Abstract
A six year old Labrador mix dog, born in Spain, was imported to Germany in young age. After a period of vague abdominal pain and negative laboratory results, the dog was referred to a local veterinary clinic for laparotomy, where the tentative diagnosis of echinococcus alveolaris was made and the dog was euthanized. At necropsy, many cystic structures and a massive proliferative peritonitis were visible. Furthermore a few solid larval cestodal stages were found in the peritoneal and chest cavity. Histological the cysts contained a small eosinophillic tegument, a cell poor stroma, basophilic somatic nuclei and a variable amount of amorphous bodies. The solid cestodes exhibited a quite similar composition with neither scolices nor any head structures. Histological features of the parasitic stages resembled cysts and asexual stages of Sparganum proliferum. Distinctions to other larval cestode infestations in body cavities were discussed.
A 6-year-old neutered female crossbred Labrador, born in Spain and brought to Germany at a young age (< 1 year), had nonspecific abdominal pain for approximately 6 months. Because clinical examination and laboratory results by a local veterinarian were not conclusive, the dog was referred to a private veterinary clinic for laparotomy. Severe proliferative peritonitis was observed with rough, thickened, and irregular yellow-red peritoneum and serosal surfaces of the abdominal organs, associated with diffuse abdominal and pleural thin-walled, fluid-filled cysts. Echinococcosis was assumed as a preliminary diagnosis. The dog was euthanized because of the poor prognosis and brought to the Regional Laboratory for Public Health and Veterinary Science, Saxony, Germany, for necropsy. The dog’s body was in good condition (body weight, 41.5 kg). The main findings were restricted to the abdominal cavity, which contained about 2.5 liters of serosanguineous fluid.
The peritoneum and serosal surfaces of most abdominal organs showed massive, diffuse, compact-to-spongy, yellow-to-red proliferation with tough fibrous adhesions among intestine, mesentery, and omentum (Fig. 1). Despite the serosal alterations, the cut surfaces of the liver, spleen, and kidneys were normal. Many cysts were free floating in the abdominal fluid or attached to the altered serosal surfaces (Fig. 1). The cysts measured 2 to 15 mm in diameter and contained clear, watery fluid and no other visible structures. Also in the abdominal cavity, mainly adherent to the altered serosal surfaces, were slightly segmented wormlike organisms that measured 1 to 8 cm long and 3 to 10 mm wide (Fig. 2).
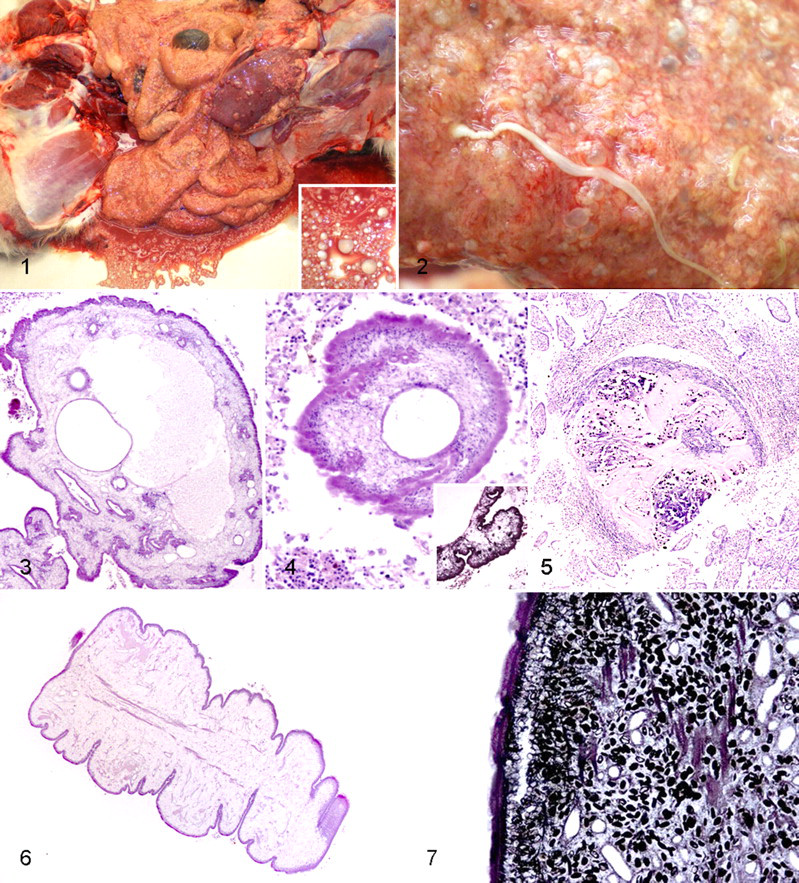
Peritoneal cavity, dog. Severe proliferative peritonitis with many cystic parasitic stages and serohemorrhagic fluid. Inset: Closer view of the cystic stages.
The chest cavity contained 1.5 to 2.0 liters of serous fluid, but serosal or pericardial proliferation or inflammation was not visible. Pleura, lungs, and heart were normal. No cysts were present, but a few wormlike organisms like those in the abdominal cavity were free in the pleural cavity or slightly adherent to the lungs. The remaining organs of the dog appeared normal.
Differential Diagnoses
Differential diagnoses included parasitic infestation with intermediate stages of the pseudophyllidean cestodes Spirometra spp (plerocercoidosis) and Sparganum proliferum (sparganosis), cyclophyllidian cestodes of the genus Mesocestoides (tetrahyridiosis), and Pentastomidae (pentastomiasis).
Microscopic Findings
The mesentery, omentum, and serosal surfaces of the abdominal organs showed massive mesothelial hyperplasia and proliferating chronic granulation tissue consisting of fibroblasts, fibrocytes, and capillaries, often forming fingerlike projections. Diffuse inflammatory infiltrations were dominated by numerous foamy macrophages, neutrophils, and siderocytes and, to a lesser extent, lymphocytes, plasma cells, and eosinophils. Giant cells were not visible. In the spleen, a mild follicular hyperplasia and an increased infiltration with macrophages were detectable. The parenchyma of the other abdominal organs appeared normal. In contrast, in the chest cavity, the pleura and pericardium showed only few foamy macrophages and neutrophils but no signs of tissue proliferation. Intact cysts (Figs. 3, 4) were covered by an amorphous, eosinophilic tegument 10 to 12 μm thick. A few shallow invaginations of the tegument were detectable, which partly formed tortuous parenchymal cavities, often filled with intensely eosinophilic granular substance. The cysts also had a variable portion of a loose, fibrillar stroma. Beneath the tegument, basophilic nuclei were located in a parallel row, sometimes in a multilayer fashion. Only a few nuclei were distributed within the cysts stroma (Fig. 4). The cystic appearance resulted from variable numbers of optically empty cavities, diffusely distributed and of varying shape and size. Most often, there was one large cavity (up to 1 mm in diameter) and several smaller ones (30–300 μm in diameter). These cavities were lined by an anucleate, eosinophilic substance of variable thickness, partly resembling the tegument (Fig. 3). Furthermore, low numbers of uniform, amphophilic, oval, calcareous bodies (dark brown to black with von Kossa’s stain) were randomly distributed in the stroma (Fig. 4, inset). Other features such as scolices and reproductive organs were not present. Some cysts were in different stages of disintegration and degradation (Fig. 5). These were characterized by the absence of tegumental structures and basophilic nuclei. Instead of stromal components, amorphous eosinophilic material was visible with variable degrees of calcification. These forms could often be found deeply embedded in the inflamed tissue (Fig. 5). The elongated larvae (Figs. 6, 7) were solid; lacked discernible scolices, suckers, or hooks; and had an amorphous eosinophilic tegument 10 to 12 μm thick that had periodic invaginations, resulting in pseudosegmentation. As in the cysts, a row of basophilic nuclei was visible beneath the tegument, and the body mainly consisted of loose stroma with scattered basophilic nuclei and several small cystic cavities (50–75 μm in diameter) lined with eosinophilic material. In contrast to the intact cysts, moderate amounts of muscle fiber bundles were present in the central part of the body and oriented parallel to the surface (Fig. 6). There were also numerous uniform, amphophilic, oval, calcareous (von Kossa’s stain) corpuscles in the stroma (Fig. 7). Reproductive and digestive organs were absent.
Diagnosis
Severe, chronic, active proliferative peritonitis with numerous parasitic cysts and larvae, consistent with proliferative sparganosis.
Discussion
In this case, the severe, chronic, active proliferative peritonitis with larval cestodes is consistent with S proliferum infestation. The larvae lacked scolices and other head structures, a digestive tract, and reproductive organs, and calcareous bodies were present. Calcareous bodies and lack a digestive tract are characteristic of cestodes, 6,7 and lack of scolices and suckers is characteristic of plerocercoid larvae of pseudophyllidean cestodes. Multiple histologic sections are recommended to ensure that neither scolices nor suckers and hooks are present. 4,6,15 Body cavity infestation with massive asexual replication is described for S proliferum, 3,11 a pseudophyllidean cestode, the most likely diagnosis in our case. The larvae are characterized by asexual branching and budding, which results in cysts that disperse in the host body and replicate. This proliferative form, or sparganosis, is often fatal. 8,10,12 Another pseudophyllidean of the genus Spirometra spp can occur in body cavities or in tissues of intermediate or paratenic hosts. 4,6,18,19 The larvae have the same morphological features as the larvae in our case, but asexual replication with resulting aberrant cystic structures does not occur. Infestation with Spirometra spp (mainly, Spirometra erinaceieuropaei or Spirometra mansonoides) is much more frequent than sparganosis but mostly caused by only one or few larvae without replication in the host, 4,8,9 inducing a nonproliferative form (plerocercoidosis). 15 It is possible that ingestion of large numbers of procercoids or plerocercoids of Spirometra spp can result in severe abdominal lesions resembling those in our case, but to our knowledge this has not reported and the procercoids and plerocercoids of Spirometra do not form cysts. 4 Thus, the predominantly cystic pattern in our case is more consistent with a diagnosis of sparganosis, although definitive confirmation would require genetic analyses of the larvae. Tetrahyridiosis, caused by larval stages of cyclophyllidean cestodes of the genus Mesocestoides, is another differential diagnosis. 4 –6 Tetrahyridia reproduce within the host but have four suckers on the anterior end and muscle separating the medullary and cortical region, features that distinguish them from plerocercoids. 4 –6,17 Pentastomidiasis is a less likely differential diagnosis. These parasites have two pairs of hooks surrounding the mouth and, in contrast to cestodes, have a multicellular intestine bordered by acidophilic glands. Furthermore, sclerotized openings in the body wall appearing as eosinophilic rings are essential features of this parasite. 1,4,6 Although pentastomids can invade body cavities of mammalian hosts, the morphology of hooks, intestine, and sclerotized openings clearly separate them from the parasitic stages in our case.
Sparganosis is a rare parasitic disease in an accidental intermediate mammalian host. 4,8,12,15 In 2001, S proliferum was phylogenetically identified as a new species in the order Pseudophyllidea. 10,13 The life cycle, including the definitive host, of S proliferum is unknown but thought to be similar to that of Spirometra spp. The definite hosts of Spirometra spp are carnivores, which shed eggs in feces. After embryonating in the environment, the eggs hatch in water and release coracidia, which are ingested by copepods. The coracidia develop into procercoids. Second intermediate hosts, including fish, reptiles, and amphibians, ingest infected copepods and acquire procercoid larvae. In the second intermediate host, the plerocercoids develop and are infective for predators of the secondary host. 4,8 Humans and other mammals, including apes, pigs, dogs, and cats, can serve as paratenic or second intermediate hosts. 2,4,8,12 Plerocercoidosis and, probably, sparganosis develop accidentally after ingesting procercoids or plerocercoids with contaminated water or infected intermediate hosts. The larvae penetrate the intestine wall and disperse in the body. A direct infection of open wounds or mucous membranes with plerocercoids has been described in humans. 4,8 Sparganosis occurs mainly in South East Asia, but a few cases are reported from the southern United States. 2 –4,15 In Europe, approximately 10 cases of mainly nonproliferative plerocercoidosis in humans have been reported; 14,16,18,19 however, there are no reports of animal disease. Sparganosis is usually associated with exposure to aquatic environments, but it can be acquired through ingestion of contaminated intermediate hosts. The source of infection in our case could not be determined, but given the history, it is unlikely to have come from outside Europe. Currently, there is no effective treatment for sparganosis, and the prognosis is poor. 3,11
Footnotes
Acknowledgements
We thank C. Zimmermann and M. Kopania for technical assistance and Ch. Hoffmann for her helpful advice.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
