Abstract
Twenty cattle (14 Holstein-Friesian, 3 Japanese Black, 3 Aberdeen Angus) ranging in age from 3 months to 8 years exhibited, at slaughter, emphysematous thickening of the ruminal submucosa owing to the appearance of numerous, contiguous, small gas bubbles. Microscopic changes in the ruminal submucosa consisted of (1) multiple cystic (emphysematous) lymphangiectasis that was frequently lined or occluded by granulomatous inflammatory infiltrates including macrophages, multinucleate giant cells, and eosinophils; (2) intralymphatic phagocytosis by macrophages and giant cells of eosinophils that showed positive labeling with the terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end-labeling assay; and (3) an inflammatory infiltrate extending from the area of lymphangitis into surrounding tissue, as well as edema, hemorrhage, fibrin exudation, fibroplasia, or capillary proliferation throughout the lesional submucosa. In addition, 15 (75%) of the cattle had globular leukocyte infiltrates in the mucosal epithelia of the rumen.
Twenty cattle (case Nos. 1–20), which originated from 20 farms, were slaughtered conventionally at an abattoir over a 10-month period. Among these cattle, 14 were Holstein-Friesian (70%), 3 Japanese Black (15%), and the remaining 3 Aberdeen Angus (15%). The age ranged from 3 months to 8 years (mean, 2.8 years; 1 male, 5%; 12 castrated males, 60%; and 7 females, 35%). In the rumen of each animal, a whitish opaque area was recognized through the serosal surface, which appeared to develop along the course of blood vessels and varied in extent among animals from 20 × 20 cm diameter to approximately one-half of the dorsal (or ventral) sac of the rumen. The affected area was more easily identified when the rumen wall still maintained its own tension than when it was examined after opening the rumen. It proved, on cut surfaces, to be emphysematous thickened submucosa that displayed a spongy appearance due to numerous, contiguous gas bubbles <3 mm in diameter, distorting the normal submucosal architecture. Gas bubbles were enclosed by a very thin, smooth wall and easily ruptured by digital pressure, discharging a small amount of gas and/or translucent fluid. The gaseous contents were colorless, odorless, and possibly pressurized because they appeared to distend the surrounding submucosal tissue. In general, the mucosal surface overlying lesional submucosa revealed a rough appearance due to irregular arrangement, discoloration, or atrophy of the ruminal papillae, some of which were firmly matted together by compostlike rumen contents. In 3 cattle (case Nos. 2, 7, 12), the rumen contents appeared to be frothy and voluminous. Other forestomachs, abomasum, and intestine were unremarkable.
In 10 cattle (case Nos. 1–5, 13, 14, 16, 19, 20), the pH values of rumen fluid—calculated within 40 minutes of death by use of a pH meter with an electrode—ranged from 6.0 to 7.3, estimated to be nearly within reference intervals. 7 Within 1 hour of death, tissue samples or swabs for bacterial culture were obtained aseptically from lesional ruminal submucosa. Samples from 11 (case Nos. 1–5, 13–16, 19, and 20) of 20 animals were plated on sheep blood agar and MacConkey agar and incubated aerobically and anaerobically. There was no significant bacterial growth. For histopathology, tissue samples were collected from the rumens of all 20 cattle. Samples were fixed in 10% buffered formalin and embedded in paraffin. Sections 4 µm thick were stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Mallory’s phosphotungstic acid–hematoxylin (PTAH), toluidine blue, alcian blue (pH 2.5), Gram, and Ziehl-Neelsen (ZN) stain. For in situ detection of DNA strand breaks, in situ TUNEL (terminal deoxynucleotidyl transferase–mediated deoxyuridine triphosphate nick end labeling) staining was applied on selected serial paraffin-embedded tissue sections. An in situ apoptosis detection kit (Takara Biomedicals, Shiga, Japan) was used for the TUNEL assay according to the manufacturer’s instructions. As controls, intact rumen tissues were taken from 20 cattle (2 to 8 years old; 10 Holstein-Friesian, 5 Japanese Black, and 5 Aberdeen Angus; 11 castrated males and 9 females) that originated from 20 farms different from those which raised the 20 cases associated with lesional rumens. Rumen tissue samples from the controls were processed by the aforementioned method, and prepared sections were stained with HE and toluidine blue.
Histopathology of the rumens of 20 cattle revealed a remarkably consistent series of lesions in the submucosa. Lesions were identified as multicystic lymphangiectasis accompanied by granulomas (Fig. 1). Dilated lymphatics were occasionally lined by a single layer of endothelial cells and contained either no fluid or a small amount of basophilic to eosinophilic serous fluid. No erythrocytes were recognized elsewhere in the lumens of such structures. Frequently or sometimes, the walls of dilated lymphatics were partially or totally lined by macrophages and multinucleate giant cells of the foreign body type. These granulomatous inflammatory infiltrates were frequently intermingled with a moderate to large number of eosinophils and partially to completely filled the lumens of dilated lymphatics (Fig. 2). ZN and Gram stains failed to reveal any bacteria in dilated lymphatics, macrophages, giant cells, or elsewhere. Instead, macrophages/giant cells had not only 1 or more vacuoles occasionally containing cell debris but also phagocytosed eosinophils that usually ranged in number from 1 to 2 per 1 macrophage/giant cell (Fig. 3). While these eosinophils still retained their characteristics to a sufficiently distinctive degree to allow differentiation from other cells of the granulocyte lineage by the identification of specific cytoplasmic granules, they displayed occasional pyknotic (shrunken) or fragmented nuclei. In addition, those eosinophils undergoing phagocytosis by macrophages/giant cells stained positively with TUNEL assay; labeling was confined to the nucleus and what appeared to be nuclear debris (Fig. 4). A marked lymphoplasmacytic infiltrate—intermixed with small numbers of scattered TUNEL-positive eosinophils, large numbers of TUNEL-negative (intact) eosinophils, and fewer macrophages—was present in the submucosal connective tissues, particularly around or adjacent to dilated lymphatics; however, there was no evidence of neutrophil infiltrates. Also present were moderate numbers of mast cells with cytoplasmic granules, which stained positively with alcian blue (pH 2.5) and metachromatically with toluidine blue. The numbers of mast cells were almost equal to or higher than those detected in intact submucosa, as well as in the ruminal submucosa of control cattle (n = 20). Mast cells were scattered throughout the lamina propria overlying lesional or intact submucosa. In addition, varying degrees of edema, hemorrhage, fibrin clot formation, fibrous tissue proliferation, or capillary proliferation were observed throughout the lesional submucosa. Arterioles that displayed fibrinoid degeneration of the tunica media, with or without mural erythrodiapedesis, were present in 2 cattle (case Nos. 3, 16). Veins/venules were unremarkable. Neither mycotic/protozoal infection nor parasitic infestation could be seen in the submucosa of the rumens.
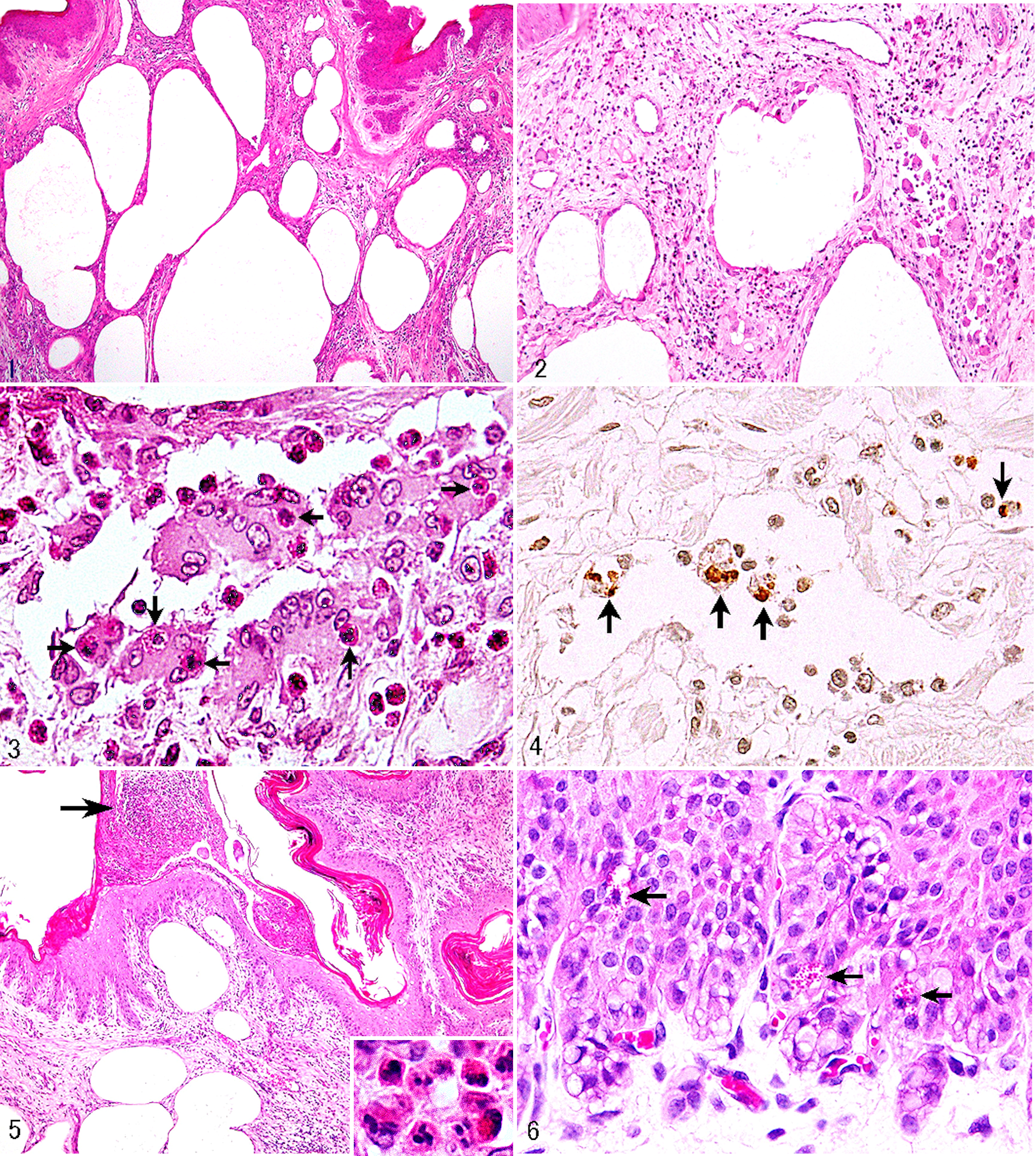
Emphysematous eosinophilic lymphangitis, cattle, rumen.
The ruminal mucosa revealed orthokeratotic or parakeratotic hyperkeratosis of the superficial layer of the stratified squamous epithelia in 7 cattle (case Nos. 2, 4–8, 16). Neutrophil-rich subcorneal pustules or microabscesses were disseminated in the epithelia of the ruminal papillae of 12 cattle (case Nos. 1, 2, 5, 8, 10, 11, 13, 15–18, 20) but did not always extend into the underlying submucosa (Fig. 5). In all 20 cattle, epithelial cells showed vacuolar degeneration, most evident in or restricted to the deep epithelia. No viral inclusions were demonstrated. A variety of these nonspecific, albeit less extensive, epithelial changes were seen in the rumens of 14 (70%) of 20 control cattle. With the exception of 5 (case Nos. 1, 2, 6, 16, 20), the other 15 cattle had globular leukocytes, which contained large numbers of approximately 1- to 1.5-µm round cytoplasmic granules and infiltrated into the epithelia of the ruminal mucosa (Fig. 6); in general, these cells were found in the deep epithelia. Cytoplasmic granules were brilliant red with HE and stained brownish black with PTAH. PAS staining affinity was positive. Toluidine blue staining failed to demonstrate a metachromatic nature to these granules. The numbers of globular leukocytes in the rumen of these 15 cattle and control cattle (n = 20) differed strikingly, none being found in the latter.
The present study reports spontaneous lesions of focal or extended accumulations of cystic dilations associated with eosinophil infiltrates and granulomas in the ruminal submucosa of cattle. These dilations proved to be multiple cystic (emphysematous) lymphangiectasis. The lesions had many similarities with the condition documented for intestinal emphysema in pigs, 1,4,9 in which emphysematous lymphangiectasis with eosinophilic granulomatous lymphangitis is a hallmark change, as in the condition described here, and production of gas by bacteria has been tentatively implicated. In addition, the lesions were superficially similar to those described in emphysematous abomasitis due to clostridial infection in calves 10 and lambs. 3 However, no microbial agents capable of producing gas were found histologically or cultured from the rumen lesions. The ruminal submucosa had neither neutrophil infiltrates indicative of bacterial infection nor changes due to mycotic, protozoal, or parasitic infection; in addition, no underlying infectious disease was recognized at slaughter in any of the 20 cattle. Seven (35%), 12 (60%), and 20 (100%) cattle exhibited hyperkeratosis of the ruminal epithelia, focal suppurative rumenitis, and epithelial degeneration of the ruminal mucosa, respectively. However, it is considered unlikely that these nonspecific epithelial changes in the rumens by themselves allowed gas passage through the lamina propria into the submucosa because of the presence of similar epithelial changes in 70% of control cattle (n = 20). Further molecular and chemical approaches are warranted to identify the gaseous components within emphysematously dilated lymphatics.
It should be noted that the rumens examined had histopathologic characteristics favoring hypersensitivity reactions, including eosinophilic, mastocytic, lymphoplasmacytic, and granulomatous infiltrates in the submucosa that concurrently represented evidence suggestive of an increased vascular permeability, such as edema, fibrin exudation, or erythrodiapedesis. Among these, not only do mast cells play a critical role in the induction of inflammation, but eosinophils have also been regarded as being effector or immunoregulatory cells in hypersensitivity-related conditions. 12 Furthermore, there were 2 episodes that may suggest an immune-mediated mechanism for the development of specific lymphangiocentric granulomatous lesions in the ruminal submucosa: one was the identification of globular leukocytes through conventional (HE, PAS, PTAH) staining techniques; the other, apoptotic eosinophils via the TUNEL assay. First, globular leukocytes migrated into the ruminal epithelia of as many as 75% of 20 cattle, while the rumens of control cattle (n = 20) had no intraepithelial globular leukocyte infiltrates. This might suggest a significant relationship between the appearance of globular leukocytes and the induction of lymphangial inflammatory lesions. Indeed, it has been shown that increased numbers of globular leukocytes are most likely related to the immune response against gastrointestinal parasitism in cattle. 8 Second, macrophages and giant cells in the lumens of dilated lymphatics phagocytosed TUNEL-positive eosinophils, indicating a specific phenomenon related to removal of senescent eosinophils with nuclear DNA fragmentation suggestive of apoptosis. 2,11 Phagocytosis of apoptotic eosinophils by macrophages/giant cells may be a feature demonstrating the ultimate fate of this type of granulocyte. 2,5,6,11,13 Apoptosis is recognized as an important mechanism for the selective elimination and phagocytic removal of eosinophils, promoting the resolution and repair process of eosinophilic inflammation of allergic origin in humans, including asthma, seasonal allergic rhinitis, allergic sinusitis, and sinonasal polyps. 2,5,6,12,13
Because none of the 20 cattle exhibited clinical evidence of forestomach (rumen) dysfunction and because normal pH values of the rumen fluid, even if calculated postmortem, probably excluded ruminal acidosis/alkalosis, 7 it appears that this naturally occurring condition involving the bovine rumen is subclinical. However, the gross appearance of the ruminal papillae that exhibited irregular arrangement, discoloration, or atrophy associated with compostlike rumen contents in all 20 cattle—together with the presence of leukocytic epithelial infiltration, especially neutrophilic pustules, in 12 (60%) of 20 cattle—was suggestive of recent or acute rumen acidosis even if the pH at time of slaughter was not below the reference range.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
