Abstract
Asthma is a chronic inflammatory disease of the lung that is a leading cause of morbidity and mortality in children worldwide. Most infants who experience wheezing episodes also exhibit evidence of an ongoing respiratory viral infection. Respiratory syncytial virus (RSV) is the leading cause of lower respiratory tract infection in children and is a common cause of wheezing in infants and young children. In the past several decades, a number of studies have demonstrated a relationship between infants with severe RSV infections and the subsequent development of asthma later during childhood. This review provides an overview of data that suggests a severe RSV infection early in childhood is linked to development of asthma later in life. In addition, the current and potential future use of various animal models to gain additional insight into the relationship between RSV and asthma is discussed.
Keywords
Asthma is a common chronic inflammatory disease of the respiratory tract that is characterized by narrowing of the airways that recurs throughout an individual’s lifetime. Prevalence rates for asthma are at historically high levels and remain a significant public health problem. 2 Asthma attacks are often characterized by recurring episodes of symptoms such as coughing, wheezing, shortness of breath, and chest pain. Acute asthma episodes can be triggered by a number of factors, including allergens, chemical irritants, drugs, infection, strenuous exercise, or sudden changes in weather. It is currently unclear what factors lead to the initial development of asthma. It is likely that asthma inception is due to a combination of both genetic predisposition and environmental cues. Virus infection may represent a critical environmental trigger for the development of asthma. RNA from common respiratory viral infections can be detected in 90% of nasal lavage samples from children aged 1 to 3 years experiencing wheezing episodes. 35 Rhinovirus and respiratory syncytial virus (RSV) are the most common viruses detected in children experiencing wheezing. 35 A study of children with wheezing symptoms revealed a strong correlation with a concurrent rhinovirus and/or RSV infection. Children with wheezing episodes who were infected by either rhinovirus or RSV in the first 3 years of life had a greater tendency to develop asthma by age 6 years compared with children not infected by either virus. 35 This suggests that exposure to common respiratory viral infections such as RSV during early childhood may increase the risk of developing asthma.
RSV is a negative-sense single-stranded RNA virus of the Paramyxoviridae family and a common cause of bronchiolitis in infants. RSV is the leading cause of lower respiratory tract infections in infants and young children. 27,77,93 By the age 2 years, nearly all children have been infected by RSV at least once. 22 While RSV is a significant health care burden, there is currently no licensed vaccine available. Over the past 20 years, there have been a number of studies demonstrating a positive correlation between severe RSV infection early in childhood and a greater risk of developing asthma. 29,53,55,80,81 This review explores the relationship between severe RSV infection and the subsequent development of asthma. Furthermore, it describes the use of BALB/c mouse models to explore this relationship and discusses the potential utility of other animal models of RSV and asthma that could be used to shed further light on this important topic.
Relationship Between Severe RSV Infection and Asthma
Epidemiology
RSV is a seasonal virus in temperate climates with infection rates peaking during winter months. However, in tropical climates, RSV infection can occur year-round. 8 While virtually all children eventually become infected with RSV, only 0.5% to 2% of infants require hospitalization during their initial exposure. 76 Hospitalizations during RSV infection are a result of airway dysfunction caused by inflammation of the bronchioles resulting in bronchiolitis. Reinfection by RSV can occur throughout one’s lifetime as sterilizing immunity is not generated following infection. 22,25,28 However, subsequent RSV exposures are believed to be less severe in healthy adults.
Link Between Severe RSV Infection and Development of Recurrent Wheezing or Asthma
A large number of studies have identified a link between severe RSV infection and an increased likelihood to develop either recurrent wheezing or asthma.* The Avon Longitudinal Study of Parents and Children by Henderson et al 29 revealed that children admitted to the hospital for RSV-induced bronchiolitis prior to the age 1 year were at increased risk of developing recurrent wheezing at 30 to 42 and 69 to 81 months compared with the control group (28.1% vs 13.1% and 22.6% vs 9.6%, respectively). Furthermore, 38.4% of the severe RSV group developed asthma by age 7 years compared with only 20.1% of the control group. Similar results were observed in a long-term Swedish study of 140 children by Sigurs et al. 80 Children who experienced a severe RSV infection prior to 1 year of age were at a much greater risk of developing asthma by age 7.5 years (30% vs 3% of control group). In addition, recurrent wheezing symptoms were more likely to occur in the children who experienced RSV-induced bronchiolitis (28%) compared with the control group (11%). A 5-year and a 10-year follow-up study of the same cohort revealed similar trends. Children who had experienced a severe RSV infection during infancy exhibited a significantly greater incidence of either recurrent wheezing (30% vs 16.3%) or asthma (37% vs 5.4%) by age 13 years. 81 The incidence of recurrent wheezing (30% vs 1%) remained significantly higher in children with an early severe RSV infection by age 18 years. 79 The authors also noted that children who had experienced an early severe RSV infection were more likely to develop allergic sensitization to common inhaled or food allergens at any of the 3 age time points investigated. 79 –81 Since allergen sensitization is a major risk factor associated with asthma inception, severe RSV infection during early childhood may contribute to the development of asthma through sensitization to common allergens. 60,68,84,86 Overall, these studies suggest that experiencing a severe RSV infection during early childhood increases the likelihood of developing asthma later in life.
While numerous studies indicate a link between early severe RSV infection and an increased risk in developing asthma, the exact nature of the relationship is unclear. The results from the Tennessee Asthma Bronchiolitis Study (TABS) of 95 310 children by Carroll et al 10 and Wu et al 98 suggest that a causal relationship exists between severe RSV infection and the development of asthma. This study assessed the relationship between the risk of developing asthma by age 5.5 years and the time of year at which there was the greatest number of bronchiolitis-related hospitalizations (BRHs). The number of BRHs peaks every year during the winter months in temperate climates. If there was no relationship between the risk of developing asthma and severe bronchiolitis, then rates of asthma among children would be expected to be similar year-round irrespective of patterns of BRH. However, the authors observed similar patterns between the timing of the peak of BRHs and the likelihood of infants developing asthma. Children born 4 months prior to the annual peak of BRHs were 29% more likely to develop asthma compared with infants born 1 year from this time point. This relationship was similar across the 5-year study even though the peak of BRHs shifted by up to 6 weeks. It was not determined if children were infected with a specific virus such as RSV, although up to 70% of severe bronchiolitis cases are associated with RSV infection. 58 The TABS suggests that viral infection during childhood directly contributes to an increased risk of developing asthma.
Influence of Age in Longitudinal RSV and Asthma Studies
Although a number of studies indicate an increased risk of developing recurrent wheezing later during childhood following a severe RSV infection, a few longitudinal studies suggest these changes are transient. A study by Pullan and Hey 65 determined that children with severe RSV infections prior to 1 year of age experienced recurrent wheezing symptoms primarily during the first 4 years of life (38% vs 15% of control group). However, by age 10 years, there was no longer a significant difference in rates of wheezing (6.2% vs 4.5%). Similar results were observed in the Tucson Children’s Respiratory Study of 1246 children. 87,91 Infants who experienced a severe RSV infection in the first 3 years of life were at a significantly increased risk of exhibiting recurrent wheezing symptoms at ages 6 and 11 years. However, there was no significant difference in infrequent or frequent wheezing symptoms by age 13 years. These data are in contrast to the longitudinal study by Sigurs et al 81 that demonstrated a significant increase in children with asthma by age 13 years. The differential findings reported in these studies could be due to many factors, including genetic variability among cohorts, parameters used to select the cohort, how wheezing/asthma is evaluated, and differences in the relative pathogenicity of circulating RSV strains.
Palivizumab Prophylaxis Treatment and the Development of Recurrent Wheezing
An alternative approach to determining if RSV contributes to the development of asthma is to limit RSV-induced disease. Although there is currently no licensed vaccine, passive immunization has shown some promise. 24,69 In the mid-1990s, a monoclonal antibody specific for the fusion protein of RSV, palivizumab, was evaluated across the United States, United Kingdom, and Canada for efficacy in the prophylactic treatment of RSV infection. 1 Palivizumab prophylaxis reduced RSV-associated hospitalization rates by 55% (10.6% placebo vs 4.8% palivizumab). Due to cost considerations, palivizumab prophylaxis is administered only to children at high risk of developing severe RSV-induced disease, such as preterm infants. In a study by Simoes et al, 82 preterm infants who either had received or had never been administered palivizumab prophylaxis were followed for development of wheezing symptoms.
This study indicated there were long-term benefits to lessening the severity of RSV infection. The rates of recurrent wheezing were compared between infants with prophylactic treatment who were not hospitalized for severe RSV infection and children who were hospitalized for RSV-induced bronchiolitis without prior palivizumab treatment. Children from the prophylaxis group were significantly less likely to develop physician-diagnosed recurrent wheezing (8% vs 16% of no prophylaxis group) by 3.5 years of age. 82 These data further support the notion that severe RSV infection may contribute to the development of asthma.
Lung Function and Immune Response to RSV Infection
Lung Function
Infants younger than 6 months are highly susceptible to RSV-induced bronchiolitis. 21,59 This is likely due to their reduced pulmonary function since lung development continues for several years. 49,100 Length of hospital stay often correlates with the degree of lung dysfunction, and a significant number of infants with severe RSV-induced disease require mechanical ventilation. 90 If severe RSV infection increases an individual’s risk of asthma inception, there may be long-term alterations to the lung environment. In agreement, children evaluated approximately 6 years after a severe RSV infection exhibited significantly reduced respiratory capacity compared with infants who were not hospitalized for RSV infection. 63 A study by Korppi et al 37 also demonstrated reduced lung function in young adults 20 years after a severe RSV infection. Similar results were observed in a number of other studies in which children exhibited reduced lung function 7 or more years after a severe RSV infection. 11,41,50,55,81,85 These studies indicate that severe RSV infection may alter the lung environment and lead to prolonged lung dysfunction long after the infection is resolved.
Immune Response
Infection with RSV results in a significant increase in proinflammatory cytokines and chemokines such as interleukin (IL)–1α, IL-1β, IL-6, IL-8, interferon (IFN)–γ, tumor necrosis factor (TNF)–α, monocyte chemotactic protein 1 (MCP-1), macrophage inflammatory protein (MIP)–1α, and MIP-1β. Since asthma is a chronic inflammatory disease, severe RSV infection may promote a prolonged inflammatory environment within the lung that leads to the development of asthma. Nasopharyngeal aspirates obtained from asthmatic infants infected with RSV by age 2 years revealed significantly elevated IL-6 protein levels compared with controls. 56 Furthermore, in murine models of allergic inflammatory disease, protein levels of proinflammatory chemokines MIP-1α and CCL5 (RANTES) were elevated and significantly contributed to allergic airway inflammation. 36,71 Increased levels of RANTES protein also have been correlated with increased RSV disease severity. Studies also suggest that an imbalance in the type of CD4 T-cell response further exacerbates allergic responses. Infection by RSV at an early age tends to induce a type 2 helper T-cell response (Th2) that produces IL-4, IL-5, and IL-13. 6,14 A Th2-biased immune response is known to contribute to disease in murine models of allergic airway inflammation and is strongly associated with asthmatic patients. 13,17,32,73,78 Therefore, infection by RSV at an early age may induce a Th2-biased immune response that promotes asthma inception.
Recent work also suggests a role for IL-17–induced pathogenesis in a subset of asthmatic patients. IL-17 is secreted by type 17 helper T cells (Th17) and can induce the chemotaxis of granulocytes such as neutrophils or directly signal through cells such as fibroblasts and macrophages to produce proinflammatory cytokines IL-1β, IL-6, and TNF-α. 51 Levels of IL-17 are elevated in the sputum and bronchoalveolar lavage (BAL) of asthmatic human patients. 15,51 A Th17-biased immune response and subsequent neutrophilia is also observed in mice following allergic sensitization to ovalbumin (OVA). 97 Furthermore, both IL-17 secretion and neutrophil influx were necessary to mediate granulocytic chemotaxis, mucus hypersecretion, and airway hypersensitivity following allergic sensitization. 51,97 These studies indicate that Th17 responses play a critical role in mediating particular forms of asthma. In addition, levels of IL-17 are elevated in both humans and mice with severe RSV infection. 52 Neutralization of IL-17 signaling during RSV infection significantly reduced granulocytic infiltrate, airway resistance, and mucus hypersecretion. 52 These data suggest that elevated Th17 responses following RSV infection may increase an individual’s susceptibility to developing asthma.
Genetic Predisposition to Asthma and the Influence of RSV Infection
Two-Hit Hypothesis
It is possible that children who are genetically predisposed to developing asthma will eventually develop chronic lung inflammation and are more likely to experience greater morbidity from RSV infection. Children who exhibit bronchiolitis prior to 1 year of age show signs of reduced lung function at 5 weeks old. 100 However, as discussed, a number of studies suggest that severe RSV infection may increase an individual’s risk of developing asthma later in life. Children younger than 6 months exhibit the greatest risk of severe morbidity from an RSV infection. 21,59 It is unclear how RSV infection would promote asthma, a chronic lung disease, years after the primary infection has been resolved.
It has been proposed that the development of asthma may require a “2-hit” scenario as illustrated in Fig. 1. 31,42 This hypothesis suggests that asthma inception depends on the interaction of at least 2 genetic, developmental, or environmental factors. An example may be a polymorphism in a cytokine that predisposes an individual to develop allergic responses. If an individual with this polymorphism experiences a severe RSV infection, he or she is more likely to exhibit either long-term symptoms or chronic inflammation such as asthma. The 2-hit hypothesis also predicts that if only one factor is present, then either disease or allergic responses would be transient. This may explain why not all children who experience severe RSV-induced bronchiolitis develop asthma as well as why some children with early childhood wheezing exhibit resolution of their illness by their adolescence. 46,65,87,88 A number of gene polymorphisms are associated with both asthma and susceptibility to severe RSV-induced disease, as discussed below.
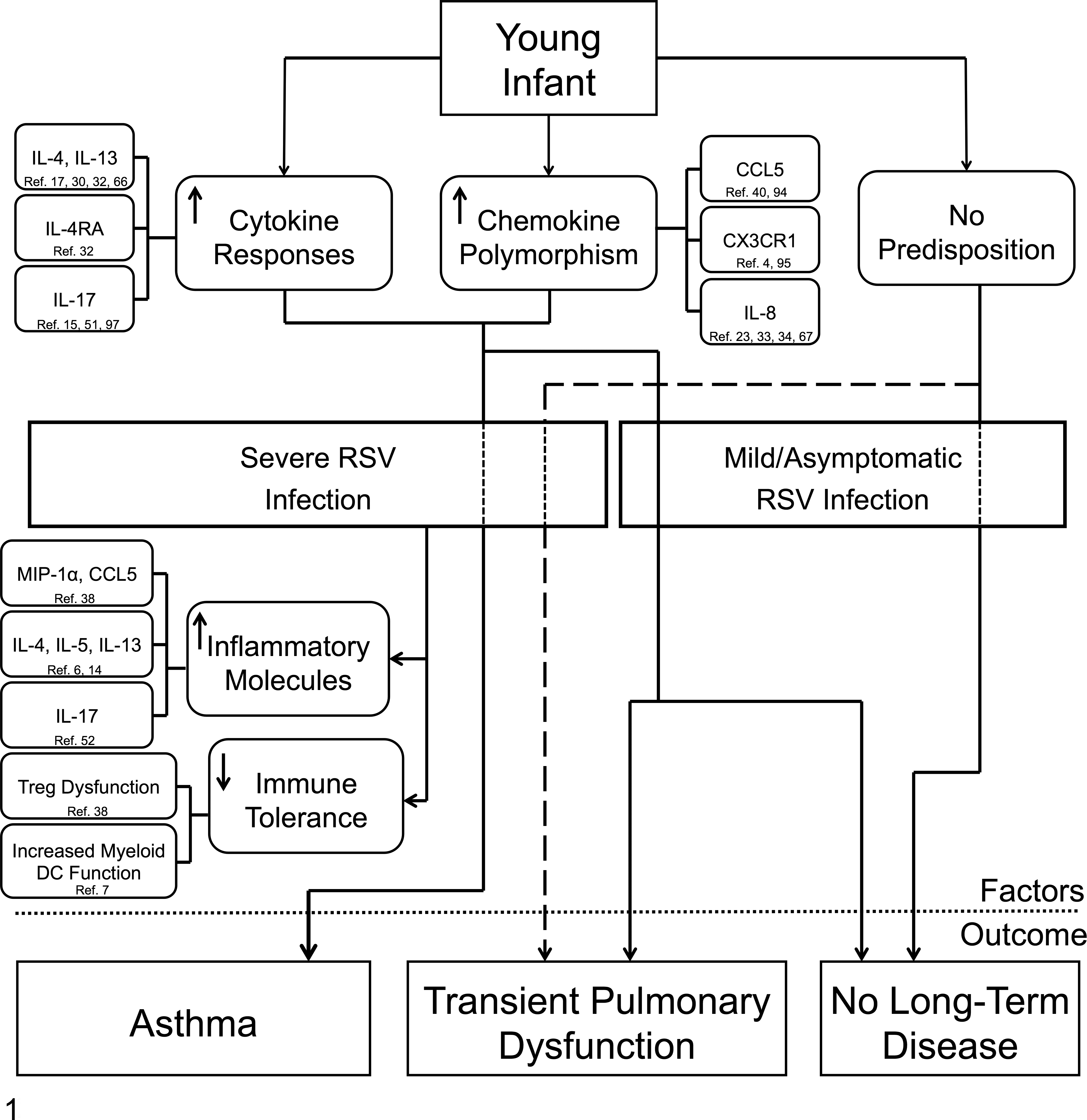
Proposed model of severe respiratory syncytial virus (RSV) infection and asthma inception based on the “2-hit” hypothesis. A young infant must have a combination of 1 or more genetic factor(s) and RSV-related immunological factor(s) to develop asthma as suggested by the 2-hit hypothesis. An infant with a genetic predisposition to allergen sensitization can develop asthma following a severe RSV infection. For example, an infant with a gain-of-function gene polymorphism for the cytokine interleukin (IL)–4 may exhibit a Th2-biased immune response following a severe RSV infection. In the absence of either a genetic factor or a severe RSV infection, thus not fulfilling a 2-hit scenario, the infant will likely experience either transient pulmonary dysfunction or no disease at all. A child with a gain-of-function
Cytokines
Cytokines play a significant role in influencing the type of immune response that develops following infection. Cytokines such as IL-4 and IL-13 are primarily produced by Th2 CD4 T cells and play a crucial role in the induction of some allergic immune responses. Children with gain-of-function polymorphisms in both
Chemokines
Chemokines are critical for the recruitment and adhesion of leukocytes during infection. Polymorphisms in either chemokines or their receptors may predispose an individual toward chronic pulmonary inflammation and increase disease severity following respiratory viral infection. The chemokine CCL5, also known as RANTES, is an important chemotactic agent for Th2-associated immune cells, including Th2 cells, eosinophils, and basophils. Gain-of-function polymorphisms in the promoter region of the
Attachment of the RSV virion to the host epithelial cell is mediated through the G protein of RSV, which is necessary for optimal infection.
43,92
While the G protein of RSV typically binds to highly sulfated heparin-like glucosaminoglycans, it also contains a chemokine motif similar to CX3CL1 or fractalkine.
19,96
The CX3CL1 mimicry function of RSV likely allows the virus to alter the host immune response.
12,96
A T280M mutation in
IL-8 is a chemokine secreted by epithelial cells and macrophages that recruits primarily neutrophils but also targets macrophages, mast cells, endothelial cells, and keratinocytes. Therefore, IL-8 is an important mediator of the innate immune system to initiate inflammation. Two separate studies by Hull et al
33,34
demonstrate that gain-of-function polymorphisms in the promoter region of
Immune Tolerance
RSV infection may also increase the susceptibility to develop allergic immune responses by breaking immune tolerance to allergens early in life. Regulatory T cells (Tregs) play an essential role in mediating immune tolerance and prevention of immune responses to allergens. 44 Tregs can regulate the immunogenicity of dendritic cells (DCs) by reducing DC expression of the costimulatory molecules CD80 and CD86 as well as expression of major histocompatibility complex (MHC) class II. 44 However, infection of mice with RSV following OVA sensitization significantly inhibits the suppressive capacity of Tregs. 38 The impaired Treg response correlates with increased DC expression of the costimulatory molecules CD40 and CD86 as well as MHC class II. This suggests that if immune tolerance is broken by RSV infection, infants may be more susceptible to the development of chronic inflammation to allergens and subsequent asthma. Furthermore, RSV infection may differentially affect the function of various DC subsets. RSV infection of myeloid DCs, but not plasmacytoid DCs, leads to the upregulation of maturation markers and an increased capacity to induce the proliferation of OVA-specific T cells in vitro. 7 Previous work has indicated that immunogenic myeloid DCs are important for promoting allergic sensitization, whereas plasmacytoid DCs are tolerogenic. 57 Together, these studies suggest that impaired regulation of allergic immune responses following RSV infection can contribute to the development of asthma.
Use of Murine Models to Explore the Relationship Between RSV and Asthma
While many human studies have indicated a role for RSV-induced inception of asthma, far less work has been performed using animal models to explore the underlying immunological mechanisms that link RSV infection and the induction of asthma. This is due to the complexity of asthma and the lack of animal models that reflect the entire disease spectrum observed in RSV-infected humans. 9,16 However, murine models of allergen sensitization have been used extensively to investigate the impact of RSV infection in the induction of asthma. 62,75 BALB/c mice can be infected with RSV either prior to or following allergic sensitization to OVA, as illustrated in Fig. 2A and Fig. 2B, respectively. Both models lead to increased airway resistance in comparison to OVA-sensitized mice not infected with RSV. Studies using these 2 models have yielded several significant pieces of information.
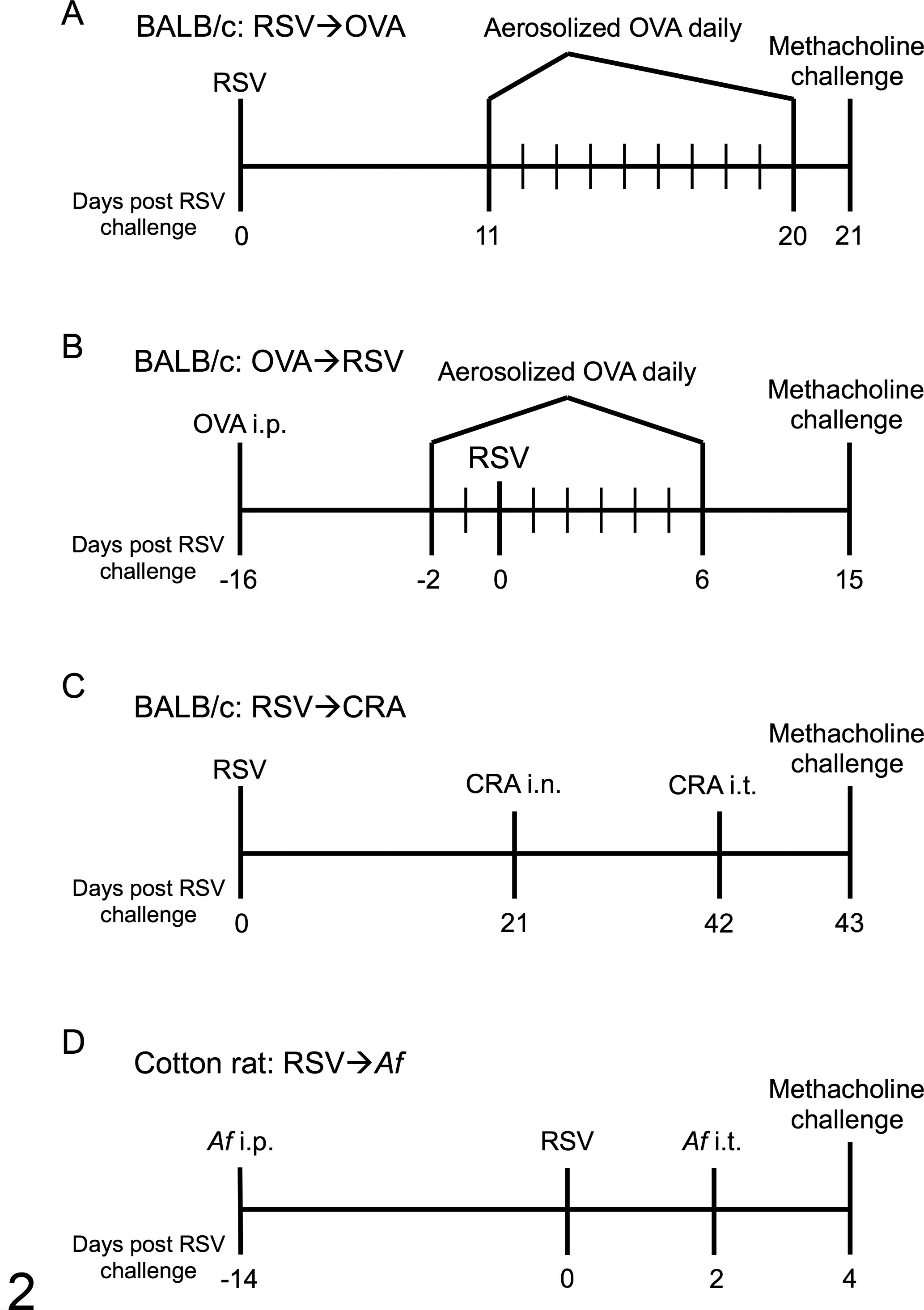
Murine models of allergen sensitization and respiratory syncytial virus (RSV) infection. (A) BALB/c mice are sensitized to OVA following RSV challenge and analyzed on day 21 postinfection (PI).
75
(B) BALB/c mice are sensitized to OVA prior to RSV challenge and assessed on day 15 PI.
62
(C) BALB/c mice are infected with RSV prior to sensitization against cockroach antigen (CRA) and challenged with methacholine 43 days PI.
45
(D) Cotton rats are sensitized to
Using the model of allergic sensitization followed by RSV infection, Peebles et al 62 reported that RSV-infected mice exhibit greater airway resistance for up to 2 weeks following infection compared with the OVA sensitization alone or acute RSV infection groups. Interestingly, the elevated airway hyperreactivity did not correlate with an elevated Th2 immune response since Th2 CD4 T cells and Th2 cytokine protein levels were similar to the OVA sensitization alone group. 61 In contrast, the Th2-associated cytokines IL-4 and IL-5 play a crucial role in mediating the elevated airway hyperresponsiveness in the murine model of RSV infection followed by OVA sensitization. 74 Similar results were observed by Lukacs et al 45 in BALB/c mice infected with RSV prior to cockroach antigen (CRA) sensitization, as illustrated in Fig. 2C. Neutralization of the Th2-associated cytokine IL-13 in BALB/c mice sensitized against CRA following RSV infection significantly reduced airway hyperreactivity. Furthermore, mice lacking IL-17A signaling exhibited significantly greater numbers of eosinophils and lymphocytes as well as increased levels of IL-13 protein in the lung following RSV infection of OVA-sensitized mice. 54 In addition, the IL-17A–deficient mice had significantly elevated airway resistance compared with wild-type (WT) mice. These data suggest that, while Th2-biased immune responses may be critical to mediate RSV-induced inception of asthma, Th17 responses limit airway hyperreactivity potentially by inhibiting a pathogenic Th2 immune response.
The development and use of alternative animal models is a crucial step to further investigate the relationship between severe RSV infection and asthma. While mice are an appealing model due to their availability and relatively low cost, mice are semi-permissive for RSV replication. In contrast, cotton rats exhibit approximately 100-fold greater peak viral titers compared with BALB/c mice following RSV infection but do not show overt clinical signs of illness.
64,70
A study by Hassantoufighi et al
26
has established a cotton rat model to investigate the relationship between RSV and asthma as outlined in Fig. 2D. Infection of cotton rats with RSV following sensitization to the fungus
Nonhuman primates, specifically macaque monkeys, represent another animal model for RSV infection and show mild signs of clinical illness.
48,83
In addition, rhesus monkeys (
Summary
RSV infection is the leading cause of lower respiratory tract infections in children worldwide. It is a significant cause of morbidity and mortality in infants. To date, there are no licensed vaccines for RSV, and only limited supportive care is provided for severe cases. A number of studies have established a strong correlation between severe RSV infection and the development of asthma during childhood. While genetic mutations may contribute to susceptibility toward asthma inception, it is highly likely that other environmental factors such as an early RSV infection may act as an initial trigger event. However, additional studies are required to elucidate the underlying immunological mechanisms of how RSV infection contributes to the subsequent induction of asthma. These studies must take into consideration the use of large cohorts over several decades with precise assessment of any genetic predispositions to accurately determine if early severe RSV infection has a causal relationship to the development of asthma. Further use of prophylactic treatment with palivizumab may also help determine if infants with a genetic predisposition to allergic responses still develop asthma independent of having a severe RSV infection early in childhood. Overall, these studies would help ascertain whether RSV prophylaxis is a viable option to indirectly prevent asthma inception in children with family histories of such disease.
Further investigation of these questions needs to be vigorously pursued using animal models. The accessibility, relatively low cost of maintenance, and broad availability of reagents and genetic mutations in mice will allow for thorough investigation of all of these questions. While models of allergen sensitization and RSV infection have been defined, there has been little investigation of the cellular composition and changes in pulmonary architecture in these murine models. The development of new models in other animals such as cotton rats and nonhuman primates would also be beneficial since RSV replicates to a greater extent in these animals compared with mice. Together, nonhuman models will help tease apart the immunological mechanisms associated with severe RSV infection that may mediate asthma inception, which cannot be evaluated in human studies. Furthermore, not only will this help to determine if RSV prophylaxis/vaccination is beneficial in the prevention of asthma, but it will also allow for the development of novel strategies to limit chronic disease following the development of asthma.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
