Abstract
Extramedullary hematopoiesis (EMH) is the formation and development of blood cells outside the medullary spaces of the bone marrow. Although widely considered an epiphenomenon, secondary to underlying primary disease and lacking serious clinical or diagnostic implications, the presence of EMH is far from incidental on a molecular basis; rather, it reflects a well-choreographed suite of changes involving stem cells and their microenvironment (the stem cell niche). The goals of this review are to reconsider the molecular basis of EMH based on current knowledge of stem cell niches and to examine its role in the pathophysiologic mechanisms of EMH in animals. The ability of blood cells to home, proliferate, and mature in extramedullary tissues of adult animals reflects embryonic patterns of hematopoiesis and establishment or reactivation of a stem cell niche. This involves pathophysiologic alterations in hematopoietic stem cells, extracellular matrix, stromal cells, and local and systemic chemokines. Four major theories involving changes in stem cells and/or their microenvironment can explain the development of most occurrences of EMH: (1) severe bone marrow failure; (2) myelostimulation; (3) tissue inflammation, injury, and repair; and (4) abnormal chemokine production. EMH has also been reported within many types of neoplasms. Understanding the concepts and factors involved in stem cell niches enhances our understanding of the occurrence of EMH in animals and its relationship to underlying disease. In turn, a better understanding of the prevalence and distribution of EMH in animals and its molecular basis could further inform our understanding of the hematopoietic stem cell niche.
Extramedullary hematopoiesis (EMH) is the formation and development of blood cells outside the medullary spaces of the bone marrow. The ability of blood cells to home, proliferate, and mature in the extramedullary organs of adult animals involves pathophysiologic alterations in stem cells and their microenvironment, which include extracellular matrix, stromal cells, and local and systemic chemokine production. EMH involving single or multiple cell lineages is observed frequently by veterinary pathologists in tissues examined by fine-needle aspiration or histopathology, yet studies of the prevalence and underlying causes of EMH in animals are limited.
Why Is Understanding EMH Important?
Part of the reason for a lack of studies on EMH is that it is widely considered an epiphenomenon: secondary or accessory to underlying primary disease, sometimes occurring independently or without an obvious underlying cause, and lacking serious clinical or diagnostic implications. Indeed, EMH in animals rarely has been associated with clinical signs. We are aware of only 1 report, in which neurologic signs were attributed to EMH within the choroid plexus of 5 dogs having no apparent blood, cerebrospinal fluid, or brain abnormalities. 7 This contrasts with humans, 63% of whom have symptoms associated with nonhepatosplenic EMH, mainly neurologic deficits due to nodular EMH located near or within the spinal canal, or hemorrhagic effusion due to EMH within the thoracic, abdominal, or pericardial cavity. 9,53,73,125 Because of a lack of clinical signs associated with EMH in animals, it is sometimes considered to be an incidental finding of no consequence to the animal; its importance lies mainly as a potential indicator of underlying hematologic disease.
EMH only occasionally presents a diagnostic challenge to veterinary pathologists. In histologic sections, EMH sometimes can be difficult to differentiate from myeloproliferative disease or inflammation. 64 Inflammation contains primarily mature myeloid cells, so it can usually be distinguished from EMH, which contains many developmental stages. 64 Differentiation between EMH and polycythemia vera or chronic myeloid leukemia can be more problematic, since most or all developmental stages of cells can be found in these neoplasms. In a study of human splenic hematopoietic proliferations, higher numbers of CD34+ CD117+ (immature hematopoietic) cells were significantly associated with neoplastic myeloid proliferations such that immunochemistry might be helpful in distinguishing myeloid neoplasia from EMH. 79 The presence of erythroid precursors and megakaryocytes also can help differentiate EMH from chronic myeloid neoplasms or from inflammation. Given the low prevalence of chronic myeloid and erythroid leukemias in animals and the availability of routine hematologic data and immunophenotypic markers, distinguishing EMH from these diseases is not a frequent diagnostic problem.
In the absence of major clinical and diagnostic implications, why then is it important to know more about the pathophysiologic mechanisms of EMH? Recent developments in our understanding of stem cells and the stromal and chemical mediators of stem cell proliferation have the potential to newly inform our understanding of EMH, its pathophysiology, and its relationships to underlying disease and tissue microenvironment. Although most research is focused on medullary hematopoiesis, similar molecular and cellular mechanisms underlie embryonic/fetal hematopoiesis as well as the establishment or reactivation of hematopoietic stem cell (HSC) niches leading to EMH. Diagnostic interpretation of EMH is aided by understanding the current theories underlying its development, including recent novel studies on the occurrence of EMH at sites of tissue injury, inflammation, and repair. It is clear that EMH is far from incidental on a molecular basis, even if a full understanding of its development remains to be elucidated.
The goal of this review is to critically synthesize new and emerging concepts about hematopoiesis and stem cells with current theories on the pathophysiologic mechanisms of EMH and its occurrence in various diseases, conditions, and lesions in animals. In addition, we hope to stimulate and help focus new research on the prevalence, distribution, and molecular basis of EMH in animals, which in turn will inform our knowledge of stem cells and their microenvironment.
Terminology
Terminology used to describe EMH can be confusing and potentially misleading. EMH is sometimes referred to, especially in medical pathology, as “myeloid metaplasia” or “agnogenic myeloid metaplasia.” 61 These terms originated to describe primary myelofibrosis in humans (which causes marked EMH) and sometimes are used to refer to EMH caused by other myelofibrotic or myelodysplastic syndromes. 64 The International Working Group on Myelofibrosis Research and Treatment recently rejected use of these terms since their meaning no longer is applicable within the context of the disease. In other contexts, the term myeloid metaplasia is sometimes used to refer to EMH with granulocytic (myeloid) predominance. 64,113 In one study, myeloid metaplasia was used to refer to diffuse proliferation of hematopoietic elements, whereas EMH was defined as discrete foci of hematopoiesis. 101
Because of potential confusion associated with these various terms and usages, we will solely use the term EMH to refer to any hematopoiesis outside the bone marrow. In light of current knowledge about EMH and to avoid ambiguity, we recommend that the term myeloid metaplasia be discontinued altogether as a synonym for any type of EMH.
HSCs and Microenvironments
The concept of a stem cell “niche” was first proposed in 1978 by Schofield, who hypothesized that a HSC in bone marrow acts as a fixed tissue cell whose further maturation is prevented by contact with the surrounding cellular environment. 97 The stem cell niche or microenvironment plays a central role in hematopoietic cell homing and function in prenatal and postnatal hematopoiesis. Embryonic hematopoiesis sets the stage for adult hematopoiesis and for the potential development of EMH. However, most of what is currently known about the cellular and chemical interactions in stem cell niches is based on bone marrow hematopoiesis in adults. Therefore, details of molecular interactions will be emphasized in the section on medullary hematopoiesis, although they may be relevant to embryonic and EMH.
Embryonic and Fetal Blood Cell Production: Differentiation of Stem Cells and Their Niche
The development (ontogeny) of blood and blood-forming organs in the embryo and fetus sets the stage for the multipotency and dynamics of the processes involved in medullary (and extramedullary) hematopoiesis in adult animals. Differentiation of HSCs and microenvironments in prenatal sites of hematopoiesis subsequently determines the characteristic tissue distribution, or “footprint,” of the most common sites of EMH in adult animals. Most of what is known about the ontogeny of blood cell production is based on studies of murine, avian, and human embryos. More recently, the zebrafish has been used as an experimental model to dissect the molecular basis of hematopoiesis via genetic screening and imaging techniques. Readers are referred to a comprehensive recent review of hematopoietic development in the zebrafish. 82
Embryonic hematopoiesis originates in the yolk sac with the hemangioblast, a progenitor cell having both hematopoietic and vascular potential. 19,23 Selective proliferation of subsequent mesodermal cells results in “primitive” and “definitive” stages of hematopoiesis as well as the development of adjacent sinuses and vessels. 41 Primitive hematopoiesis develops within blood islands (angioblastic cords) and results in formation of primitive erythroid cells, while definitive hematopoiesis includes both erythropoiesis and myelopoiesis. 32 However, HSC precursors have been demonstrated in the yolk sac, 94 and current evidence now supports the likelihood of multipotential hematopoiesis in the yolk sac as monocytic and megakaryocytic progenitors have been isolated from the presomite stages of mouse embryos. 65
Definitive erythroid progenitors and committed myeloid precursors produced in the yolk sac subsequently colonize the liver. In addition, pluripotential HSCs continue to be generated in the yolk sac as well as the para-aortic splanchnopleure and murine placenta. 21,94 Fetal liver does not generate HSCs de novo but instead serves as a site for expansion and maturation of HSCs and hematopoietic progenitor cells (HPCs). Hematopoiesis shifts from liver to spleen in a time-dependent manner that varies with species and cell lineage. In avian embryos, HSCs and progenitor cells colonize the spleen early in embryonic development, and splenic hematopoiesis continues through birth. 122 The spleen emerges as an important hematopoietic organ in human, bovine, and likely equine fetuses in mid- to late gestation and contributes somewhat less in dogs and cats. 35,69,92,107
In neonatal mice, humans, dogs, cats, cattle, and pigs, persistent residual sites of fetal hematopoiesis are retained—mainly in spleen but also in liver and lymph nodes—that regress with growth and disappear prior to adulthood. 45,78,108,113 In mice (and, to a lesser extent, rats and hamsters), the spleen continues to play an active role in hematopoiesis throughout adulthood. 106 The shift of hematopoietic activity to spleen late in fetal development, with retention of minor residual hematopoiesis in other hemic organs (liver, lymph nodes), is consistent with the primary tissue distribution of EMH in adult animals.
Studies in mice have shown that the developmental stage of the microenvironment can influence the developmental stage of transplanted HPCs, highlighting the microenvironment as a key factor in the homing, retention, support, and differentiation of hematopoietic cells in embryonic and fetal hematopoiesis. 28,72 For example, when adult marrow HSCs were injected into blastocysts, the cells reverted to an embryonic/fetal differentiation course of gene transcription, and vice versa. 28 In another study, distinct patterns of gene expression and the ability to support hematopoiesis were found in stromal cells isolated from fetal hematopoietic tissues at various developmental stages and from adult bone marrow. 119 Studies also have suggested that there are limitations to the ability of the microenvironment to influence hematopoietic cell fate. For example, adult HSCs were unable to produce fetal-type T cells when transplanted into fetal thymus, indicating a fundamental difference in developmental potential between fetal liver and adult marrow HSCs independent of microenvironment. 42 Even as the rapid pace of technology expands the experimental evidence in this area, differentiation of the hematopoietic microenvironment has emerged as a key factor in blood cell ontogeny and in creation of the stem cell niches that support medullary hematopoiesis in adult animals.
Current Concepts of Stem Cell Niches in Medullary Hematopoiesis
The shift to bone marrow hematopoiesis occurs late in fetal development or at birth, with the establishment of intramedullary stem cell niches to which circulating HSCs home and establish residence for self-renewal and differentiation. At present, 2 main types of HSC niches in the bone marrow are proposed: an osteoblastic (endosteal) niche and a vascular niche. In any stem cell niche, hematopoietic or nonhematopoietic, the conserved central elements are as follows: stromal cells that promote cell-cell adhesion and secrete soluble factors that signal stem cells in a paracrine manner; extracellular matrix proteins that anchor stem cells and provide a physical scaffolding; blood vessels to provide nutritional support and systemic signals and to convey stem cells into the periphery; and neural inputs that integrate signals from different organ systems. 24 The stromal cell components of the bone marrow microenvironment include osteoblasts, fibroblasts, adipocytes, chondrocytes, and vascular cells (endothelial and abluminal cells, vascular smooth muscle cells, and pericapillary pericytes). Most of these stromal cell populations derive from mesenchymal stem cells (MSCs) in the bone marrow. 13,118 Marrow stromal cells create a microenvironment supportive of hematopoieis via elaboration of growth factors, chemokines, and extracellular matrix proteins, including hemonectin, fibronectin, laminin, proteoglycans, and collagen fibers.
The osteoblastic niche
An osteoblastic niche was first implied by homing studies of transplanted HSCs that tended to lodge close to the endosteal surface of bone, while lineage-committed and terminally differentiated cells predominantly distributed to the central marrow region. 76,77 Osteoblasts likely play an important role in regulating medullary hematopoiesis. They produce hematopoietic growth factors, including granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), hepatocyte growth factor, and interleukin-6 (IL-6). 109,110 Conditional ablation of osteoblasts results in reduction of hematopoietic cells, including HSCs. 115
Osteoblasts and stromal cells synthesize and secrete stromal cell-derived factor-1 (SDF-1; also known as CXCL12), an α-chemokine that binds to the G-protein–coupled receptor CXCR4 on the surface of hematopoietic cells and other cell types, including some tumor cells. Deficiency of either SDF-1 or CXCR4 causes embryonic lethality, as SDF-1/CXCR4 signaling is essential for marrow hematopoiesis as well as cardiovascular formation and neurogenesis. 74,131 Of note, fetal liver hematopoiesis is not impaired by deletion of SDF-1 or CXCR4. Production of SDF-1 by marrow osteoblasts and other stromal cells induces homing of CXCR4+ hematopoietic cells to the bone marrow, 86 and SDF-1/CXCR4 signaling is required to maintain HSCs within the marrow. 75,104
Critical interactions between osteoblasts and HSCs result in retention of HSCs within the marrow, including formation of an N-cadherin/β-catenin adhesin complex. 130 Other interactions involve the secretion of Jagged protein by osteoblasts, the triggering of HSC signaling via the Notch receptor, and expression of angiopoietin-1 on osteoblasts. The latter activates Tie2 on the HSC surface, thereby promoting tight adhesion of HSCs within the niche. 2 Angiopoietin/Tie2 is a tyrosine kinase signaling pathway that is crucial to remodeling of vasculature in the embryo and increasingly reported as an important pathway in hematopoiesis. 54 Additionally, a population of nestin-positive MSCs has been identified in bone marrow in close association with HSCs and adrenergic nerve fibers. 67 These cells express genes for HSC maintenance that are downregulated during induced HSC mobilization.
The osteoblastic niche is characterized by a hypoxic environment that plays a fundamental role in the preferential concentration of HSCs in the region and in regulating their quiescence; hypoxia favors self-renewal rather than proliferation and is thought to protect the cells from damaging oxygen radicals. 85,90 Low perfusion and low oxygen tension in the endosteal region exist despite high vascularity, presumably because blood velocity in the endosteal sinusoids is one-tenth of that in central medullary capillaries. 59 Overall, cellular and molecular interactions within the osteoblastic niche appear to induce the quiescent microenvironment that supports long-term survival of HSCs.
The vascular niche
Consistent with their close apposition in the developing embryo, HSCs have been identified in association with sinusoidal endothelium in bone marrow and spleen, leading to the classification of a vascular niche. 52,55 The vascular niche is a relatively new concept, and factors leading to establishment of HSCs within the niche are not well understood. However, studies on HPCs have shown that recruitment of progenitor cells, including megakaryocytic progenitors, to the vascular niche involves signaling through SDF-1 and fibroblast growth factor 4 (FGF-4). 4,52,55 FGF-4 upregulates very late antigen 4 on the surface of megakaryocytes to promote adhesion to endothelial vascular cell adhesion molecule-1 (VCAM-1). Stromal “reticular” cells producing high levels of SDF-1 and nestin-positive MSCs are often located perivascularly, and bone marrow endothelium produces SDF-1 to draw megakaryocytes toward sinusoidal junctions. 4,52 Bone marrow endothelial cells are required for megakaryocyte maturation and thrombopoiesis, consistent with an active role of vascular endothelium in hematopoiesis.
Experiments also have shown that endothelial cells derived from various sites release soluble “angiocrine” factors that promote HSC expansion. 17 Angiopoietin/Tie2 signaling, as described earlier in the context of the osteoblastic niche, plays a role in bone marrow neovascularization and hematopoietic recovery from myelosuppression, suggesting potential importance of this interaction in supporting hematopoiesis in the vascular niche. 54 Microvascular endothelial cells have recently been shown to restore hematopoiesis following bone marrow irradiation, supporting a role for endothelium in self-renewal of adult HSCs. 60 The bone marrow vascular niche may act not only to sustain hematopoiesis but also to promote transendothelial migration of HSCs. Cells adjacent to sinusoidal endothelium may sense circulating soluble factors and respond by quickly mobilizing to the periphery. 52
An interactive relationship between the vascular and osteoblastic niches within the bone marrow is proposed and may be critical in regulation of HSC mobilization. While long-term HSCs favor the hypoxic osteoblastic niche in the endosteal region, more active proliferating HSCs and differentiating hematopoietic cells reside in the well-perfused vascular niche in the central medullary region of the marrow where they can access necessary oxygen and nutrients. Recruitment of HSCs from the osteoblastic to the vascular niche appears to rely on increasing gradients of oxygen and FGF-4 encountered by cells migrating into the vascular niche. 128 HSCs are stimulated to enter the cell cycle and differentiate as well as migrate toward the vascular niche via conversion of membrane-associated c-kit ligand (stem cell factor) into soluble c-kit ligand due to the activity of SDF-1 and vascular endothelial growth factor (VEGF) on matrix metalloproteinase 9. 37 Although the osteoblastic and vascular niches confer a general spatial organization to the distribution of hematopoietic cells in the marrow, these areas overlap anatomically, so a clear distinction is simplistic; of more importance is the unique balance of cells and signals, including hypoxia, within each cellular niche.
Erythroblastic islands
Erythroblastic islands are essentially microniches for red blood cell production in the bone marrow (also in the spleen during marked EMH). Readers are referred to an excellent recent review of the erythroblastic island niche by Chasis and Mohandas. 14 An erythroblastic island includes a central resident macrophage (CD11c+ CD18+) that physically and chemically provides a microenvironment for erythroblast development, enucleation, and phagocytosis of extruded nuclei. The central macrophage and developing erythroblasts are connected via adhesive interactions that facilitate signaling and provide regulatory feedback. One important molecular mediator is erythroblast-macrophage protein. In facilitating the attachment of erythroblasts to macrophages, erythroblast-macrophage protein inhibits apoptosis and promotes terminal maturation of erythroid precursors, leading to the eventual enucleation of normoblasts (metarubricytes). 36 The potential role of macrophages as facilitators or mediators of EMH in spleen, liver, lymph nodes, and other tissues where macrophages are increased in association with disease processes has not been fully explored but warrants further study.
Activation of Stem Cell Niches in Extramedullary Sites
The environmental conditions and cells (including resident populations of relevant stromal cells and possibly HSCs) in hemic tissues such as spleen and liver remain favorable for the initiation or reactivation of a stem cell niche. However, EMH—and, therefore, presumably the requisite stem cell microenvironment—can occur in any tissue. 64,78 The activation of vascular niches appears to be important in establishing and sustaining hematopoietic activity in extramedullary sites such as the spleen and other tissues (and possibly neoplasms) in association with endothelium or vessels (Fig. 1). In a sense, vasculature serves as an extension of the bone marrow vascular niche and supports the homing, expansion, and differentiation of HSCs in a variety of tissues on an as-needed basis to meet systemic and local needs. Development of an extramedullary vascular niche is facilitated by the release of soluble factors by endothelial cells that promote HSC expansion and, possibly, by resident macrophages. 17 Activated macrophages are considered by some to be crucial in the development of EMH because of their widespread and often perivascular distribution in tissues, their ability to induce stromal proliferation, and their close morphologic and functional relationships with other cells. 7,70,93
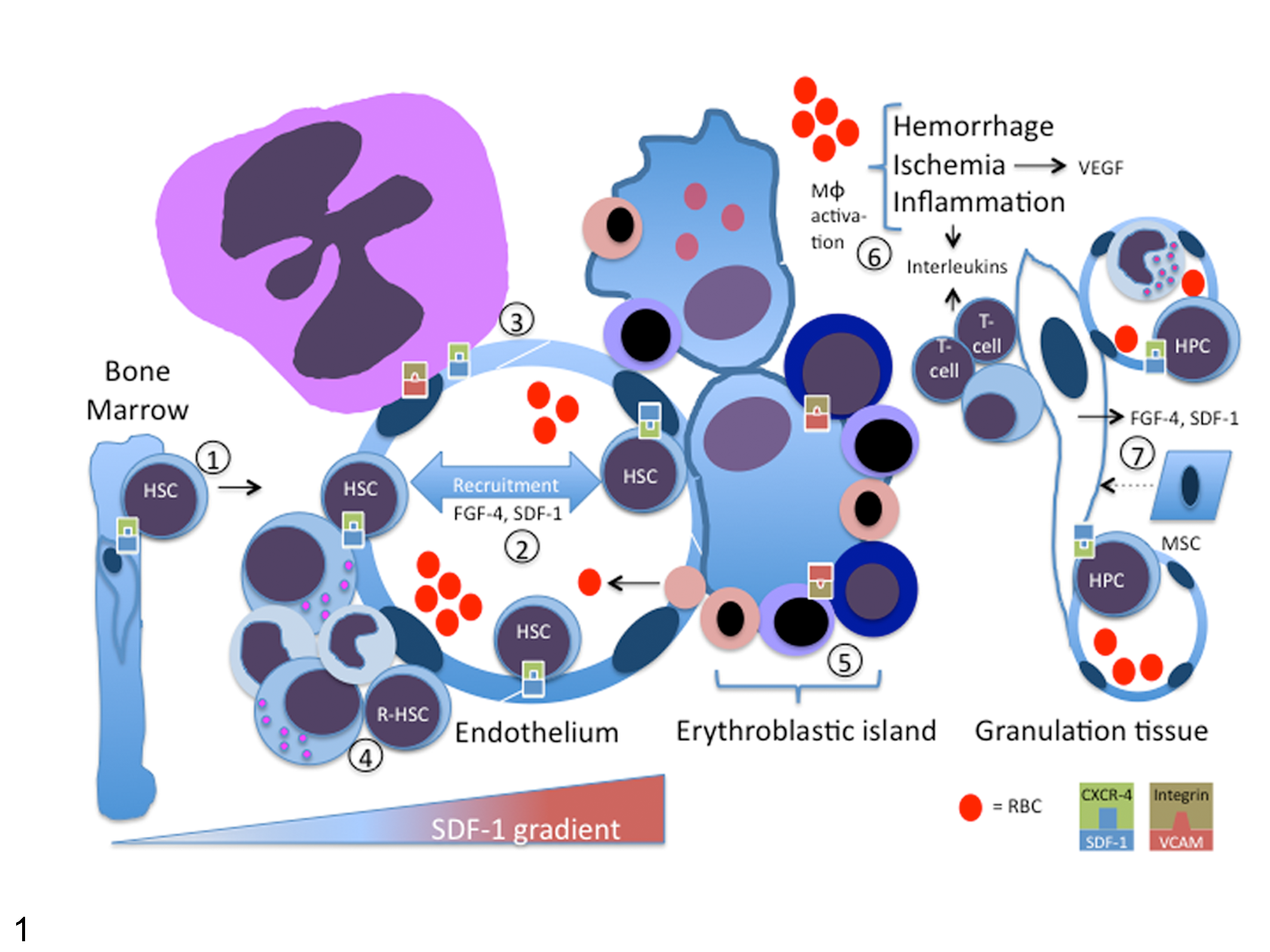
Pathogenesis of vascular niche formation in extramedullary hematopoiesis (EMH). Endothelial cells, hematopoietic cells, and macrophages are the key cellular components of a vascular niche, where a myriad of cytokines, growth factors, signaling pathways, and adhesion molecules promote the proliferation and differentiation of hematopoietic cells. (1) Insults such as hypoxia and myelostimulation alter stromal cell–derived factor 1 (SDF-1) gradients by decreasing levels of SDF-1 in bone marrow and/or increasing production at sites of tissue inflammation or injury, resulting in a gradient shift favoring mobilization of hematopoietic stem cells (HSCs) and hematopoietic progenitor cells (HPCs) from the marrow. Disruption of the SDF-1/CXCR4 interaction between HSCs and stromal cells is mediated in part by matrix metalloproteinase 9 (MMP-9), leukocyte proteases, soluble Kit ligand, and granulocyte colony-stimulating factor (G-CSF). (2) Expression of SDF-1 and production of fibroblast growth factor-4 (FGF-4) by endothelial cells promotes HSC and HPC homing and adhesion to the vascular niche and stimulates their division and maturation. (3) SDF-1 is also chemotactic for megakaryocytes and, with FGF-4, enhances expression of integrins (very late antigen 4 [VLA-4], binding vascular cell adhesion molecule 1 [VCAM-1]) that stimulate megakaryocyte adhesion to endothelium and subsequent maturation. (4) In tissues such as spleen, resident HSCs are also present that are stimulated to divide and mature in the developing niche environment, in this case leading to a focus of myelopoiesis. (5) Erythroblastic islands form around tissue macrophages such as splenic macrophages and Kupffer cells. Macrophages and erythroid precursors express adhesive molecules such as erythroblast macrophage protein, VCAM-1, and α1 β1 integrin that facilitate maturation and egress of mature cells into the circulation. (6, 7) Macrophages (especially erythrophagocytic macrophages), fibroblasts, and activated lymphocytes contribute to the microenvironment of a vascular niche at sites of tissue hemorrhage, ischemia, inflammation, and repair through secretion of interleukin-3, -4, -5, and -6. These and other interleukins and cytokines contribute to the recruitment and homing of HSCs and mesenchymal stem cells (MSCs) and their local proliferation and maturation. Stromal cells constitutively express SDF-1 and produce FGF-4. Hypoxia contributes to an optimal HSC microenvironment and leads to vascular endothelial growth factor (VEGF) production, which enhances endothelial-mediated EMH.
It has been long known that hematopoietic precursors are present in the circulation; 124 in fact, HSCs likely travel into and out of the circulation many times, which provides an immediately available pool that can be recruited rapidly for EMH. Bone marrow–derived HSCs and HPCs have been found within vessels and adjacent to endothelial cells in spleen, liver, myocardium, lung, kidney, skeletal muscle, small intestine, and adipose tissue. 3,34,38 Many of these tissues are frequently reported sites of EMH in adult animals, along with the adrenal glands and central nervous system. 7,15
The area adjacent to sinusoidal endothelium in the spleen appears to provide a sufficiently hypoxic environment to support a resident population of HSCs (similar to the osteoblastic niche in the marrow). Preferential homing and expansion of this HSC pool would be expected in systemic hypoxia, contributing to increased EMH (see stress erythropoiesis below). In adipose tissue, a hypoxic environment and complex vasculature surrounded by stromal cells likely favor residence of HSCs and HPCs. 34 EMH is thought to occur preferentially in the adrenals because of their reticular meshwork of cells, which could provide the stromal factors necessary for hematopoiesis; 113 however, we are unaware of studies that address this. Interestingly, HPCs express receptors for dopamine and epinephrine, and neurotransmitters induce hematoproliferation and mobilization via the Wnt and matrix metalloproteinase signaling pathways. 102 As mentioned earlier, matrix metalloproteinases are involved in cleavage of signaling molecules and resultant mobilization of HSCs and HPCs. 37,66 However, adrenal lesions in which EMH is observed are not always functional such that the importance of these pathways in the development of EMH in this organ is unknown.
Consistent with a vascular niche, splenic EMH occurs in the red pulp, both within the stroma and intrasinusoidally. 111 In histologic sections, megakaryocytes are found near the muscular trabeculae. 64 In cytologic specimens, nucleated red blood cells (NRBCs) alone do not conclusively indicate splenic erythropoiesis, since low numbers of cells are released prematurely from the bone marrow and sequestered here. 16 However, high numbers of NRBCs, the presence of early or all stages of differentiation, and the presence of mitotic figures involving erythroid precursors would support erythroid EMH in cytologic specimens of spleen. Hepatic EMH is usually localized within the sinusoids (a site of fetal hematopoiesis) 78 but also is reported perisinusoidally, 64 in portal areas (eg, in acute myeloid leukemia), 113 and in larger portal veins. 64 Interestingly, hepatic EMH has been described in fine-needle aspiration specimens of transplanted livers in humans, which apparently have an altered microenvironment that is favorable for hematopoietic cell proliferation. 96 Kupffer cells have been identified as capable of supporting erythroid EMH in the liver in mice in which bone marrow macrophages were depleted. 81 In lymph nodes, EMH is found initially in medullary cords and progresses to the sinuses. 113 In addition to localization in sinuses or vascular tissue, EMH can be diffuse, nodular, or multifocal, possibly reflecting the underlying mechanism of niche formation (whether arising from systemic factors or a local microenvironment) as well as the architecture of the underlying tissue.
The role of the osteoblastic/endosteal niche in the development of EMH is likely limited to sites where extramedullary bone exists, as in osseous metaplasia. However, MSCs at sites of EMH could exert similar effects to those of osteoblasts in the marrow, including expression of high levels of angiopoietin-1 and VCAM-1. 67 Hematopoiesis can form in bone marrow cavities associated with osseous metaplasia, which is common in the cervical and lumbar dura mater of dogs over 2 years of age; 15 however, whether similar chemical and architectural relationships exist, as in medullary hematopoiesis, is unknown.
Species differences in predilection for EMH have been noted, but with the exception of mice, in which splenic EMH persists throughout adulthood, potential mechanisms have not been well investigated, especially in domestic animals. Splenic EMH has been reported in adult musk shrews. 26,98 In cotton-eared marmosets, apparent “spontaneous” EMH was found in mesenteric lymph nodes, spleen, liver, kidney, adrenal gland, and cerebral choroid plexus. 46,80 Marmosets and African hedgehogs (Atelerix albiventris) with a variety of underlying disorders also develop EMH in many tissues. 46,80,88 Of 14 African hedgehogs, 13 (91%) had splenic hematopoiesis at necropsy, regardless of the underlying condition. 87 The marmoset, tree shrew, and lesser hedgehog tenrec are important laboratory animal models such that future research using these species might help shed light on underlying mechanisms of EMH.
Major Theories of Development of EMH
Conceptually, EMH can develop wherever and whenever a stem cell niche/microenvironment emerges in a tissue other than bone marrow; the development or reactivation of an extramedullary stem cell niche in different underlying disease processes may be induced by the mobilization and stimulation of HSCs and MSCs, by stromal proliferation, and/or by stimulation of cytoadhesive and chemokine signaling pathways that induce homing, activation of stem cells, or a combination of these factors (Fig. 1). The choreography of stromal, humoral, and stem cell interactions is a 3-dimensional interplay that relies on much more than physical space. As noted earlier, a residual fetal niche environment persists physiologically in hemic tissues in neonates; these sites are also readily activated and expanded in various disease states.
Although a complete understanding of the reasons underlying EMH in all disorders is not yet elucidated, 4 major theories encompass most of the pathophysiologic causes: (1) bone marrow failure; (2) bone marrow stimulation (myelostimulation); (3) tissue inflammation, injury, and repair; and (4) abnormal systemic or local chemokine production. Although EMH is usually trilineage, consistent with the multilineage potential of embryonic hematopoiesis and HSCs, 1 or 2 cell types may predominate, depending on the underlying pathogenesis. 65,113
Bone Marrow Failure (Downregulation or Ablation of Medullary Hematopoiesis)
Bone marrow disease or ablation causing damaged, disrupted, or inadequate medullary spaces and altered architecture, signaling, and microenvironment results in failure of normal hematopoiesis and the mobilization and/or activation of HSCs and HPCs to extramedullary sites, resulting in marked EMH in multiple organs. 78 Such bone marrow diseases include myelofibrosis, myelophthisis (replacement of hematopoietic bone marrow by abnormal tissue), myelodysplasia, drug or plant toxicities, radiation, immune-mediated disease, necrosis, infection, and metastatic neoplasia.
Primary myelofibrosis: prototypical EMH
Primary myelofibrosis in humans is a rare clonal myeloproliferative disease resulting in the mobilization and disordering of marrow space, and it is characterized by extensive trilineage EMH in tissues throughout the body, especially spleen and liver. 62 Primary myelofibrosis is the prototypical example of bone marrow failure associated with EMH. The disease arises from the transformation and clonal proliferation (hyperproliferation) of a single HSC in the marrow, with mobilization of stem cells to new or reactivated niches in the spleen and liver, shifting the balance and location of hematopoiesis and resulting in an imbalance between the osteoblastic and vascular niches. 58
The cellular abnormalities and disordered marrow that characterize primary myelofibrosis are accompanied by changes in the synthesis of numerous chemokines that stimulate collagen synthesis and decrease collagenolysis, including increased production of thrombopoietin, platelet-derived growth factor, collagenase inhibitors, transforming growth factor β (TGF-β), and possibly calmodulin. TGF-β increases biosynthesis of collagen types I, II, and III. The net result of these changes is the deposition of connective tissue (polyclonal fibrosis) within the bone marrow and subsequent sclerosis. 44 A direct relationship has been shown between serum thrombopoietin concentration and the amount of fibrosis, and there is a direct relationship between the cytokines required for fibrogenesis and the α-granules of megakaryocytes and platelets.
While primary myelofibrosis is characterized by aberrant proliferation of HSCs, it was recently observed that certain molecular alterations in microenvironment alone can result in myeloproliferative disease. In one study, deletion of retinoblastoma protein, a central cell-cycle regulator, or of retinoic acid receptor gamma resulted in myeloproliferative syndromes in genetically altered mice. 116,117 Disease occurrence was shown via transplantation experiments to not be intrinsic to the hematopoietic cells themselves, proving that microenvironment can be the sole foundation for hematopoietic disease. This fascinating observation opens the door to a range of novel initiating mechanisms for clonal hematopoietic disorders.
Documented reports of presumptive spontaneous primary myelofibrosis in animals are rare and the molecular basis remains unknown. 39,40 Two case series of dogs diagnosed antemortem with presumed primary myelofibrosis have been described. 40,114 In another case series of female Beagle dogs with myelofibrosis, EMH was found in liver and spleen at necropsy. 89 A case of idiopathic myelofibrosis was reported in a male marmoset in which trilineage EMH was found throughout the intestinal serosa. 51 Primary myelofibrosis (and myelosclerosis) also was reported as a primary congenital disease in Pygmy goats. 11 The disease manifested as severe progressive pancytopenia beginning soon after birth, with death occurring at 6-12 weeks and marked EMH in spleen, liver, gall bladder, lymph nodes, tonsil, thymus, heart, lung, and testicular tunics.
Other causes
Myelofibrosis secondary to other disorders also can result in EMH because of myelophthisis, with loss of marrow architecture and presumptive disruption of osteoblastic and vascular niches. In dogs, pyruvate kinase deficiency is the most frequently reported cause of secondary myelofibrosis, which occurs secondary to marked persistent stimulation of erythropoiesis (see section on myelostimulation) and/or as a response to iron overload. 121,129 Other causes of secondary myelofibrosis in animals include treatment with high doses of recombinant human erythropoietin, as well as feline leukemia virus infection. 120,121 Myelofibrosis and the potential to induce EMH also can occur concurrent or secondary to bone marrow necrosis (Figs. 2, 3), infectious diseases, and drugs or toxicities. 39,89,120 The exact mechanism linking necrosis and fibrosis has not been studied. Myelophthisis in animals is usually associated with hemic neoplasia (leukemia and lymphoma). 20
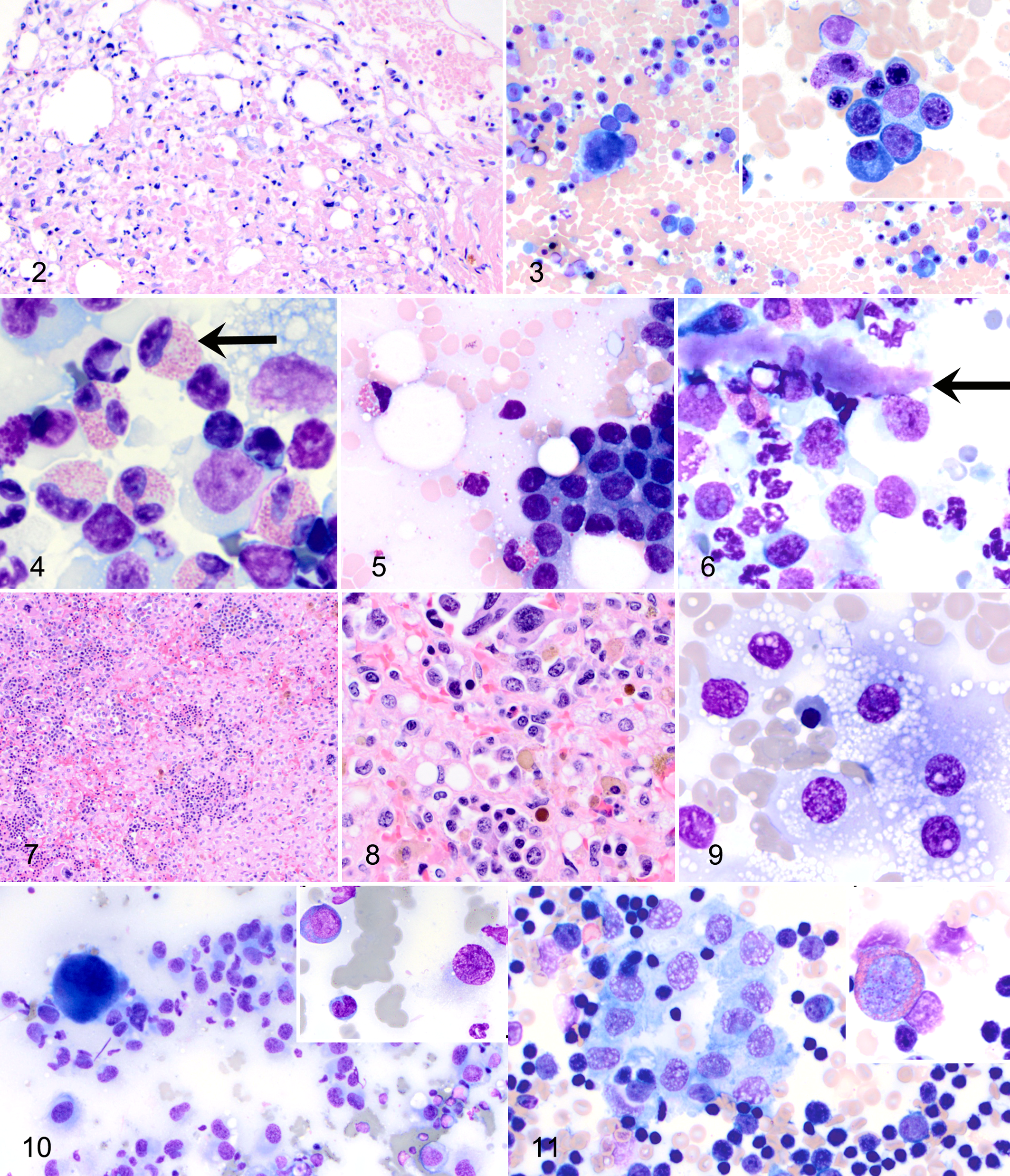
Bone marrow, pancytopenic dog. Severe necrosis with widespread hypocellularity; all hematopoietic cell lines are shifted toward immaturity. HE.
Myelostimulation (Upregulation of Medullary Hematopoiesis)
In the myelostimulatory theory of EMH, sometimes called compensatory reactivation or reactive EMH, upregulation of hematopoiesis in the bone marrow results in stem cell mobilization, increased synthesis of chemokines and growth factors, and subsequent upregulation of hematopoiesis in embryonic sites (mainly spleen and liver) and sometimes in other tissues. Myelostimulation is a natural homeostatic response to the need for increased blood cell production that occurs in hematologic and inflammatory disorders such that concurrent blood and bone marrow evaluation is important in making a diagnosis. Because adult bone marrow has a large capacity for expanded hematopoietic activity, EMH does not occur simply because available marrow space has been exceeded (although humans with hereditary spherocytosis develop severe paravertebral EMH and subsequent extrusion of vertebral marrow). Rather, a more complex situation exists in myelostimulatory EMH that involves the reactivation of stem cell niches in embryonic sites of hematopoiesis. Myelostimulatory EMH usually results from disorders associated with chronic or marked accelerated erythropoiesis, but other cell lines occasionally are affected. 62
Marked accelerated erythropoiesis
Hypoxia caused by severe hemorrhagic or hemolytic anemia is the primary stimulus for myelostimulatory EMH, whereby increased erythropoietin production in the kidney stimulates the proliferation and maturation of erythroid precursors. Circulating HSCs home to and seed embryonic sites of hematopoiesis, and erythropoietin and other chemical mediators stimulate proliferation of preexisting extramedullary HPCs in these organs. 31,91 Hemolytic anemia caused by pyruvate kinase deficiency, babesiosis, immune-mediated hemolytic anemia, zinc toxicosis, and other disorders that shorten erythrocyte lifespan are reported causes of myelostimulatory EMH in animals, especially dogs. 123
Splenic erythropoiesis secondary to hypoxia has been studied extensively in mice and is often referred to as “stress EMH.” Stress erythropoiesis involves the rapid, hypoxia-mediated expansion and differentiation of specialized stress erythroid progenitor cells in the spleen (especially) and liver. Stress erythropoiesis is mediated by bone morphogenetic protein 4, a member of the TGF-β superfamily, which regulates proliferation, differentiation, and apoptosis in many tissues. Stress erythropoiesis is characterized by increased homing of HSCs, proliferation of MSCs, and chemokine production in the spleen. Bone morphogenetic protein 4 works in concert with stem cell factor and hedgehog and is a potent regulator of hepcidin production, 112 which in turn regulates iron metabolism and heme synthesis. 83 Erythroblastic islands can be observed with marked erythroid EMH in the spleen, contributing further to the establishment and maintenance of a suitable niche for erythropoiesis.
Marked accelerated granulopoiesis
Stimulation of granulopoiesis in marked inflammatory disease occasionally can cause EMH, mainly myelopoiesis. Canine pyometra, for example, causes marked neutrophilia and granulopoiesis that can result in EMH, especially in the kidney; why specifically renal EMH is prominent is unknown. 64 In humans, use of therapeutic G-CSF and GM-CSF is a rare cause of EMH (mainly myelopoiesis) in the spleen and other embryonic sites of hematopoiesis. 78 Recently, G-CSF was found to mobilize HSCs and HPCs from bone marrow to adipose tissue, expanding the extramedullary pool of stem cells available for EMH. 34 Sustained exogenous treatment of dogs with G-CSF has been reported to cause EMH in dogs; however, this conclusion was based on finding increased numbers of neutrophils and bands in the spleen, which more likely reflected peripheral blood concentrations. 127
Marked accelerated thrombopoiesis
In humans, accelerated thrombopoiesis in the bone marrow as a response to immune-mediated thrombocytopenia and increased thrombopoietin has been reported to result in secondary EMH (primarily megakaryocytopoiesis) in the spleen and other organs. 95 Although accelerated thrombopoiesis is also a potential cause of EMH in animals, we are unaware of studies in which this has been documented. Thrombopoiesis appears to occur most often together with erythropoiesis in sites of EMH, consistent with stimulation of erythropoietin receptors on mixed erythroid/megakaryocyte progenitors. 10,68
Tissue Inflammation, Injury, and Repair (De Novo Sites of Hematopoiesis)
The cellular and stromal elements in granulation tissue, resolving hematomas, areas of ischemia and neovascularization, and sites of inflammation can result in the development of a niche environment capable of supporting EMH. This emerging concept posits that molecular changes associated with some types of local inflammation, injury, and tissue repair mimic those involved in hematopoiesis. Numerous cytokines and inflammatory mediators can increase the number of HSCs and MSCs in the peripheral circulation that subsequently can home to affected organs and tissues to participate in local regenerative or inflammatory responses. 124 This theory of EMH is supported by key studies of myocardial infarcts and allergic airway disease in humans but is also applicable to sites of inflammation and regeneration in other tissues and in animals.
Myocardial infarcts
In a study of human myocardial tissue, multiple foci of EMH were found in 15 of 207 biopsy specimens (7.2%) and 22 of 598 autopsy specimens (3.7%), and most (24 of 37, 65%) were associated with infarcts > 72 hours old, in hearts with abundant granulation tissue and large numbers of fibroblasts, macrophages, and lymphocytes. 38 In only 2 cases was an underlying hematopoietic disorder diagnosed. In another report, EMH was found adjacent to zones of myocardial infarction in 4 patients, also without bone marrow disorders. 29 Immunohistochemistry for glycophorin A, myeloperoxidase, and factor VIII–related antigen was performed on selected cases to document cells of erythroid, myeloid, and megakaryocytic origin, respectively. 29,38 These findings suggest that healing infarcts provide both the architectural components (loose connective tissue with neovascularization) and the cellular components of a microenvironment conducive to hematopoietic cell homing and proliferation.
Cells in healing infarcts are sources of hematopoietic growth factors, such as IL-3 (activated T cells), GM-CSF (macrophages, T cells, endothelial cells, fibroblasts), and IL-6 (macrophages). The margination of immature myeloid and erythroid cell clusters in intramyocardial capillaries and adjacent stroma support the hypothesis that EMH originates from circulating stem cells that localize to the tissue because of inflammation and repair-associated trophic factors. Hypoxia in ischemic tissue creates an optimal microenvironment for HSCs.
EMH occurs occasionally in myocardium and epicardium of animals, but we are unaware of studies to assess its prevalence or association with underlying disease. Perivascular and epicardial EMH were documented in a subset of marmosets in one study. 12 We also have observed hematopoietic precursor cells in pericardial fluid samples.
Atopic airway injury
In studies of allergen-mediated airway disease (asthma) in humans, active recruitment of stem cells from the bone marrow has been identified as a source of local hematopoiesis, primarily eosinophilopoiesis. 22 A circulating common progenitor of eosinophils and basophils (measured via in vitro colony-forming units) was detectable in greater numbers in the peripheral blood of asymptomatic atopic patients compared with controls. These progenitors are responsive to activated T cell–derived cytokines and chemokines, especially IL-5. During the inflammatory process, eosinophil and basophil progenitor cells, HSCs, and MSCs are recruited from marrow and home to inflamed respiratory mucosa where local chemokine production (eg, eotaxin, SDF-1, and IL-5) by activated T cells and other cells can occur. Recently, it was shown in mice with chronic allergen challenge that bone marrow homing of HPCs was decreased concurrent with decreased CXCR4 expression, allowing them to leave the marrow and be recruited to sites of inflammation in the airways, resulting in EMH. 84
Locally elaborated hematopoietic growth factors and Eo/B-CFU (eosinophil/basophil progenitor cells) also have been detected within nasal polyps. 22 In addition, activated CD34+ cells containing IL-5 and/or expressing IL-5 receptor messenger RNA have been detected in sputum and lung biopsies from atopic asthmatic patients. 1,22 Upregulation of IL-5 favors increased traffic of progenitor cells from marrow to peripheral circulation in response to eotaxin and SDF-1. Resultant blood, nasal, and/or pulmonary eosinophilia in models can be blocked by antibodies to IL-5. Overall, these studies indicate an active participation of bone marrow and related systemic hematopoietic events in the development and maintenance of allergic inflammation in local tissue, including early initiation and release of eosinophil and basophil progenitors and their progeny from the bone marrow and recruitment to the respiratory mucosa and other tissues in atopic individuals.
In animals with eosinophilic inflammation (eg, feline asthma) and other inflammatory conditions, bronchoalveolar lavage specimens often contain what appear to be eosinophil myelocytes (Fig. 4). We also found the so-called eosinophil myelocytes within a papillary carcinoma of the lung of a dog (Fig. 5). It is possible but as of yet unknown whether these eosinophil precursors represent local eosinophilopoiesis associated with local production of hematopoietic growth factors and stem cell homing, as described above.
Megakaryocytes or megakaryocyte nuclei are frequently observed in histopathologic sections of lung tissue in animals (estimated at 1 to 2.5/cm2 in dogs). 47,99 Rarely, proplatelets (Fig. 6) and evidence of megakaryocytopoeisis can be found in bronchoalveolar lavage samples. It is thought that circulating megakaryocytes and proplatelets enter the circulation and embolize in the lung; their contribution to local thrombopoiesis is unclear, and there is no correlation between peripheral platelet counts and the number of pulmonary megakaryocytes. 47,48 It would be interesting to assess whether the “embolization” or “filtration” of megakaryocytes in the lung is in fact VCAM-1-mediated adhesion facilitated by endothelial cells through SDF-1 and FGF-4 signaling in a vascular niche environment. Pulmonary megakaryocyte numbers can also increase secondary to increased marrow thrombopoiesis and/or altered pulmonary vascular filtration in various conditions, including shock. 105 In one study, the density of megakaryocytes in human lung sections was increased in conjunction with pulmonary pathology caused by smoking, decreased lung function, and cardiovascular disease such that ischemia, stromal proliferation, and inflammation could play a role in expanding the pulmonary niche environment for hematopoiesis. 99
Splenic inflammation, hematomas, and thrombosis
Retrospective studies of dogs and cats indicate that EMH occurs most often in the spleen in association with degenerative and inflammatory conditions, including lymphoid hyperplasia, hematomas, and thrombosis. 5,100,101 In a retrospective study of 1480 histologic specimens of spleens from dogs, EMH concurrent with nodular lymphoid hyperplasia and hematomas was found in 43% of specimens. EMH (defined as discrete foci) or myeloid metaplasia (defined as diffuse proliferation that disrupted follicular and sinusoidal organization) alone was diagnosed in ∼7% of specimens. 101 In cats, EMH was found in 4% of 455 histologic specimens; EMH concurrent with nodular lymphoid hyperplasia and hematomas was much less common (5% of cases) because of the lower incidence of those disorders in this species. 100
Trilineage EMH (mainly megakaryocytic and erythroid) is often observed in cytologic specimens from animals with plasmacytosis, stromal proliferation, and histiocytic hyperplasia associated with erythrophagocytosis, increased iron, or inflammation. 16 In one study, EMH was observed more often in cytologic than histologic sections from dogs and cats and was concurrent with fungal splenitis, reactive lymphoid hyperplasia, histiocytic hyperplasia, and neoplasia. 5 Splenic EMH often is observed in animals without obvious hematologic abnormalities, suggesting that local tissue injury, inflammation, and repair are a major underlying pathogenesis of EMH in this organ.
Other tissue sites
Trilineage EMH was found in the anterior and posterior uvea of enucleated eyes from 3 of 6 cats with ocular disease. 43 The cats had a history of corneal degeneration during the neonatal period, leading to corneal destruction and extensive prolapse of uveal tissue, suggesting that EMH was secondary to tissue trauma and repair; mild lymphoplasmacytic inflammation also was noted. Histologically, EMH was found in the iris and, to a lesser extent, the adjacent ciliary body and choroid, with mature granulocytes, mainly eosinophils, seen within clusters of precursor cells. Metaplastic bone tissue was not observed.
Resolving hematomas or hemorrhage in various tissues appears to initiate formation of a microenvironment conducive for hematopoiesis. For example, EMH in the spinal and cranial dura matter has been reported in conjunction with subdural hematomas, 25,56 and we have observed adrenal EMH in conjunction with hemorrhage (Figs. 7 –9). The relatively hypoxic environment of a resolving hematoma would be conducive to the homing of HSCs and HPCs. The stimulatory and support role of activated macrophages in the development of EMH in tissues with hemorrhage, other forms of tissue injury and repair, and histiocytic hyperplasia and in histiocytic sarcoma (see below) warrants further study. In addition, trilineage EMH in the mandibular lymph nodes of monkeys infected with chimeric simian human immunodeficiency virus was proposed by the authors to be the result of local macrophage activation and IL-6 synthesis. 103
Abnormal Systemic or Local Chemokine Production
A rare cause of EMH is the generation of abnormal cytokines or other hematopoietic growth factors that induce stem cell populations to differentiate into hematopoietic cells and/or simulate the marrow microenvironment. 53,78 Sometimes (confusingly) referred to as the “redirectional differentiation” theory, abnormal cytokines may function systemically or locally. These selective occurrences of excessive chemokine production should be differentiated from increases in chemokine synthesis that occur with EMH secondary to bone marrow failure, myelostimulation, and tissue inflammation, injury, or repair.
The constitutive overexpression of IL-5 in transgenic mice causes colonization of the spleen by mesenchymal/stromal progenitor cells, which provide the necessary microenvironment for establishment of hematopoiesis here and in other extramedullary sites. HSCs and HPCs migrate to the new microenvironment to proliferate and differentiate toward the eosinophil lineage. 49 This mouse model has been useful for studying the effects of a single cytokine. IL-5 is a primary regulator of eosinophil differentiation, but its effects on hematopoiesis clearly are broader as overexpression also results in increased numbers of committed progenitors for macrophage, B-lymphocyte, and granulocyte-macrophage lineages in spleen and peripheral blood.
Cutaneous EMH has been reported at the site of IL-3 injections in monkeys. 50 Small, firm nodules developed at injection sites and histologically were found to contain trilineage hematopoietic cells; eosinophilic cells were overrepresented within the myeloid population as a whole.
EMH Within Neoplasms
EMH can be found within neoplasms involving both hemic and nonhemic tissues. In humans, EMH has been reported in association with cerebellar hemangioblastoma, astrocytoma, hemangioma, hepatoblastoma, leiomyoma, pilomatricoma, hepatic angiosarcoma, endometrial carcinoma, meningioma, hepatic adenoma, spindle cell lipoma, liposarcoma, myofibroblastic tumors, and renal tumors. 78 EMH has been reported in up to 42% of hemangiosarcomas in dogs, 6 in a benign mixed mammary tumor in a dog, 30 and in histiocytic sarcomas in mice and dogs. 57,71 We have observed EMH in cytologic specimens from histiocytic sarcomas in dogs (Fig. 10), as well as in a thymoma in a rabbit (Fig. 11). The paucity of reports on EMH in neoplasms in animals could reflect the perceived unimportance of the finding rather than its rarity.
The molecular mechanisms of intratumoral EMH are diverse and can differ within and between neoplasms. EMH in hemangiosarcomas could be related to the common stem cell origin of endothelial and blood cells and to the proliferation of a conducive vascular niche environment, including the production of chemokines by neoplastic endothelial cells. 6 In hepatoblastomas, production of numerous hematopoietic growth factors by tumor cells is postulated to result in EMH. The causes of EMH in other neoplasms are not well understood but likely include local hypoxia (ischemia), local tissue injury or repair, and production of growth factors—for example, by endothelial cells in areas of neovascularization. Local hypoxia in neoplasms could create an environment conducive to the homing of HSCs.
Tumor-derived circulating VEGF recently was shown in a mouse model to suppress bone marrow hematopoiesis and induce EMH in the spleen and liver together with altered vessel architecture and increased expression of VEGF receptors on endothelial cells. 126 VEGF is a well-known angiogenic factor that is induced by hypoxia; its complete absence causes embryonic lethality because of a failure to establish angiogenesis as well as hematopoiesis. In addition, as noted earlier, SDF-1 binds to the G-protein–coupled receptor CXCR4 on the surface of some tumor cells (reviewed in Furusato et al 27 ), potentially promoting trafficking and metastasis of CXCR4+ cancer stem cells to organs expressing high levels of SDF-1, such as lymph nodes, lung, and bone. HSCs and HPCs expressing surface CXCR4 may traffic to the SDF-1-producing site in concert with tumor cells, leading to EMH within tumors.
Histiocytic sarcoma has been associated with EMH. 57 In mice, 89% of histiocytic sarcomas had hepatic (portal) EMH, and EMH was often found within a tumor nodule; bone marrows were normocellular and not obviously affected by the neoplasm. Fifty percent of the tumors had marked erythrophagocytosis; however, immunochemical staining was not done to further characterize them, and variants of histiocytic sarcoma have not been fully defined in mice. In dogs, hemophagocytic histiocytic sarcoma of CD11c+ CD18+ macrophages can cause EMH in the affected spleen, with diffuse infiltration of red pulp cords and sinuses by neoplastic histiocytes and erythroid precursors. 71 Erythrophagocytic histiocytic sarcomas originate from macrophages in the splenic red pulp or bone marrow, with early intravascular metastasis to liver and lung. 71 Dogs with hemophagocytic histiocytic sarcoma usually have profound anemia such that hypoxia may be the primary stimulus for the EMH. We have observed EMH in cytologic specimens from both erythrophagocytic and nonerythrophagocytic histiocytic sarcomas involving liver and spleen. It is possible that in dendritic cell (CD11d+) histiocytic sarcoma, other mechanisms could elicit EMH, such as activation of resident macrophages or T cells and/or tumor cell production of chemokines.
Myelolipomas are rare benign tumors of animals and humans composed of approximately 50% well-differentiated adipose tissue and 50% hematopoietic tissue. 113 In humans, myelolipomas usually are found in the adrenal gland, retroperitoneally, or in the mediastinum. In animals, myelolipomas occur as a focal mass in spleen or liver. 113 The hematopoietic component of myelolipomas is trilineage but differs significantly from that of bone marrow in cellular composition, cellularity, and stromal elements. 8 Myelolipomas in animals usually are incidental findings, and there are no reports of malignant transformation.
Nucleated RBCs in Peripheral Blood: Potential Misinterpretation as EMH
EMH is reported as a cause of circulating NRBCs and immature neutrophils (leukoerythroblastosis) in peripheral blood as a result of “passive incontinence” of hematopoietic cells from sites of EMH in the spleen and other organs; however, we were unable to find evidence to support this contention. 62,121 Ultrastructural and histologic evidence for intrasinusoidal and intravascular EMH in the spleen and the presence of HPCs within vessels at sites of tissue injury (ie, an extramedullary vascular niche) support the localization of EMH within vessels. 111 However, to our knowledge, it has not been demonstrated that release of precursors from these sites into the circulation occurs more readily or prematurely compared with the vascular niche within the bone marrow.
A potential association between NRBCs and EMH appears to have originated in a 1966 review by Clifford, 18 in which he posits that it “seems probable” that circulating immature cells are derived from sites of EMH where tissue organization and control of cell egress may be less effective than in marrow. However, most of the cases described in that review were associated with severe bone marrow disease (eg, myeloproliferative disease, metastatic carcinoma) or hypoxia, which can result directly in inappropriate release of NRBCs from marrow. Similarly, in a recent study of 100 children with NRBCs, greater than 80% had an underlying explanation for NRBCs (40% had hypoxia), and the remaining children were candidates for bone marrow transplantation because of major hematologic disorders. Two studies in animals, 1 in cats 33 and 1 in dogs, 63 are often cited to support an association between leukoerythroblastosis and EMH. However, none of the 313 cats or 280 dogs evaluated in those studies had documented EMH. Thus, while it is possible that altered systemic factors that affect egress of cells from the marrow in hypoxia and hematologic diseases could similarly affect egress of cells at intravascular or other sites of EMH, at present, evidence that EMH alone results in leukoerythroblastosis is lacking.
Conclusions
In conclusion, EMH develops with local or systemic factors that induce or activate a stem cell niche outside the bone marrow, generally in conjunction with endothelial cells and often involving resident macrophages. Niches may be induced by the mobilization and stimulation of HSCs and HPCs, by the mobilization and stimulation of MSCs and stromal proliferation, and/or by stimulation of cytoadhesive and signaling pathways. As theories and knowledge regarding stem cell niches and microenvironments evolve, our understanding of the pathophysiology of EMH will evolve accordingly. More research is needed to better understand the prevalence of EMH in animals and the mechanisms of EMH in different species, tissues, and disease conditions. Insights gained about EMH could in turn inform our understanding of HSCs and their microenvironment.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
