Abstract
High-throughput in vitro models lack human-relevant complexity, which undermines their ability to accurately mimic in vivo biologic and pathologic responses. The emergence of microphysiological systems (MPS) presents an opportunity to revolutionize in vitro modeling for both basic biomedical research and applied drug discovery. The MPS platform has been an area of interdisciplinary collaboration to develop new, predictive, and reliable in vitro methods for regulatory acceptance. The current MPS models have been developed to recapitulate an organ or tissue on a smaller scale. However, the complexity of these models (ie, including all cell types present in the in vivo tissue) with appropriate structural, functional, and biochemical attributes are often not fully characterized. Here, we provide an overview of the capabilities and limitations of the microfluidic MPS model (aka organs-on-chips) within the scope of drug development. We recommend the engagement of pathologists early in the MPS design, characterization, and validation phases, because this will enable development of more robust and comprehensive MPS models that can accurately replicate normal biology and pathophysiology and hence be more predictive of human responses.
Keywords
Successful drug development costs approximately $2.6 billion per molecule 29,30 and takes 12 to 15 years. 23 Failure of drugs during clinical trials is largely due to toxicity or lack of clinical efficacy. 31 Nonclinical toxicology studies are known to account for about 40% of drug failures. 94 Hence, there is a need for a more human-predictive paradigm for screening and testing of new drugs.
In drug development, initial proof-of-concept studies are often conducted in traditional 2-dimensional (2-D) human cell culture models and in animal models. The 2-D cell culture models are useful for investigating simpler hypotheses in a presumptively human-relevant way with higher throughput and lower cost. Alternatively, animal models have been the long-standing primary approach for modeling clinical efficacy and safety of novel therapeutics because of the remarkable anatomical and physiological similarities with humans. 7 However, a growing body of literature highlights the translational weaknesses of these 2 systems 17,46,63 ; hence, there is a requirement to develop new models to fill this gap.
The US Food and Drug Administration’s 21st-century predictive toxicology roadmap proposes use of alternative cell-based model systems to assess the safety and effectiveness of regulated products. Use of these model systems could improve the clinical predictivity of nonclinical assessments while reducing animal testing 27 by replicating complex human biology. These alternative models are expected to reduce the time to market for medical products as well as prevent products with safety liabilities from entering the market. 85
One such alternative cell-based model, which has risen to the forefront as a viable option, is the microphysiological system (MPS) using induced pluripotent stem cells (iPSCs). These iPSC models can differentiate along a growing number of specialized phenotypic pathways and can be engineered into small-scale devices to recreate the dynamic in vivo cellular environment by incorporating components of the extracellular matrix, cell interactions, cell morphology and architecture, blood or blood components, and mechanical forces.
This article discusses the potential roles for pathologists, as members of a multidisciplinary team, to contribute significantly to the design of MPS, to evaluate and interpret the resulting complex data, and to develop human-relevant disease models that can be a viable and more predictive alternative to the current nonclinical toxicology testing paradigms.
2-D and 3-D Cell Culture Models
In drug discovery, cell-based models are valuable tools, as they provide simple, fast, and cost-effective means to screen compounds. 31 Traditionally, these models complement in vivo studies because of the ease in manipulation of cellular functions and processes.
2-D cell culture models are monolayers of cells cultured on flat and rigid surfaces. These culture systems have historically used either immortalized cells or primary cells that are harvested from live or deceased human patients or animals. Both cell sources (primary and immortalized) have been extremely valuable in studying the effects of countless physical, chemical, and infectious perturbations and as systems for early compound screening by pharmaceutical industries. However, primary cells can be difficult to procure and have a limited life span and limited proliferation ability. Alternatively, immortalized cell lines remain viable in vitro for longer periods of time and are easy to culture and transfect, but since they have been in culture for years, they often differ genetically, phenotypically, and morphologically from their tissue of origin. This is commonly evidenced in hepatic cell lines in which cells that are serially passaged lose their in vivo characteristics (cell-cell communication), which impairs their ability to accurately predict drug metabolism and toxicity. 2,67,78
The 3-D cell culture models are more dynamic and better recapitulate the in vivo environment the cells reside in, and they can be maintained for longer periods of time without losing their viability and metabolic activity. The 3-D cell culture models include self-aggregated spheroids, multicellular organoids, 3-D–printed tissues, and the more sophisticated microfluidic-based MPS in vitro models (organ-on-a-chip) that can be monocultures or represent multicellular systems with supporting parenchymal cells. Bioprinted human tissue models—especially liver models—which incorporate both parenchymal and nonparenchymal cells, are useful in understanding the progression of liver injury in response to xenobiotics. However, for the purpose of this article, we will focus on microfluidic “microphysiological” systems (aka organs-on-chips).
MPSs are small, fabricated devices that integrate biology and bioenegineering to recapitulate organ-like functions in vitro. 12 These models can be developed from pluripotent stem cells (PSCs), which are capable of self-renewal. 73 There are 2 types of PSCs depending on their origin: embryonic stem cells and iPSCs. Embryonic stem cells can be maintained indefinitely and propagated in a pluripotent state, whereas iPSCs can differentiate from a pluripotential state to one expressing cell-specific phenotypes with prolonged survival capabilities. 73 iPSCs derived from adult somatic cells are particularly useful because of the availability of their associated medical/health histories and fewer ethical concerns (as compared with fetal sources that are typical for embryonic stem cells). The donor cells can be programmed by molecular manipulation to yield iPSCs, which share common features of embryonic stem cells such as proliferation, morphology, and gene expression. Although this creates a new way for cells to differentiate and offers a powerful tool for drug development, disease modeling, and transplantation medicine, 14 there are some issues that need to be addressed. For example, somatic cell programming is a slow process, with only a small fraction (0.1%-3%) of the transfected cells becoming iPSCs. In addition, there is evidence of genetic and epigenetic variations between different iPSCs depending on the donor somatic cells, reprogramming process, or the extensive culturing of cells, which could alter the functional activities of the iPSCs. These limitations can be overcome by optimizing reprogramming processes and variability in culture conditions. 73
Some examples of these MPS models are pliable devices that allow the distension and contraction of a “breathing” airway (lung-on-a-chip), 42,43,65 a curling “thin film” induced by contraction of cardiomyocytes and integrated pegs deflected by a contracting cardiomyocyte bundle (heart-on-a-chip), 84 or cell-lined tubular structures resembling renal proximal convoluted tubules (kidney-on-a-chip). Continuous or pulsatile fluid perfusion induces in vivo–like shear forces with induction and maintenance of phenotypic cellular maturation and polarity for extended periods of time. 37,51 Currently, semipermeable membranes lined on either side by different cell types that model in vivo barrier functions, such as the blood-brain barrier, are being explored. 49,93 Aside from being able to support tissue-level organization, these 3-D devices also enable collection of a spectrum of meaningful biological, transcriptional, proteomic, biochemical, and imaging endpoints. For example, acute exposure of nanoparticles (silica) using a lung-on-a-chip model demonstrated increased production of intracellular reactive oxygen species from the alveolar epithelial cells by simulation of the physiological breathing motions. In addition, the movement of nanoparticles can be visualized across the alveolar-capillary interface. 41 Another example of a 3-D model is the kidney model expressing KIM, CLU, HO-1, and SPP1 biomarkers for toxicity induced by cisplatin and doxorubicin exposure. The response pattern was in agreement with the clinical endpoints. 51,100
Context of Use for MPS
The development of MPS for improved predictivity of specific human responses and outcomes is predicated on our ability to identify near- and long-term applications for these modeling platforms that complement or replace current approaches. MPS models hold the promise of humanizing our testing strategies by aiding in the understanding of human biological processes and responses to injury. Although not likely to replace (at least in the near term) the high-throughput capacity of simpler cell-based systems, a few key applications are possible with a likely evolution over time as confidence builds in the data derived. In addition, it is unlikely that these systems will be able to detect all of the lesions and functional effects that can be assessed by in vivo models. However, these models will likely improve our understanding of the underlying physiological and pathological processes and potentially fill gaps that cannot be currently assessed with standard 2-D cell culture models.
Replicating diseases at the microphysiological or microanatomic level offers opportunities for both basic biomedical researchers and drug developers. Human-relevant disease models (eg, pulmonary edema on a chip, Crohn’s disease on a chip, etc) could affect various stages of the drug development process, including target identification, target validation, and identification and prioritization of lead compounds (ie, lead optimization). Several examples of MPS disease models are described in detail later in this review. Linking different organ chips via microfluidic channels may provide a way to model the physiological interplay between different organs; for example, linking the gut and liver to understand the interplay between transporters and drug-metabolizing enzymes for determination of drug absorption, distribution, metabolism, and excretion properties. 40 Another example of combined gut- and liver-model MPS has been described 82 and was used to model the pharmacokinetics of diclofenac and hydrocortisone. 81
The temporal and phenotypic resolution offered by MPS provides significant opportunity to address discrete mechanistic questions at the cellular and/or tissue level. For example, a human liver-kidney MPS model was used to elucidate the mechanisms of aristolochic acid nephrotoxicity. 18 In drug development, that application could allow detailed study of a drug’s mechanism of pharmacologic or toxicologic action at a very specific anatomic site, thus facilitating a more informed assessment of drug-target interaction, tissue response (beneficial or toxic), and associated biomarkers of those activities. The human origin of these systems could also help to differentiate species-specific responses that currently undermine confidence in the clinical predictivity of animal studies. 5,26 For example, it is challenging to interpret the human relevance when toxicity is identified in only 1 of 2 species used for preclinical studies. Recreating the preclinical finding in MPS model systems, for the sensitive species, would give greater confidence of the predictivity of the outcome in the corresponding human model systems. 87
Another application of MPS in drug development could be as screening tools for drug development–limiting safety liabilities (drug-induced toxicity). Safety liabilities in lead drug candidate molecules are most often identified in animal studies or during clinical development. In the full scheme of drug development, this is late and costly. Earlier identification of safety liabilities (eg, during lead optimization prior to animal safety studies) would offer better prioritization of candidate molecules or opportunities to guide molecular design of chemical or peptide structures with less safety liability. The most impactful MPS screening strategy would be one in which assessments of target-mediated pharmacodynamics and safety are integrated such that a balance of efficacy and safety is designed into a candidate drug by first intent (Fig. 1). A limitation of this approach is the inability to fully replicate all possible anatomic sites of toxicity even with a portfolio of MPS representing different tissues. However, published and unpublished drug safety attrition analyses reveal common target organs of toxicity (eg, liver, heart, and kidney), enabling an evidence-based prioritization of target tissues that might constitute a relevant portfolio. 52 No definitively successful examples of MPS use in drug candidate safety screening exist in the current scientific literature. Mainly, works addressing this context of use include characterization and proof-of-context data from prototypical compounds. For example, screening early drug candidates has been proposed with models such as the blood-brain barrier on a chip. 19 This model showed strong barrier integrity similar to that observed in vivo and documented the expected compartmentalization of FITC-dextran, caffeine, cimetidine, and doxorubicin as a proof of concept. 19 One of the challenges for this application is the generally higher cost and lower throughput of MPS than standard 2-D cell culture models. The use of more relevant chemistry that is similar to compounds used in current drug portfolios would provide a more accurate assessment of model value. However, this is challenging for chip developers who do not generally have access to pharma chemical libraries.
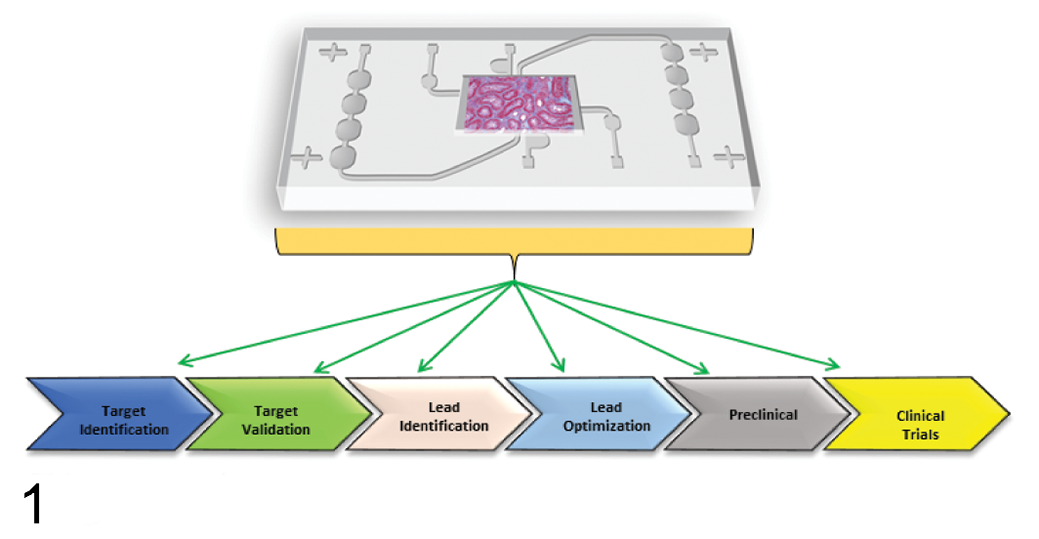
The utility of microphysiological systems in various stages of the drug development process.
Another application for MPS could be in the field of personalized medicine, especially in oncology. 91 In essence, if an MPS can eventually mimic individual patient pathophysiology and allow for molecular- and cellular-scale analysis, it would open up possibilities for personalized medicine. Each cancer type, depending on the individual, has a unique molecular signature, which, when integrated with the morphologic subtype (depending on each patient’s unique pathology, physiology, and pathophysiology), will pave the way to tailor treatment strategies that are specifically optimized for an individual’s condition.
Biomarker identification is another potential use for human MPS. 60 These systems can be used to identify translatable human biomarkers of drug-induced injury or disease. This offers substantial advantage, as drug programs with clinical phamacodynamic and toxicity biomarkers have a 3-fold greater chance of success in the clinic. 21 For example, human biomarkers of aristolochic acid–induced renal injury were identified using a human-based microphysiological model in an effort to identify the mechanisms of toxicity. 18 Similarly, a small airway (human lung) on a chip model was used to identify the inflammatory cytokine macrophage colony-stimulating factor as a potential new biomarker of chronic obstructive pulmonary disorders. 11
Examples of Biological Applications
Despite the increased complexity of MPS over 2-D cell culture models, current MPS models represent only a fraction of the complexity of the tissues they represent. A good example of this is evident when considering characterizations of “kidney chip” capabilities in defining renal disease or toxicity and the spectrum of morphologic changes traditionally characterized by animal studies.
Kidney
The kidney is a complex organ with primary roles in the excretion of metabolic waste, maintenance of water and acid-base balance, blood pressure, vitamin D metabolism, calcium-phosphorus balance, and erythropoiesis. These roles are accomplished through intimate functional integration between nephron units and a highly vascularized interstitium. Although the proximal renal tubule is the most sensitive site for toxic renal injury, other segments of the nephron and renal interstitium can also be potential targets of toxicity and disease (Table 1). This breadth of lesions and the variation in their morphologic manifestations demonstrate a lofty bar that MPS would need to demonstrate in order to replace animal testing for renal toxicities. Nonetheless, a physiologically relevant model of the renal proximal tubule offers an important opportunity to screen for toxicity and even elucidate mechanisms of toxicity. 37 Although initial kidney models mainly focused on the proximal tubule, additional models have been created for the glomerulus and distal tubule/collecting ducts as well. 3 No models of the complete nephron have yet been produced. In addition to the kidney, there are many examples of MPS that lack the complexity of critical in vivo organ functions such as the lack of bile production and flow in liver chips, valvular functions in cardiac models, bone deposition and reabsorption in a bone model, and immune cell maturation in lymphoid models such as the thymus.
Microscopic Lesions in the Renal Tubules Observed in Animal Models.

Proximal tubule on a chip models do indeed provide some advantage over current 2-D renal proximal tubule epithelial cell cultures. For example, a proximal tubule on a chip model possesses critical transporters and metabolic enzymes for vitamin D synthesis that function properly. 62,75,96 In addition, this model appears to be sensitive to appropriate toxicants and express injury markers such as KIM1. 57 In contrast to 2-D cultures, the flow system stimulates polarization of the tubular epithelial cells, 96 and the microvascular component is sensitive to cyclosporin A, causing platelet and erythrocyte adherence. 57 This system has also been linked to other tissues such as the liver to demonstrate sensitivity of the renal proximal tubular cells to hepatic metabolites of xenobiotics. 18,88
Current 2-D in vitro models are not sufficiently sophisticated to accurately detect many pathophysiological changes. In addition, the current regulatory approval process for drugs relies largely on conducting preclinical safety testing in animals, as the alternative testing paradigms are still in their infancy and need to be validated. Despite these challenges, key opportunities are currently available that may be more focused in their objective. This perspective was shared by the cross-pharma Innovation and Quality Consortium. 32 Therefore, the initial context of use for MPS by the Pharmaceutical Industry and Health Authorities for drug safety evaluation would include the investigation of mechanisms of toxicity to understand target relatedness and species specificity. Once these systems are qualified, they should have a significant impact for early screening of lead compounds in drug discovery for identification of potential liability and safety concerns, which are not well addressed by other in vitro models.
Liver
Another biological application of MPS is its utility in evaluating liver fibrosis, which is a significant safety issue and thus extremely important to detect prior to the start of any human clinical trials. In many instances, fibrosing liver disease can be detected in animal testing; however, in cases of human-specific liver toxicity, a human liver MPS system may be useful. Standard 2-D primary human hepatocyte cultures do not include the immune system and in particular the stellate cells that may be involved in hepatic fibrosis. A human liver MPS was used to demonstrate early indication of fibrosis in response to 30 nM of methotrexate. 89 Methotrexate toxicity in the liver is not selective to humans, 61 but testing in this model shows that it can recapitulate the process of hepatic fibrosis. This model is one of the most complex as it contains primary human hepatocytes as well as endothelial, immune, and stellate cells in proportions similar to human liver and could be maintained for at least 28 days. Models like this offer the complexity and human specificity that may be needed to identify human-specific toxicity. Detection of a fibrosis signal in a human liver chip, without confirming with animal data, may be difficult to interpret until sufficient confidence is gained to make decisions based solely on MPS data. However, a positive signal in human MPS in the near term could trigger stringent monitoring in human subjects with biomarkers confirmed in the MPS model. If animal and human MPS were available, this may be useful to further understand species-specific toxicities.
Numerous multicellular liver models with various features have been developed. 6,13,45 A significant challenge for multicellular MPS models is capturing and maintaining the proper proportions of key cell types. For example, within the rat liver, hepatocytes comprise 60% to 65% of cells, stellate cells comprise 8%, liver sinusoidal endothelial cells comprise 16%, and Kupffer cells comprise 12%. 6,25,58 Even if cells are seeded in proper proportions into an MPS, maintaining these proportions during culture may be difficult as growth media may be more favorable to specific cell types.
Nervous System
Many opportunities exist for MPS models of neurologic disease. For example, iPSCs from amyotrophic lateral sclerosis (ALS) patients were used to develop in vitro models of ALS. 72 Indeed, iPSCs have been differentiated into several ALS-relevant subtypes including both motor neurons and astrocytes. 28,59,74 In addition, differentiation into oligodendrocytes, microglia, Schwann, and muscle cells has been demonstrated. 1,24,53,66 A National Institutes of Health (NIH)–funded collaboration between Cedar-Sinai and Emulate, Inc is now creating an MPS disease model for ALS and Parkinson disease. 79 MPS may be particularly suited to modeling diseases with a genetic component via the use of iPSC-derived or primary cells from patients with these conditions. Genetically modified iPSC-derived cells represent further disease-modeling opportunities, as described above. 72 Further, disease conditions that are difficult to reproduce in animal models, such as Alzheimer disease, may be reproducible in complex in vitro systems such as MPS. These models offer some promise in identifying useful markers of efficacy for human patients, which have thus far been elusive. Other MPS disease models created with iPSCs include those of the blood-brain barrier and neurodegeneration, 33 cardiomyocytes 77 skin, 36 liver, 3 and neurovascular disorders. 15
Hemostasis
A noteworthy example of the use of MPS is in the area of thrombus formation, which poses a safety issue for patients and oftentimes goes undetected or underdiagnosed in preclinical animal studies because of species differences. 44 In in vivo preclinical animal toxicity studies, thrombus formation may be identified only if thrombi occur in the preclinical species and are present in microscopic sections of tissues routinely evaluated by pathologists. Evaluation of thrombus formation within an MPS offers several advantages. In this system, the microfluidic device was lined by a chemically fixed human endothelium to demonstrate the ability of the endothelial tissue to modulate hemostasis under continuous flow to replicate critical determinants of hemostasis, platelet function, and shear stress. For hemostasis, signaling between vascular endothelium and platelets is essential. Endothelium lining the microfluidic chamber can initiate the formation of platelet-rich thrombi in the presence of physiological shear stress. These conditions are not replicated in standard primary endothelial monolayer cultures, demonstrating an advancement by adding the complexity of microfluidics. 44 While other models of endothelium exist, this model is the only one currently described that has been characterized for the study of thrombosis. With the endothelial components of other tissues and endothelial-specific models, it is likely that other models will eventually be characterized for this purpose as well.
Cancer
Several MPS disease models (Table 2) have been developed to study primary cancers and metastases. 47,56,64,89 Clark et al developed a liver MPS to assess tumor cell dormancy and inflammatory responses. This platform used parenchymal and nonparenchymal liver cell types in a 1:1 ratio, which were then seeded with breast cancer cells (MDA-MB-231 or MCF7) to study spontaneous dormancy following metastatic outgrowth of breast tumor cells. 97 The liver is a common site of metastasis, due to high blood flow and a fenestrated endothelial barrier; therefore, this model has utility in the study of metastatic cell dormancy. Spontaneous dormancy was observed in a subpopulation of breast cancer cells. Dormant cells resided primarily within the 3-D tissue, while proliferating cells predominated along the polystyrene scaffold. When the hepatic model was modified to include a PEGDa-SynKRGDa hydrogel matrix, the number of cells spontaneously entering dormancy was increased, and the hepatic “tissues” secreted less proinflammatory analytes and were more responsive to an inflammatory stimulus (lipopolysaccharide). 10 This model has been used to study the early microenvironment of metastatic breast cancer cells in the liver and is anticipated to support the identification of new therapeutic targets and assessment of efficacy. 97 Other types of malignancy, including prostate cancer and melanoma, have also been successfully used in this model. 22
Current Established Microphysiological System Models of Disease.

Abbreviations: hMSC, human mesenchymal stem cells; iPSCs, induced pluripotent stem cells; MDA-MB-231 or MCF7, MDAnderson or Michigian cancer foundation-7 cell lines for breast cancer.
Another MPS model has been reported to reproduce Ewing’s sarcoma, an aggressive and osteolytic pediatric bone tumor. 90 Ewing’s sarcoma stimulates osteoclast differentiation and activation, leading to osteolytic lesions that culminate in broken bones, pain, and hypercalcemia. 90 A model was developed with osteoclasts and osteoblasts within a 3-D–engineered mineralized bone matrix, and in this culture system, bone mineralization was shown to occur. Adding Ewing’s sarcoma cell aggregates to the model reproduced the expected decreases in bone density, connectivity, and matrix deposition seen in human patients with Ewing’s sarcoma. In addition, treatment with zoledronic acid inhibited bone resorption in this model, consistent with results from Ewing’s sarcoma patients, thus demonstrating the potential use of the model in identifying novel therapeutic agents for this cancer.
In addition to the models listed in Table 2, some MPS models (disease and drug/chemical safety assessment models) that are in development and funded by the NIH and National Center for Advancing Translational Sciences are listed in Table 3.
NIH-Funded MPS Models of Human Diseases in Development.
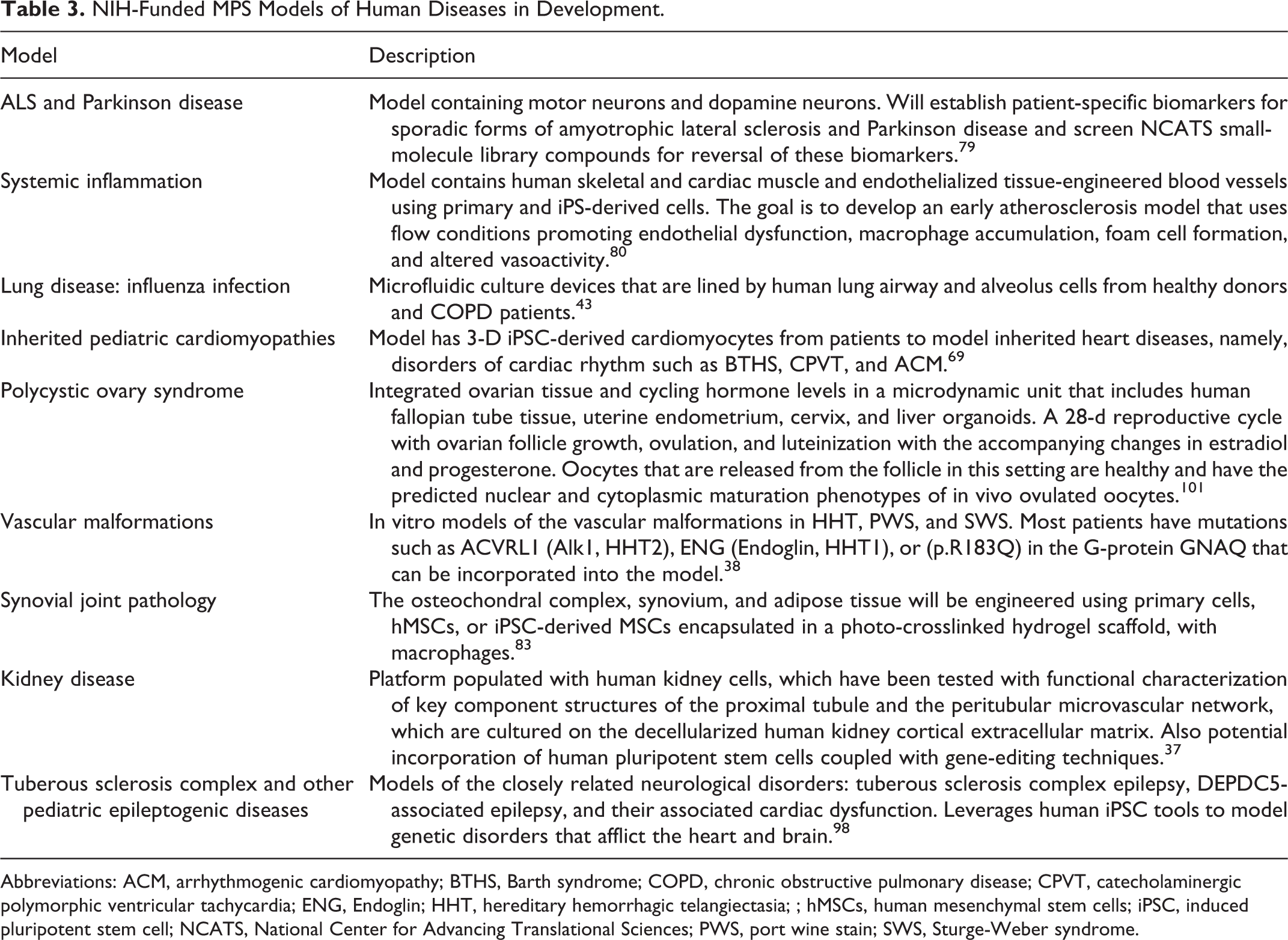
Abbreviations: ACM, arrhythmogenic cardiomyopathy; BTHS, Barth syndrome; COPD, chronic obstructive pulmonary disease; CPVT, catecholaminergic polymorphic ventricular tachycardia; ENG, Endoglin; HHT, hereditary hemorrhagic telangiectasia; ; hMSCs, human mesenchymal stem cells; iPSC, induced pluripotent stem cell; NCATS, National Center for Advancing Translational Sciences; PWS, port wine stain; SWS, Sturge-Weber syndrome.
Rare Diseases
MPS offers a promising alternative to studying rare diseases and screening for efficacy of therapeutics. Rare diseases (generally <4 cases per 10 000 individuals at risk) are a significant challenge because preclinical animal models often do not exist, and the lack of understanding of the diseases makes it a challenge to develop appropriate models. Accordingly, models of rare diseases are an important future niche for MPS. 54 –56 MPS can be seeded with cells, including iPSCs from patients with rare disease. Therefore, they offer the potential to accurately re-create human disease conditions in vitro, even in cases in which the underlying genetic cause is not clearly known. 56 Several MPS disease models are discussed as follows.
An MPS model is reported for cardiomyopathy related to Barth syndrome, 92 a rare disease associated with structural and functional cardiac abnormalities thought to result from mutations in the gene for tafazzin. Tafazzin is an acyltransferase that is responsible for acylation of cardiolipin, a major phospholipid of the inner mitochondrial membrane. Abnormalities in cardiolipin have been associated with cardiac dysfunction, aging, and ischemic disease. 20 The authors used cardiomyocytes derived from iPSCs from Barth syndrome patients. These cells contained sparse and irregular sarcomeres that contracted weakly, 92 and they contained normal levels of adenosine triphosphate but produced high levels of reactive oxygen species. Cas-9 gene-editing techniques were used to restore tafazzin function, which resulted in normal morphology and contraction of cells in the MPS, thus confirming the link between the tafazzin mutation and the disease.
Infectious Diseases
Infectious diseases is another area in which MPS models may be useful. Many infectious diseases are species specific or have unique species-specific pathogeneses (particularly viral diseases). A recently developed human intestinal enteroid model was composed of multiple epithelial cell types that were physiologically active. Although this model is not microfluidic, it represents an example of an advancement of in vitro models for a common contagious infectious disease. This model evaluated the susceptibility of different cell types to human rotavirus infection. Human rotavirus is a major cause of diarrheal disease worldwide, but there is little to no knowledge of host susceptibility and pathophysiology. In the human intestinal enteroid model, it was found that the virus preferentially infects differentiated enterocytes and enteroendocrine cells of the intestine at their villus tips while sparing the intestinal crypts. There was also swelling of the human intestinal enteroid as a consequence of enterotoxin production. This model can potentially be used to study human intestinal pathogens to better understand host-pathogen interactions at the cellular level. 76
Roles for Pathologists
An aspiration of many researchers developing MPS is the complete replacement of animal studies. Although a noble aspiration, our understanding of how to extrapolate in vitro biology and pathobiology to the situation in vivo is likely too rudimentary to support that objective in the near term. However, MPS offer the opportunity to fill substantial gaps as near-term “preanimal” complements to animal studies. In turn, these experiences of in vitro findings extrapolated to in vivo realities would further inform model development and perhaps replacement of animal studies.
The complexity of these MPSs, albeit biologically relevant, comes with challenges in technical reproducibility, cost, and throughput. Differentiation protocols for iPSCs can be lengthy, and it can be challenging to define protocols to attain the expected pattern of cellular differentiation of some cell types. Microfluidic systems generally use incredibly small volumes and flow rates in which air bubbles in the system pose a constant challenge to its normal functioning. 19,70,99 Bubble traps have been employed in the microfabrication of systems composed of synthetic polymers, but these substances have a varying capacity for gas permeability, and test compounds, chemicals, or perfusate components may nonspecifically adhere to these synthetic polymers and impair the drug’s bioavailability. This was evidenced while testing cardiac drugs using polydimethylsiloxane (PDMS), wherein variability in absorption was noted depending on the hydrophobicity of the molecules. 86 This technology is still in its infancy; hence, high-volume manufacture of MPS devices has not reached a cost-effective industrialization phase. Throughput for some of the more complex (and most physiologically relevant) systems can be very low (ie, single to 10s of compounds).
Traditionally, the role of pathologists in the efficacy and safety assessment of new therapeutics has been to identify and interpret microscopic and biochemical changes in animal studies. The advent of MPS offers pathologists an opportunity to apply their knowledge of anatomy, pathophysiology, toxicology, and molecular biology to shape these clinically relevant model systems and move well beyond the realm of the microscope (Fig. 2). Pathologists can link histomorphology to disease mechanisms based on their expertise in tissue microarchitecture, response to injury, and systems biology, thus supporting MPS model development. Early engagement of pathologists during development of MPS will ensure that the cell types essential for maintaining organ function (based on knowledge of histology and immunohistochemistry) are included in the MPS and that the morphology of these cells is assessed as a form of quality assurance. This is important because of the structural and functional complexity of living tissues and organs. For example, to develop a relevant MPS model of the gastrointestinal tract, one should emulate its structural and functional characteristics, with an appropriate epithelial lining that includes the enterocytes, Paneth cells, stem cells, and goblet cells and that achieves the expected barrier function. 32
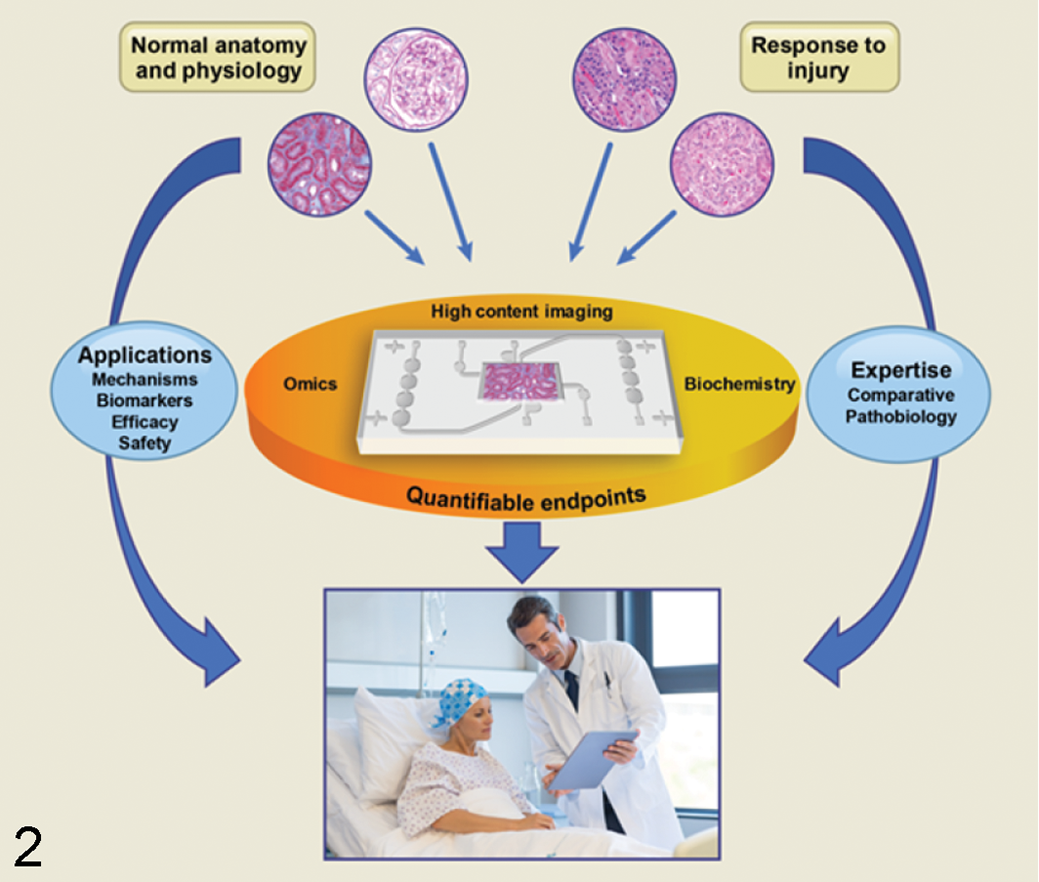
Microphysiological systems can be a platform for convergence of disciplines such as anatomy, physiology, pathology, biochemistry, and omics for developing therapeutic strategies. Pathologists can aid in the design of systems that reflect tissue-level biology and pathobiology (illustrated in the images at the top of the figure), identify endpoints (imaging and nonimaging) that link histologic and mechanistic changes, and relate the outcomes to the species of primary interest: human patients.
Recent advances in the MPS field have led to the development of cellular and functional units of organs “on chips,” such as lung, liver, heart, kidney, and brain.* The critical function of the alveolar-capillary interface of the human lung was reconstituted on a microfluidic chip and used to study toxic and inflammatory responses to nanoparticles. Results were reproduced in mouse models, thus validating the physiological relevance. 41 As progress is made in developing more complex MPS systems, researchers are now working toward mimicking gas exchange between alveoli and blood. As these systems evolve, a multitude of mechanistic data would be generated, for which meaningful interpretation and linkage to adverse outcomes in humans could be provided by trained pathologists.
Another impactful opportunity is the identification and validation of biomarkers of cell and tissue injury. Clinical pathologists apply their broad training in pathology by assessing biochemical and cytologic markers of health and disease. Analysis of the tissue culture medium in MPS can aid in the identification of novel biomarkers specific to that MPS model, which needs to be validated given that it can vary with assay quality and reference standards. The diagnostic or predictive value of a biomarker is most compelling when confirmed by the morphological changes in the associated tissues (histopathology). Cystatin C, for example, was proposed as a biomarker for glomerular filtration rate in the early 1980s, but it took years of research before it could be confirmed as a validated biomarker. 16,35 Using a combination approach can improve the predictability of outcomes of novel therapeutic strategies in the context of toxicology and risk sciences. For example, when histopathology is used in combination with proteomic and immunological techniques, it can aid in defining various disease processes including drug-induced toxicities, which is especially important in drug-induced idiosyncratic toxicity such as drug-induced liver injury. 4,34,95
Pathologists, with their broad cross-species training in the initiation and progression of disease, as part of a diverse group, can contribute to the development and use of the MPS. Stand-alone molecular MPS data may have limited value but when taken together with the histomorphology and clinical chemistry parameters, will provide a strong perspective on biology and pathophysiology.
Conclusion
Successful development of MPS models that accurately reflect in vivo biology requires close collaboration between scientists across numerous disciplines including but not limited to bioengineers, cell biologists, and pathologists. Pathologists, in particular, have a significant and unique role in defining in vivo biology at the tissue, organ, and system levels, including dynamic physiology, responses to injury, and appropriate measures for extrapolation to in vivo responses. Useful applications of these systems include studying fundamental mechanisms of disease, toxicity, drug efficacy, and even novel biomarker identification.
Although there is an aspiration by many to replace animal studies with a more human-relevant modeling platform such as MPS, there is a lack of sufficient breadth of understanding of the complexity of biology and direct human applicability to accomplish that in the near term. However, MPSs have a significant opportunity to enhance and decrease our dependence on animal studies as well as contribute to a better understanding of differences in animal and human responses. Using MPS in a tiered approach to complement the existing testing systems, while actively engaging pathologists, would enable better understanding of the underlying pathophysiological processes and hence allow for targeted drug development.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosures
R.S. and T.V.V. are employees of AbbVie and may own AbbVie stock. B.R.B. is an employee of the National Toxicology Program, National Institute of Environmental Health Sciences. AbbVie and National Toxicology Program/National Institute of Environmental Health Sciences sponsored and funded the study, contributed to the design, and participated in writing, reviewing, and approval of the final publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
