Abstract
An unusually “lumpy” lobster, Homarus americanus, was presented to the Atlantic Veterinary College Lobster Science Centre for evaluation. The lobster was weak with numerous pale, raised, and flat areas (diameter, 3–15 mm) on the exoskeleton, some of which were ulcerated. On postmortem examination, the pale areas corresponded to accumulations of viscous to free-flowing white material, which was found in only the subcuticular connective tissues. No internal organs were affected. Direct light examination of nonstained impression smears of the material showed abundant crystals resembling uric acid, amorphous urates, and sodium urate, which were readily soluble in 1 M potassium hydroxide. Wright-Giemsa stained imprints showed numerous fine, rounded, nonstaining granules free in the background and within individual round cells. Fourier-transformed infrared spectroscopy confirmed the presence of urates or mixed urate salts. Hemolymph plasma urea (1.7 mmol/liter) and uric acid (287 μmol/liter) concentrations were slightly higher than those seen with 36-hour emersion. Histologic sections showed aggregates of vacuolated mononuclear cells in the loose subcuticular connective tissue occasionally infiltrating between underlying muscle fibers. Grossly visible urate deposits are occasionally documented in land crabs and rarely reported in the blue crab; none, however, are associated with deformation of the cuticle. Possible etiologies include increased uric acid intake or production or decreased excretion. Anecdotal reports of similarly affected lobsters have been received but are intermittent and undocumented.
To date, there are only rare reports describing the pathological deposition of material in tissues of the American lobster (Homarus americanus). 4,7 Bowser and Rosemark observed globular, calcium-containing mineral deposits on and in the inner surface of the shed exoskeletons of many juvenile lobsters (H. americanus and H. americanus × H. gammarus hybrids) during an artificial diet trial. 4 The lobsters died during or shortly after molting with dietary stress considered a contributing factor. Dove et al described “excretory calcinosis” as a condition of extracellular calcium carbonate deposits affecting a significant number of lobsters in 2002. 7 The condition involved the antennal glands and gills, which showed marked inflammation and occasionally necrosis in more chronic lesions. 7 Increased bottom water temperature was suggested as a possible cause.
The current report describes a case of multifocal, nodular, intracellular, subcuticular urate deposition in an American lobster occasionally associated with deformation of the overlying exoskeleton. Inflammation was noted only in areas where there was secondary surface abrasion of the nodules. The cytological, histological, and hemolymph biochemistry findings associated with this condition are presented. The etiology remains undetermined; however, it is likely to involve one or more of the following factors: increased dietary nitrogen (uric acid) intake, increased uric acid production, or decreased uric acid excretion.
Case History
A 718-g, intermoult, hardshell, sexually mature, female American lobster was submitted (overnight shipping packed in wet seaweed) to the Atlantic Veterinary College Lobster Science Centre for examination. The lobster had been caught off of the south shore of Nova Scotia, Canada, in May 2008 and held (specifics of holding conditions not provided) for 1 week prior to being shipped.
Initial examination revealed a weak animal with numerous hard, pale, flat, and raised nodular areas of the exoskeleton of varying sizes (up to 15 mm in diameter) (Figures 1–3). The apex of some nodules was depressed and/or ulcerated. In other areas, external lesions were limited to pale discolouration of an otherwise normal exoskeleton. Hemolymph was collected (3-ml syringe, 22G needle) from the ventral abdominal sinus after surface disinfection with 70% alcohol, immediately centrifuged (3,500 × g for 5 minutes, 4°C) and the cell-free supernatant (“plasma”) analyzed for urea, uric acid, and total protein concentrations, using an automated biochemistry analyser (Cobas c501, Roche Diagnostics Corporation, Indianapolis, Indiana). Hemolymph plasma osmolality was determined by freezing point depression (μOsmette, Precision Systems Inc, Natick, Massachusetts). The lobster was euthanized by intrasinus potassium chloride injection as approved by the Animal Care Committee at the University of Prince Edward Island. 1
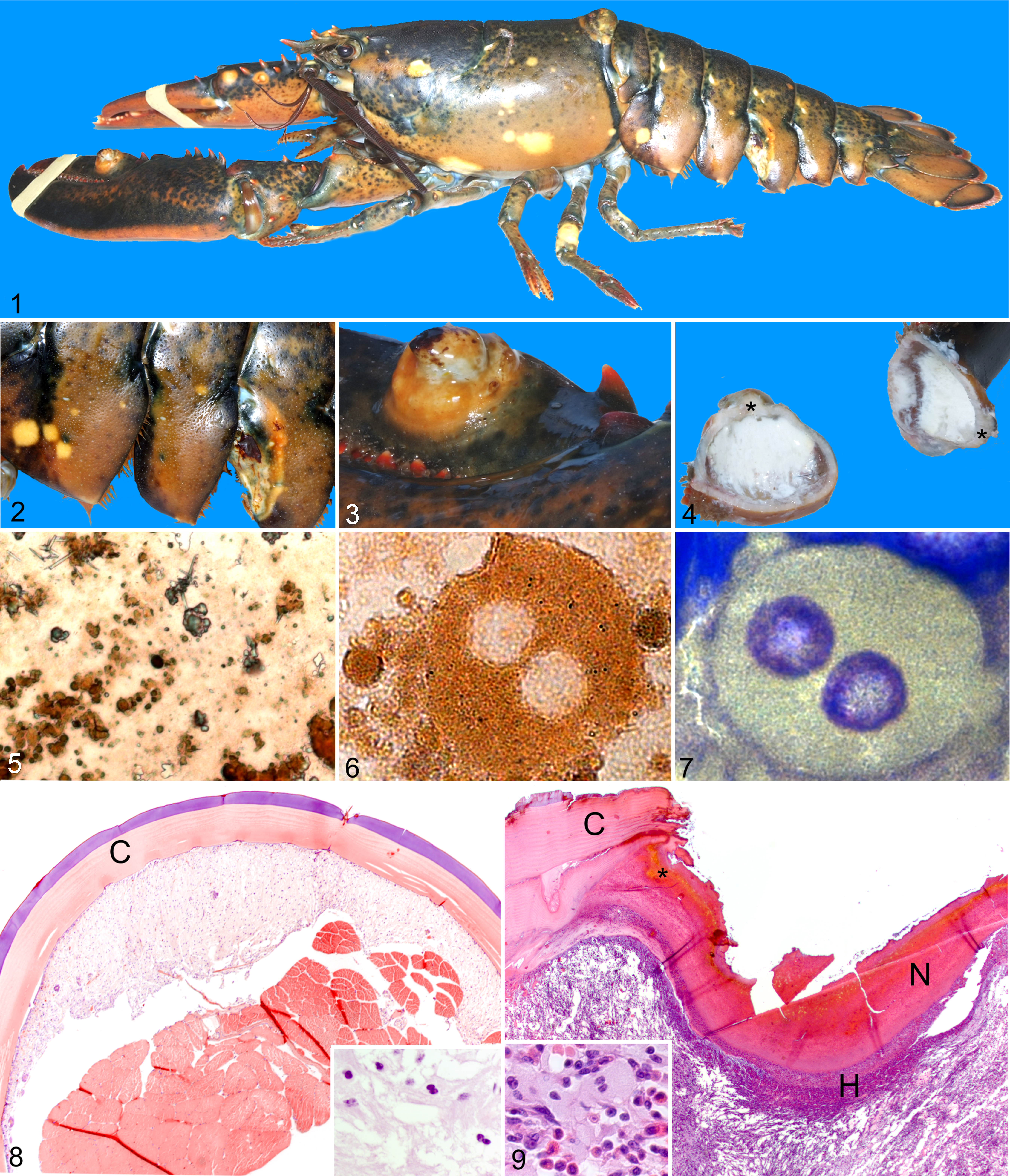
On postmortem examination, the external nodules corresponded to focal areas of white to pale yellow, viscous to free-flowing, slightly granular material limited to the subcuticular soft tissues. The interior surface of the shell in affected areas was smooth. No other significant lesions were noted. Impression smears of the material were made and left to air-dry for cytological evaluation of unstained and Wright-Giemsa stained slides. Representative tissue samples of heart, gill, hepatopancreas, muscle, intestine, antennal gland, ventral nerve cord, gonad, and multiple examples of the affected areas were collected and placed into 1G:4F fixative. 10 Tissues were embedded in paraffin, cut into 5-μm sections, and stained with hematoxylin and eosin for histological evaluation. A fixed sample of one of the larger deposits was submitted to the Canadian Veterinary Urolith Centre for Fourier transformed infrared (FTIR) spectroscopic, scanning electron microscopic, and x-ray radiographic analyses.
Plasma biochemistry results of the case lobster were compared to values from 27 male and female lobsters freshly caught in southwest Nova Scotia in May 2009 and a group of 18 male and female lobsters exposed to air (emersed) for 36 hours from separate studies (Battison and Lavallée, unpublished results). Uric acid and urea levels in the case lobster were above the other 2 groups, while osmolality values were comparable (Table 1).
Hemolymph plasma biochemistry data.
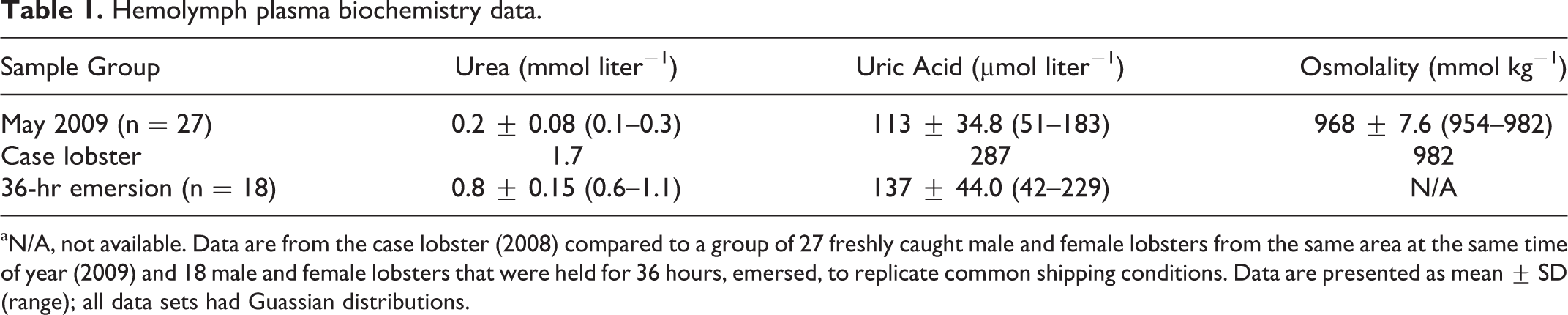
aN/A, not available. Data are from the case lobster (2008) compared to a group of 27 freshly caught male and female lobsters from the same area at the same time of year (2009) and 18 male and female lobsters that were held for 36 hours, emersed, to replicate common shipping conditions. Data are presented as mean ± SD (range); all data sets had Guassian distributions.
Examination of unstained air-dried impression smears by direct light microscopy revealed abundant crystalline material, often with a golden-brown hue, filling the background and occasionally noted within cells. The crystals grossly resembled uric acid, amorphous urates, and sodium urate (needle form) (Figure 5) and were refractile under polarised light. Addition of 1 M potassium hydroxide resulted in immediate and near complete dissolution of the crystals. Larger urate crystals formed over a period of hours after addition of 10% glacial acetic acid. These solubility characteristics are typical of those reported for urates. 17 Mono- and binucleate individual round cells with intracellular crystals were also present in low numbers (Figure 6).
Examination of Wright-Giemsa-stained impression smears revealed a background filled with rounded amber crystals, which were only slightly refringent under polarized light. Groups of deeply basophilic, uninuclear, columnar epithelial cells forming acinar arrangements (interpreted as tegmental glands) were noted occasionally. Individual, round, binucleate cells, similar to those noted in the unstained preparations, were also identified (Figure 7).
Histologic lesions were limited to the subcuticular connective tissue in areas below the gross areas of pallor in nonulcerated lesions. Irregularly arranged, vacuolated (variably sized, irregularly shaped vacuoles), lightly eosinophilic cells lacking readily defined cell borders formed nodular, unencapsulated aggregates, which extended from directly below the cuticular epithelium into the underlying loose connective tissue (Figure 8). The cells surrounded tegmental glands and striated muscle fibres. Nuclei were round to irregular and heterochromatic. Crystalline material was not evident using direct or polarized light. Special stains for glycogen, fungi, calcium and Mycobacteria species (periodic acid–Schiff, Gomori’s methanamine silver, von Kossa, and acid fast stains, respectively) were negative.
Ulceration occurred with only a few of the larger, protruding nodules and a ventral-lateral area of a segment of the tail. In these areas, the superficial periphery of the lesion was surrounded by ragged edges of the remaining cuticle (Figure 9). The subcuticular epithelium and cuticle at the center of the lesion was replaced by a thick layer of eosinophilic proteinaceous material admixed with nuclear debris, small amounts of a golden to red-brown pigment with low numbers of unidentified mixed bacteria and occasional protozoa on the surface. The pigment reduced silver (Fontana-Masson stain) consistent with identification as melanin. The underlying loose connective tissue contained a marked, diffuse mixed infiltrate of hemocytes with high numbers of large eosinophilic cytoplasmic granules (“large granular hemocytes”) and hemocytes with fewer, smaller, eosinophilic to amphophilic granules (“small granular hemocytes”) and low to moderate amounts of randomly dispersed nuclear debris. Small areas containing bundles of elongated cells with poorly defined cell borders (fibroblast-like) were also noted. Low to moderate numbers of round, occasionally binucleate, cells with finely granular eosinophilic cytoplasm were distributed throughout the hemocyte infiltrate in the area corresponding to the gross nodule. The granular cytoplasmic material was also negative upon staining for Mycobacteria (acid fast stain), calcium (von Kossa stain), glycogen (periodic acid–Schiff stain), or fungal agents (Gomori’s methanamine silver stain).
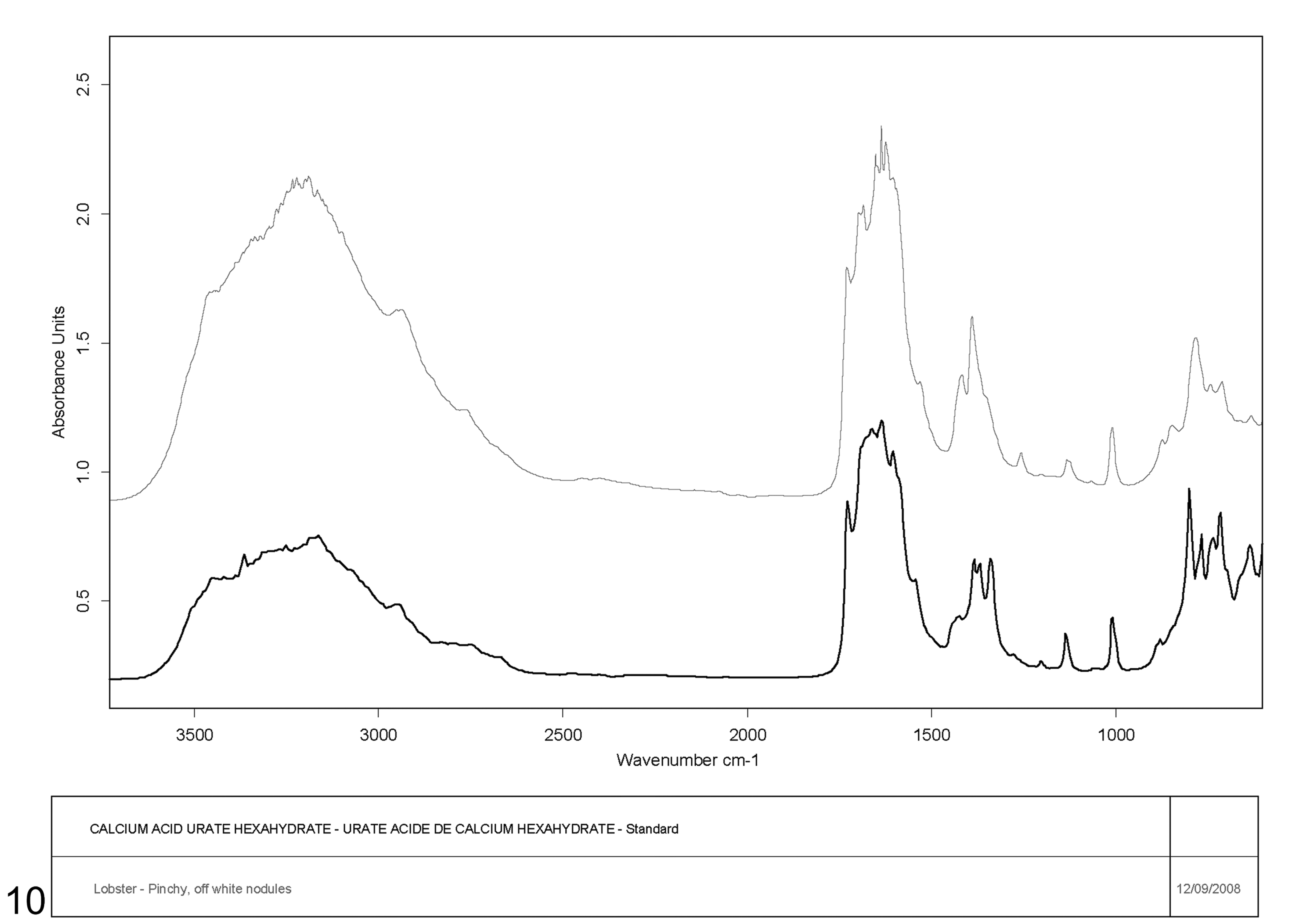
The Canadian Veterinary Urolith Centre identified 3 grossly distinct crystal types for analysis: “off-white nodule”, “tan crystal”, and “white crystal”. The first 2 crystals most closely matched the urate group of stones based on scanning electron microscopic/x-ray analysis. The FTIR spectroscopic analysis matched most closely with calcium acid urate hexahydrate although relatively little calcium with more magnesium and sodium was detected, suggesting the presence of other forms of urate stones or salts not present in the FTIR library (Figure 10). The “white crystal” was determined to be a form of urate but did not match any of the human, canine, or feline standards in the FTIR library.
Copyright 1992 Bruker Analytische Messtechnik GmbH.
Discussion
The nodular lesions observed in this lobster were due to intracellular accumulations of uric acid salts (urates) limited to the subcuticular tissues. It was interesting to note that the nature of the crystals was most easily determined by direct light microscopic examination of the unstained white material in the lesions. Processing steps required for cytological and histological examination resulted in progressive dissolution of the crystals—from rounded edges to near total absence, respectively. The finely granular nature of the material was, however, somewhat preserved in histologic sections of the areas of inflammation associated with the ulcerated lesions.
Uric acid is a type of purine, a group of nitrogen-containing organic compounds that includes the nucleic acid bases adenine and guanine, as well as hypoxanthine and xanthine, caffeine, and theobromine. Hemolymph uric acid can be of dietary origin or the result of catabolism of nucleic acids. 6,12 Uric acid can be a precursor for urea following the actions of uricase, allantoinase, and allantoicase. 6,13 Urea can be subsequently hydrolysed by urease to ammonia and CO2. 6,12
American lobsters, as other aquatic crustaceans, are ammonotelic—ammonia is the main nitrogenous waste product, with only minimal amounts of urea and uric acid produced under normal conditions. 5 The ammonia is excreted across the gills, directly into the external environment, down the favorable concentration maintained by continual replacement of the water in the gill chamber during respiration. 9 In contrast, the antennal glands, despite producing a “urine,” function primarily in an osmotic and ionic regulatory capacity. 3
Grossly evident urate deposits have been reported infrequently in other crustaceans; however, the current case is distinguished by the very restricted subcuticular distribution of the deposits. Intracellular urate deposits in the land crab Gecarcoidea natalis were found in spongy connective tissues throughout the body, including the subcuticular tissues, within specialized urate storage cells. 13 Accumulations could be quite extensive and were positively correlated with dietary nitrogen content. The authors speculated that the urates were being absorbed by and stored in the cells rather than being directly synthesized, as there was little in the way of cellular organelles present. 13 The deposits were also considered as a potential storage site for nitrogen reserves. Notably, these deposits did not deform the overlying exoskeleton. In a separate study, the authors clearly showed that G. natilis was capable of de novo purine synthesis using [ 15 N] glycine studies, which was consistent with findings in Artemia species but in contrast to earlier suggestions that crustacea in general may lack this capability. 6,14,15 Diffuse intrahemocoelomic urate deposits, often adherent to organ surfaces, were described for the land crab Cardisoma guanhumi. 8 These deposits were noted to be subjectively greater in laboratory-reared crabs. Intracellular, pericardial, urate deposits have also been described in the blue crab (Callinectes sapidus). 11
Further work by Linton and Greenaway in Gecarcoidea natalis described a route of uric acid synthesis and transport by demonstrating the presence of some of the required enzymes for uric acid synthesis (xanthine oxidoreductase) and catabolism (uricase, urease) in the hepatopancreas, spongy connective tissues, and gills. 12 They suggested that the hepatopancreas is the primary site of uric acid synthesis with some synthesis also occurring in spongy connective tissue. The latter could serve as a temporary storage site when the crab is unable to excrete its nitrogenous waste as ammonia. Urates are then catabolized to allantoic acid, allantoin, or, most likely, urea for transport via hemolymph to the gills. Urease in the gills would then catabolize urea to ammonia and CO2 for excretion. A similar process has been suggested for Nephrops norvegicus where both hemolymph urea and uric acid levels increased with emersion for 24, 48, and 72 hours, followed by above-normal ammonia excretion rates upon reimmersion. 2
Studies in the Kuruma shrimp Marsupenaeus japonicus have shown, via tissue biochemical analysis, that uric acid accumulates (2.2-fold increase) in the epidermis as part of the physiological response to environmental salinity being increased from 18 to 42 parts per thousand. 5 The overall response to increased salinity was a depletion of hepatopancreatic uric acid with a concomitant increase in epidermal uric acid and increased hemolymph urea and uric acid levels. Neither gross nor histologic findings were reported in the study.
Multifocal deposition of uric acid within the epidermal cells in insects such as the Dysdercus species is used intentionally to create a pigmentation pattern. 16 In the current case, while the uric acid crystals were located below the epidermis in the loose connective tissue, they may have had a similar effect.
Plasma biochemistry analysis in the present case showed changes that could be consistent with prolonged emersion (increased urea and uric acid). Unfortunately, urea and uric acid values from the case lobster under immersion conditions were unavailable for comparison to the reference values for immersed lobsters from a similar area. These results may have provided clues as to the normal route of nitrogen excretion in the case lobster.
The etiology of the lesions in this case remains undetermined. Others have shown that crustaceans have the capacity to switch to uric acid production under emersion stress. However, it is unlikely that the lobster in the current case experienced repeated and prolonged emersion to the extent to cause the accumulation of the large amount of urates seen in the tissue. Given the proposed normal route of synthesis and excretion of nitrogenous products in other crustacean species, intracellular accumulation in the connective tissues in the current case is not entirely unexpected. Possible causes to consider include increased dietary nitrogen intake or altered nitrogen metabolism. The latter could include a wide range of conditions, such as decreased uric acid catabolism, for example, due to a lack of uricolytic pathway enzyme(s) resulting in a form of “storage disease”; inappropriately increased uric acid production; or response to salinity stress. It is speculated that the nodular lesions were a result of urate deposits accumulated in the inter- or premoult period under a hardened exoskeleton. With the subsequent moult, the new, soft exoskeleton may have adopted the nodular shape upon hardening. As only relatively few of the lesions were ulcerated, this was attributed to mechanical abrasion of the raised surfaces and considered a secondary change.
According to anecdotal evidence, this is not a unique case, with similar lobsters being observed sporadically in the fishery. Further investigation of this disease will include determining factors such as the regional and seasonal distribution patterns, associated environmental conditions (eg, water temperature, salinity, oxygen), signalment (sex, size, moult stage, etc), in addition to characterization of plasma biochemistry parameters (eg, ammonia, urea, uric acid, and activities of enzymes involved in nitrogen metabolism) and ammonia excretory capacity of affected lobsters.
Footnotes
Acknowledgements
Special thanks go to the Canadian Veterinary Urolith Centre for analyzing the crystals and to Shelley Ebbett (Atlantic Veterinary College) for her photographic skills.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the Atlantic Lobster Moult and Quality project delivered by the AVC Lobster Science Centre represents a consortium of private sector companies, fishermen’s organisations, First Nations and provincial and federal government agencies, including the Atlantic Innovation Fund through the Atlantic Canada Opportunities Agency.
