Abstract
The prostate is the only accessory male genital gland described in cetaceans. Although few studies describe the gross and histologic anatomy of the prostate in cetaceans, there is no information on pathological findings involving this organ. The prostate glands of 45 cetaceans, including 8 different odontocete species (n = 44) and 1 mysticete, were evaluated. The main pathologic diagnoses were verminous prostatitis, septic prostatitis, viral prostatitis, benign prostatic hyperplasia, and prostatitis of unknown etiology. Verminous prostatitis (n = 12) was caused by nematodes of the genus Crassicauda, and different presentations were observed. Septic prostatitis, identified in 2 cases, both involved nematode infestation and Clostridium spp coinfection. One case of viral prostatitis was identified and was associated with morbillivirus infection. In prostatitis of unknown cause (n = 7), varying degrees of prostatic lesions, mostly chronic inflammation, were identified. Impacts at individual levels (eg, localized disease, loss of reproductive capacity) and population levels (eg, decreased reproductive success) are plausible. Our results indicate a high occurrence of prostatic lesions in free-ranging odontocetes. For this reason, the prostate should be routinely inspected and sampled during necropsy of odontocete cetaceans.
Keywords
The male reproductive system of cetaceans has unique morphologic and physiologic adaptations to aquatic life. These include intra-abdominal gonads, a retractile fibroelastic penis with a sigmoid flexure, and specialized vascular modifications (ie, arteriovenous countercurrent heat exchangers) that optimize hydrodynamics and ensure correct temperature for sperm production. 24 The prostate is the only accessory male genital gland in cetaceans and has undergone particular adaptations through evolution (eg, development of the prostate compressor muscle). 5
In men and terrestrial animals, the prostatic fluid is rich in citrate and zinc, which provides a nutritive and protective transporting medium for spermatozoids during ejaculation. 9,14 Lesions in the prostate therefore may lead to decreased viability of spermatozoids, causing infertility. 14,19
While few studies describe the gross and histologic anatomy of the prostate of dolphins, knowledge on pathological conditions is lacking in cetaceans. Furthermore, the existing literature is rather confusing and difficult to access, and images are scarce. 5,12,20,30 In delphinids, the prostate is located between the 2 vestigial pelvic bones in the penis root and supported by the ischiocavernosus and bulbocavernosus muscles. 5 The organ consists of a diffuse part and a lobular part, and it is surrounded by the prostate compressor muscle. 20
To fill in the gap of knowledge on cetacean prostatic pathology, this study aimed to describe, characterize, and categorize gross and histopathological findings involving the prostate of different species of odontocete cetaceans.
Materials and Methods
Data Recording and Autopsy Examination
For this study, we performed retrospective evaluation of prostatic tissues present in the Marine Mammal Tissue Bank (Cetacean Pathology Division, Institute of Animal Health [IUSA], University of Las Palmas of Gran Canaria) and prospective postmortem examinations of cetaceans stranded, including meticulous dissection and inspection of the prostate (January 2014 to December 2016). Animals examined included male free-ranging cetaceans live- and dead-stranded in the Canary Islands (n = 42) and in Mallorca (Balearic Islands; n = 1), as well as captive animals (n = 2). The inclusion criterion for this study was the evidence, grossly or histologically, of lesions in the prostatic tissue.
In all cases, a complete standardized autopsy was performed. Life history data (age class, nutritional condition, decomposition code, morphometry, stranding condition) were recorded systematically (Suppl. Table S1). Age class was determined attending to total body length and sexual maturity (morphologic and histologic appearance of the gonads), resulting in these categories: neonate, calf, juvenile/subadult, and adult. 11,13 Decomposition code was classified as very fresh, fresh, moderate autolysis, advanced autolysis, or very advanced autolysis. 11,13
A detailed description of the protocol used for prostate examination and sampling is provided in the Supplemental Materials. For microbiological analysis, fresh pieces or swabs of prostate gland were collected and submitted to an external laboratory for culture. For parasitological analysis, gross specimens were collected and preserved in 70% ethanol, and identification was based on morphological features.
Histopathology, Histochemistry, and Immunohistochemistry
Prostate tissue samples were fixed in 10% buffered formalin, embedded in paraffin wax, sectioned at 4 μm, and stained with hematoxylin and eosin. Periodic acid–Schiff (PAS), PAS-diastase, Masson’s trichrome, Gram/Twort, Alcian Blue, and Von-Kossa stains were used as needed. In suspected cases of morbillivirus or herpesvirus infection, immunohistochemistry (IHC) was performed in prostatic sections as previously described. 8,23
Results
The study included 45 prostates from 44 odontocetes and 1 mysticete. Of these, 21 (47%) had lesions and were reported in this study. Life history data are provided in Supplemental Table S1. Age categories were calf (4/21; 19%), juvenile/subadult (5/21; 24%), and adult (12/21; 57%). Prostatic lesions were observed in free-ranging animals stranded in the Canarian archipelago and the Balearic archipelago (Spain).
Based on gross and microscopic findings, prostatic lesions were classified into 5 categories: verminous prostatitis (n = 12), septic prostatitis (n = 2), viral prostatitis (n = 1), benign prostatic hyperplasia (n = 1), and prostatitis of unknown etiology (n = 7). Supplemental Table S2 summarizes the morphologic diagnoses and pathologic categories.
Verminous Prostatitis
Verminous prostatitis was diagnosed in 12 of 21 (57%) cases, including 5 striped dolphins (Stenella coeruleoalba), 4 spotted dolphins (Stenella frontalis), 2 short-beaked common dolphins (Delphinus delphis), and 1 bottlenose dolphin (Tursiops truncatus). Five (42%) animals were juvenile/subadult, 4 (33%) were adults, and 3 (25%) were calves.
Lesions were often observed on gross dissection of the prostate. Swelling and mucocutaneous edema and erythema of the genital folds were evident in case No. 4 (Fig. 1). Externally, the anogenital area of case No. 9 was bulging and firm. Main gross findings included multifocal, soft, moderately well demarcated, yellow nodules (Fig. 2) and, less frequently, multifocal mineralized foci in the penis root and prostate (case Nos. 7 and 9). In case Nos. 9 and 12, abundant suppurative exudate was observed on cut surface of the prostate and prostatic urethra.
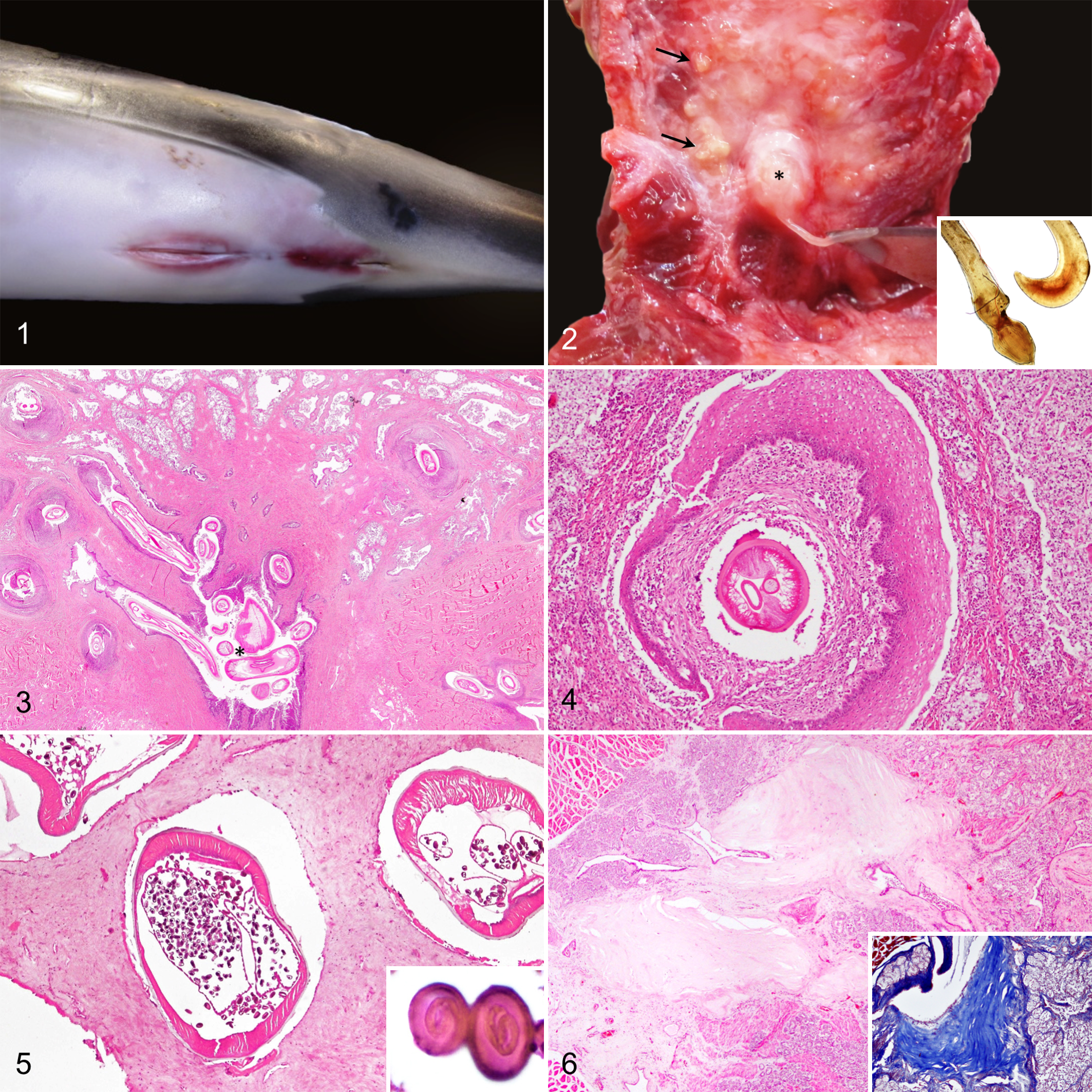
Histopathologic findings were dominated by an inflammatory response of varying severity, extent, and chronicity in association with intralesional (prostatic gland or urethral) adult nematodes, larvae, or eggs. In early lesions, nematodes appeared to reach the urethra and main prostatic ducts by passing through the penis root musculature, prostate compressor muscle, and glandular prostatic parenchyma (Fig. 3), causing necrosis with suppurative inflammation and frequent Splendore-Hoeppli material (n = 4). Neutrophils were seen associated with necrotic debris and infiltrating the prostatic interstitium. Occasionally, the epithelial lining of prostatic acini became metaplastic with squamous differentiation (Fig. 4).
In subacute/early chronic stages, the pyogranulomatous reaction predominated. Diffuse lymphoplasmacytic inflammation surrounded the parasitized main prostatic ducts and urethra. Frequently, the transitional epithelium of the urethra and main prostatic ducts underwent squamous metaplasia. Granulation tissue partially replaced the prostatic parenchyma.
In late chronic stages, the nematodes were mostly found in the urethra and main prostatic ducts. The reparative process was dominated by fibrosis, with intense replacement of the prostatic glandular parenchyma and discrete lymphoplasmacytic inflammatory foci within the remaining glands, prostate compressor muscle, periprostatic tissues, or the interstitium of the main prostatic ducts. Lymphoid follicle formation was observed in chronically inflamed areas in case Nos. 11 and 18. Other common findings included periductal fibrosis (Fig. 6), dilated prostatic ducts filled with desquamated epithelium and glandular secretion, and cysts lined by a simple cuboidal epithelium. Glandular atrophy and acinar replacement by granulation tissue were also seen in various cases (n = 4). In case Nos. 9 and 12, Crassicauda nematodes were found forming nests in the periphery of the gland. These nests consisted of 1 or various coiled nematodes surrounded by dense fibrous tissue, with minimal rimming inflammatory response (Fig. 5) or, occasionally, targeted by pyogranulomatous inflammation. Crassicauda eggs were frequently observed in the parasitic nest, within the glandular or periglandular parenchyma, or in the lumen of the urethra and main prostatic ducts. When located in the tissue, they were usually accompanied by an intense pyogranulomatous reaction. In case No. 20, they were observed in the glandular prostatic parenchyma without associated inflammation.
In all cases, intralesional nematodes were identified as genus Crassicauda, based on morphological criteria. 16,22 The nematodes varied in size and stages of development. They ranged from 300 μm to 1 mm in diameter. Immature life stages with no mature gonads were histologically evident in serial sections. Adult females had fully developed intrauterine larvated eggs (Fig. 5), while males had sperm. When present, the eggs were ovoid and measured 34 to 43 μm and typically contained a coiled embryo inside (Fig. 5, inset). In most cases, the terminal end of the nematodes was located in the lumen of the prostatic and/or the penile urethra, but nematodes were also found in the main and secondary prostatic ducts, glandular parenchyma, and periprostatic areas.
Septic Prostatitis
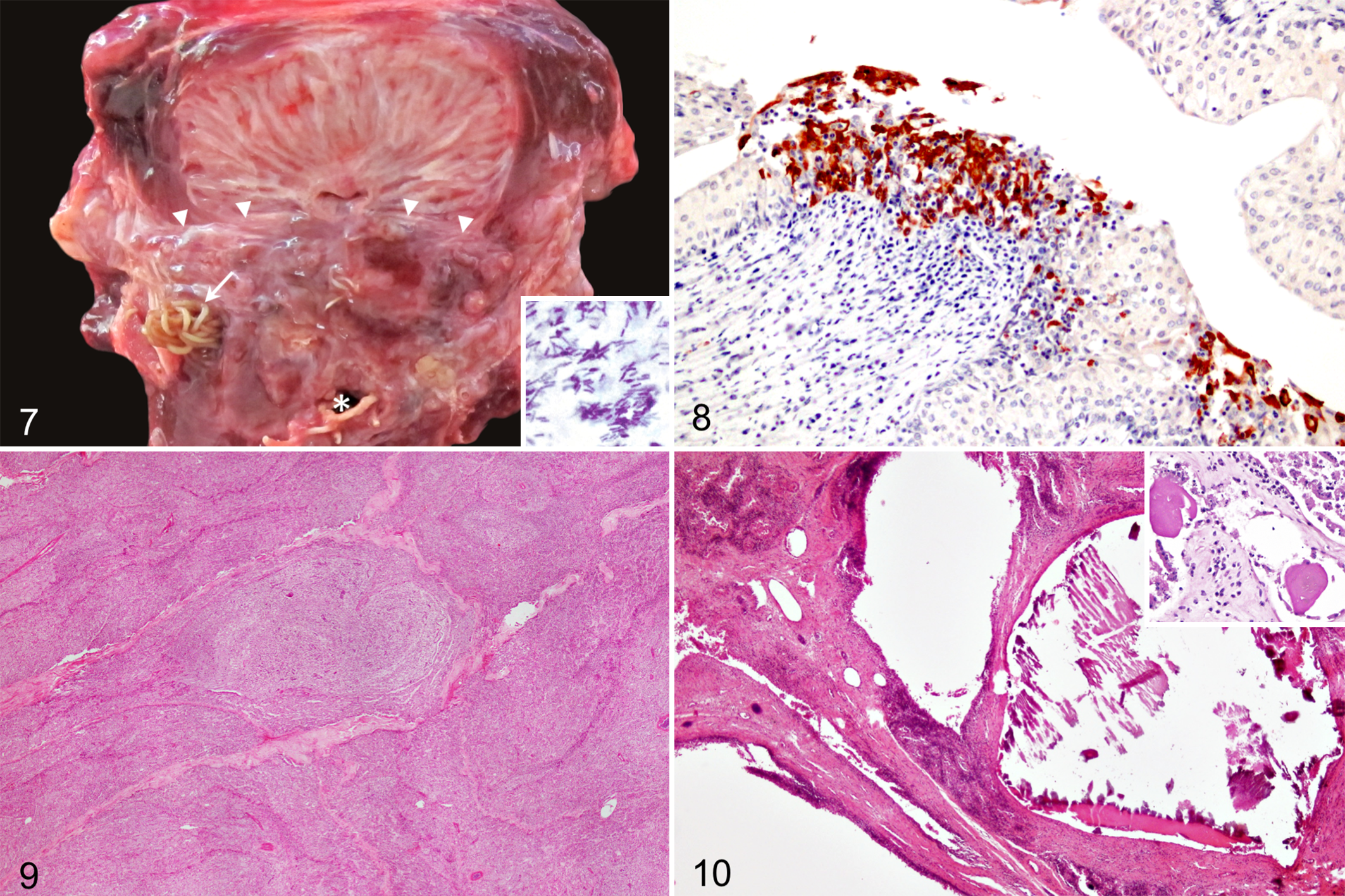
Septic prostatitis was diagnosed in case Nos. 9 and 12. In case No. 9, upon dissection of the urogenital area, suppurative exudate oozed from the preputial fold submucosa, muscles of the penis root, and prostate. In case No. 12, severe, focal, locally extensive necrosis and suppurative exudate of the penis root, prostatic urethra, and main prostatic ducts were observed (Fig. 7). Severe transmural necrosuppurative cystitis was also noted in this case. Both cases presented verminous prostatitis by Crassicauda sp concurrently. Histologically, numerous intralesional gram-positive bacterial rods were present in areas of neutrophilic inflammation in both cases (Fig. 7, inset). Clostridium sordellii and Clostridium perfringens were isolated from fresh prostatic tissue in case Nos. 9 and 12, respectively.
Viral Prostatitis
A short-finned pilot whale calf (Globicephala macrorhynchus; case No. 8) had discrete multifocal lymphoplasmacytic infiltrates in the glandular parenchyma, main prostatic ducts, and prostatic urethra. The glandular epithelium within inflamed areas, as well as the transitional epithelium of the main prostatic ducts, had multifocal single-cell necrosis and numerous 1- to 2-μm diameter intracytoplasmic, acidophilic inclusion bodies. Immunohistochemical analysis for morbillivirus antigen revealed strong intracytoplasmic and membranous positive immunolabeling in glandular prostatic epithelium, transitional epithelium of the main prostatic ducts (Fig. 8), and prostatic urethral epithelium.
Benign Prostatic Hyperplasia
Benign prostatic hyperplasia was diagnosed in an adult striped dolphin (case No. 1). Grossly, the prostate was diffusely enlarged, and on cut surface, multiple 1- to 3-mm diameter, white, poorly demarcated nodules expanded the glandular parenchyma. Microscopically, these nodules consisted of a proliferation of the glandular prostatic epithelium supported by a thin fibrovascular stroma (Fig. 9).
Prostatitis of Unknown Cause
Prostatitis of unknown etiology was diagnosed in 7 (33%) animals, including 3 spotted dolphins, 2 Cuvier’s beaked whales (Ziphius cavirostris), 1 common dolphin, and 1 pygmy sperm whale (Kogia breviceps). All these animals except case No. 10 were adults. In this category, inflammatory changes consistently involved the prostatic parenchyma and, to a lesser extent, the periprostatic tissues. Morbillivirus and herpesvirus were not identified by IHC performed on formalin-fixed, paraffin-embedded tissues.
None of these animals, except case No. 13, presented gross abnormalities in the prostate. Histologically, the most common pathologic finding was multifocal lymphoplasmacytic infiltrates within the prostatic parenchyma or surrounding the main prostatic ducts (n = 6). In case No. 3, multifocal eosinophilic infiltrates were also noted in the periphery of the main prostatic ducts. Lymphoplasmacytic prostatic urethritis was observed in case Nos. 6 and 19. In case No. 13, the prostatic glandular interstitium was thickened and diffusely infiltrated by numerous lymphocytes and plasma cells. The glandular acini were atrophied and extensively replaced by fibrous tissue. Multiple corpora amylacea were observed within the prostatic ducts (Fig. 10, inset). In addition, this case contained numerous 125- to 500-μm cystic dilations lined by a simple cuboidal to flattened epithelium that expanded the prostatic parenchyma (Fig. 10). These cysts often contained intraluminal Von-Kossa–positive mineral aggregates.
Discussion
This study found a high occurrence of prostatic pathology (47%) in free-ranging cetaceans. Verminous prostatitis, involving nematodes of genus Crassicauda, was the most prevalent prostatic condition (57%). Other pathological categories were septic and viral prostatic infections, benign prostatic hyperplasia, and prostatitis of undetermined etiology.
The genus Crassicauda, including 14 different species, parasitizes cetaceans exclusively 7,16 and typically affects soft tissues. 1,2,4,16,17,18,28 Crassicauda has been observed in the subcutis, skeletal and diaphragm muscle, pleurae, mammary gland, and cranial sinuses of different mysticete and odontocete cetaceans, often associated with a localized suppurative to granulomatous inflammatory reaction. 2,4,28 Crassicauda boopis has been described in the kidneys, renal veins, colonic, cranial mesenteric and renal arteries, aorta, mammary gland, and lungs of mysticetes, 16 whereas C. crassicauda infects the lower urinary tract in the same species. 17 Although in most cases, Crassicauda parasitosis appears to bear little pathogenic significance, fatalities have been identified, including C. boopis in mysticetes 16,17 and Crassicauda sp in Cuvier’s beaked whales. 7
The life cycle of Crassicauda is unresolved. Case Nos. 6 and 9 were calves in which Crassicauda nematodes were found also in the epaxial musculature. We have commonly observed migrating nematode larvae in lymph nodes and kidneys in cetaceans with Crassicauda sp infestation. Although final identification of these migrating larvae has not been achieved, they present a similar aspect to those found in larvated eggs of Crassicauda in this work. The fact that adult parasites were also found in very young animals supports Lambertsen’s hypothesis 17 of vertical transmission of Crassicauda. In our experience, mastitis due to Crassicauda is very frequent in delphinids from the Canary Islands. We have observed parasitism even in very young animals, in which the mammary gland was not fully developed, as this work reports in the prostate of the male counterpart. Crassicauda might have a predilection for apocrine epithelium in dolphins, as we found more nematodes in the prostate and prostatic urethra than in other tissues. This may represent an evolutionary adaptation to improve transmissibility through milk, semen, and urine. In the most advanced phases of verminous prostatitis, Crassicauda was found in the urethra and main prostatic ducts, associated with only minimal inflammation. Immune evasion may be another potential explanation for such anatomical tropism of the parasite.
Septic prostatitis was diagnosed in 2 spotted dolphins. In these cases, necrosuppurative inflammation of the prostate and periprostatic tissues was observed in association with numerous intralesional gram-positive rods. Both individuals also had heavy prostatic and urethral Crassicauda infestation. In case No. 12, there was also severe ascending necrosuppurative cystitis. In these cases, nematode parasitism might have favored bacterial colonization via mechanical transmission or trauma, as reported in conditions such as bacillary hemoglobinuria or infectious necrotic hepatitis in domestic ruminants. 3 Ascending bacterial prostatitis is not uncommon in dogs and humans, with Escherichia coli most frequently involved. 9,14
To our knowledge, case No. 8 represents the first report of viral prostatitis involving morbillivirus in a cetacean. This animal had systemic morbillivirus infection, as suggested by histopathological, IHC, and molecular analyses. Pilot whale morbillivirus strain was identified by real-time reverse transcription (RT)–polymerase chain reaction (PCR) in the brain, pharynx, lung, multiple lymph nodes, and testicles. 26 Furthermore, morbillivirus antigen was detected by IHC in testicle and epididymis. These findings suggest potential sexual transmission of morbillivirus, as suspected previously. 27 In humans, few cases of viral prostatitis have been described, mostly involving immunosuppressed individuals with secondary cytomegalovirus, herpesvirus simplex, or Zika virus infection. 6,21,25,31 In mice, cytomegalovirus and mouse mammary tumor virus have been detected in prostatic tissue by electron microscopy but not linked with prostatitis. 6
In this study, case No. 1 represents, to our knowledge, the first description of benign prostatic hyperplasia in cetaceans. It involved an aged striped dolphin based on gross and histomorphologic features (eg, total body length, wear and loss of teeth, widespread neuronal lipofuscinosis, and multiorgan arteriosclerosis and arteriolosclerosis). Benign prostatic hyperplasia (BPH) is frequent in men and dogs. 9,14 Although the pathogenesis of BPH is not fully resolved, androgens are considered to play a fundamental role. Estrogens may also elicit prostatic hyperplasia. 9 In man, a common associated sequela of BPH is urinary obstruction, whereas in dogs, constipation due to rectal compression is most common. In case No. 1, hyperplastic prostatic nodules were considered mild, so neither urinary nor rectal obstruction were likely.
Most cases with prostatitis of unknown etiology involved adults. Histologically, most cases of prostatitis of unknown etiology had multifocal lymphoplasmacytic inflammatory infiltrates of the gland or diffuse lymphoplasmacytic infiltration surrounding the prostatic urethra or main prostatic ducts. Immunohistochemical investigation for morbillivirus or herpesvirus was negative. The etiology in these cases remains unknown; however, resolved parasitism or chronic antigenic stimulation from lower urinary tract pathogens are possibilities. No intact nematodes or nematode cuticle debris were noted in the prostate or urinary tract of these individuals. Case Nos. 10 and 14 were Cuvier’s beaked whales with severe chronic fibrosing arteritis and granulomatous nephritis, lesions associated with Crassicauda sp and C. magna infestation in this species. 7 In case No. 13, an adult pygmy sperm whale, Crassicauda nematodes were found in the “false gill” (a glandular organ situated in the cervical region in this species). This seems to be the definitive location for the parasite in the pygmy sperm whale. 1 Prostatic changes in this case were characterized by multiple intraprostatic cysts and marked diffuse glandular atrophy. In humans, prostatic atrophy is considered a senile change, 14 whereas in domestic animals, the most common cause is deprivation of androgenic stimulus due to castration. Prostatic atrophy has also been observed in geriatric intact dogs. 9 Multiple corpora amylacea, interpreted as an incidental finding in this case, are frequent in the prostate of humans and animals and are not necessarily linked to prostatic disease. 9,14 Several contaminants (eg, flutamide, 1,1-dichloro-2,2-bis(p-chlorophenyl)ethylene [DDE]) may alter prostatic function, in some cases causing prostatitis. 32 No toxicological analyses were performed in the animals of this study; however, high levels of persistent contaminants, including DDE, have been detected in free-ranging dolphins of the Canary Islands. 10 Future studies focused on potential detrimental effects of xenobiotics on reproductive tissues may help to elucidate their role in prostatitis of unknown etiology in cetaceans.
In humans and animals, the prostate plays a significant role in spermatic viability by providing nutrients and transportation media during ejaculation. 9,14 In men, prostatic diseases contribute to spermatic deterioration due to reduction of antioxidant properties of the seminal fluid. 19 The consequences of prostatic lesions in cetaceans remain unknown, but they could negatively affect reproduction and population health. In those populations of cetaceans that fail to recover without a recognized explanation, such as the beluga whales of the St Lawrence estuary and the southern resident killer whale population, 15,29 male fertility should be assessed, including evaluation of the prostate.
In summary, our results document a high occurrence of prostatic lesions in free-ranging odontocetes. Thus, the prostate should be routinely inspected during autopsy of odontocete cetaceans. Negative impacts at the level of the individual (eg, localized disease, loss of reproductive capacity) and population (eg, decreased reproductive success) are plausible and merit further investigation.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818755252 - Prostatic Lesions in Odontocete Cetaceans
Supplemental Material, DS1_VET_10.1177_0300985818755252 for Prostatic Lesions in Odontocete Cetaceans by Cristian M. Suárez-Santana, Eva Sierra, Josue Díaz-Delgado, Daniele Zucca, Yara Bernaldo de Quirós, Raquel Puig-Lozano, Nakita Câmara, Jesús De la Fuente, Antonio Espinosa de los Monteros, Miguel Rivero, Manuel Arbelo, and Antonio Fernández in Veterinary Pathology
Footnotes
Acknowledgements
We thank all the people who indirectly participated in this work, including volunteers and personnel of the Government of Canarias. We greatly acknowledge Ana Afonso Almeda and Idaira Felipe Jiménez for technical assistance, the partner nongovernmental organizations SECAC and Canarias Conservation, and Binter Canarias. This study is part of a PhD program supported by the University of Las Palmas of Gran Canaria through a predoctoral fellowship for students (Contrato Predoctoral Convocatoria del 2012 programa propio de la ULPG, BOULPGC Año VI num. 6). Partial funding came from the national project “Embolic Pathology in Cetaceans” (ref. CGL2015-71498-P).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
