Abstract

Keywords
It wasn’t very long ago that the only disease of amphibians that students might hear about in veterinary school was “red leg syndrome,” attributed to infections with the bacterium Aeromonas hydrophila. And perhaps if you were a pathology resident, you would learn about a few other conditions—especially for the board examination—such as the classic Lucké renal adenocarcinoma in northern leopard frogs caused by an oncogenic herpesvirus (Ranid herpesvirus-1) or mycobacteriosis, which unfortunately in African clawed frogs (Xenopus laevis) can histologically mimic a lymphoma well enough to confound research studies. But otherwise, except for the work of a few specialists, the study of amphibian disease did not receive a lot of attention—at least in comparison to other vertebrate groups.
This would begin to change with recognition of the “Amphibian Extinction Crisis.” At the First World Congress of Herpetology in 1989, informal discussions about mysterious disappearances of previously thriving populations of frogs and salamanders revealed a global problem on a scale that has been estimated at 200 times the historical background rate of extinction for amphibians. 4,14,20 A 2004 assessment by the International Union for the Conservation of Nature (IUCN) estimated that nearly one-third of all known amphibian species were threatened and over 400 (7.4%) are critically endangered—on the brink of extinction—compared to 1.8% of birds and 3.8% of mammals. 19 Like for so many other species, factors such as habitat loss could be implicated; the most concerning population declines were termed enigmatic, occurring in areas with seemingly pristine habitat and some of the richest amphibian biodiversity.
The role of emerging infectious diseases (EIDs) in these enigmatic amphibian population declines is bringing amphibian pathology into the forefront and beyond red leg syndrome. The most important of the EIDs in regard to amphibian population declines is chytridiomycosis caused by the zoosporic fungus, Batrachochytrium dendrobatidis or Bd for short (Fig. 1). 1,4,13 Discovered in declining populations of stream-dwelling amphibians in high-elevation locations in Central America and Australia, Bd is a generalist pathogen that when introduced to new geographic locations with susceptible species causes mass mortality events and rapid population crashes. Chytridiomycosis has been described as the “most devastating disease on record to impact vertebrate biodiversity” and has changed perceptions on the potential of infectious diseases to contribute to population declines and even species extinctions. 1
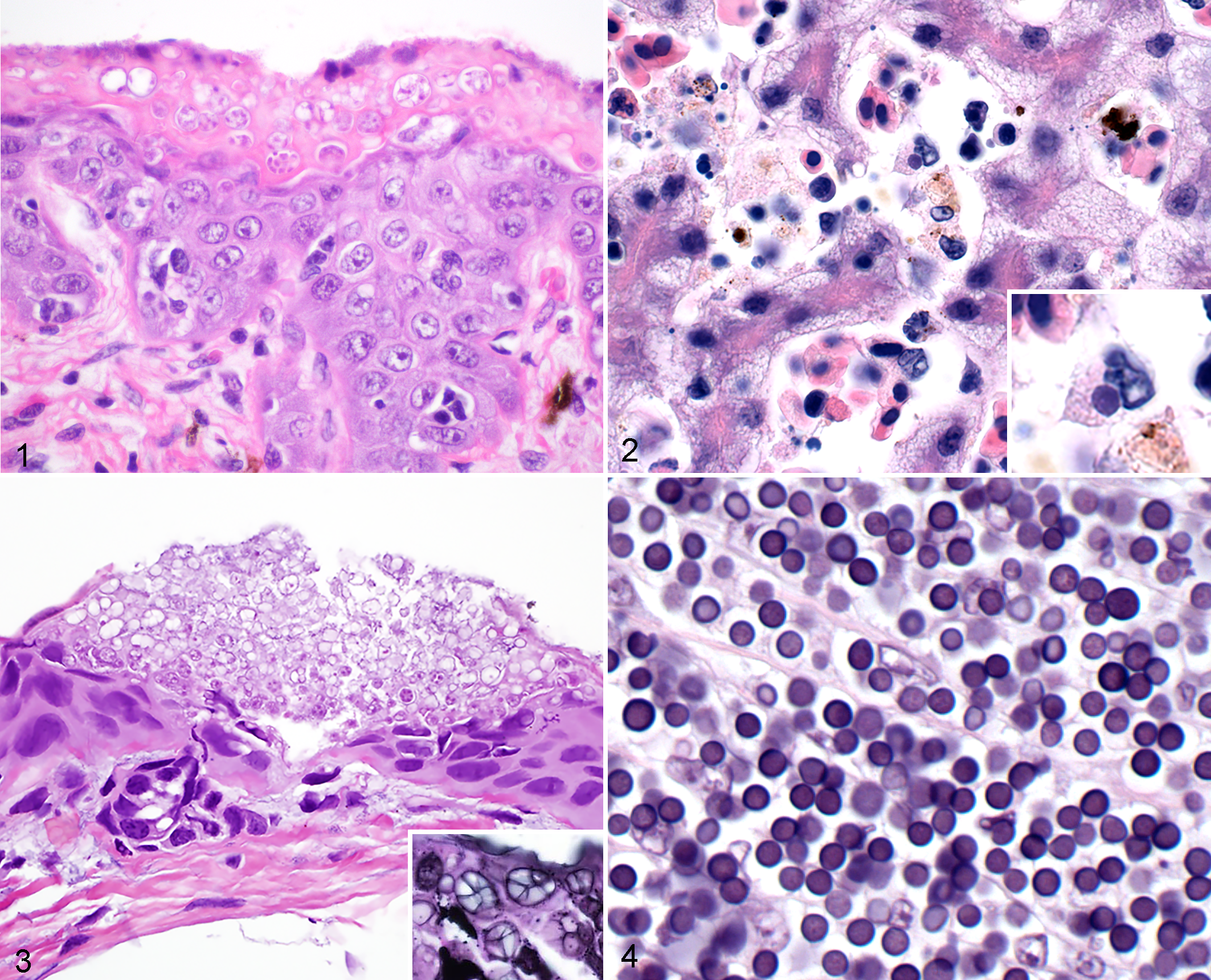
Chytridiomycosis due to Batrachochytrium dendrobatidis, skin, Wyoming toad (Anaxyrus baxteri). There is epidermal hyperplasia and orthokeratotic hyperkeratosis with numerous intracorneal chytrid fungal thalli. Hematoxylin and eosin (HE).
In this issue of Veterinary Pathology, Forzán et al, using experimental infections of a Ranavirus, Frog Virus 3 (FV3), in wood frogs (Rana sylvatica), provide a new and well-characterized model that advances our understanding of the other major EID in amphibians. 8 The ranaviruses were discovered in the mid-1960s as part of efforts to characterize the herpesvirus-associated renal carcinomas, but for a long time their association with natural disease was limited to American bullfrog (Lithobates catesbeianus) tadpoles with generalized edema and hemorrhagic lesions grossly resembling red leg syndrome. 22 In the 1990s, Ranavirus-associated mass mortality events in native salamanders and frogs from North America 4,9 and common frogs (Rana temporaria) 5 from Europe significantly raised the profile on these viruses. Despite the observation of mass mortality events, ranaviruses are not as clearly linked to amphibian population declines as chytridiomycosis. However, there are legitimate concerns about the impact of ranaviral mass mortality events on small amphibian populations—many endangered species exist only in a few isolated locations—and the potential for more generalist ranaviruses such as the common midwife toad ranavirus (CMTV) to cause declines across a broader range of amphibian species. 7,16 Further concerns include the ability of ranaviruses such as FV3 and the Bohle iridovirus (BIV) to cause mortality events in reptiles and fish and infections of endangered amphibians held in captive “survival assurance populations” as part of conservation efforts. 3,12,21 The work of Forzán et al provides guidance to veterinary pathologists on recognizing the sometimes subtle lesions of ranaviral infection (Fig. 2) as well as new insights into approaches for diagnostic sampling—most notably, the poor performance of oral, skin, and cloacal swabs for PCR detection of early preclinical infections, an approach that has seen wide empirical use for disease surveillance of amphibian populations.
The emergence of chytridiomycosis and ranaviriosis has resulted in an explosion of amphibian disease research and pathogen discovery. Recently, a second species of chytrid fungus, Batrachochytrium salamandrivorans, was identified as the cause of mortality and population declines in European salamanders with attendant concerns about movement of this newly recognized pathogen to new locations via amphibian trade (Fig. 3). 18 Other infectious diseases receiving recent attention include unnamed alveolate protozoa responsible for mortality events of ranid tadpoles in North America (Fig. 4), 2 hepatitis and encephalitis in Australian native frogs due to the myxzoan parasite Cystodiscus axonis, 10 infections with mesomycetozoa (organisms at the animal-fungal divergence) such as Amphibiocystidium and Ichthyophonus-like organisms, 17 and a report that imported salamanders were harboring spring viremia of carp virus. 11
Finally, as mentioned by Forzán et al, there is anecdotal evidence that many historical mortality events of wild amphibians attributed to red leg syndrome—usually based on gross observation of erythematous skin combined solely with bacterial isolation—were instead outbreaks of other diseases such as ranaviriosis. And indeed, secondary bacterial infections are common with ranaviral disease. 5,15 Today, there are similar pitfalls with the wide use of PCR assays that allow anyone to easily detect ranaviral or chytrid fungal DNA during amphibian mortality events but have the great potential to miss other significant (or new) disease problems if not used as part of comprehensive disease investigations that include methods such as histopathology. 6 Therefore, the need for veterinary pathologists to direct and or provide support for studies into amphibian disease has never been greater.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
