Abstract
Marek’s disease (MD) is a disease of chickens that occurs worldwide and has serious economic consequences. MD can present as one of several forms, with the most commonly occurring forms being the lymphoproliferative diseases. Under experimental conditions, an early mortality syndrome has been recognized following infection by some but not all strains of MD virus (MDV). This is the first report of a confirmed case of mortality due to naturally occurring MDV infection in 1-week-old, nonvaccinated, chickens. Necrotizing lesions were observed in the bursa of Fabricius, lung, duodenum, jejunum, and proventriculus, and large intranuclear inclusion bodies were a striking feature in tissues with lesions in all birds. Immunohistochemical staining for the pp38 protein of MDV revealed abundant pp38 antigen in the affected tissues, confirming the presence of MDV within the lesions. PCR yielded an amplicon with 97% homology to the meq gene of MDV. No evidence of co-infection by either of the immunosuppressive agents chicken anemia virus and infectious bursal disease virus was detected.
Marek’s disease (MD) is a disease of chickens that occurs worldwide and has serious economic consequences. It is caused by Marek’s disease virus (MDV), a herpesvirus of the genus Mardivirus, within the subfamily Alphaherpesvirinae. Serotype 1 MDVs were recently renamed as Gallid herpesvirus 2 (GaHV-2). 18 MD can present as one of several forms, with the most commonly occurring forms being the lymphoproliferative diseases. 10 These are characterized by the development of mononuclear inflammatory nerve lesions and/or visceral lymphomas, or sometimes by mononuclear inflammatory cell or lymphomatous infiltration of the skin or eye. 18 Under experimental conditions, an early mortality syndrome has been recognized following infection by some, but not all, strains of MDV.17,20,23,24 This early mortality syndrome was characterized clinically by depression and paralysis, and deaths occurred as early as 5 to 10 days after infection. Histologically, there was marked atrophy of thymus and bursa of Fabricius, with necrosis of lymphoid cells and occasional herpetic inclusions. There were no lymphomas or lymphoproliferative lesions in the affected birds. Illness and death were attributed to the cytolytic phase of MD, which occurs during the productive viral replication that starts about 3 to 6 days post infection. 24 To date, there have been no confirmed reports of naturally occurring early mortality associated with MDV infection. Herein, we describe a case of mortality in 1-week-old chickens that had extensive necrotizing lesions and intranuclear inclusions associated with naturally occurring MDV infection.
Three dead 1-week-old Rhode Island Red chickens were submitted to the Connecticut Veterinary Medical Diagnostic Laboratory (CVMDL) (Storrs, Connecticut) with a history of drooping wings and depression. The chickens were from a small backyard flock that had had an ongoing low-level mortality problem among chickens up to 2 months of age for the previous 2 to 3 months. The submitted chickens had been hatched on site and had not been vaccinated against MD.
At necropsy, all 3 chickens had a mottled spleen, small thymic lobes, dilated proventriculus with multiple petechial hemorrhages in the mucosa, and a pale heart. One chicken also had petechial hemorrhages in the mucosa of the esophagus. This chicken, and 1 other, had small amounts of digested blood in the colon.
Samples of esophagus, crop, proventriculus, ventriculus, duodenum, jejunum, colon, pancreas, liver, kidney, bursa of Fabricius, spleen, trachea, lung, heart, skeletal muscle, bone, bone marrow, skin, and brain were fixed in 10% neutral buffered formalin and routinely processed to paraffin. Five-micrometer sections were stained with hematoxylin and eosin (HE) for histologic evaluation.
Paraffin-embedded sections of bursa of Fabricius, lung, intestine, proventriculus, and ventriculus from 1 bird were submitted to Michigan State University’s Diagnostic Center for Population and Animal Health for immunohistochemical staining for MDV. This was performed on the Bench Mark Automated Staining System (Ventana Medical Systems, Tucson, AZ) using the Ultra View-Red Detection (alkaline phophatase red) System (Ventana Medical Systems). Antigen retrieval was achieved using the Ventana Medical Systems Retrieval Solution CC1 for 60 minutes. An MDV-specific mouse monoclonal antibody H19 (pp38) (ADOL, East Lansing, MI) was applied at a concentration of 1:1,000 for 10 minutes. Sections were counterstained with hematoxylin. Paraffin sections of known MDV positive tissues were included as a positive control. For negative control purposes, the primary antibody was replaced with buffer. Separate paraffin-embedded sections of bursa of Fabricius, proventriculus, gizzard, small intestine, colon, lung, and heart from all 3 birds were immunostained separately using mouse monoclonal antibody with reactivity to multiple infectious bursal disease virus (IBDV) strains 19 and rabbit polyclonal antibody to Newcastle disease virus (NDV) (Veterinary Science Division, AFBINI, Belfast, Northern Ireland), respectively. Antigen retrieval was achieved by incubation in 0.1% protease 14 (Sigma-Aldrich) in Tris-buffered saline, for 5 minutes at room temperature, and immunolabeling was completed using the Dako REAL Envision Detection System Peroxidase/DAB+, Rabbit/Mouse detection system (Dako, Glostrup, Denmark) according to the manufacturer’s instructions except that sections were incubated with the polymer for 30 minutes. Paraffin sections of known NDV- and IBDV-positive tissues were included in the study for positive control purposes.
PCR was performed in CVMDL for detection of MDV DNA. 1 Briefly, total DNA was extracted from fresh proventriculus and lung tissue from 1 bird, using a DNeasy Tissue kit (Qiagen, Valencia, CA). DNA extraction quality was assessed by real-time polymerase chain reaction (PCR) on the Roche LightCycler 2.0 system using the Qiagen SYBR Green Master Mix kit (Applied Biosystems, Foster City, CA) with primers targeting 18S ribosomal DNA (18e 5′-CTGGTTGATCCTGCCAGT-3′ and 18i 5′-TTTCTCAGGCTCCCTCTCCGGAATCGAACCCT-3′). 14 Cycling conditions included a 15-minute enzyme activation followed by 45 cycles of 94°C for 15 seconds, 60°C for 30 seconds, and 72°C for 20 seconds. Both samples passed this quality assessment and were further analyzed. MDV DNA was amplified by using a primer set that targets the meq gene of MDV—MDV1-meq_F 5′-GTCCCCCCTCGATCTTTCTC-3′ and MDV1-meq_R 5′-CGTCTGCTTCCTGCGTCTTC-3′ 6 —and a HotStarTaq Master Mix (Qiagen) kit. Cycling conditions included a enzyme activation step at 95°C for 15 minutes, followed by 35 cycles of 94°C for 30 seconds, 52°C for 30 seconds, 72°C for 30 seconds, and a final elongation step at 72°C for 5 minutes. Amplified PCR products (189 bp) were resolved in 2% agarose gels and amplicons were purified using a QIAquick Gel Extraction Kit (Qiagen). Purified products were sequenced using specific forward and reverse primers described above (MWG, Huntsville, AL). Sequences were assembled with Sequencher software (Gene Codes Corp, Ann Arbor, MI). Sequence identities were determined using the National Center for Biotechnology Information (NCBI) software Basic Local Alignment Search Tool (BLAST) (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi).
After confirming the presence of RNA (concentration 303 ng/μl and 113 ng/μl, respectively) in the above extracted samples using a NanoDrop 1000 (Thermo Scientific, Wilmington, DE), reverse transcription (RT) real-time PCR was performed on the extracted samples, according to the protocol from the National Veterinary Services Laboratory (Document No. SOP-AV-1505.05. Real-Time RT-PCR for Detection of Virulent Newcastle Disease Virus in Clinical Samples. 021810 RNA), which is as described by Wise et al. 22
PCR for detection of chicken anemia virus (CAV) was performed on each of the 3 birds, using DNA extracted from the paraffin-embedded tissues using a DNeasy Tissue kit (Qiagen, Valencia, CA) according to the manufacturer’s instructions. The quality of the DNA recovered from the tissue blocks was confirmed by real-time PCR on the Roche LightCycler 2.0 system using the Qiagen SYBR Green Master Mix kit (Applied Biosystems, Foster City, CA) with primers targeting 18S ribosomal DNA as described above. The PCR reactions for detection of CAV was carried out by the Texas Veterinary Medical Diagnostic Laboratory System, College Station, Texas.
Histologic lesions were present in bursa of Fabricius, spleen, proventriculus, duodenum, colon, and lung. In the bursa of Fabricius of all chickens, the medulla of most lymphoid follicles was undergoing acute necrosis, as characterized pyknosis and karyorrhexis of virtually all lymphocytes (Fig. 1a). Segments of follicular cortices were similarly affected. Occasional clusters of follicles were heavily infiltrated by heterophils and macrophages and had few remaining lymphocytes. In these follicles, some cells had large, round to very elongated nuclei with marginated chromatin and a large amphophilic, 5- to 12-μm-diameter inclusion body, which usually filled the nucleus (Fig. 1a, inset). Most of these cells were thought to be reticular epithelial cells and macrophages, but intranuclear inclusions were also recognized in occasional lymphocytes. The interfollicular connective tissue was infiltrated and expanded by numerous macrophages and heterophils, some of which were necrotic. In these areas, scattered cells with enlarged nuclei, marginated chromatin, and a single nuclear inclusion as described above were observed.
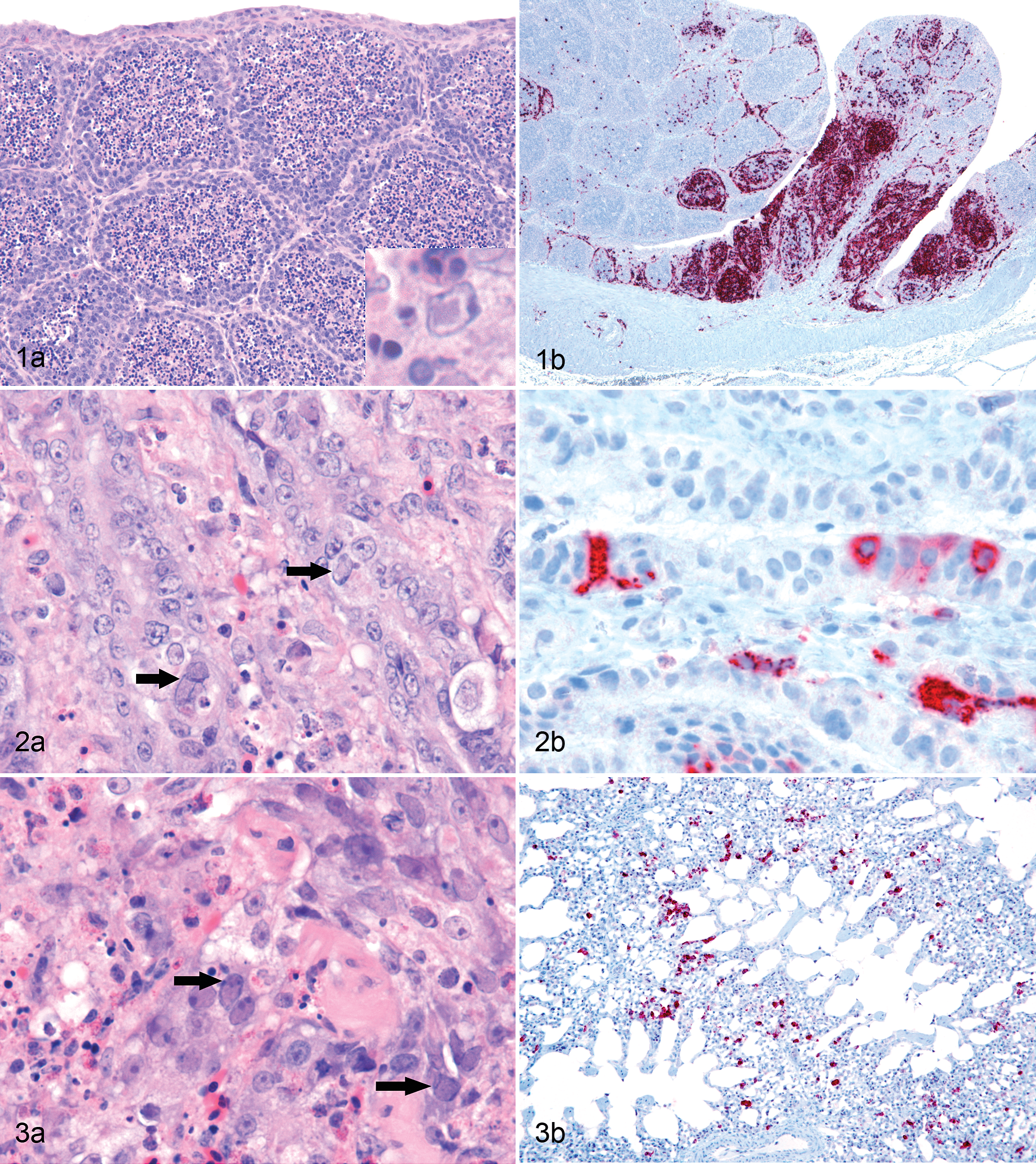
The white pulp of the spleen was moderately expanded. There were few lymphocytes, many very prominent pale staining reticular cells, and moderate to marked heterophilic and histiocytic infiltrate. Large intranuclear inclusion bodies similar to those described in the bursa of Fabricius were found in small to moderate numbers of cells in the white pulp.
In the superficial mucosa of the proventriculus of all chickens, there were multiple foci of coagulative necrosis, congested capillaries, and small hemorrhages. Deep in the mucosa, between the base of the mucosal glands (sulci), there were moderately dense infiltrates of macrophages and heterophils admixed with small numbers of lymphocytes. Occasional hemorrhages were also present. In numerous epithelial cells of the mucosal glands and also among inflammatory cells, there were markedly enlarged nuclei with marginated chromatin, which contained a large amphophilic inclusion body (Fig. 2a) as described in the bursa of Fabricius. The submucosal glands were unaffected.
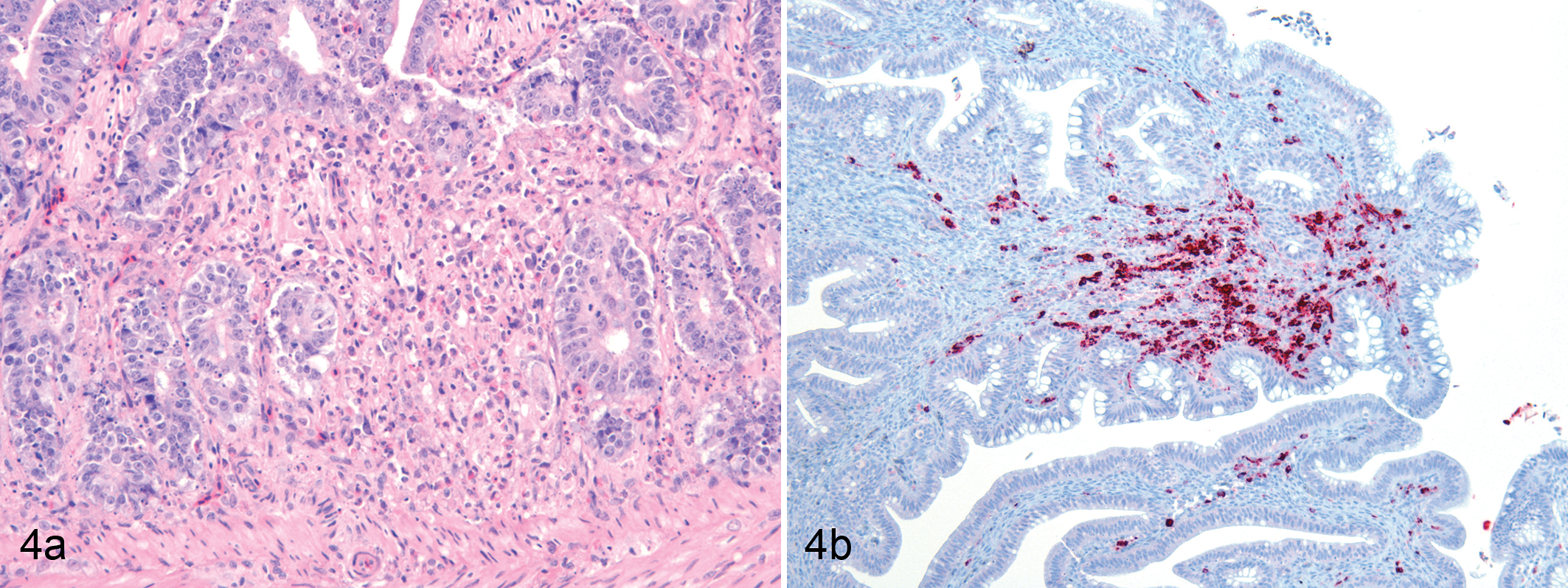
(a) Duodenum; chicken No. 1. The lamina propria is infiltrated by macrophages and heterophils, and there is pyknotic and karyorrhectic cellular debris. Necrosis and loss of crypts are evident in the central area of this field. HE. (b) Colon; chicken No 1. Cells in the lamina propria are immunopositive (red label) for pp38 of MDV. Mayer’s hematoxylin counterstain.
Multifocally, in the lungs of all 3 chickens, there was similar inflammatory cell infiltration of the interstitium with effacement of parabronchi and surrounding air capillaries and obliteration of airspace. Numerous large nuclei with marginated chromatin and a single large intranuclear inclusion as described above were present in these areas (Fig. 3a). Some of the affected cells were respiratory epithelial cells, whereas others were inflammatory cells, most likely macrophages. Additionally, there were occasional clusters of 2 to 5 cells with abundant cytoplasm, undergoing coagulative necrosis.
There was focally extensive heterophilic and histiocytic infiltration of the lamina propria of the jejunum of 2 birds and of the duodenum of 1 bird. In these regions, there was loss of crypts (Fig. 4a), and large intranuclear inclusion bodies were present within large oval-shaped nuclei in the lamina propria. Bone marrow was normal.
Immunolabeling with antibody to the pp38 protein of MDV revealed the presence of pp38 antigen in multiple tissues. In the bursa of Fabricius, pp38 antigen was mainly located in the medulla of the lymphoid follicles and in the interfollicular tissue (Fig. 1b). For the most part, the cortex of the follicles contained scattered positive cells, but there were some foci in which numerous cortical cells were positively immunostained. In the proventriculus, pp38 antigen was observed throughout the mucosa but was especially abundant in the deeper regions the mucosa. It was present in both epithelial cells and in cells within the lamina propria and was present in nuclei and/or cytoplasm of positive cells (Fig. 2b). In the lung, there were moderately abundant single cells or clusters of cells with strong cytoplasmic and/or nuclear immunolabeling for pp38 (Fig. 3b). Again, these cells were found both in epithelium and in the interstitium. In the intestine (colon), single cells or small clusters of cells in the lamina propria were immunolabeled (Fig. 4b). Spleen, duodenum, and jejunum were not tested by immunocytochemistry for MDV. No immunolabeling of IBDV or NDV was present in the tissues of any of the 3 birds.
On PCR for MDV, amplicons of the expected size (189bp) were obtained from the lung and proventriculus of the examined bird. The nucleotide sequence had 97% identity to the meq gene of GaHV-2 (serotype 1 MDV). RT real-time PCR for NDV proved negative whereas the positive control sample yielded a positive result. PCR for CAV proved negative, whereas the corresponding positive control yielded an amplicon of the expected size.
The pathogenesis of MD can be divided into 4 well-characterized phases: (1) acute cytolytic infection; (2) latency phase; (3) secondary cytolytic phase, and (4) lymphoproliferative disease.10,18 Disease usually becomes manifest during the fourth phase. However, after experimental infection of chickens with some strains of MDV, early deaths occurred that coincided with the acute cytolytic stage of infection and occurred between 5 and 21 days post inoculation.23,24 This process was named early mortality syndrome (EMS). The acute cytolytic phase occurs 3 to 4 days post infection, when there is cytolysis of B-lymphocytes and activated T-lymphocytes.7–9 By days 7 to 13 post infection, atrophy of bursa of Fabricius and thymus is present. 24 During the cytolytic phase, there is abundant production of nonenveloped virus particles.5,20
In these 1-week-old chickens from a backyard flock, severe necrotizing lesions with herpesvirus-like intranuclear inclusions in macrophages, reticular cells, epithelial cells, and less commonly, lymphocytes were present in multiple tissues of all 3 birds. The intranuclear inclusions were a striking feature in all of the birds, and based on their herpesviral-like appearance, the early cytolytic phase of MDV infection was suspected. We were able to confirm the presence of large amounts of pp38 MDV antigen in the lesions in our case. Pp38 is an early protein of MDV and is highly specific to MDV.4,11 It is necessary for MDV cytolytic infection of B cells and maintenance of the transformed status of lymphocytes by preventing apoptosis. 11 It is expressed in the cytoplasm of lytically infected cells and is a good indicator of MDV cytolytic infection, 16 but has also be seen in nuclei of infected cells. 11 Both cytoplasmic and nuclear pp38 antigen was found in our birds. Previous studies describe and illustrate immunopositivity in the bursa of Fabricius of chickens experimentally infected with MDV.4,18 These findings are very similar to those demonstrated in our birds, although additionally in our case, pp38 immunolabeling was detected in the nucleus and/or cytoplasm of both epithelial cells and cells of the interstitium of the lung and proventriculus, in the intestinal lamina propria, and to a lesser extent in intestinal epithelial cells. It is reported that intranuclear inclusions are sometimes found in cells associated with lymphodegenerative lesions in experimentally induced MD.6,18,20 In EMS of experimentally induced MD, intranuclear inclusions were found in the bursa of Fabricius and thymus and in the kidney associated with necrotizing lesions,6,20 although they were not reported in the necrotizing lesions that were present in the pancreas, proventriculus, liver, spleen, and heart. In contrast, in our case, inclusion bodies were numerous in all affected tissues. The presence of MDV was confirmed in our case not only by the detection of abundant pp38 antigen of MDV within the lesions but also by PCR amplification of product with 97% homology to the meq gene of MDV. During co-infection by MDV and either of the immunosuppressive agents CAV and IBDV, the disease process induced by each infecting virus was exacerbated.15,21 However, no evidence of co-infection by CAV or IBDV was found in any of the 3 birds of the present study. These findings, together with the severity of the MDV-induced histologic lesions, suggest that our case was a naturally occurring case of early mortality due to MDV infection.
To date there has only been 1 report of a naturally occurring, spontaneous necrotizing, bursitis and thymusitis with intranuclear herpesviral inclusions in young chickens. 13 The nature of the herpesvirus in that case was not determined. The case was later attributed to MDV infection, 10 and although this seems highly probable given the similarity to the lesions in the present case, to our knowledge it was not definitively confirmed as MD.
In the present case, necrotizing lesions with inclusion body formation were found in lymphoid organs as well as in the proventriculus, intestine, and lung. In contrast to the present case, no lesions were present in the lung and intestine in the previously described herpesviral infection, 13 and necrotizing lesions with inclusion body formation were not present in the proventriculus.
The lung is considered to be the portal of entry of MDV.2,3,12 In an inhalation study with MDV particles, half of the birds had MDV antigens in the spleen as early as 3 days post exposure (post infection), and proventriculus and skin were positive as early as 5 to 7 days post infection. 3 The similarity of this antigen distribution to the findings in the present case, together with the very young age of our birds, suggests that our birds were infected very close to the time of hatching. This concurs with the recently reported contention that in the field, challenge of chickens occurs as soon as they hatch, 12 and it emphasizes the need for control programs to minimize the possibility of early exposure of nonprotected chickens.
Under experimental conditions, EMS only occurred with some strains of MDV, and could occur, even in the presence of maternally derived antibody, albeit at reduced incidence. 24 In the present case, it seems likely that the birds were infected by such strains, perhaps with a high infectious dose, and that they may well have had little or no maternally derived antibody to MDV.
In conclusion, the histopathologic, PCR, and immunohistochemical results presented here were all consistent with death due to MDV infection. To our knowledge, this is the first confirmed case of naturally occurring early mortality due to MD in chickens.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
