Abstract
Cancers in humans and animals can be caused by viruses, but virus-induced tumors are considered to be poor sites for replication of intact virions (lytic replication). Fibropapillomatosis (FP) is a neoplastic disease associated with a herpesvirus, chelonid herpesvirus 5 (ChHV5), that affects green turtles globally. ChHV5 probably replicates in epidermal cells of tumors, because epidermal intranuclear inclusions (EIIs) contain herpesvirus-like particles. However, although EIIs are a sign of herpesvirus replication, they have not yet been firmly linked to ChHV5. Moreover, the dynamics of viral shedding in turtles are unknown, and there are no serological reagents to confirm actual presence of the specific ChHV5 virus in tissues. The investigators analyzed 381 FP tumors for the presence of EIIs and found that overall, about 35% of green turtles had lytic replication in skin tumors with 7% of tumors showing lytic replication. A few (11%) turtles accounted for more than 30% cases having lytic viral replication, and lytic replication was more likely in smaller tumors. To confirm that turtles were actively replicating ChHV5, a prerequisite for shedding, the investigators used antiserum raised against F-VP26, a predicted capsid protein of ChHV5 that localizes to the host cell nucleus during viral replication. This antiserum revealed F-VP26 in EIIs of tumors, thus confirming the presence of replicating ChHV5. In this light, it is proposed that unlike other virus-induced neoplastic diseases, FP is a disease that may depend on superspreaders, a few highly infectious individuals growing numerous small tumors permissive to viral production, for transmission of ChHV5.
Introduction
Cancers in humans 28 and wildlife 27 can be associated with a diversity of viruses, including papilloma- and polyomaviruses, herpesviruses, hepadnaviruses, and retroviruses. One of these cancers, fibropapillomatosis (FP), is a disease of marine turtles with a global distribution. 40 FP is most common in green turtles (Chelonia mydas), less so in loggerhead (Caretta caretta) and olive ridley (Lepidochelys olivacea) turtles, and rare in hawksbill (Eretmochelys imbricata), leatherback (Dermochelys coriacea), flatback (Natator depressus), and Kemp’s ridley (Lepidochelys kempii) turtles. 14 In Hawaii, FP is the most important cause of stranding in green turtles, the most abundant species of marine turtle in the archipelago. 6,11 Prevalence of FP in Hawaii peaked in the mid-1990s and has declined ever since for unknown reasons; 6,11 however, the disease continues to persist in the eastern United States. In green turtles in Florida 10 and Hawaii, 41 where most is known about the disease, FP results in external tumors that can impede vision and, if large enough, increase drag and impede locomotion. Green turtles also get tumors in various internal organs such as the heart, kidney, and lungs; 41 however, oral tumors that impede breathing are common in Hawaii 2,3 but rare in Florida. 5
There is strong evidence that a herpesvirus is closely associated with FP. Areas of ballooning degeneration with eosinophilic epidermal intranuclear inclusions (EIIs) had evidence of herpesvirus-like particles on electron microscopy, 18 turtles with FP from Florida but not Hawaii developed antibodies against undefined viral proteins in those inclusions, 16 specific herpesviral DNA was consistently detected by polymerase chain reaction (PCR) in external and internal tumors in affected turtles from Florida 23 and Hawaii, 34 herpesviral DNA has been localized to tumors in turtles from Puerto Rico using insitu hybridization, 20 and herpesvirus DNA was not detected in biopsies of normal skin by PCR. 33,34 In contrast, serological evidence for a causal role of herpesvirus has been less clear. Turtles with FP from Florida but not Hawaii developed antibodies against EIIs based on analysis using immunohistochemistry; 15 however, serologic assays using baculovirus-expressed ChHV5 glycoprotein H revealed seroconversion in turtles with and without FP in both FP-enzootic and FP-free areas, 17 suggesting that factors other than virus infection are necessary for tumor development.
Despite these data, we know little of the basic epidemiology of FP such as how ChHV5 is spread or how the virus interacts with tissues. The former could shed light on dynamics of virus transmission and could explain different regional patterns of disease in Hawaii versus Florida. The latter could explain pathogenesis and the role of the virus in tumor formation. For example, we suspect that shedding occurs through EIIs in the skin but know nothing about how often this occurs. The continuing inability to culture ChHV5 has prevented attempts to completely fulfill Koch’s postulates 42 and has impaired attempts to understand the pathogenesis. Inability to culture the virus makes access to viral antigens for development of diagnostic tests problematic. Other than PCR and in situ hybridization, there are no widely available laboratory tests to detect ChHV5. PCR 34 does not allow for visualization of gene expression at the cellular level, and in situ hybridization 20 is cumbersome and requires use of toxic reagents (formamide), although the specificity of current serological reagents to identify ChHV5 in tissues 15 is unknown. Reagents that could help trace development of the virus at the morphologic level would greatly aid our understanding of the pathogenesis and pathology of FP. The recent publication of the genome of ChHV5 1 has opened the door to developing F-VP26 and corresponding antibodies that could be useful in helping confirm presence of the virus in tissues.
Our objective was to use presence of EIIs in turtle tissues to (1) assess the dynamics of virus shedding in Hawaiian green turtles and (2) confirm the presence of ChHV5 in EIIs using antiserum to F-VP26, a capsid protein of ChHV5 that localizes to the host cell nucleus during viral replication. 36 We chose EIIs as an explicit and biologically meaningful index of virus shedding in this study because EIIs have been confirmed to show replicating whole virions by electron microscopy 19 that are present in skin epidermis, a cell layer known to shed herpesvirus in other animals like chickens. 30
Methods
Tissues originated from green turtles that had stranded in Hawaii from FP. Turtles underwent a complete external and internal examination including recording of straight carapace length (nearest 0.1 cm), carcass weight (nearest 0.1 kg), and size and location of tumors. Surface area of each tumor was estimated using the formula for a sphere or an ellipsoid for irregularly shaped tumors depending on whether length and width of tumors was equal or not, respectively. Tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Tumor sections were examined histologically for presence/absence of EIIs associated with ballooning degeneration indicative of herpesviral lytic replication 16,18 (Fig. 1). We used Spearman correlation to assess the relationship between number of tumors examined and the number EII-positive, and we used regression to test the relationship between tumor size and EII status after testing data for normality using Shapiro-Wilks test. All analyses were done with R. 35
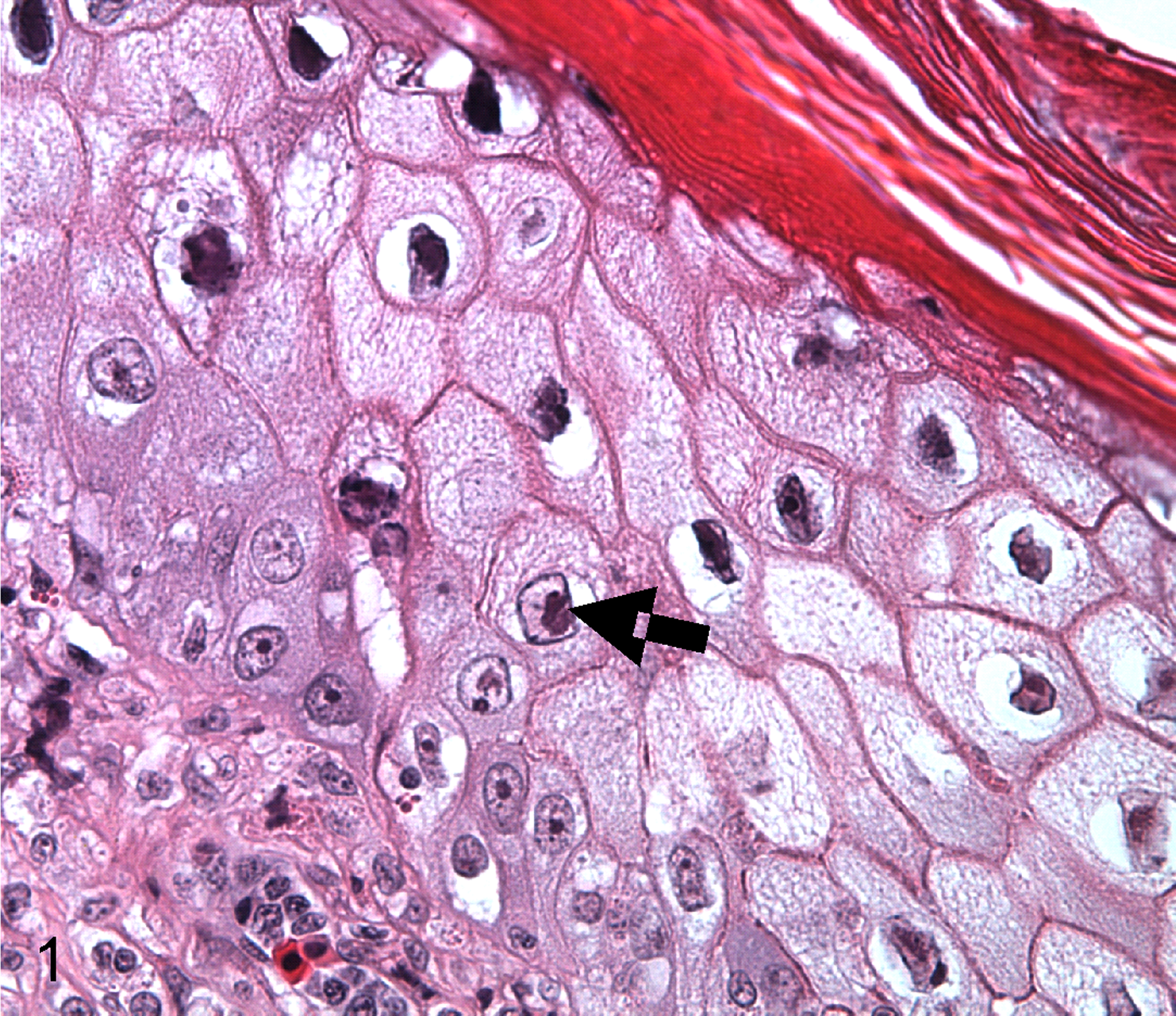
Fibropapilloma, skin, green turtle (Chelonia mydas). Ballooning degeneration of epidermal cells with intranuclear inclusions (arrow). Hematoxylin and eosin (HE).
Viral Shedding
To understand the dynamics of viral shedding among tumors and between turtles, most or all tumors were collected from a subset of 17 necropsied turtles with FP, and a single tissue section from each tumor was examined histologically for presence of EIIs. To understand the variation of virus shedding within tumors, selected individual tumors collected from an additional 3 turtles with FP were sliced into approximately 0.5-cm-wide sections, and the entire surface of all tumor slices was examined histologically for presence of EIIs.
Synthetic Peptides and Rat Immunizations
After preimmune sera were collected, rats were immunized by a commercial supplier (BioGenes GmbH, Berlin, Germany). Briefly, the animals were kept and handled according to internationally accepted animal use guidelines of the supplier and were vaccinated 4 times at 2-week intervals with 0.2 ml of an equimolar mixture of the 2 antigens (ie, synthetic peptides 10 mg/ml; 80% purity by high pressure liquid chromatography and mass spectroscopy) conjugated to bovine thyroglobulin (2 mg/ml) as carrier protein. The final bleed was taken at day 90. The sequences of the synthetic peptides were derived from the predicted amino acid sequence of F-VP26 (encoded by F-UL35) and complemented with an N-terminal cysteine to facilitate conjugation to the carrier protein. Specifically, the amino acid sequences were C-LQVGDQQELRRALA and C-GAMKVDPEATRDHG (Suppl. Fig. S1).
Protein Expression and Western Blot
The entire F-UL35 open reading frame without its stop codon was amplified by PCR (Gateway cloning system, Invitrogen) using attB-flanked primers (forward 5′-GGGGACAAGTTTGTACAAAAAAGCAGGCT
Upon emergence of strong fluorescence in EYFP-transfected cultures, the V5-transfected cells were collected by scraping and low-speed spinning, and protein lysates were extracted using sodium dodecyl sulfate buffer suitable for polyacrylamide gel electrophoresis. The proteins were then transferred to membranes by using the iBlot Dry blotting system (Invitrogen Ltd, Carlsbad, California) according to the manufacturer’s instructions. Membranes were probed with rat sera used at a dilution of 1:100. Mouse-anti-V5 monoclonal antibody (Invitrogen) was used at 1:5000. 12 The Vectastain (Vector Laboratories, Burlingame, California) Peroxidase ABC system was applied (supplier’s protocol) for visualization of the bands, making use of secondary biotinylated goat anti-rat IgG or horse anti-mouse IgG, respectively, and 4-chloro-1-naphthol as substrate. Controls included unrelated EYFP- and V5-fusion proteins, preimmune sera, and rat antiserum blocked with an equal volume of 4 mg/ml stock solutions of synthetic peptides for 15 minutes at room temperature (blocked antiserum used 1:100 dilution).
Immunohistochemistry
Skin tumors manifesting EIIs were used for immunohistochemistry. Slides were deparaffinized in xylene and rehydrated in ethanol series. Sections were then incubated for 30 minutes at 97°C in preheated EnVision FLEX target retrieval solution, pH9 (Dako), rinsed in EnVison FLEX wash buffer (Dako), blocked for 5 minutes with Envision FLEX block solution, washed briefly with buffer, and then blocked with 5% nonfat milk in borate buffer for 30 minutes. Sections were then incubated 30 minutes with rat anti-VP26 antiserum diluted 1:100 (in Dako antibody diluent), with preimmune serum, or with “blocked” rat serum. Sections were then washed, incubated 30 minutes with horseradish peroxidase conjugated goat-anti-rat serum (Cat. No. 3050-05; Southern Biotech, Birmingham, Alabama) diluted 1:1000 in Dako antibody diluent, washed twice, visualized with diaminobenzidine (Dako) for 4 minutes, counterstained with Vector hematoxylin QS solution (H-3404; Vector Laboratories) for 30 seconds, dehydrated with ethanol and xylene, and coverslipped with Cytoseal 60 (Richard-Allan Scientific, Kalamazoo, Michigan).
Results
Viral Shedding
Six of 17 turtles (35%) had EIIs in at least 1 tumor, with each turtle having anywhere from 0% to 37% of tumors EII-positive. Of 381 tumors examined from these 17 turtles, 27 (7%) were EII-positive (Suppl. Table S1). When present in a tumor, EIIs encompassed a single focus of epidermis no larger than about 1 mm in diameter. Estimated surface area of tumors ranged from about 3 to 500 cm2 (about 0.25-13 cm diameter). The frequency distribution of the sizes of tumors was skewed toward smaller tumors (Fig. 2a). When tumors with EIIs were quantified by location on the turtle (number of tumors examined or percentage EII-positive), EIIs were most often seen in the scutes (n = 4 or 25%), mouth (n = 19 or 16%), neck (n = 78 or 8%), eye (n = 42 or 7%), flippers (n = 214 or 6%), or oral cavity (n = 17 or 6%), with none in the tail (n = 7). Smaller tumors were significantly (F = 8.3, P < .05) more likely to be EII-positive (Fig. 2b). There was no evident seasonal pattern to virus shedding (Suppl. Table S1). The number of tumors examined did not significantly affect the likelihood of detecting EIIs (R = 0.16, P > .05).
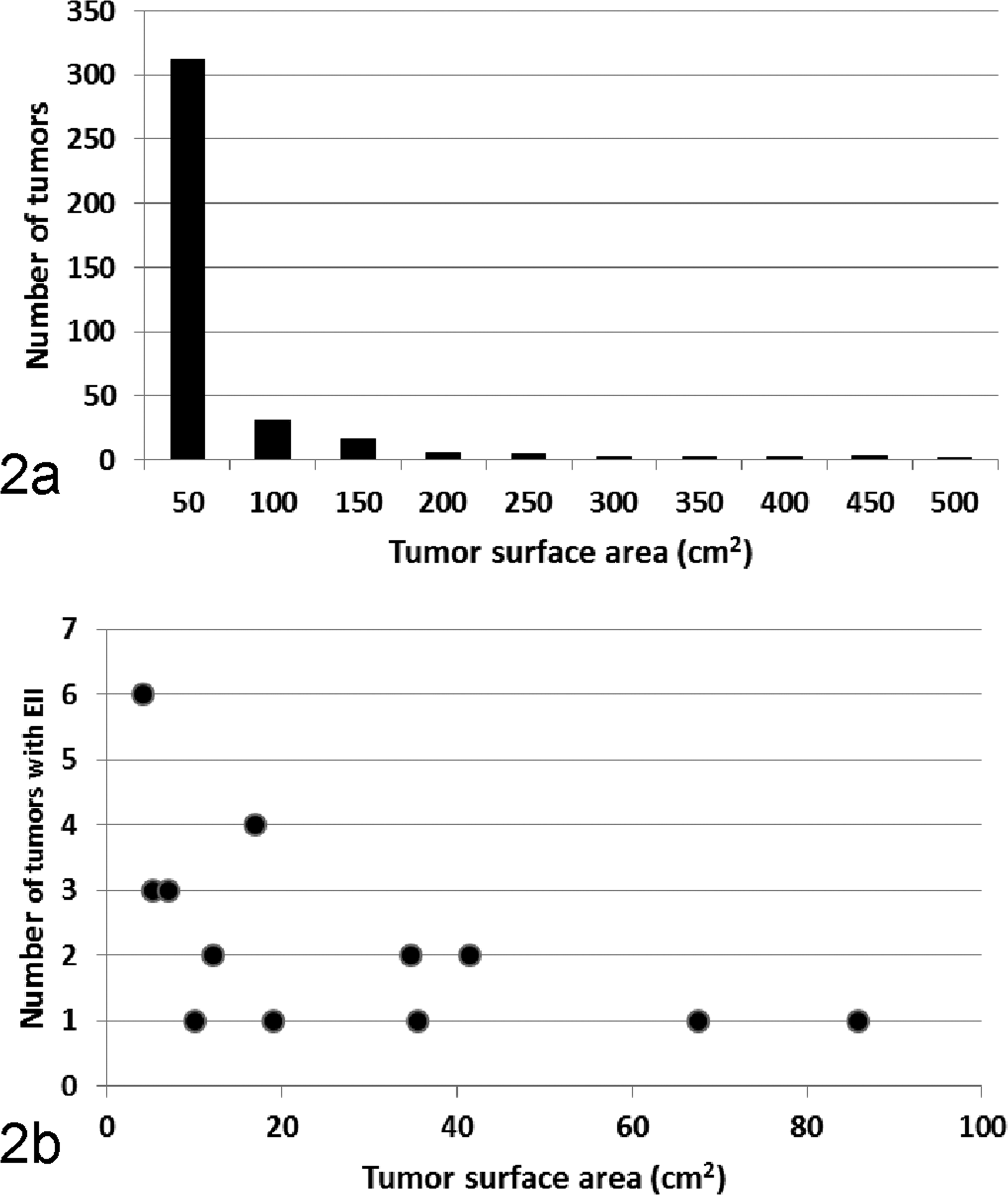
(a) Frequency distribution of sizes (surface area) of 381 tumors originating from 17 green turtles in Hawaii. (b) Scatterplot relating the surface area of tumors to the number with epidermal intranuclear inclusions (EIIs).
Twenty tumors originating from the flippers (n = 11), neck (n = 5), oral cavity (n = 2), mouth, or eye were examined in detail by taking histologic sections of the entire surface of serial 0.5-cm-wide slices of each tumor. Of 310 tissue sections examined microscopically, 2 had EIIs, each from a different tumor. Tumors ranged in size from 4 to 111 cm2, and number of histological sections examined per tumor ranged from 4 to 138.
Identification of F-VP26 and Its Antiserum
VP26 is a capsid component in many herpesviruses that typically has no known nuclear translocation signal and remains cytoplasmic in the absence of ongoing viral replication. However, in the presence of other viral factors produced during active replication, VP26 translocates to the nucleus to form hexamers on the outside of newly assembled viral capsids prior to tegumentation, envelopment, and release. 36 To assess the intracellular localization of F-VP26 in the absence of replicating ChHV5, HEK223 T cells were transfected with pHSV-VP26-EYFP. After overnight culture, a random punctate bright yellow fluorescence was seen in the cytoplasm of many cells, suggesting successful transfection and extranuclear localization and synthesis of F-VP26 (Fig. 3). Notably, F-VP26 neither translocated to the nucleus nor colocalized with concanavalin A-stained cellular membranes. Mock-transfected cells remained without fluorescence, whereas an unrelated EYFP-fusion protein (F-US8, a predicted viral membrane protein) provided a reticulate fluorescence pattern within cytoplasm particularly along the nuclear membrane, the endoplasmic reticulum, and the external cellular membrane, which indicated that the EYFP tag did not rule over the cellular localization of its fusion protein (Fig. 4). On Western blot, the V5 monoclonal antibody detected a greater than 15-kDa band (predicted 17.3 kDa) in extracts from pcDNA6.2-VP26-cLumio Dest transfected cells and an approximately 35-kDa band in extracts from cells transfected with an unrelated V5-fusion protein (F-US4, predicted 32.9 kDa), indicating that the observed mobility of the respective V5-fusion protein depended on its fusion partner. The rat anti-F-VP26 serum recognized the same 17-kDa protein as the V5 monoclonal antibody (Suppl. Fig. S2). In contrast, preimmune and “blocked” rat sera did not react. In all FP tumor sections from Hawaii and Florida that were tested (n = 3), rat anti-F-VP26 specifically stained intranuclear EIIs, but preimmune or blocked sera did not (Figs. 5 and 6). We did not observe any staining in tumored tissue without inclusions. The nuclear localization of F-VP26 in the tumors strongly suggested that ChHV5 was in a state of active, lytic replication in tumor cells with EIIs. Thus, these EIIs may be considered as viral factories, an essential prerequisite for viral shedding.
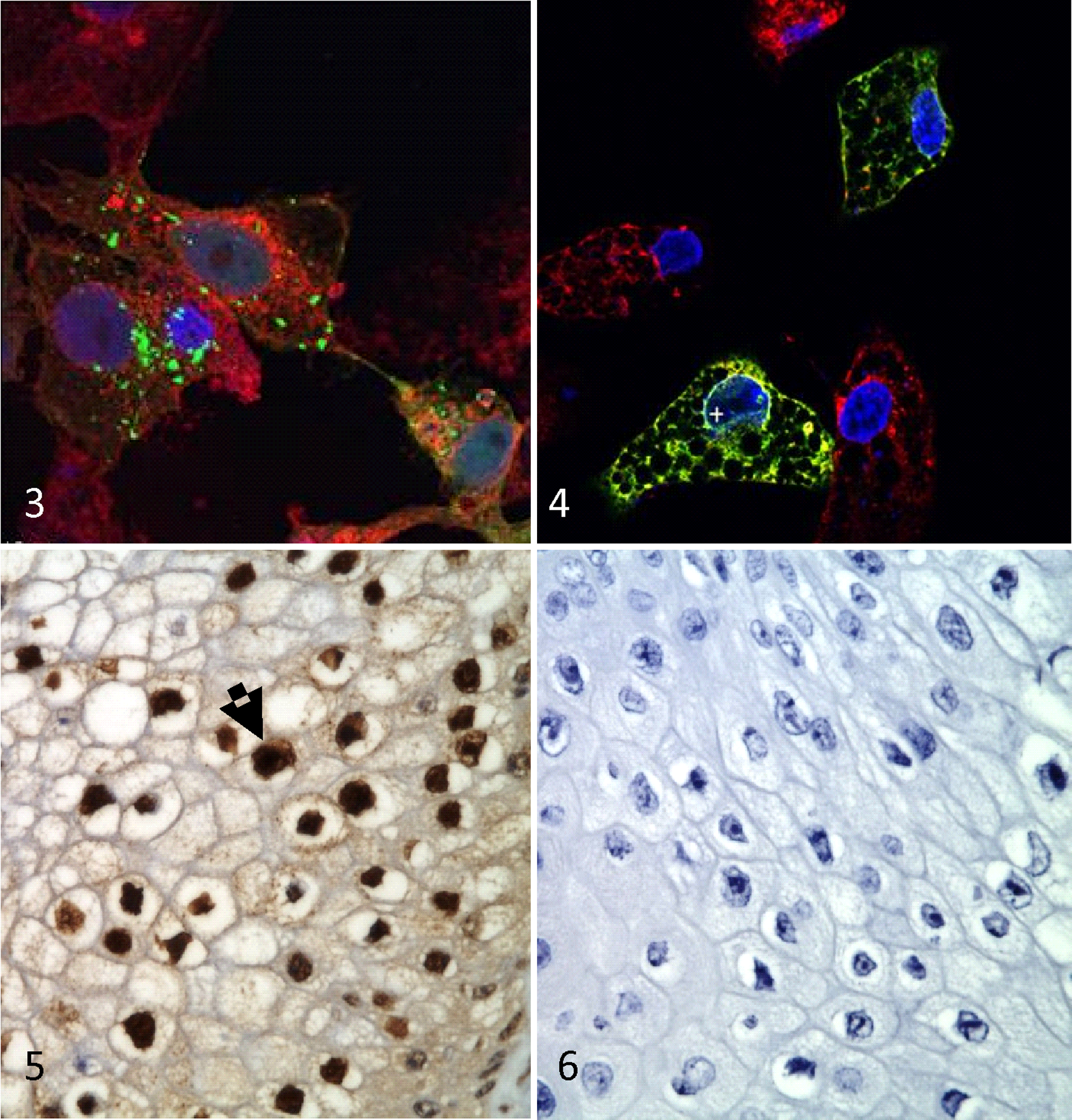
Vero 2.2 cells transfected with an expression plasmid encoding the F-VP26-EYFP fusion protein. The fusion protein is located in cytoplasm (green dots), 4′,6-diamidino-2-phenylindole (DAPI) (blue) stains nuclei and concanavalin A (red) stains cellular membranes.
Discussion
Our study reveals 2 important aspects of FP in Hawaiian green turtles. First, the frequency distribution of EIIs in tumors and shedding pattern in turtles suggests that transmission of ChHV5 may depend on superspreaders. Second, we link for the first time presence of the capsid protein F-VP26 to EIIs in skin tumors, revealing replicating ChHV5 and a viable route of viral transmission.
Virus shedding by epithelia in turtles with FP appears to be sporadic, with a low percentage (7%) of all tumors positive with EIIs. Sixty-five percent of animals had no detectable virus shedding at the time of sampling, and when animals did shed, the proportion of tumors with inclusions was relatively low (<40%) with 2 animals each at less than 10%, 10% to 20%, or 30% to 40%. The pattern seen here provides an index of intensity of shedding that follows an exponential distribution with mostly negative animals, a few positive ones, and a few animals where shedding is most intense (>30% tumors positive). This implies that superspreaders, 21 that is, a few infectious individuals in a population that contribute disproportionately to secondary infections, 37 could play an important role in the transmission dynamics of ChHV5. The potential role of superspreaders in the epizootiology of FP would fit the epizootic curve of the disease in Hawaii, where the peak of disease prevalence was broad and spread over a long time period, 6 a characteristic of epidemic curves where superspreaders drive transmission of pathogens. This contrasts with shorter and sharper epidemic curves where they do not. 21
If superspreaders do not play a role in the spread of FP, then the contact rate between animals that shed virus and susceptible hosts would have to compensate for the low percentage of animals that shed virus. For example, assuming that approximately 35% of turtles will have at least 1 tumor shedding virus, and if the percentage of tumors on a shedding turtle ranges from 5% to 37%, then the probability of encountering an animal shedding virus at any particular time ranges from 2% (0.35 × 0.05) to 13% (0.35 × 0.37). Furthermore, the surface area of tumors actually shedding virus was extremely small and ranged anywhere from 0.0001% to 0.2% of the total surface area of tumor, further decreasing the odds of transmission. Proposed mechanisms of transmission of ChHV5 include direct acquisition of viral infection from virus-laden seawater, direct contact between turtles, or indirect contact through cleaner fish that incidentally bite tumors 25 or leeches, 13 both of which have been found to be PCR-positive for ChHV5. Other herpesviruses that cause nonneoplastic skin diseases in green turtles such as gray patch disease can persist in viable infective state in seawater for long periods. 7 Turtles do periodically aggregate at cleaning stations, so presumably some contact transmission occurs either directly between turtles or with virus-contaminated substrate. 43 However, given the scarcity of EIIs on the surface of tumors, it is hard to believe that sufficient amounts of virus could be shed to still allow for efficient transmission. In contrast, cleaner fish, leeches, or other as yet unidentified vectors may play a role in virus transmission, and this needs further exploration.
Another unknown variable is how long animals shed virus. Determining this using tumor biopsies would be difficult given how infrequently EIIs are detected histologically, so identifying ways to do this for live animals will require further research. However, our newly developed antisera against F-VP26 may contribute to culturing the virus by discriminating between replicating and nonreplicating ChHV5, a feature that is not trivial in light of ChHV5 not being able to replicate in cell cultures. 42 The F-VP26 capsid protein is known to lack an independent nuclear translocation signal 1 and will localize, similar to VP26 of the herpes simplex virus type 1, to the nucleus only in the presence of ongoing viral replication. 36 For example, this technique was used to confirm the in vivo replication in sheep airway epithelial cells of another herpesvirus, OvHV-2, that causes malignant catarrhal fever in various cloven-hoofed ungulates and that does not replicate in conventional cell cultures. 38 Moreover, our findings of intranuclear localization of F-VP26 indicating productive, lytic viral replication in EIIs concur with similar studies using in situ hybridization 20 where mRNA encoding the ChHV5 DNA polymerase (F-UL30) was detected in EIIs, suggesting that ChHV5 actually transcribed a lytic viral gene in those cells. The cell culture expression and Western blot results confirmed that our rat antiserum specifically recognized F-VP26. In the absence of other viral proteins, F-VP26 localized randomly to the cytoplasm of transiently expressing cells, whereas it assumed an intranuclear localization in the slides with EIIs. In other herpesviruses, VP26 forms hexameric structures on the outer face of capsids and plays a functional role in newly infected cells transporting incoming capsids from the periphery toward nuclear pores, where the viral DNA is released to initiate a new cycle of infection. 8 Therefore, VP26 is added late in productive infection to newly assembled herpesvirus capsids but prior to nuclear egress, tegumentation, envelopment, and release. Accordingly, nuclear localization of F-VP26 may be considered as strong evidence that ChHV5 actually replicated in a productive and lytic fashion within these lesions. Thus, one may assume that EIIs in those tumors actually represent sites of ChHV5 replication and shedding.
Virus shedding in smaller, and presumably younger, tumors would fit the pattern that ChHV5 is temporally associated with development of tumors in that virus replication occurs early in tumor formation, and virus becomes latent as tumors grow. This might also explain the preponderance of smaller tumors in turtles afflicted with FP, because they would provide a renewable source of replicating virus. Quantitative PCR for herpesviral DNA polymerase gene failed to detect a relationship between tumor size and viral DNA loads. 42 However, it is unclear whether testing for this gene by quantifying DNA would be the best marker of viral shedding for ChHV5. For instance, measurement of viral RNA expression of the T0.7 and T1.1 proteins was used to detect shedding of human herpesvirus 8, the virus associated with Kaposi’s sarcoma in humans. 32 Our understanding of basic gene expression for ChHV5 in tumors is rudimentary and merits further exploration.
The shedding patterns of ChHV5 in green turtles have similarities and contrasts with other tumor-inducing herpesviruses. Tumors in sea turtles appear to play an active role in virus shedding. In contrast, in other virus-induced neoplastic diseases such as Epstein Barr virus or Kaposi’s sarcoma in humans, tumor tissues are considered a dead-end for lytic virus replication; 28 rather, virus replication occurs in normal epithelium to varying degrees. For example, virus shedding in humans infected with the tumorigenic human herpesvirus 8 associated with Kaposi’s sarcoma is limited to rare (1%) epithelial mucosa cells in small (3%) percentages of people. 39 In contrast, over 90% of humans infected with human herpesvirus 4 (Epstein Barr virus) shed virus in their saliva, 26 and chickens infected with Marek’s disease virus shed infected skin cells continuously in large numbers, with transient (2-3 weeks) shedding in animals that do not develop visceral tumors and continuous shedding in those that do. 29 We think it unlikely that virus is shed in normal skin of green turtles with FP, because EIIs have never been seen microscopically in normal skin, 16,42 nor has virus been routinely detected by PCR in normal skin. 23,34 However, more recent studies suggest that this may not be the case in all locations where FP occurs, because normal skin from green turtles without tumors from Puerto Rico was recently shown to be positive for herpesviral DNA using nested PCR. 31 We caution that this technique has resulted in false-positive results with other herpesviruses, 24 and amplicons in the Puerto Rico study were not consistently sequenced to confirm ChHV5, so independent confirmation of such findings would be informative.
Contrasting virus shedding in green turtles from Florida, where FP continues to persist in wild populations, to those of Hawaii, where the disease is waning, may illuminate the importance of this parameter in persistence of FP and its management. The strains of viruses infecting turtles vary geographically, 9,34 and so likely do the host immune responses to the virus and tumorigenesis. 4 Host manifestation of FP differs in turtles from Florida versus Hawaii, with those from Florida more prone to liver tumors 14 whereas oral tumors are common in Hawaii. 2,3
The findings herein have potential management implications. If skin contact is an important mechanism of virus transmission, and animals with many tumors act as superspreaders, systematic removal of these individuals from the wild may, theoretically, help reduce incidence of disease and might explain the differences in prevalence of disease between Hawaii and Florida. For instance, in Hawaii, where FP is declining in prevalence, facilities to rehabilitate turtles with tumors do not exist, and stranded turtles with many tumors and with poor prognosis for survival have been permanently removed from the population. Whether removal of such animals is truly a viable management option to limit the spread of FP awaits confirmation, and implementing such a measure would require careful forethought. For example, one might perhaps target animals with large numbers of small tumors or tumors that shed the most virus such as oral or scute tumors. Our findings might also help attempts to isolate ChHV5 in the laboratory. For instance, knowing that smaller tumors are more likely to have EIIs might allow for more efficient targeting of tissues for virus isolation. Finally, if a rapid and high throughput sampling scheme to detect virus shedding (eg, swabs) could be developed, surveillance efforts would be optimized by focusing on smaller tumors.
Footnotes
Acknowledgments
Thanks to Bob Rameyer, Renee Breeden, Shandell Eames, Devin Francke, and Shawn Murakawa for technical assistance. Mention of product or trade names do not imply endorsement by the US government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Mathias Ackermann’s contribution to this research was supported by the Wyss Charitable Endowment.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
