Abstract
Clostridium difficile is a well-documented cause of enterocolitis in several species, including humans, with limited documentation in New World nonhuman primates. We report several cases of C. difficile–associated pseudomembranous enterocolitis, including a case in a Geoffroy’s spider monkey (Ateles geoffroyi) and several cases in common marmosets (Callithrix jacchus). The histologic lesions included a spectrum of severity, with most cases characterized by the classic “volcano” lesions described in humans and several other animal species. C. difficile was isolated from the colon of the spider monkey, while the presence of toxin A or toxin B or of the genes of toxin A or B by polymerase chain reaction served as corroborative evidence in several affected marmosets. C. difficile should be considered a cause of enterocolitis in these species.
Keywords
Clostridium difficile–associated disease (CDAD) is an emerging cause of illness and nosocomial infection in animals and is one of the most important causes of antibiotic-associated diarrhea and nosocomial infections in humans. 2,4,8,10,19,20,22,23 Antibiotic-associated pseudomembranous colitis due to C. difficile infection has been documented rarely in cotton-top tamarins, a marmoset, and an orangutan, but reports of the pathology of natural C. difficile infection in New World primates are very limited. 7,12,13
We report 2 colonies of New World primates affected by outbreaks of C. difficile enterocolitis. The first was a zoo population. Five members of a group-housed population of Geoffroy’s spider monkeys (Ateles geoffroyi) developed lethargy and diarrhea. A 26-year-old male intact animal died within 2 days after having lethargy, loose stool, and weight loss.
At necropsy, the dilated large intestine contained a large amount of homogeneous, brown, turbid, thick fluid. The most aborad ileal mucosa, the cecal mucosa, and large portions of the colonic mucosa were covered by pale brown membranous material that was easily removed. Underneath this material, the mucosa was reddened. Within the cecum, the mucosa was nearly uniformly coated by this material, while in the colon, the material was distributed multifocally in numerous round, up to 3 to 4 mm in diameter, evenly spaced convex deposits (Fig. 1).
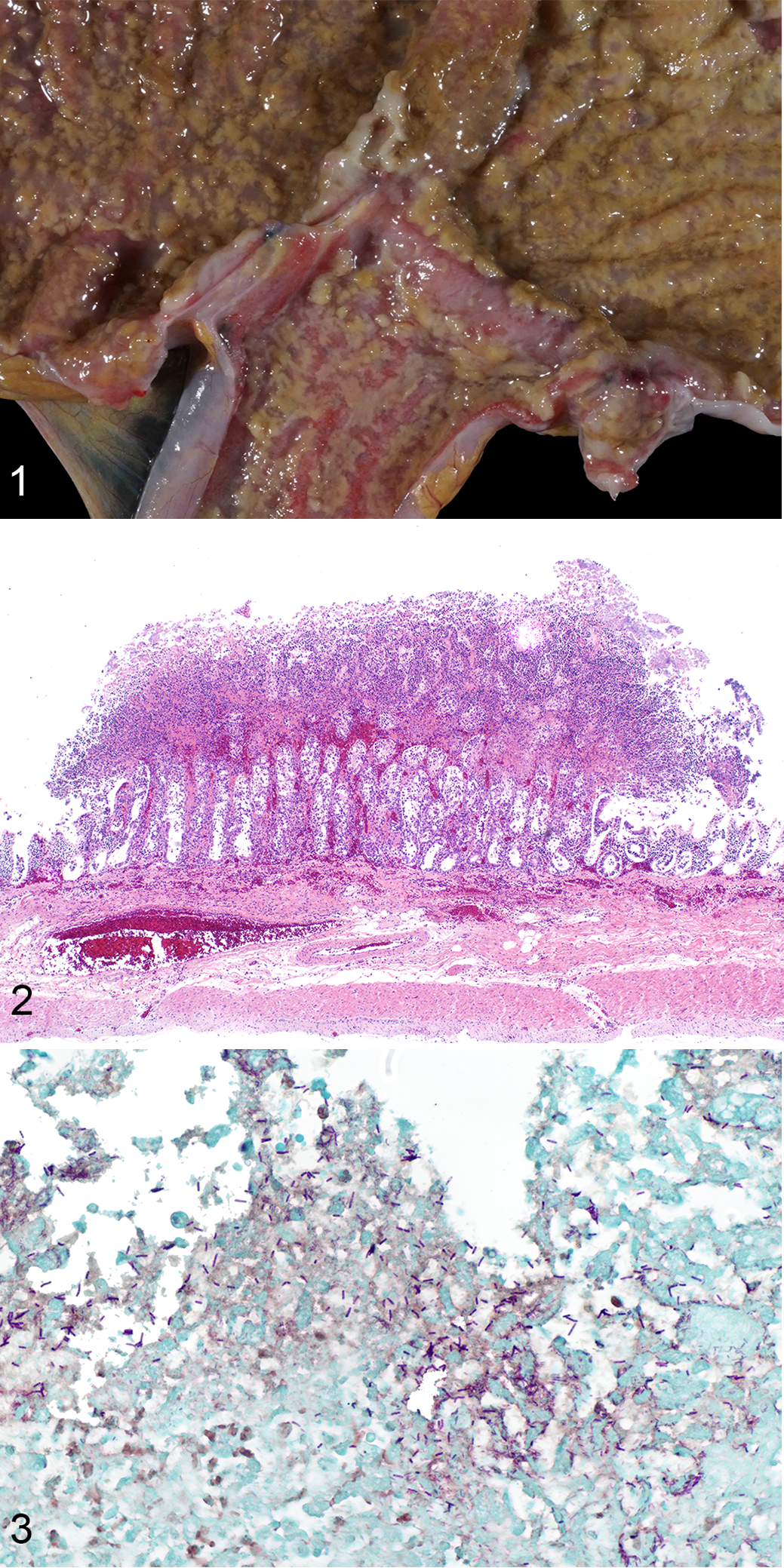
Clostridium difficile enterocolitis, cecocolic junction, spider monkey.
Histologically within the cecum and colon, there were “volcano lesions” composed of aggregations of sloughed epithelial cells, numerous degenerate and nondegenerate neutrophils, few erythrocytes, and fibrin within and extending from the underlying eroded epithelium and lamina propria into the lumen (Fig. 2). Along the luminal aspect of these aggregates, frequent individual and clustered, rod-shaped, straight, pale basophilic bacteria up to 10 μm long, occasionally with a central clear vacuole (oval spores), were admixed with cocci and shorter rod-shaped bacteria. The rods were gram positive (Fig. 3). The epithelium beneath the aggregates was partially to completely necrotic, with multifocal extension of inflammatory infiltrates composed of plasma cells, lymphocytes, and degenerate neutrophils into the lamina propria and underlying submucosa. Multifocal regions of partial-thickness necrosis with sparing of the crypts were present. Full-thickness necrosis of the mucosa was frequently present overlying the Peyer’s patches within the ileum and cecum. The lumina of several capillaries of the lamina propria and submucosa were occluded by thrombi.
C. difficile was isolated from the feces, though a C. difficile toxin A and B (TcdA and TcdB) enzyme-linked immunosorbent assay (ELISA; C. difficile toxin A/B II kit; Techlab, Blacksburg, VA) using frozen small intestinal content was negative for TcdA and TcdB. The C. difficile isolate was negative for the binding component of the binary toxin (cdtB) gene by polymerase chain reaction (PCR) performed at the Minnesota Department of Health, but the isolate was positive for the pathogenicity locus gene. 5,21 Due to high clinical suspicion, fecal testing was also performed on the diseased cohoused animals. Feces from 3 of the 4 cohoused animals was positive for C. difficile by culture and by C. difficile ELISA for toxin (C. difficile toxin A/B II kit; Techlab). 16S ribosomal RNA (rRNA) sequencing confirmed the agent as C. difficile. Aerobic culture, anaerobic culture (using media without reduced oxygen tension to isolate Clostridium perfringens), and Salmonella spp. enrichment culture of small intestine and colon failed to isolate significant agents. Fecal flotation yielded negative results and Cryptosporidium spp. and Giardia spp. were not detected by antigen-specific immunofluorescent antigen testing. Colonic contents were negative for viral particles based on transmission electron microscopic examination.
The second cluster of cases involved 19 members of a research colony of common marmosets (Callithrix jacchus) that presented between June 2015 and July 2017 with either acute and rapidly progressive fulminant diarrhea, weight loss, and failure to thrive over a period of days or with a prolonged clinical course of intermittent diarrhea, weight loss, and clinical decline over the course of several weeks to months. The animals were from several cohorts (different shipments and periods of time in housing) from a colony at a single facility. The 5 cases confirmed by PCR testing for toxins A and B included 3 females and 2 males that ranged from 1 to 4 years of age and had weight loss and/or chronic diarrhea that did not resolve with symptomatic treatments (ie, suggestive of recurrent CDAD). The marmosets were euthanized (n = 2) or found dead (n = 3). Two of the marmosets had been previously diagnosed with Giardia infection based on an ELISA (3 and 6 months prior to death, respectively) and were treated with tinadazole (62.5 mg per os [PO] followed by 31.25 mg 5 days later) with complete resolution of clinical disease based on a negative follow-up ELISA test 1 month after the positive result and at the time of necropsy. Two of the marmosets had tested positive for Campylobacter spp. on fecal culture approximately 6 months prior to necropsy but were asymptomatic at the time of testing and no treatment for Campylobacter infection was provided (1 of these animals included a Giardia-positive animal mentioned above).
The marmosets were necropsied by investigative, veterinary, and pathology staff at the University of Pittsburgh within the Laboratory Animal Resource Division, with formalin-fixed tissues at a minimum provided to the pathologists. Gross lesions in the marmosets were similar regardless of the clinical history and consisted of dilation of the colon, with a swollen, edematous appearance (Fig. 4). The colonic lumen often contained large amounts of clear, thick, gelatinous mucoid material (Fig. 5), which was adherent to the mucosa and could be removed in a distinct layer. Subjacent to this material, there were irregular patches of coalescing, cream-colored mucosal nodularity.
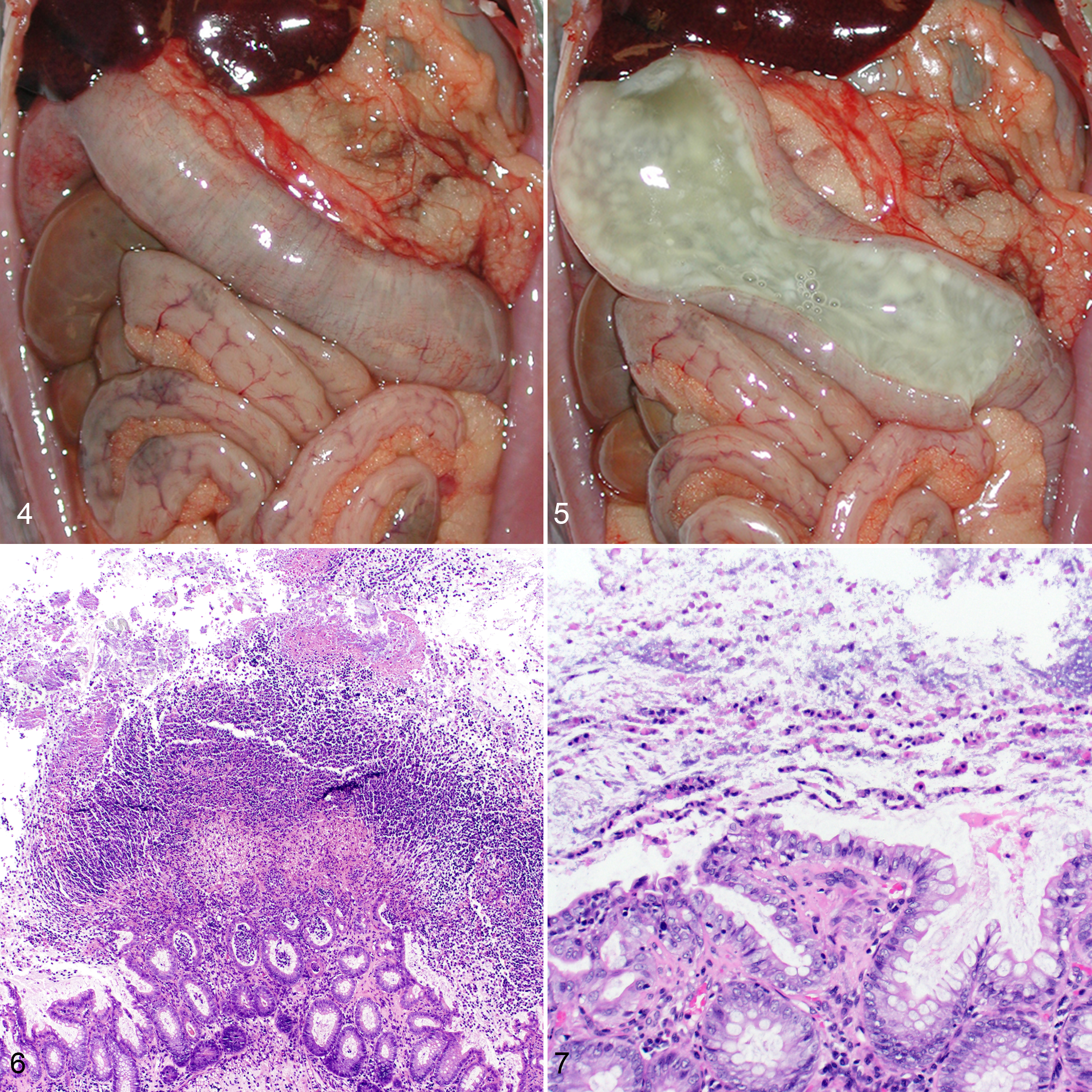
Clostridium difficile enterocolitis, colon, marmoset.
Histologically, cases were characterized by similar lesions to those observed in the spider monkey, with variation in distribution from distinct multifocal areas of mucosal necrosis (“volcano” lesions; Fig. 6) to more extensive, diffuse necrosis of the superficial mucosa, with a thick layer of fibrin, mucus, and leukocytes (predominantly neutrophils) admixed with nuclear debris along the mucosal surface. In several areas, a characteristic striated pattern was formed by columns of polymorphonuclear cells and mucin (Fig. 7). In addition, the submucosa and, to a lesser extent, the muscularis were often expanded by edema and scattered inflammatory infiltrates, with prominent dilations of the veins and lymphatics.
A full standard enteric pathogen workup was done for all marmosets, including a fecal culture panel for Salmonella spp., Shigella spp., Escherichia coli 0157: H7, and Campylobacter spp., as well as fecal flotation for pinworms and other parasitic disease. PCR testing of feces for clostridial toxins A and B was performed at a commercial laboratory (Zoologix, Chatsworth, CA). Of the affected marmosets, confirmatory PCR was available in 5 animals, identifying TcdA in 1 case and TcdB in 4 cases. All additional testing was negative and no comorbidities were identified.
The gross and histologic findings were consistent with the well-described characteristics of CDAD in humans and various animal species, notably including pseudomembranous colitis and characteristic histologic “volcano” lesions composed of neutrophils, fibrin, and necrosis in several cases. 16,18,20 While C. difficile–associated lesions have been reported in several animal species, the distribution and severity of the lesions within the intestinal tract can be variable by species and by age, with adult horses and neonatal piglets more often developing lesions in the cecum and ascending colon and foals more often developing small intestinal lesions. 7 In both the spider monkey and the marmosets, the cecum and colon were predominantly affected. The 1 previously published report of naturally occurring CDAD in nonhuman primates described lesions affecting the colon. 15
The traditional gold standard for identification of C. difficile is bacterial culture followed by a cytotoxin neutralization assay in cell culture that detects TcdB and, in some cases, TcdA. 1,11,14 PCR for the toxin genes is considered highly sensitive and specific for the genes encoding these toxins, and ELISA for TcdA and TcdB is highly specific for the presence of these toxins. 16 The spider monkey was positive by pathogenicity locus PCR of small intestinal content, with positive ELISA results for TcdA and/or TcdB from feces of clinically affected, cohoused animals. The marmosets were positive for either TcdA or TcdB by PCR. The diagnosis of CDAD in these cases was based on the presence of clinical disease and gross and histologic lesions in conjunction with the positive ancillary testing results.
TcdA and TcdB cause apoptosis of enterocytes and disrupt the cell-to-cell adhesion mechanisms, increasing permeability of mucosal surfaces. Considerable effort within the scientific community has been devoted to determining the relative contribution to disease of TcdA and TcdB, with strains producing TcdA alone, TcdB alone, and both TcdA and TcdB implicated in disease in various models. 3,6 Recently, an actin-ADP-ribosylating toxin has also been described in certain C. difficile strains (variably named C. difficile transferase, Clostridium difficile toxin [CDT], or binary toxin). The role of binary toxin in the pathogenesis of CDAD has not yet been elucidated. 6 One of the challenges of diagnosing CDAD is that toxigenic strains of C. difficile may be isolated from healthy humans and animals. 8,17 Identification of the presence of either TcdA or TcdB via ELISA is useful in confirming active disease, as toxin-negative strains are nonpathogenic, but the low sensitivity of available TcdA/TcdB ELISA tests precludes the toxin-detecting ELISA from being used as a stand-alone diagnostic for CDAD. 1,8,9,17 Confirmation of the toxin-related genes via PCR differentiates toxigenic from nontoxigenic strains but does not discriminate whether toxin is actually being produced. Therefore, this test has in rare cases been positive in asymptomatic carriers. 1,17 The cytotoxin neutralization assay is still considered the gold standard to confirm CDAD, given its ability to confirm cytotoxicity in cell culture, but its use has declined in light of increasing availability of faster, more easily performed tests. 1,16
The cases reported herein represent naturally occurring CDAD. Disturbance of the intestinal microbiome with overgrowth of toxigenic C. difficile is considered essential for development of CDAD. However, significant risk factors such as antibiotic use or extraordinary stressors (eg, construction near or in the exhibits/holding areas of the animals or changes in caretaker personnel) were not apparent in the present cases. The spider monkey and the marmosets had no history of antibiotic treatment within at least the 6 months prior to development of clinical disease attributed to C. difficile. The source of the infection remains uncertain, but asymptomatic carriers among the monkeys or their caretakers and intruding wildlife such as rodents in noncontained habitats may be considered. Strain analysis of the various human and animal populations involved in the outbreaks based on whole-genome sequencing, PCR-based toxinotyping, and ribotyping in future cases could help in clarifying the origin of the isolate in outbreaks in nonhuman primates.
Both outbreaks described herein underscore the importance of C. difficile on the individual and population level. While only the 5 marmoset cases with the most complete postmortem workups were included in our study, the affected colony has been experiencing significant CDAD-associated mortality (19 suspected cases) over a period of 3 years, and an effective preventive/eradication strategy has not yet been developed. Based on our findings, C. difficile is an important cause of enteritis in these species. Study of CDAD in primate colonies may further elucidate the pathogenesis and diagnostic and treatment modalities.
Footnotes
Acknowledgements
We thank the animal keepers and Dr. Micky Trent from the Como Park Zoo for providing the medical history of the spider monkey and Dr. Julia Oluoch at University of Pittsburgh for the care and provision of the medical history of the marmosets. We gratefully acknowledge the contributions of the University of Pittsburgh and University of Minnesota Veterinary Diagnostic Laboratory histology, bacteriology, and molecular diagnostic laboratories for their contributions, along with the Minnesota Department of Health. This research received no specific grant support from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
