Abstract
Norway (Rattus norvegicus) and black rats (Rattus rattus) are common commensal pests, yet little is known about the ecology of wild rats, including their natural diseases. We describe microscopic cardiovascular pathology in a subset of a sample of 725 wild urban rats. Changes observed in the pulmonary blood vessels (n = 199) included arteriolar medial hypertrophy (20.1%, n = 40) and blood vessel mineralization (19.1%, n = 38). Microscopic changes in the heart (n = 200) included myocarditis (33.5%, n = 67), fibrosis (6.0%, n = 12), mineralization (9.5%, n = 19), myocardial degeneration (22.0%, n = 44), and right ventricular hypertrophy (4.5%, n = 9). Rats with myocarditis, fibrosis, or myocardial degeneration were grouped into a composite variable: cardiomyopathy. Statistical analysis showed that the odds of being affected by cardiomyopathy were greater in male rats (odds ratio [OR] = 2.49; 95% confidence interval [CI] = 1.12-5.55) and heavier rats (OR = 1.14; 95% CI = 1.09-1.20). The odds of pulmonary arteriolar medial hypertrophy was greater in sexually mature rats (OR = 2.35; 95% CI = 0.75-7.36), while the odds of pulmonary vessel mineralization were greater in heavier rats (OR = 1.07; 95% CI = 1.03-1.11) and in black rats (OR = 5.35; 95% CI = 1.62-17.69) compared to Norway rats. To our knowledge, this is the first detailed description of pathology in the cardiovascular system of wild rats and demonstrates that cardiovascular disease is common. The impact of these lesions on individual and population health remains to be investigated.
Keywords
Norway and black rats (Rattus norvegicus and Rattus rattus) are among the most abundant and widespread mammalian species, inhabiting all continents except Antarctica. 9 These rats are especially well adapted to living in close proximity to humans, making use of human harborage, refuse, and agricultural crops. 9 This adaptation has resulted in a number of negative consequences for people, ranging from economic losses through food consumption to health risks associated with the transmission of zoonotic diseases. 9,14 Despite humankind’s long-standing association with Norway and black rats, there is still a number of significant gaps in our understanding of the ecology of these species. Perhaps the most notable gap is related to causes of natural morbidity and mortality.
The life span of wild urban rats is short, with few individuals living beyond 1 year. 9 In contrast, the expected life span of laboratory rats ranges from 2 to 3 years. 22 The reason for this rapid population turnover in wild rats is unclear. Research has shown that predation does not significantly reduce population size. 9 Similarly, long-term population effects of rat control activities, such as trapping and poisoning, are limited because of these rodents’ high fecundity rates. 9 Additionally, competition for food, habitat, and other resources has been suggested as a cause of rat mortality, but this fails to explain the loss of mature animals, which would be expected to outcompete juvenile rats. 9
Little work has been done to characterize natural diseases in wild rats, which may be a key component to understanding morbidity and mortality in these species. Understanding the causes of mortality in wild rats is important for a variety of reasons. For example, insight into natural population turnover is helpful for designing effective ecologic rodent control strategies. 28 Additionally, some natural rodent mortality events can signal a health risk to people. In plague outbreaks, for example, death in wild rats typically precedes human mortality. 16 An understanding of the full range of natural disease in rats can help researchers and health officials to differentiate mortality events caused by zoonotic pathogens from those that are normally occurring in rat populations. Finally, documentation of wild rat pathology may be useful in understanding laboratory rat disease. Since laboratory rats are a domestic strain of R. norvegicus, wild rats can serve as a useful comparison for background conditions, including spontaneous cardiovascular lesions.
The objective of this study was to describe and categorize cardiovascular pathology in the myocardium and pulmonary vasculature among a population of urban Norway and black rats.
Methods
Trapping and Rat Collection
This study was conducted as part of a larger project, the Vancouver Rat Project (www.vancouverratproject.com), which focused on characterizing the ecology of zoonotic pathogens in urban rat populations. Given that rat ecology is a primary determinant of zoonotic pathogen dynamics within rat populations and the risk of pathogen transfer to humans, 14 this project also sought to characterize the ecology of urban rats populations, including natural causes of morbidity and mortality. Rats were trapped in 43 contiguous city blocks within an inner-city urban neighborhood of Vancouver, Canada (N49°17′/W123°6′), as well as on one property within an international shipping port that forms the northern border of the study area. The port was included to capture any potential incursion of foreign rats and their pathogens. 2 Pairs of blocks and the port site were randomly allocated to one 3-week trapping period over the course of 1 year (September 2011–August 2012).
Briefly, 15 to 20 Tomahawk Rigid Traps for rats (Tomahawk Live Trap LLC, Hazlelhurst, WI, USA) were set out in an alleyway that ran the length of the block behind the buildings. At the port, traps were placed in areas where port staff had observed rats. Traps were prebaited with bacon fat, peanut butter, and oats, with doors fixed open for 1 week to acclimatize rats to trapping equipment and bait, followed by 2 weeks of active trapping. Rats were anesthetized using isoflurane before blood collection via cardiac puncture and euthanasia by intracardiac injection of pentobarbital. At the port site, rats trapped by a private pest control professional using snap-type lethal traps were also collected. A total of 725 rats was collected over the course of the study.
Data collected in the field included species (determined by external morphology), sex, weight, the number of cutaneous bite wounds, and sexual maturity. Females with an open vaginal orifice and males with scrotal testes were considered sexually mature. Rats were stored at –30°C and sent to the Animal Health Centre, British Columbia Ministry of Agriculture, Abbotsford, British Columbia, for further analysis. Due to the number of rats in this study and the geographic distance between the study site and laboratory, immediate autopsy of fresh specimens was not possible. This study was approved by the University of British Columbia’s Animal Care Committee (A11-0087).
Autopsy and Tissue Collection
Rats (n = 725) were thawed at 4°C before undergoing a complete standardized autopsy and tissue collection protocol. For each rat, representative samples of the lungs and heart were immersed in 10% neutral buffered formalin. Livers were examined for the presence of grossly evident multifocal tortuous white foci consistent with Capillaria hepatica infection. 26
Histopathology
Microscopic tissue screening was conducted on all rats with gross lesions in any organ system (n = 111). Also included was a random selection of 89 rats with no visible gross lesions (selected via the “sample” function in R; R Development Core Team, Vienna, Austria) to ensure detection of lesions present at ≥ 3% prevalence (Survey Toolbox, Australian Centre for International Agriculture Research, Canberra, Australia). A total of 200 heart and 199 lung samples were examined histologically (lung from 1 black rat was not available for histologic examination).
Transverse lung lobe sections (n = 1–3) and 1 transverse section of heart (through both ventricles immediately apical to the atrioventricular valves) were prepared for microscopic examination. Following routine processing, these tissues were embedded in paraffin and 5-μm tissue sections were stained with hematoxylin and eosin. Based on initial microscopic findings of the stained slides, additional sections of selected cases were stained with Von Kossa and Masson’s trichrome stains.
Lesion Categorization
The pulmonary blood vessels and heart from each rat were screened for morphologic cardiovascular abnormalities, which were recorded. A system for defining and categorizing lesions (Table 1) was created on the basis of changes observed in the initial screen, in combination with a review of lesions commonly described in the heart and pulmonary blood vessels of laboratory and wild rats. 6 –8,11,15,18,20,21,23,25,27,31 Inclusion and exclusion criteria were defined for each lesion to enhance objectivity. Stringent binary classification schemata were used to accurately and conservatively evaluate the presence and apparent prevalence of microscopic abnormalities in this sample of rats. Lesion severity and scoring were not assessed. Once the classification scheme was developed, all tissues were re-evaluated and categorized by these criteria.
Categories and Apparent Prevalence of Microscopic Cardiovascular Changes Used to Screen Wild Urban Rats (Rattus norvegicus and Rattus rattus).

aSample size was 200 rats for all heart categories and 199 for lung categories (lung from 1 black rat was not available for histologic examination). n = rats that had the lesion in question.
bConfirmed in a subsample of rats with Masson’s trichrome stain.
cConfirmed in a subsample of rats with Von Kossa stain.
dPercentage medial thickness = 2 (medial thickness) × 100/external vessel diameter. 3 Measurements taken on circular arteriole profiles using Olympus DP 71 Camera System.
Statistics
Bivariate associations between each microscopic change in the heart and those in the pulmonary blood vessels were assessed with Fisher exact test or simple logistic regression. Lesions with relationships that were both statistically significant (P ≤ .05) and supported by the literature were then combined to create a new composite variable. Any rat that was positive on 1 or more of the component variables was considered positive on the composite variable.
Cardiovascular lesions were then independently used as the outcome in logistic regression analyses investigating the relationship between the aforementioned changes and a variety of explanatory variables: season (September–November = fall, December–February = winter, March–May = spring, June–August = summer), sex, sexual maturity (immature vs mature), body condition as assessed by volume of internal fat stores (subjective score of 0 – 2), weight (considered in 10-g increments), species, presence or absence of cutaneous bite wounds, and number of bite wounds. Presence or absence of grossly evident C. hepatica infection was also included in the model examining associations with medial hypertrophy of pulmonary arterioles. 31
Variables that were significantly associated with the outcome of interest at an alpha level of ≤0.10 on bivariate analysis were considered for inclusion in a multivariate generalized linear mixed model (GLMM) controlling for clustering by block of origin. The goal of the model-building strategy was to identify the most parsimonious set of explanatory variables that predicted the outcome. Individuals were excluded for which data were missing for 1 or more of the variables under study.
To evaluate variations in demographic characteristics among rats with and without gross lesions, the relationships between weight and sex and the presence of gross lesions (vs no gross lesions) were evaluated with a 2-sided t test and χ2 test, respectively. All statistical analyses were conducted with R.
Results
Population Demographic Characteristics
Of the 725 rats trapped, 685 (94.5%) were Norway rats, and 40 (5.5%) were black rats. Among the Norway rats, 381 (56.3%) were male, and 397 (63.9%) were sexually mature; sex and maturity could not be determined for 8 and 64 rats, respectively. Median weight was 142.2 g (range = 20.0–466.2 g). Among the black rats, 19 (47.5%) were male, and 21 (61.8%) were mature; sexual maturity could not be determined for 6 rats. Median weight was 76.4 g (range = 24.4–259.8 g).
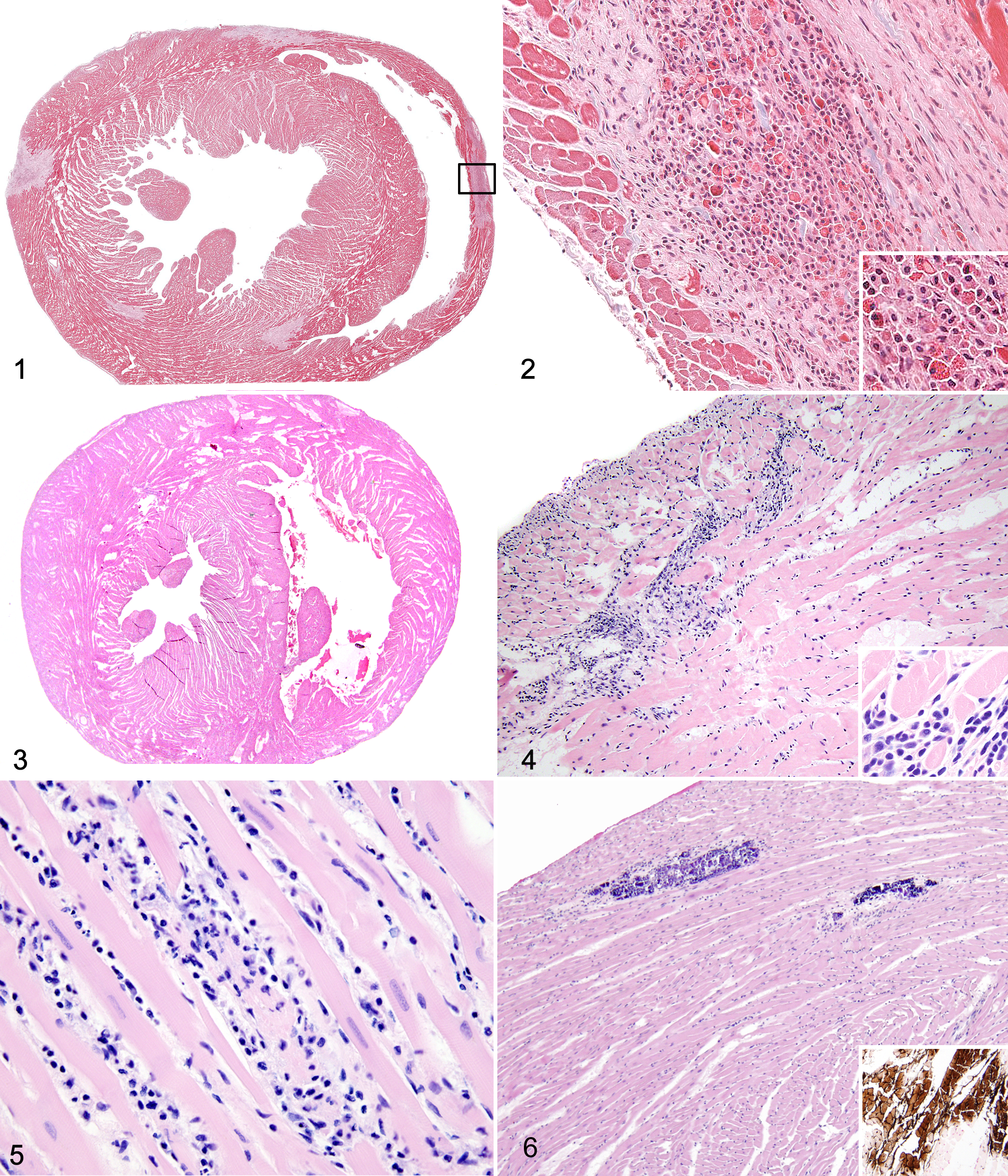
Among the 200 rats included in the histologic analysis, 184 (92.0%) were Norway rats, and 16 (8.0%) were black rats. Of the Norway rats, 107 (58.2%) were male, and 141 (76.6%) were sexually mature; sexual maturity could not be determined for 11 rats. Median weight was 231 g (range = 27–458 g). Among the black rats, 9 were males (56.3%), and 12 (74.0%) were sexually mature; sexual maturity could not be determined for 1 black rat. Median weight was 152 g (range = 26–260 g).
There was no significant difference in the number of male and female rats with and without gross lesions. However, rats with gross lesions were more likely to be heavier (P < .001) than rats without gross lesions.
Gross and Histopathologic Examination
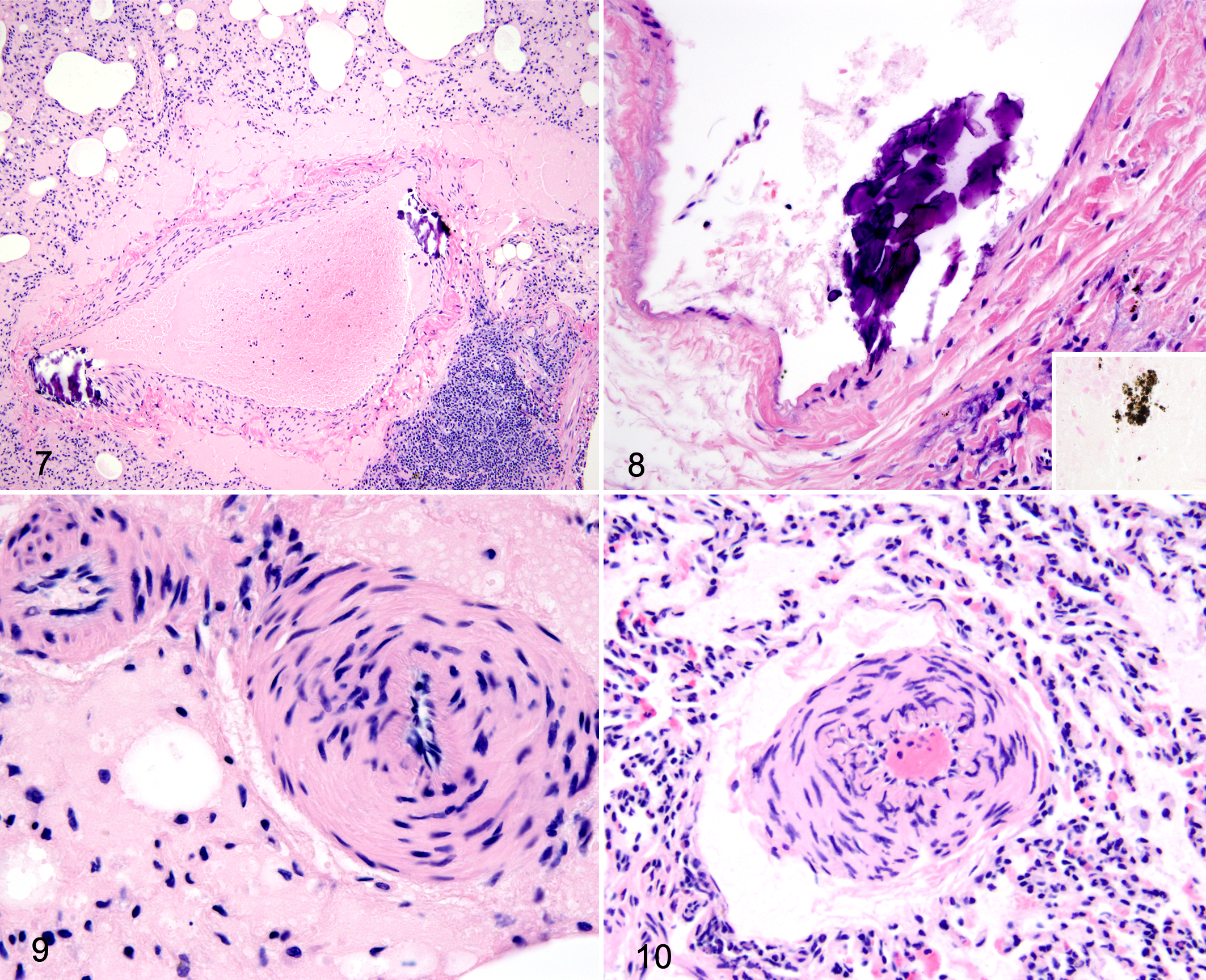
Grossly evident vascular lung or heart lesions were not identified in any of the rats examined (n = 725). Microscopic cardiovascular changes detected in the study population included fibrosis, mineralization, inflammation, cardiac myocyte degeneration, right ventricular hypertrophy, and pulmonary vessel mineralization and medial hypertrophy of pulmonary arterioles (Table 1). Pulmonary arteriolar medial hypertrophy and pulmonary vessel mineralization were present in 40 (20.1%) and 38 (19.1%) of 199 Norway and black rats, respectively (lung from 1 black rat was not available for histologic examination). The most frequently encountered changes in the myocardium were myocarditis (n = 67 of 200, 33.5%) and myocardial degeneration (44, 22.0%). Right ventricular hypertrophy (9, 4.5%), fibrosis (12, 6.0%), and mineralization (19, 9.5%) were found less frequently.
Myocardial fibrosis, myocardial degeneration, and myocarditis were strongly related (P ≤ .001 for all comparisons); odds ratios (ORs) could not be determined because extreme concordance led to zero values in 1 or more cells. These changes are recognized components of a syndrome termed cardiomyopathy in laboratory rats and were therefore combined to create a new composite variable: cardiomyopathy. 7,11,15,21,23,27 Cardiomyopathy was present in 67 of 200 (33.5%) rats examined.
Cardiomyopathy was associated with right ventricular hypertrophy (OR = 7.64; 95% confidence interval [CI] = 1.78-52.32; P = .013) and myocardial mineralization (OR = 9.30; 95% CI = 3.20-33.83; P < .001); however, these changes were not included in the composite variable, because they are inconsistently reported in laboratory rats and could have differing pathophysiology.
Cardiomyopathy was weakly associated with pulmonary vessel mineralization (OR = 2.70; 95% CI = 1.31-5.59; P = .007) and not associated with pulmonary arteriolar medial hypertrophy (OR = 0.94; 95% CI = 0.44-1.93; P = .86). There was no significant association between pulmonary vessel mineralization and pulmonary arteriolar medial hypertrophy (OR = 0.88; 95% CI = 0.33-2.07; P = .77) or myocardial and pulmonary vessel mineralization (OR = 1.15; 95% CI = 0.31-3.39; P = .82).
Statistical Demographical Associations
Results of bivariate analysis are presented in Supplemental Tables 1 and 2 (available online at http://vet.sagepub.com/supplemental). For the cardiomyopathy variable, the final GLMM included only sex and weight. Specifically, the odds of an individual rat having cardiomyopathy increased with male sex (OR = 2.49; 95% CI = 1.12-5.55) and increased weight (OR = 1.14; 95% CI = 1.09-1.20 per 10 g; Supplemental Table 2). Among male rats, 43.1% (50 of 116) had lesions consistent with cardiomyopathy, compared to 20.2% (17 of 84) of female rats.
The final GLMM predicting medial hypertrophy of pulmonary arterioles included only sexual maturity (OR = 2.35; 95% CI = 0.75-7.36), with mature animals having greater odds of experiencing the outcome. The final GLMM predicting pulmonary vessel mineralization included weight (OR = 1.07; 95% CI = 1.03-1.11 per 10 g) and species (OR = 5.35; 95% CI = 1.62-17.69), with heavier rats and black rats being more likely to experience the outcome. Among black rats, pulmonary vessel mineralization was present in 5 of 15 (33.3%) compared to 33 of 184 (17.9%) Norway rats.
Discussion
Myocarditis was the most common cardiovascular change, and it was often accompanied by varying degrees of myocardial fibrosis and degeneration. When considered together, these lesions were found in 67 of 200 (33.5%) rats. Previous studies of heart lesions in wild rats are sparse and have contrasting results with regard to the presence and prevalence of myocardial lesions. A small study of 11 wild rats identified some animals with diffuse interstitial myocarditis (rat species and exact number of rats affected were not stated). 19 In contrast, no myocardial lesions were identified in 31 wild Norway rats killed and examined as controls in another study. 24
The combination of myocarditis, myocardial fibrosis, and myocardial degeneration is characteristic of cardiomyopathy in laboratory rats, which are a domesticated strain of R. norvegicus. 7,11,15,23 Cardiomyopathy is a common spontaneous change that most often affects older males, although lesions can develop in rats as young as 3 months of age. 27 This is consistent with the results in this study population, in which the odds of cardiomyopathy increased with increasing weight (a proxy for age) 9 and male sex. The nature of the cardiomyopathy changes in this study population is also similar to laboratory rats, with inflammatory infiltrates seen more frequently than fibrosis or cardiac myocyte degeneration. 7
In males of certain laboratory rat strains, prevalence of cardiomyopathy can reach 100%, and this condition is the most important background change in nonclinical cardiovascular toxicology studies. 7 The lower apparent prevalence in this study population (33.5%) could suggest that cardiomyopathy is less common in wild populations, but it might also reflect the inclusion of very young rats in this study. Additionally, a past study has shown that examination of a single cross section of heart (vs multiple sections), as done in this study, can miss up to 28% of cases of cardiomyopathy in laboratory rats, 7 suggesting that the true prevalence of cardiomyopathy may have been underestimated in this study. Alternatively, the prevalence may have been overestimated by the inclusion of heavier rats, as these were more likely to have gross lesions in any system.
The effect of cardiomyopathy on individual wild rats is unknown. Since only live-trapped wild rats were included in this study, we were unable to evaluate natural mortality in this population; however, macro- and microscopic changes attributable to heart failure (hepatic passive congestion, ascites, etc) were not identified. It is reasonable to speculate that wild rats with severe cardiomyopathy would be less competitive compared to unaffected rats. Despite the high frequency of cardiomyopathy in laboratory rats, morbidity and mortality attributed to this condition is infrequently described. However, a long-term study of male Sprague-Dawley rats fed ad libitum attributed 7% of mortality to cardiomyopathy. 17
Environment, stress, and diet have been found to affect the age of onset and severity of cardiomyopathy in laboratory rats. 20 For example, laboratory Sprague-Dawley rats fed ad libitum had increased severity and incidence of cardiomyopathy, but the mechanism for this remains undetermined. 17 Although access to food resources was not evaluated in our study, it is expected that the heaviest male wild rats would be socially dominant and thus have greater access to food resources. They might also be subject to greater frequency and intensity of social stressors related to competition for territory and mates. 9 Thus, the behavioral characteristics that lead to foraging success, attainment of heavy weight, and social dominance also may contribute to degenerative heart disease. Exposure to cardiotoxic substances cannot be ruled out but would be unlikely to occur only in heavy males and over such a wide geographic area (likely encompassing multiple discrete colonies). 9
The histologic lesions noted in this study were not consistent with cardiotropic infectious agents that are classically associated with heart lesions, such as Clostridium piliformis (the causative agent of Tyzzer disease), Streptococcus pneumoniae, or other bacteria 4 ; thus, additional specific ancillary testing was not conducted. The pathogenesis of cardiomyopathy in laboratory rats is suspected to be the result of focal ischemia from vascular function abnormalities, since lesions are associated with regions of the myocardium most sensitive to hypoxia. 11 The cause of cardiomyopathy in this population of wild rats remains undetermined.
Right ventricular hypertrophy was rare, with only 9 of 200 (4.5%) rats affected. Although it was slightly associated with cardiomyopathy, the low number of affected rats hinders accurate assessment of this possible association. Associations between right ventricular hypertrophy and cardiomyopathy are inconsistently described in the laboratory rat literature. 11,23 Studies of the physiology of wild Norway rats have found that heart weight was higher in males and females in the winter compared to the summer. 1,13 In one study, this increased heart weight was attributed to ventricular hypertrophy and was thought to be a physiologic adaptation to cold weather and/or the presence of renal pathology, although factors such as age and sexual maturity were not controlled for in the analysis. 1 These studies do not describe the microscopic appearance of the heart. 1,13
Mineralization in the myocardium was identified in 19 of 199 (9.5%) rats. This change was associated with cardiomyopathy, albeit less strongly than was myocardial fibrosis and degeneration, suggesting that it may or may not be a component of cardiomyopathy in wild rats. The association between cardiomyopathy and mineralization in laboratory rats also is inconsistent, 11,15 and some researchers have considered mineralization to be an independent lesion. 11,27
There was no relationship between mineral in pulmonary blood vessels and mineral in the myocardium, suggesting differing pathogenesis for these 2 changes. Blood vessel mineralization is common in older laboratory rats, with males more frequently affected than females, and it may be associated with chronic renal disease or parathyroid gland hyperplasia. 27 One study of laboratory rats found spontaneous pulmonary vessel mineralization in 46% of rats, often in blood vessels that had no other changes, although the relationship between vessel mineralization and other lesions was not evaluated. 29 Pulmonary vessel mineralization was found to be positively associated with high body weights in this study but not with sex. Subjectively, associations with microscopic renal disease or parathyroid gland hyperplasia were not appreciated in this study population. It is interesting that black rats were overrepresented among rats with vessel mineralization, despite the relatively small sample size, compared to Norway rats. Given that black rats have differing biology compared to Norway rats, 9 a number of unidentified factors could be contributing to this species variation.
Cholecalciferol-based rodenticides can cause mineralization of tissues 6,12 and could be considered a potential cause for the heart and blood vessel mineralization observed in these rats. Although chemical analysis for exposure to rodenticides was not done in this study, cholecalciferol rodenticides are not currently licensed for use in Canada. 10
Medial hypertrophy of pulmonary arterioles was present in 20.1% of rats and was positively associated with sexual maturity. A previous study in wild Norway rats described medial hypertrophy in 11 of 89 rats (12.4%) and made an association between medial hypertrophy and infection with the liver parasite C. hepatica. 31 However, age and sexual maturity were not assessed by these researchers, and they could confound the relationship between arteriolar hypertrophy and C. hepatica. There was no statistical association between liver infection with C. hepatica and medial hypertrophy of pulmonary arterioles in our study. In laboratory rats, medial hypertrophy of pulmonary arterioles has been induced by hypertension, chronic hypoxia, and monocrotaline, and is considered a rare spontaneous lesion. 11 The findings of this study suggest that it is a common lesion in wild rats; however, the cause and reasons for association with sexual maturity remain unclear.
One limitation of this study was the method of tissue collection and preparation. For example, the use of a single cross section of heart, rather than multiple cross sections, limited the scope of the survey. Structures that were not specifically examined include the valves and conduction system. Future studies should seek to expand this work by evaluating aspects of the cardiovascular system not included here. An additional limitation was the low number of black rats included in the study, although the proportion of black rats in this study is consistent with the overall sample population. Biased trapping methodology—specifically, ground-based outdoor traps—may have resulted in black rats being underrepresented in the trapped population. This can be explained by habitat use whereby black rats typically inhabit the upper levels of humanmade structures. 9 Also, Norway rats have been known to completely displace black rats where the 2 species are sympatric 5,30 ; therefore, black rats may truly be rare within the study area. Future studies should seek to focus on black rats to determine if the extent and nature of cardiovascular pathology is similar to that of Norway rats.
Conclusion
To our knowledge, this is the first detailed description of myocardial and pulmonary vascular pathology in a large sample of wild urban rats. Lesions in the myocardium and pulmonary vessels were common in this population. The high frequency of lesions in wild rats suggests that similar lesions found in laboratory rats may be typical for these species, rather than induced by extrinsic factors associated with domestication and captivity. The prevalence and variety of cardiovascular lesions also suggest that degenerative cardiovascular disease may be one of the factors contributing to the short lifespan of wild urban rats.
Footnotes
Acknowledgements
This study was made possible by the efforts of K. Parsons, A. Feng, V. Chang, D. Rempel, T. Urness, H. Anholt, and the Vancouver Area Network of Drug Users. I. Shirley, C. Stewart, and K. Brown provided invaluable technical assistance. This study was undertaken as part of a master of veterinary science research project, and appreciation is given committee members: S. Gomis, T. Bollinger, and B. Kidney.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Canadian Institutes of Health Research (MOP 119530, CGV 104833) and the Western College of Veterinary Medicine Interprovincial Graduate Student Fellowship.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
