Abstract
A 10-year-old female Papillon dog that had previously developed a mammary tumor was admitted for treatment of a hypoglycemic attack. Blood examination showed severe hypoglycemia and decreased blood insulin concentration. Computed tomography indicated multiple tumors in the cranial and caudal lobes of the right lung. These tumors were resected surgically and diagnosed as pulmonary adenocarcinomas by histopathologic examination. Hypoglycemia was temporarily improved after the resection, but a hypoglycemic event occurred 2 months after the surgery. Immunohistochemistry of the tumor demonstrated the expression of insulin-like growth factor 2 in tumor cells. Western blot analysis revealed the expression of high-molecular-weight (big)–insulin-like growth factor 2 in the tumor region. Insulin-like growth factor 2 mRNA expression was also confirmed in the tumor using reverse transcription–polymerase chain reaction. These findings indicate the diagnosis of non–islet cell tumor-induced hypoglycemia caused by big-insulin-like growth factor 2 produced by the tumor in the dog. This report provides information on differentiating tumors that cause paraneoplastic hypoglycemia.
Keywords
Hypoglycemia is a common medical emergency, mostly resulting from endocrine disease or complications of insulin treatment in patients with diabetes. In rare cases, hypoglycemia could be a manifestation of neoplastic diseases and can be a paraneoplastic syndrome. Paraneoplastic syndromes are described as clinical symptoms and signs caused by the systemic effects of tumors. 9 Generally, hypoglycemia as a paraneoplastic syndrome is observed in pancreatic insulinoma, ectopic insulin-producing tumors, and tumors expressing various factors that interfere with glucose metabolism, such as insulin receptor antibodies, tumor necrosis factor–α, interleukin-1 and -6, catecholamines, and insulin-like growth factor (IGF). 5,13 The lattermost type is known as non-islet cell tumor hypoglycemia (NICTH) and mostly caused by overexpression of partially processed pro-IGF-2 (big-IGF-2) due to impaired posttranscriptional processing of IGF-2. 12 IGF-2 is initially synthesized as pre-pro-IGF-2, which is converted to pro-IGF-2 by various convertases. 6,19 Normally, pro-IGF-2 is processed to mature IGF-2 (about 7.5 kDa) through several steps of degradation. 5 In the tumor tissues of NICTH, however, the proper peptidase processing of pro-IGF-2 is considered to be deficient, resulting in aberrant production of a relatively stable high-molecular-weight IGF-2 (big-IGF-2), ranging in size from 11 to 18 kDa. 5 NICTH caused by big-IGF-2 is rare in humans and is reported in mesenchymal tumors such as mesothelioma, leiomyosarcoma, and fibrosarcoma and in epithelial tumors such as hepatocarcinoma, gastric cancer, and colon cancer. 7 In dogs, there have been several reports of suspected NICTH 1 ; however, only 1 case has demonstrated big-IGF-2-production in a leiomyoma of the stomach. 3 Here, we describe a case of pulmonary adenocarcinoma in a dog showing hyperglycemia and confirm the production of big-IGF-2 in the tumor region.
A 10-year-old spayed female Papillon dog (body weight 5 kg) was brought to the local clinic with signs of general weakness. At presentation, a blood test revealed hypoglycemia, and multiple tumors were identified in the right lung by radiography. Although the hypoglycemia was stabilized by oral glucose administration, it recurred and gradually worsened, and the patient experienced a seizure. Therefore, the patient was sent to the Nippon Veterinary and Life Science University (NVLU) Veterinary Medical Teaching Hospital. There was no history of diabetes treatment. However, the patient had a mammary gland tumor diagnosed as grade 2 tubulopapillary carcinoma and treated by resection 21 months earlier. 8
The results of the blood tests conducted at NVLU showed a glucose level of 47 mg/dL (reference value: 75 to 128 mg/dL) and insulin <0.02 ng/mL (reference value: 0.27 to 0.65 ng/mL). The computed tomography examination demonstrated 3 masses in the right lung: 1 in the cranial lobe (51 × 36 × 52 mm) and 2 in the caudal lobe (29 × 15 × 28 mm and 7 × 7 × 7 mm; Suppl. Figs. 1 and 2). Furthermore, a sternal lymph node and a cranial mediastinal lymph node were enlarged, but an axillary lymph node and an inguinal lymph node were not changed. No tumor was found in other organs.
To investigate the relationship between the hypoglycemia and the lung masses, we surgically resected the right cranial and middle lobes and a solitary mass on the surface of the right caudal lobe; however, we could not completely resect the tumor inside the right caudal lobe. The tumor in the right cranial lobe expanded from the caudal surface of the lobe and invaded the middle lobe (Fig. 1). The specimens were fixed in 10% buffered formalin for histopathologic and immunohistochemical examinations. Other specimens were snap-frozen in liquid nitrogen for mRNA and protein analysis. After surgery, hypoglycemia immediately improved, and serum insulin level also increased to the normal range. The dog was discharged on postsurgical day 9, and we followed up by imaging of the remaining tumor. On postsurgical day 62, hypoglycemia relapsed and showed no improvement by oral glucose administration from postoperative day 115. A chest radiograph revealed pleural effusion, although no detectable change was observed in the remaining tumor within the right caudal lobe. However, a large number of atypical epithelial cells were found in the pleural fluid by cytologic examination, indicating dissemination of the neoplastic cells to the pleura. The hypoglycemia became difficult to control, and the dog showed frequent seizures. The owner elected euthanasia because of poor prognosis and progressive worsening of the hypoglycemic condition.
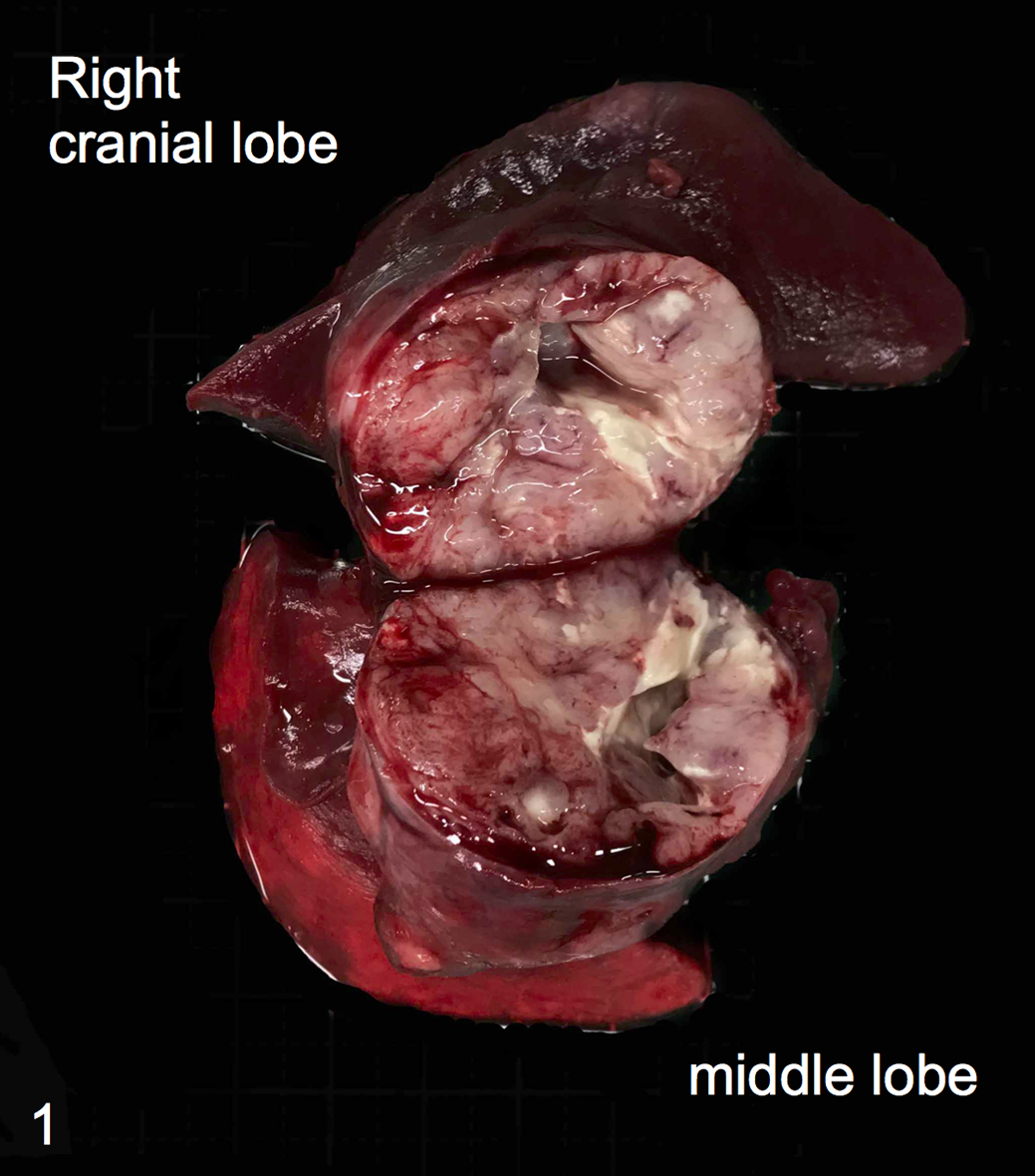
Pulmonary adenocarcinoma (resected tumor), lung, dog. Tumor in the right cranial and middle lung lobes contains mucus and has a necrotic center.
The surgically resected lung masses were routinely processed and stained with hematoxylin and eosin (Fig. 2a). Histologic examination revealed papillary adenocarcinoma. The neoplastic cells were medium sized and cuboidal to slightly flattened, with epithelial monolayers forming tubular and papillary structures, and the neoplastic cells showed extensive atypia. The nucleus of the neoplastic cells was almost uniform, oval to elliptical, with a high nuclear-cytoplasm ratio and 2 to 4 mitotic figures per high-power field (400×). The neoplastic cells secreted mucus into a lumen and had abundant necrosis, fibrosis, and aggregated lymphocytes. Neoplastic cells invaded the pleural surface, but there was no vascular invasion.
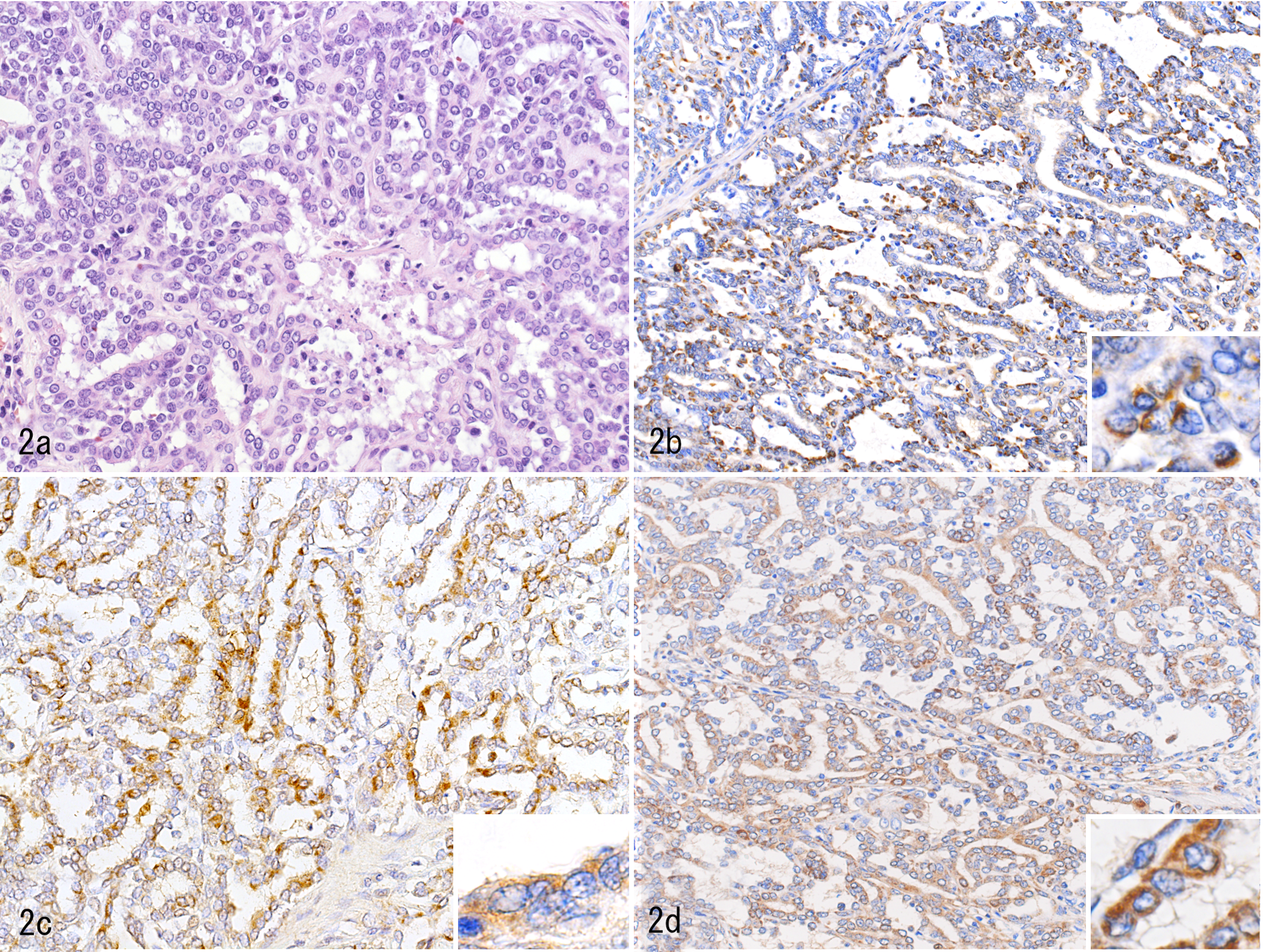
Pulmonary adenocarcinoma, lung, dog. (a) Neoplastic cells form cords and tubulopapillary structures. Hematoxylin-eosin. (b) Insulin-like growth factor 2 immunoreactivity is detected in the cytoplasm of the neoplastic cells. Immunohistochemistry for napsin A (c) and surfactant protein A (d) reveals cytoplasmic labeling of the neoplastic cells.
We conducted immunohistochemistry using antibodies against napsin A (418061, rabbit polyclonal, Nichirei Biosciences, Tokyo, Japan), surfactant protein A (SP-A; bs-10265R-TR, rabbit polyclonal, Bioss Antibodies, Woburn, Massachusetts, USA), thyroid transcription factor 1 (TTF-1; 713841, mouse monoclonal, Nichirei Biosciences), and cytokeratins AE1/AE3 (MU071-UC, mouse monoclonal, BioGenex, Fremont, California, USA). 2 Napsin A, SP-A, and AE1/AE3 immunoreactivity was detected in the cytoplasm of the tumor cells but not TTF-1 (Fig. 2c, d; Suppl. Fig. 3b). Napsin A and SP-A immunoreactivity was not detected in the mammary gland tumor that was resected 21 months earlier. Thus, we diagnosed this case as pulmonary adenocarcinoma.
To determine the cause of hypoglycemia, we conducted immunohistochemistry, Western blot, and reverse transcription–polymerase chain reaction (RT-PCR) analysis of IGF-1 and IGF-2 expression in the tumor and adjacent normal lung tissue, using normal canine testis as a positive control. 14,15 We used the protocols described in our previous work. 11,18 Immunohistochemistry and Western blot were performed using anti-IGF-1 (bs-0014 R, rabbit polyclonal, Bioss Antibodies) and anti-IGF-2 (ab 9574, rabbit polyclonal, Abcam, Cambridge, UK) antibodies using dilutions of 1:500 in immunohistochemistry and 1:2000 in Western blot analysis. Nonspecific isotype control antibodies (rabbit IgG, Abcam; mouse IgG, Nichirei) were used as negative controls for immunohistochemistry. Immunohistochemistry showed no IGF-1 immunoreactivity in either the tumor or adjacent normal lung tissue (Suppl. Figs. 3a and 4a). In contrast, IGF-2 immunoreactivity was detected in the cytoplasm of the tumor cells but not in the adjacent normal lung tissue (Fig. 2b; Suppl. Fig. 4b). Both IGF-1 and IGF-2 immunoreactivities were detected in interstitial cells in the normal testis. The Western blot analysis for IGF-1 showed an immunoreactive band at about 20 kDa in the normal testis but not in either the tumor or adjacent normal lung tissue (Fig. 3). However, Western blot analysis of tumor tissue showed IGF-2–immunoreactive bands of normal molecular weight (about 7.5 kDa) and large molecular weight (about 20 kDa), while the normal testis expressed only normal IGF-2 (Fig. 3). No specific immunoreactive band was detected in adjacent normal lung tissue.
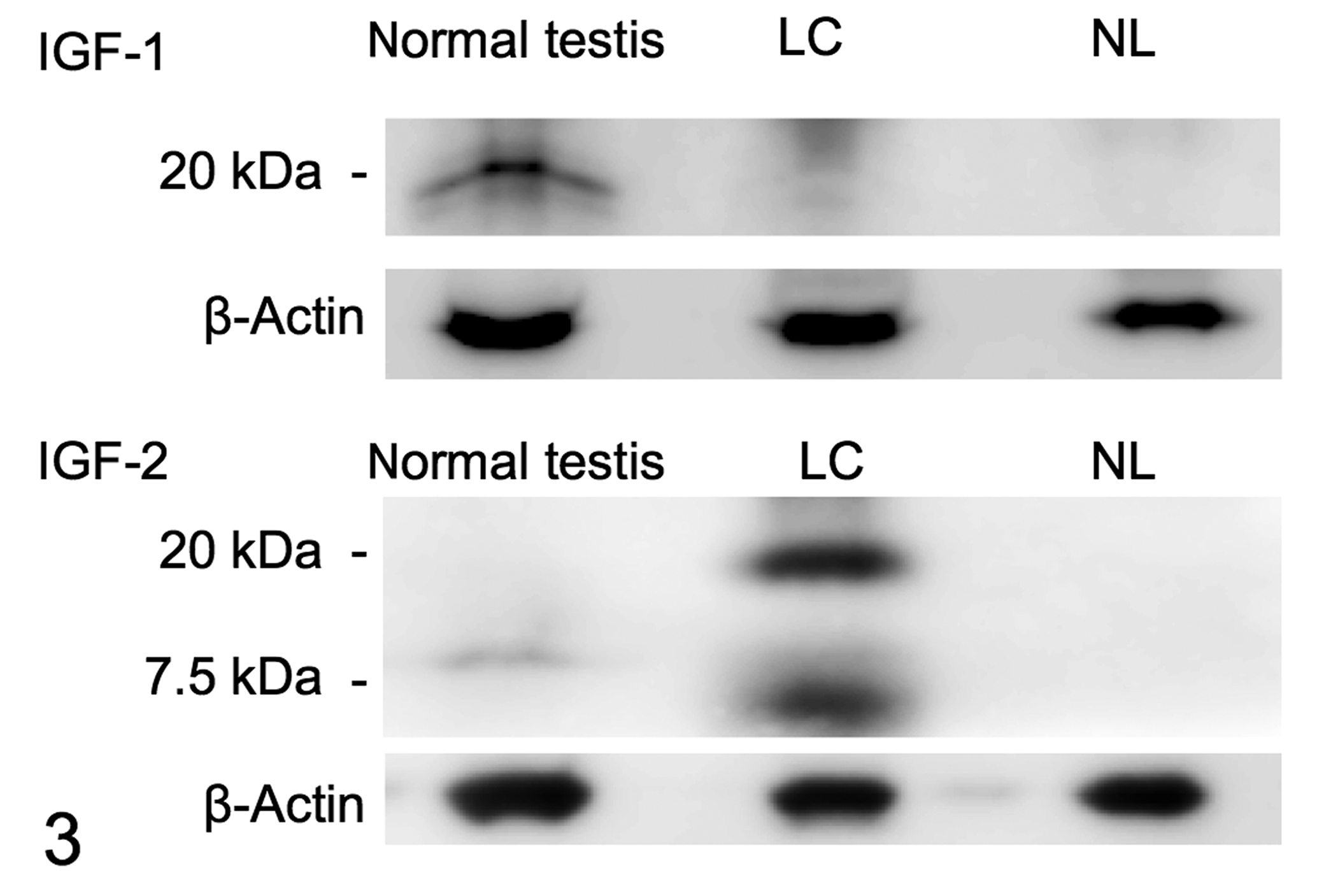
Western blot analysis of insulin-like growth factor (IGF) 1 and 2 in the lung adenocarcinoma (LC) and the adjacent normal lung tissue (NL). An IGF-1–immunoreactive band was detected in normal testis but not in LC or NL. LC showed expression of normal IGF-2 (7.5 kDa) and big-IGF-2 (about 20 kDa). Normal testis expressed only normal IGF-2. No immunoreactive band was detected in NL. β-actin was used as a loading control.
RT-PCR was performed to confirm IGF-1 and IGF-2 mRNA expression using specific primers as follows: canine IGF-1: F, ATGTCCTCCTCGCATCTCTT and R, TCCCTCTACTTGCGTTCTTC; canine IGF-2: F, TTGGCCTTCGCCTCGTGCTG and R, GGACGGTGACGCTTGGCCTCTCTG. 15 IGF-1 mRNA was expressed in both the tumor and adjacent normal lung tissue, whereas IGF-2 mRNA was detected in only the tumor tissue (Fig. 4). Normal testis showed expression of both IGF-1 and IGF-2 mRNA.
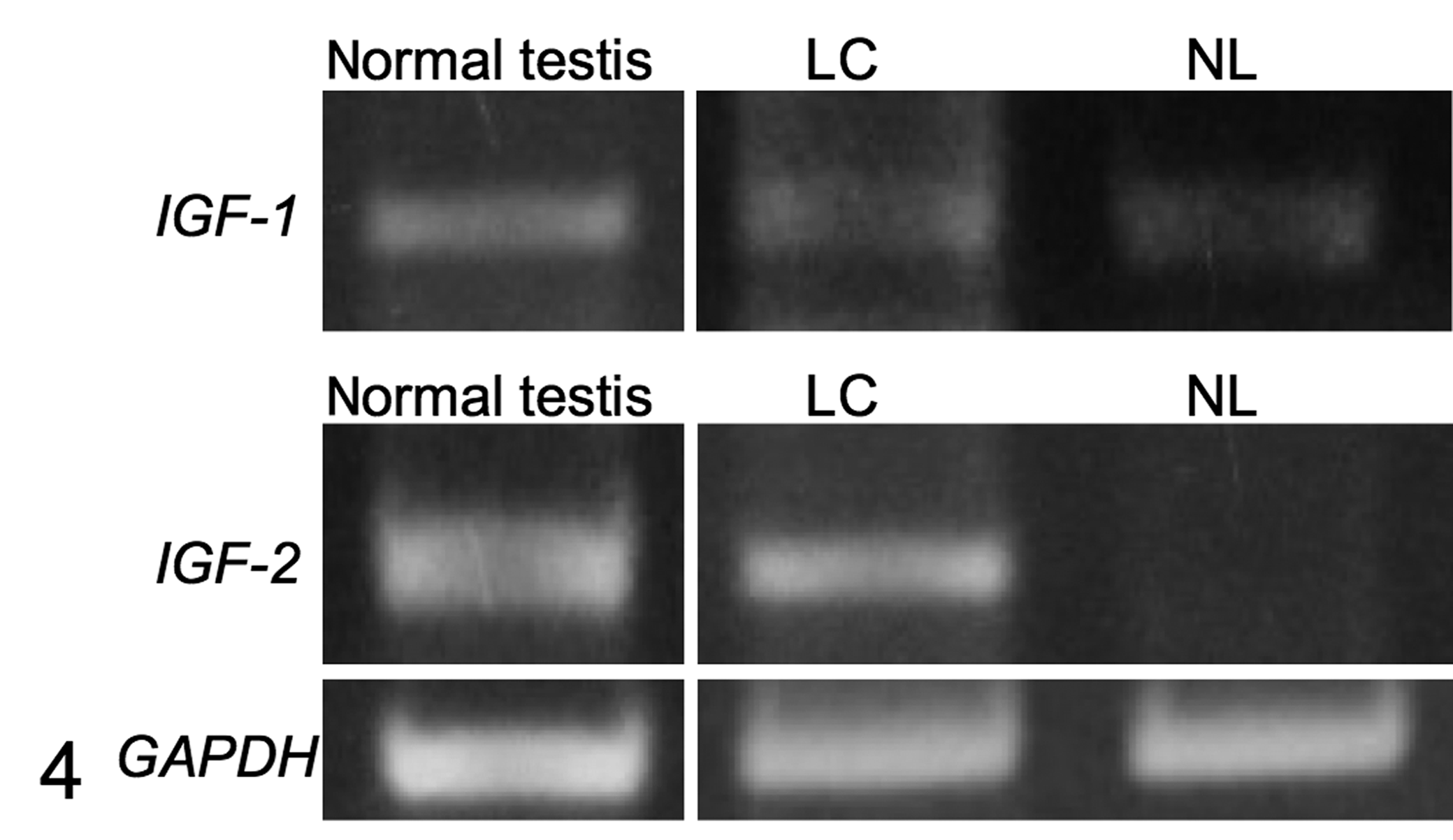
Reverse transcription–polymerase chain reaction of insulin-like growth factor (IGF) 1 and 2. IGF-1 mRNA was detected in lung adenocarcinoma (LC), adjacent normal lung (NL), and normal testis. IGF-2 mRNA was expressed in LC and normal testis. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a loading control.
In the present study, we used Western blotting to demonstrate the expression of big-IGF-2 in a pulmonary adenocarcinoma from a dog with hypoglycemia. Immunohistochemistry revealed that IGF-2 was detected in only the neoplastic cells. Furthermore, the expression of IGF-2 mRNA was confirmed in the tumor tissue using RT-PCR. IGF-1 was not detected in the tumor by immunohistochemistry or Western blot analysis. These findings indicate the diagnosis of NICTH due to big-IGF-2 production by pulmonary adenocarcinoma cells. This diagnosis is supported by the results indicating that hypoglycemia was improved after surgical resection of the lung tumor and recurred soon before cytologic detection of tumor spread within the pleural cavity. In NICTH, impaired posttranscriptional processing of pro-IGF-2 results in the accumulation of big-IGF-2, a variable-sized pro-IGF-2. 5 Big-IGF-2 is biologically active and induces a robust insulin-like effect via the insulin and IGF receptors. 5,7 Hizuka et al 10 demonstrated that an increase in big-IGF-2 expression in NICTH was caused by deficient O-linked glycosylation of pro-IGF-2, which is essential for the proper peptidase processing of pro-IGF-2 to mature IGF-2. Recently, patients with NICTH caused by solitary fibrous tumor showed an increase in big-IGF-2 expression and a decrease in proprotein convertase subtilisin/kexin type 4 (PCSK4), a critical pro-IGF-2 convertase. 12 These results indicated that an imbalance in big-IGF-2 and PCSK4 expression could lead to increased serum levels of big-IGF-2. 12 Similar mechanisms might be involved in the increase in big-IGF-2 in dogs with NICTH, but this was not examined. In veterinary cases showing hypoglycemia, it is common to preferentially suspect an insulin-producing tumor, such as insulinoma, when there is no history of insulin treatment or endocrine disease. In the present case, however, the serum insulin level was below the normal range. Hypoglycemia with a decrease in serum insulin level was described in human NICTH cases. 4,7 Most NICTH cases also involve a reduction in serum growth hormone (GH) as well as insulin and IGF-1. 20 Usually, GH increases the production of insulin and IGF-1, which downregulate GH secretion through a negative feedback loop. 20 Elevated levels of big-IGF-2 also suppress pituitary GH secretion in NICTH cases, leading to a reduction in circulating insulin and IGF-1. 16 Thus, the hypoinsulinemia in this case may be due to the decrease in GH production caused by overexpressed big-IGF-2.
In this study, we also investigated IGF-1 and IGF-2 expression using immunohistochemistry in 3 nonhypoglycemic cases of canine pulmonary adenocarcinoma. Neither IGF-1 nor IGF-2 immunoreactivity were detected in the tumor cells of these 3 cases (data not shown). Some cases of human lung carcinoma expressed IGF-2, and it has been reported that NICTH is caused by the overexpression of IGF-2. 13 However, to our knowledge, big-IGF-2 production has not been identified in pulmonary adenocarcinoma. In dogs, only 1 report demonstrated hypoglycemia due to IGF-2 expressed by a mammary gland adenocarcinoma, although big-IGF-2 production was not identified. 17 Furthermore, there is no report describing NICTH caused by big-IGF-2 in epithelial tumor cases, including pulmonary adenocarcinoma in dogs. Therefore, the present study is the first report demonstrating NICTH caused by big-IGF-2 produced in epithelial tumors in dogs.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Noguchi_et_al - Big Insulin-like Growth Factor 2–Producing Tumor in a Hypoglycemic Dog
Supplemental Material, Combined_supplemental_materials-Noguchi_et_al for Big Insulin-like Growth Factor 2–Producing Tumor in a Hypoglycemic Dog by Syunya Noguchi, Yoshiaki Kubo, Mami Araki, Miki Koh, Yuji Hamamoto, Kyoichi Tamura, Hirotada Otsuka, Akiko Yasuda, Daigo Azakami, Masaki Michishita and Satoshi Soeta in Veterinary Pathology
Footnotes
Acknowledgements
We appreciate the veterinarians and veterinary technicians at Nippon Veterinary and Life Science University Accessory Animal Medical Teaching Hospital who assisted with this case. In particular, we appreciate Dr Akihiro Mori for participating in the diagnosis and treatment. We would also like to thank Editage (![]() ) for English-language editing.
) for English-language editing.
Declaration of Conflicting Interests
The authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
