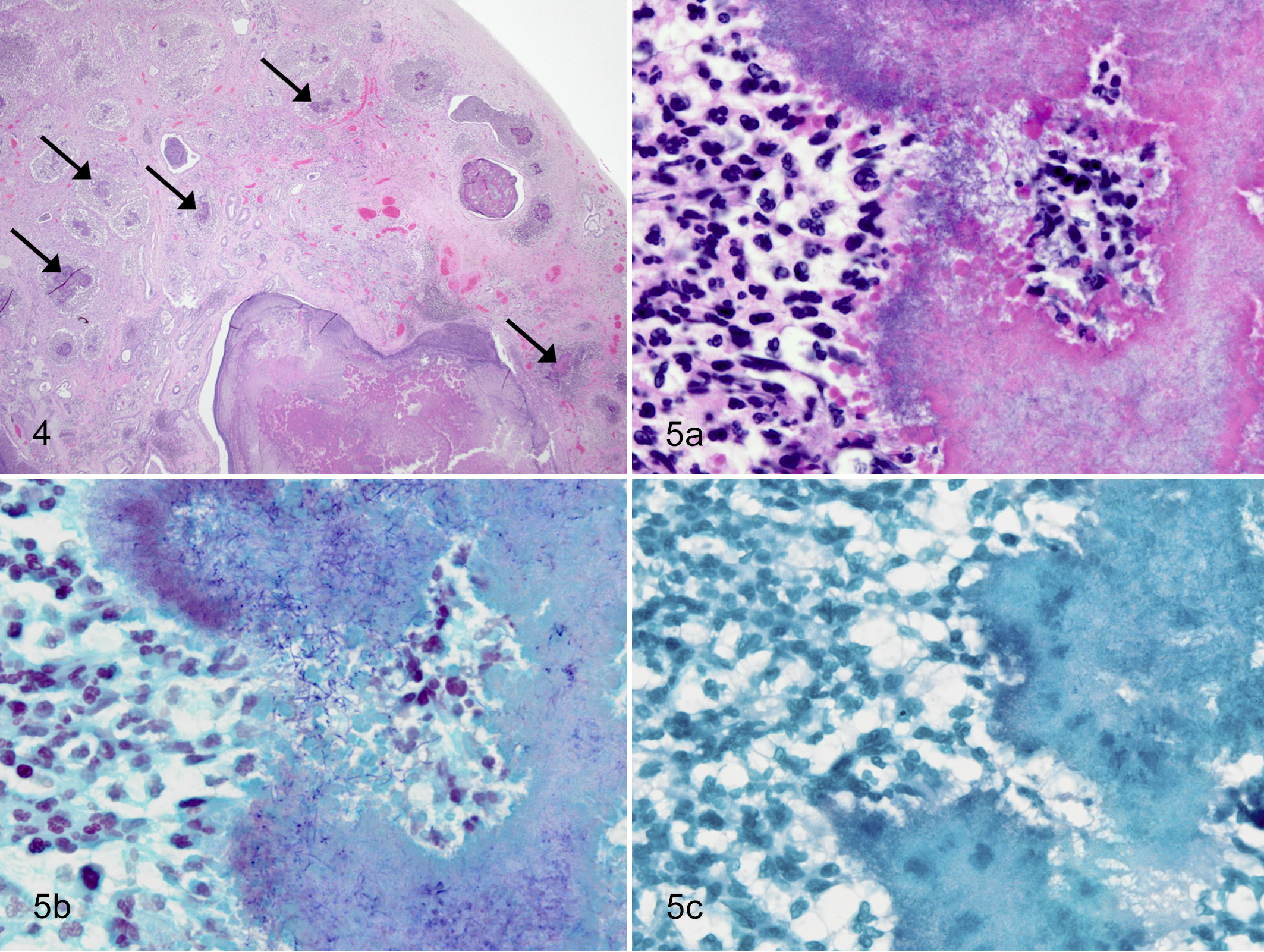
All pneumonias were characterized by a marked extensive chronic-active neutrophilic, histiocytic, and lymphocytic pneumonia (pyogranulomatous pneumonia), with variable multinucleate giant cell formation, necrosis, and fibrosis. There was concurrent intralesional Splendore-Hoeppli material with associated filamentous bacteria (Table 1 and Fig. 4 and Fig. 5). Most (16/17) cases had concurrent mild pleuritis or pleural fibrosis of the affected lobe. Filamentous bacteria were non-acid-fast ) and gram-positive or gram-variable (Fig 5). Either gram-positive cocci or gram-negative rods were also observed in 3/17 (18%) cases. In 2 cases there was plant material present (PAS positive) within inflamed and consolidated airways, confirming concurrent aspiration pneumonia. No other slides had PAS-positive elements. Alcian blue stain for detection of Cryptococcus spp. was negative. Giemsa stain did not identify Chlamydia spp. within tissue section. No viral inclusions were observed (Supplemental Table S1).
Microbiological Investigations
Microbiological culture was performed on swabs from 5 koalas. Culture showed no growth aerobically, and either pure or mixed growth in anaerobic cultures. Two of the 9 anaerobic subcultures subjected to MALDI-TOF-MS analysis were identified: Case 10—Prevotella heparinolytica; and Case 16—Porphyromonas gingivalis. The remaining 7 were not identified with MALDI-TOF MS (Supplemental Table S2) and were identified by PCR amplification and sequencing of the 16S rRNA gene (Table 2).
16S rDNA PCR Sequencing Results of the Anaerobic Isolates.

Abbreviations: PCR, polymerase chain reaction; MALDI-TOF-MS, matrix-assisted laser desorption ionization-time of flight mass spectrometry. Note: The boldface text highlights isolates that have homology to Actinomyces.
a From culture investigations, this bacterium grew anaerobically and formed black pigmented colonies. These characteristics are consistent with Prevotella heparinolytica as identified on MALDI-TOF-MS analysis (Table S2). This culture did not grow under aerobic conditions and as P. strutzeri is a facultative aerobe, this also supports the identification as Prevotella spp.
BLAST analysis (NCBI) from 16S rRNA gene sequencing of pure subcultures (n = 8) from 5 cases of pyogranulomatous lobar pneumonia identified 4 cases in which the isolate shared approximately 95% genetic sequence with Actinomyces timonensis (Table 2). The closest identified species of other culture isolates were Fusobacterium nucleatum, Fusobacterium spp., and Solobacterium moorei.
Of 14 koalas tested for koala retrovirus A, 8 (57%) were positive (Table 1). Koala retrovirus status was not significantly associated with pulmonary actinomycosis (P = .59, n = 14).
Of cases tested by PCR for C. pecorum, 5 (29%) were positive (Table 1). Detection of C. pecorum was not significantly associated with pulmonary actinomycosis (P = .09, n = 17).
Discussion
The results of this study showed pulmonary actinomycosis in this cohort of koalas was characterized by marked pyogranulomatous lobar pneumonia, most commonly affecting the left caudal lobe. Splendore-Hoeppli phenomenon and intralesional gram-variable non-acid-fast filamentous bacteria, consistent with actinomycosis, were present in all cases. Microbiological culture and 16S rRNA analysis demonstrated a gram-variable, filamentous bacterium with approximately 95% sequence homology to Actinomyces timonensis in 4 out of 5 cases subjected to testing. Although the histological lesions in case 17 were consistent with this pathogen, the negative culture may reflect the difficulty in isolating Actinomyces spp. 34 These findings support that a significant pathogen in these pneumonia cases is a novel Actinomyces species, due to less than 97% similarity to Actinomyces timonensis. Pulmonary actinomycosis has not been described in the koala previously, but the observed lesions were consistent with actinomycotic lesions observed in other species. 49,53
Actinomyces is an anaerobic or facultative aerobic, gram-positive, filamentous bacteria, which is non-spore forming and non-motile. 47 Actinomyces species can be found in normal healthy microbiota of the human oropharynx and gastrointestinal tract 55,60 and on nasal, oral, or oropharyngeal mucosal surfaces of other animals 15,47 and are most often associated with opportunistic infections. 47 Pulmonary actinomycosis is rare in animals but has been described in a small number of free-ranging species including 2 chamois (Rupicapra rupicapra) 49 and a black-tufted marmoset (Callithrix penicillata) 53 and in a captive red kangaroo (Osphranter rufus). 28
The pathogenesis of pulmonary actinomycosis in the koala is unknown and assumed to be non-contagious. In humans, aspiration has been reported as a leading initiator of pulmonary actinomycosis 55,60 with parenchymal or lobar pneumonia often affecting the lower lung lobes. 19,60 Pulmonary actinomycosis in koalas showed a similar lesion distribution, with caudal lung lobes, especially the left caudal lung lobe, affected in the majority of cases, consistent with the bipedal-like position of the koala when it climbs and rests. The lung resin study showed that the left primary bronchus followed a caudal path and did not branch until deep into the left lung, whereas the right primary bronchus branched in a more lateral direction from the trachea prior to entering the lung parenchyma, then into lobar bronchi. A less acute angle of bifurcation creates greater laminar flow. 30 Therefore, if aspiration were to occur, plant material or bacteria from the oropharynx could seed within the left caudal lung lobe more readily. Aspiration pneumonia has been described in koalas, 22 with potentially greater risk of occurrence in captive syringe-fed koalas or hand-raised joeys. 9 In the current study, there were no juvenile koalas and only one captive animal. Aspiration pneumonia, as evidenced by pulmonary inflammation around leaf fragments, was found in 2 of the 17 cases presented here, one of which had a less extensive or low-grade pulmonary actinomycosis and the other had more extensive pulmonary actinomycosis with all lung lobes affected.
Anaerobes are frequently isolated in cases of aspiration pneumonia in other species, including in humans. 4,20,21,48 The non-Actinomyces isolates identified in the current study have been described as opportunistic infections from the oral microbiome: Porphyromonas spp., 4 Prevotella spp., 48 Fusobacterium spp., 20 and Solobacterium spp. 21 In humans, concomitant species are often cultured in addition to the Actinomyces sp., and could potentiate the pathogenicity of the Actinomyces infection. 34 Investigation into these co-cultured bacteria may provide further insight into the pathogenesis of pulmonary actinomycosis in koalas. Prevotella and Porphyromonas are common isolates from the oral microbiome of many animals, 5,6,18,51 and Prevotella intermedia, Prevotella nigrescens, Porphyromonas gingivalis-like, Porphyromonas salivosa, Porphyromonas endontalis, and Porphyromonas loveana sp. nov. have been isolated from the oral microbiome of koalas. 1,6,7,42 Porphyromonas spp. have also been isolated more frequently in ageing koalas with periodontal disease. 6 We speculate that the isolate from case 16 might be Porphyromonas gingivalis-like species 42 or Porphyromonas loveana. 7 Fusobacterium necrophorum is commonly isolated from macropod progressive periodontal disease, 5,6,40 but has also been isolated from healthy red-necked wallabies (Notamacropus rufogriseus), eastern grey kangaroos (Macropus giganteus), and red kangaroos, 2,40 demonstrating that Fusobacterium spp. are within the macropod oral microbiome. Fusobacterium nucleatum is a commensal bacterium of the oral cavity and an opportunistic pathogen in humans. 20 Solobacterium moorei has been identified in humans to be associated with halitosis 21 and root canal infections. 50 A similar isolate has been identified in canine and feline periodontal disease, 25 and as sequencing showed less than 97% homology, it is likely that this is also a novel marsupial species. Poor dental hygiene has been increasingly reported in koalas 12,45 and has been recognized as a predisposing factor for pulmonary actinomycosis in humans. 55,60 Dental health scoring was not carried out with this cohort and would be a useful addition to the further investigation of koala pulmonary actinomycosis. A recent study identified increasing periodontal disease with age of koalas, 12 and given that this study showed a trend of pulmonary actinomycosis in older koalas, an association with dental disease is possible.
A reduced body condition score (emaciated, poor or fair) was significantly associated with the presence of pulmonary actinomycosis. It is likely that pulmonary actinomycosis would be a debilitating disease leading to reduced food intake in affected koalas and subsequent loss of condition, due to the fine metabolic balance koalas have between intake and output. 26,29,31 –33 Also, koala retrovirus has been associated with the presence of disease 16,52,56,59 and changes in immune function. 23,36 However, analysis of this cohort did not show any association between koala retrovirus and pulmonary actinomycosis, similar to the findings for koala retrovirus and periodontal disease. 12 Other pathogens, such as C. pecorum, Bordetella bronchiseptica, or Cryptococcus spp., cannot be ruled out as initiating pathogens, inciting micro-anaerobic conditions needed for Actinomyces to proliferate, although investigations using PCR, culture, and special stains did not find such associations. C. pecorum was found in 5 FFPE lung tissue sections and may contribute to the pathogenesis in these koalas or be due to pathogen presence in circulating mononucleocytes. 10 The majority of koalas presented with comorbidities associated with overt chlamydiosis, which may reflect the reduced fitness or compromised state of these koalas.
This case series presents the first reports of pulmonary actinomycosis in koalas. The pathogenesis likely involves aspiration. Currently this has only been recognized in South Australian koalas, which might indicate an increased susceptibility in this subpopulation. Anatomical or dental microbial origins, among others, warrant exploration as potential risk factors.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Stephenson_et_al - Pulmonary Actinomycosis in South Australian Koalas (Phascolarctos cinereus)
Supplemental Material, Combined_supplemental_materials-Stephenson_et_al for Pulmonary Actinomycosis in South Australian Koalas (Phascolarctos cinereus) by Tamsyn Stephenson, Ken Lee, Joanna E. Griffith, David J. McLelland, Anthony Wilkes, Philip S. Bird, Darren J. Trott, K. Natasha Speight, Farhid Hemmatzadeh and Lucy Woolford in Veterinary Pathology
Footnotes
Acknowledgements
Thanks to the South Australian Fauna Rescue Groups (Adelaide and Adelaide Hills Koala Rescues, Adelaide Koala Rescue, Southern Koala Rescues, and others), Adelaide Koala and Wildlife Hospital, Cleland Wildlife Park, and Dr Oliver Funnell, Aldgate Veterinary Clinic. Thanks to the Veterinary Diagnostic Lab staff for their technical assistance, especially Adrian Hines and Cheryl Day.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
