Abstract
A 10-year-old dog presented with convulsive crisis and symmetrical hyperkeratotic cutaneous lesions affecting the abdomen, inguinal area, eyelids, muzzles, both pinnae, and all the paw pads. Hypoglycemia and hyperinsulinemia were the main biochemical findings. A mass 2 cm in diameter was detected within the left pancreatic lobe by ultrasonography. It was surgically removed and histologically and immunohistochemically diagnosed as an insulin-producing pancreatic islet cell carcinoma. The animal was eventually euthanized due to lack of clinical improvement. At necropsy, metastatic nodules were observed in the pancreatic lymph nodes and liver. Histopathological findings of cutaneous lesions were highly suggestive of superficial necrolytic dermatitis and were interpreted as a paraneoplastic syndrome derived from the islet cell carcinoma. To the authors’ knowledge, this is the first report of superficial necrolytic dermatitis associated with an insulin-producing pancreatic neuroendocrine carcinoma in dogs.
Superficial necrolytic dermatitis (SND), previously known as metabolic epidermal necrosis, is a well-described skin condition in dogs. It is commonly related with idiopathic or drug-induced severe vacuolar hepatopathy (hepatocutaneous syndrome [HS]) and less often with glucagon-secreting pancreatic or extra-pancreatic neuroendocrine neoplasms (glucagonoma syndrome [GS]). One case of SND has also been diagnosed in a dog with diabetes mellitus. 4 The human counterpart, the necrolytic migratory erythema (NME), is a well-documented syndrome seen mostly in people with glucagon-secreting pancreatic neuroendocrine tumors. 4 However, 1 case was reported in association with a predominantly insulin-secreting pancreatic neuroendocrine tumor. 9 NME has also been observed in a small proportion of patients with severe liver disease, enteropathy, or chronic pancreatitis. 4 In other mammalian species, there are several reports of this condition—namely, cats, 4 black rhinoceroses, 6 and a fox. 8
Despite these different underlying causes, the clinical cutaneous findings of SND are very similar and consist of bilateral and symmetric hyperkeratosis with crusts and cracks on the feet, pressure points (elbows and hocks), flank, perineal area, muzzle, facial mucocutaneous junctions, and/or oral cavity. 4,7 Histopathological findings of SND are distinctive and can be highly suggestive of the diagnosis. The epidermis shows parakeratotic hyperkeratosis with neutrophilic serocellular crusts, intracellular edema, and necrosis of keratinocytes, leading to the distinctive pale middle layer, and hyperplasia of the basal cells. In some cases, there are scattered apoptotic cells in the stratum spinosum. In addition, secondary clefting can occur in the devitalized middle layer, which may lead to ulceration. 4
Beta cell (insulin-secreting) tumors are the most frequently occurring neoplasms arising from pancreatic islets of dogs. They are frequently hormonally functional, causing marked hypoglycemia and associated clinical signs such as periodic convulsive seizures. 2 In humans, numerous studies have demonstrated how hyperinsulinism induces relative and total hypoaminoacidemia. 1,3 In dogs and cats, a striking relation between total or relative hypoaminoacidemia and SND has also been recently shown. 4
To the best of our knowledge, no cases of insulin-secreting pancreatic tumors associated with SND have been reported in dogs to date.
Case Report
A 10-year-old male crossbred dog presented with episodic seizures due to hypoglycemia. Insulin levels were markedly increased (54.68 μUI/ml; reference values, 5–25). Abdominal ultrasonography disclosed an irregular, 2 cm in diameter hypoechoic mass within the left pancreatic lobe. No abnormalities were identified on the thoracic radiographs. Surgical resection of the pancreatic mass was performed, and no gross metastases were seen during the procedure. Postsurgical recovery was uneventful.
The pancreatic mass was submitted for histopathological examination. It was fixed in 10% buffered formalin and routinely processed. Histopathological evaluation revealed a nonencapsulated, infiltrative, highly cellular carcinoma of islet cell origin. Tumor cells were arranged in densely packed nests separated by a fine fibrovascular stroma. Cells were medium sized (approximately 15 μm) and polyhedral in shape, with distinct borders and a moderate amount of granular-eosinophilic cytoplasm. Nuclei were round to oval, paracentral, with finely stippled chromatin and 1 eosinophilic and evident nucleolus (Fig. 1). The anisocytosis and anisokaryosis were moderate, and the mitotic index ranged from 1 to 2 mitoses per high-power field. Tumor cells were present near the resection borders, but there was no evidence of blood vessel or lymphatic invasions.
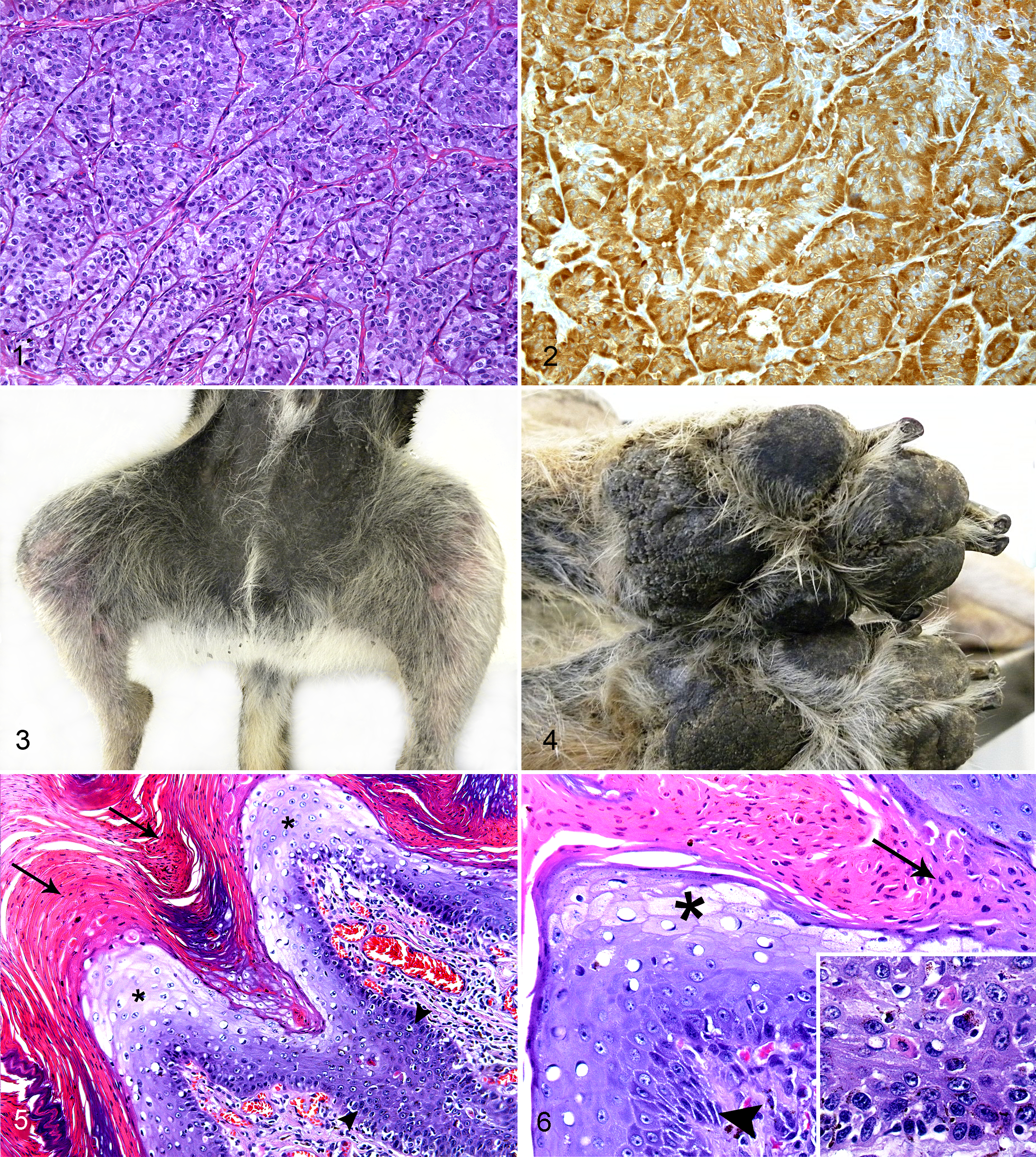
Pancreas; dog. Representative section of the islet cell carcinoma composed of polyhedral cells arranged in closely packed nests separated by a fine fibrovascular stroma. Hematoxylin and eosin (HE).
Serial sections from the neoplasm were immunohistochemically examined for 2 markers to characterize the type or hormone-producing cells. Briefly, endogenous peroxidase activity was inhibited by incubation in 3% H2O2 for 30 minutes. Sections were blocked for 60 minutes with 30% goat serum prior to application of the primary antibodies. No antigen retrieval was needed. Slides were incubated with a guinea pig polyclonal anti–insulin antibody (I-8510; Sigma-Aldrich, St Louis, MO) at a 1:200 dilution and a polyclonal rabbit anti–glucagon antibody (Ref. 219-01; Signet Laboratories, Dedham, MA) at a 1:100 dilution, overnight at 4°C. Secondary anti–guinea pig Ig horseradish peroxidase (HRP) complex (P0141; Dako, Glostrup, Denmark) and anti–rabbit Ig biotinylated complex (E0432; Dako) were used. The slides were then rinsed and incubated with an avidin-biotin complex (32020; Thermo Scientific, Rockford, IL). Reactivity was revealed with 3,3′-diaminobenzidine in a chromogen solution kit (K5007; Dako). Slides were counterstained with hematoxylin, dehydrated with graded ethanol, cleared in xylene, and then coverslipped. Negative controls were carried out with omission of the primary antibodies. For positive controls, pancreas from a healthy dog was used.
A large number of neoplastic cells (90%) had strong positive cytoplasmic reactivity for anti-insulin antibody (Fig. 2) but were negative for anti–glucagon antibody. From these results, a diagnosis of an insulin-producing beta cell carcinoma was established.
After surgery, the patient did not show clinical signs for following 12 months. During this period, 2 abdominal ultrasounds (ultrasonographic monitoring of the abdominal cavity) revealed no abnormalities, and both glucose and insulin levels (14.2 μUI/ml) were within the normal limits at the end of this period.
After 1 year free of clinical signs, the patient presented with ulcerative dermatitis and lymphadenomegaly. On dermatologic examination, hyperpigmented areas associated with moderate erythema were observed in the inguinal area. Paw pads were hyperkeratotic, and ulcers were present in the inguinal area. Superficial skin cytologies revealed the presence of neutrophils and cocci in the inguinal area and ulcerated lesions. A seric enzyme-linked immunosorbent assay (ELISA) test to detect anti-Leishmania antibodies was negative. Serum biochemical parameters were within normal reference ranges and revealed normoglycemia. Urine-specific gravity was 1015, and direct urine examination and reactive strip were normal. The patient was sent home with cephalexin (25 mg/kg/12 h per os [PO]) and 3% clorhexidine shampoo bathing every 3 days. On reexamination 21 days later, the lesions had become more pronounced and had spread to involve the eyelids, muzzle, and both pinnae. Cytologies revealed the presence of neutrophils and cocci in the affected areas. Bacterial resistance was suspected, and fusidic acid cream every 12 hours was added to the antibiotic regime. Vaseline was applied to the hyperkeratotic paw pads. Lesions partially responded to this treatment and were stable for the following 3 months. After this period, the patient showed signs of abdominal pain, his mental state changed from alert to depressed, and the cutaneous lesions became more exudative, pruritic, and painful. Yeast overgrowth was detected on cytological examination, and topical miconazole and chlorhexidine rinses (Malaseb shampoo) were prescribed. Neither complete blood count nor biochemical analysis was performed. Four weeks later, due to lack of response to the treatment, the animal was humanely euthanized, and subsequently, a complete necropsy was performed.
On gross examination, cutaneous lesions had the same appearance and distribution as previously described (Figs. 3, 4). The pancreatic lymph nodes were increased in size (1–2 cm in diameter) and in cut surface were firm and gray. In the liver, 3 dispersed, firm, whitish to yellowish nodules 1 to 3 cm in diameter were observed within an otherwise normal parenchyma. No nodules were found within the pancreatic parenchyma. Samples from pancreatic and mandibular lymph nodes, liver, pancreas, stomach, small and large intestine, adrenal glands, kidney, spleen, lung, heart, hypophysis, thyroid, brain, and skin from the affected areas and paw pads were fixed in 10% formalin and routinely processed for histopathology.
Histopathology revealed invasion of pancreatic lymph nodes. The neoplastic cell proliferation exhibited features similar to those of the pancreatic tumor tissue previously excised. There was no evidence of remaining pancreatic tumor in the studied sections of the pancreas. Hepatic nodules were consistent with metastatic lesions of the previously diagnosed pancreatic neoplasia. Glucagon and insulin immunostaining was also carried out on tissue sections from all these nodules with similar results to the initial excisional biopsy of the pancreatic mass. Skin lesions consisted of focally extensive areas of hyperplastic basal and suprabasal keratinocytes with scattered apoptotic cells and moderate hyperpigmentation. The stratum spinosum showed hydropic swelling of keratinocytes, spongiosis, acanthosis, and occasional areas of clefting. Severe parakeratotic hyperkeratosis was also observed in the corneous layer with formation of large neutrophilic crusts with few bacterial colonies. Intense exocytosis of neutrophils and slight lymphoplasmacytic perivascular infiltrate of the superficial dermis were also remarkable findings (Figs. 5, 6). These lesions were consistent with SND. No other relevant lesions were found in the other tissues studied.
Discussion
Canine functional beta cell pancreatic tumors are relatively common endocrine neoplasms in dogs. In contrast to human beings, the malignant form (carcinoma) is more frequent than adenomas 2 and can be differentiated by their larger size and their invasive and early metastatic behavior. In this case, clinical signs and laboratory findings (intermittent seizures with marked fasting hypoglycemia and hyperinsulinemia) suggested that the pancreatic nodule observed by ultrasonography was an insulin-producing tumor. This was confirmed by histopathology and immunohistochemical analyses. On the other hand, microscopic examination of cutaneous lesions revealed changes typically described for canine SND.
SND is a well-characterized cutaneous disorder of dogs that shares many clinical and histopathological features with NME of human beings. It has been attributed to different metabolic and hormone imbalances, but the inconsistency of the associated biochemical and pathological findings hampers the understanding of its pathogenesis. 7
In dogs, SND has been usually seen in association with vacuolar hepatopathy or as a paraneoplastic syndrome derived from glucagon-producing islet cell tumors. 4 Nevertheless, neither hyperglucagonemia nor hepatic dysfunction has been a constant finding in cases of SND and NME. 4,5,8 In the present case, hepatic enzymes were within the normal values, and no macroscopic or microscopic lesions were identified in the liver apart of the neoplastic metastases.
SND is typically associated with low amino acid levels. 4 It is not completely understood, but it is widely accepted that increased blood insulin levels inhibit proteolysis and stimulate proteogenesis, generating both relative and total hypoaminoacidemia. 1,3 In this case, plasma samples for amino acid determination were not available. However, prolonged high levels of insulin derived from the islet cell carcinoma may have caused the appearance of the cutaneous lesions as part of a paraneoplastic syndrome. In future cases, coupled determination of insulin and amino acid levels in blood should be undertaken for a better understanding of the role that insulin plays in the development of this syndrome.
The relevance of this study lies in it being the first reported case of SND associated with an insulin-producing beta cell tumor in dogs. Further research is needed to elucidate the pathogenesis of SND in dogs, particularly with regard to the influence of insulin and amino acid levels in blood.
Footnotes
Acknowledgements
We thank Blanca Pérez, Aida Neira, Ester Blasco, and Lola Pérez for their technical assistance with histological and immunohistochemical processing. We are also grateful to Dr Enric Vidal for his help in preparing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Servei de Diagnòstic de Patologia Veterinària (Universitat Autònoma de Barcelona).
