Abstract
Bovine odorant-binding protein (OBP) may function in olfaction and defense against oxidative injury, but its role in inflammation and defense against bacterial infection has not been investigated. Expression of OBP was discovered in the bovine lung and found to undergo changes in abundance during glucocorticoid administration and stress. OBP was localized to nasal, tracheal, and bronchial mucosal glands with immunohistochemistry, with faint expression in airway surface epithelium and none in bronchioles or alveoli. Two isoforms of OBP were identified, appearing to be differentially regulated during lipopolysaccharide-induced pulmonary inflammation, but differences between these isoforms were not revealed by matrix-assisted laser desorption/ionization–time of flight mass spectrometry. Functional studies showed no effect of OBP on in vitro growth of Escherichia coli or Mannheimia haemolytica under iron-replete or iron-depleted conditions, nor did OBP opsonize bacteria for an enhanced neutrophil oxidative burst. However, OBP did reduce the ability of supernatants from lipopolysaccharide-stimulated macrophages to induce neutrophil chemotaxis. These findings indicate that OBP may inhibit neutrophil recruitment by inflammatory mediators, and they suggest an ability to bind macrophage-derived inflammatory mediators within the airways.
Keywords
The epithelial lining fluid of the lung and major airways is a thin but complex layer that includes numerous protein and nonprotein components originating from plasma, epithelial cells of the airways and alveoli, and leukocytes within the respiratory tract. Proteins within the airway surface liquid are known to have varied functional roles in respiratory health and disease, including bactericidal and bacteriostatic activity, opsonization of particulates including pathogens, modulation of the immunoinflammatory response, protection against oxidative or proteolytic injury, and stimulation of cell proliferation and tissue repair.5,20,30 We previously identified odorant-binding protein (OBP) as an abundant component of the bronchoalveolar lavage fluid (BALF) of cattle 18 and here further characterize its expression and function.
A member of the heterogeneous widely distributed lipocalin superfamily, bovine OBP is abundantly expressed in nasal secretions3,22 and is suspected to function in olfaction as it binds a range of small hydrophobic odorant molecules. 28 However, the mechanism through which OBP enables olfaction remains unclear, and alternative functions have been suggested. Interestingly, OBP is thought to reduce insect predation of cattle by binding components in expired air, which are potent insect attractants. 24 Binding is not restricted to odorants; OBP also binds the toxic aldehydes produced by lipid peroxidation, protecting epithelial cells from oxidative damage. 11 Other lipocalins, such as neutrophil gelatinase-associated lipocalin, tear lipocalin, and lipocalin 2, limit bacterial growth by scavenging bacterial siderophores, which are necessary for nutrient acquisition.8-10
OBPs are found in many insects where their importance in olfaction has been demonstrated.15,29,31 However, OBPs may function in the acute phase response of insects. A proteomic study of Drosophila hemolymph identified the expression of OBPs as being altered by bacterial, viral, and fungal infections. 14 Furthermore, a proteomic survey of the antibacterial compounds of Sarcophaga bullata (fleshfly) larvae identified an OBP that inhibits the growth of Escherichia coli and Staphylococcus aureus. 6
We recently demonstrated the presence of OBP in BALF by mass spectrometry and Western blotting, and OBP mRNA was detected in bronchial biopsies, confirming pulmonary gene expression.18,19 Pulmonary expression of this protein was upregulated by treatment of calves with dexamethasone. 18 Two isoforms of OBP were identified in Western blots, and a reduction in density of the lower pI isoform was identified in BALF from stressed compared to nonstressed calves.18,19 Analysis of the 5′flanking region of the OBP gene suggested several putative binding sites for the transcription factors AP-1, signal transducer and activator of transcription 6 (STAT-6), and STAT-1, 18 which are implicated in the regulation of host defense protein expression in response to proinflammatory cytokines, such as tumor necrosis factor α and interferon γ.1,12 Here, we further characterize OBP expression in the lung and suggest a role for this protein in modulating inflammatory responses in the respiratory system.
Materials and Methods
Immunohistochemistry
Samples of fresh tissue from bovine nasal mucosa, trachea, bronchi, and lung obtained from a slaughterhouse were flash frozen in liquid nitrogen and stored at –80°C before processing. Following embedding in ornithine carbamoyltransferase medium (Tissue Tek, Miles Inc, Elkhart, IN), 6-μm-thick sections were cut on a cryostat and allowed to air dry. Endogenous peroxidase was quenched with LabVision peroxidase blocker (Dako Canada Inc, Mississauga, Ontario, Canada), and nonspecific staining was blocked with EnVision universal blocker (Dako Canada Inc). An affinity-purified antibody to bovine OBP was prepared from the serum of rabbits immunized 4 times with a synthetic peptide fragment of bovine OBP (C-EDEDLEKFWKLTEDKGIDKK; residues 125E–144K; based on accession No. P07435 for the full-length OBP protein; Pacific Immunology Corporation, San Diego, CA). The antibody was applied at a 1:500 dilution in phosphate buffered saline (PBS) for 30 minutes at room temperature. After repeated washing in PBS, peroxidase-conjugated anti-mouse/anti-rabbit immunoglobulin (EnVision, Dako Canada Inc) was added for 30 minutes. Visualization was achieved with Nova Red chromogen (Vector Labs, Burlingame, CA) with hematoxylin counterstain. Negative control slides were incubated with rabbit preimmune sera instead of OBP antibody.
Bronchoalveolar Lavage and Bronchial Biopsy Collection
BALF samples were collected from eight 8-month-old beef calves as previously described. 18 The fluid was retrieved from the right caudal lung lobe; then, 500 μg of lipopolysaccharide (LPS) in 10 ml PBS was delivered endoscopically into the left caudal bronchus. After 24 hours, BALF was retrieved from the left caudal lung lobe. Biopsies of second- to third-generation bronchi were harvested as previously described, 18 immediately following lavage on the first day.
Analysis of OBP Gene Expression
Primary cultures of differentiated, pseudostratified bovine tracheal epithelial cells were prepared at an air–liquid interface 17 and harvested between 14 and 21 days in culture. Total RNA was isolated from homogenized bronchial biopsies or cultured tracheal epithelial cell cultures as previously described.17,18 Briefly, 500 ng of total RNA was reverse transcribed with QuantiTect Reverse Transcriptase kit (Qiagen Inc, Mississauga, Ontario, Canada); cDNA products were diluted 1:10; and real-time reverse transcription polymerase chain reaction was performed with a Light Cycler and MasterPlus SYBR Green I master mix (Roche Diagnostics, Mannheim, Germany). Primer sequences (forward 5′-CAAGGCTACAAAGCAAGACG-3′, reverse 5′-TTTGTCTTCCGTCAGCTTCC-3′) resulted in a 211–base pair product, confirmed by gel electrophoresis and sequencing to be OBP. Initial numbers of OBP template copies present in 500 ng of total RNA from cultured tracheal epithelial cells compared to bronchial biopsies were calculated with the following formula: No = 1/2 n , where No is the initial number of OBP amplicons and n is the crossing-point threshold. 13 The fold difference was then calculated with the mean initial number of amplicons in epithelial cultures equal to 1.
Analysis of BALF Proteins
BALF supernatant was prepared and separated by 2-dimensional electrophoresis (2-DE) with previously published methods. 18 Quadruplicate gels were analyzed with Phoretix software (Version 2004, Nonlinear Dynamics, Newcastle Upon Tyne, United Kingdom) to generate normalized spot volumes. 18 For mass spectrometry, excised gel pieces containing SYPRO Ruby–stained spots were destained, cysteines reduced with dithiothreitol, and alkylated with iodoacetamide. Proteomics-grade trypsin (Sigma-Aldrich, St Louis, MO) was added overnight and peptides extracted with acetonitrile/formic acid. Peptides were concentrated and desalted using ZipTipμ-C18 (Millipore, Billerica, MA) and eluted onto the target with α-cyano-4-hydroxycinnamic acid (Sigma-Aldrich). Peptides were analyzed with a Bruker Reflex III matrix-assisted laser desorption/ionization–time of flight (MALDI-TOF) mass spectrometer (mass accuracy, ± 0.1%) in positive-ion linear detection mode, calibrated externally with adrenocorticotrophin (ACTH 18–39) and cytochrome C (Sigma-Aldrich).
Bacterial Growth Inhibition Assay
Single colonies of E coli (DH5α) or Mannheimia haemolytica obtained from a calf with pneumonia were grown overnight in brain–heart infusion broth, then washed 3 times in PBS by centrifugation at 6,000 × g for 10 minutes. Bacteria were diluted to 3.5 × 105 colony-forming units per milliliter, and 2 μl of this suspension was added to 30 μl of serially diluted (3 × 10–5 to 3 × 10–8 M) recombinant bovine OBP (rbOBP) in PBS; rbOBP was a kind gift from Prof Roberto Ramoni, University of Parma, Italy, and was prepared in E coli as previously described. 23 The rbOBP protein identity was confirmed by SDS-PAGE and Western blot with the OBP antibody described above, and the protein concentration was confirmed by colorimetric assay (2D Quant Kit, Amersham Biosciences, Uppsala, Sweden). Samples were contained in a 96-well culture plate and incubated for 2, 5, or 24 hours in a humidified incubator at 37°C, then serially seeded in triplicate onto Columbia agar B plates and incubated overnight at 37°C, with the number of colonies counted. Bacterial growth inhibition under iron-restricted conditions was performed as previously described 8 such that rbOBP and bacteria were incubated in Dulbecco’s modified Eagle's medium containing 10% fetal bovine serum.
Isolation of Peripheral Blood Neutrophils and Macrophage Culture
Blood was collected from clinically normal, mature Holstein cows, and neutrophils were isolated as previously described. 16 Peripheral blood monocytes were isolated, and macrophage monolayer cultures were generated as previously described 25 with the following alterations. The buffy coat was obtained after centrifuging whole blood at 1,200 × g for 20 minutes and was overlain on Ficoll-Paque Plus (GE Healthcare Canada Inc, Mississauga, Ontario, Canada). After centrifuging for 30 minutes at 4°C, the resultant band containing monocytes was removed and washed 4 times in PBS containing 5-mM EDTA. Cells were resuspended in RPMI containing 10% fetal bovine serum, 2 mM L-glutamine, penicillin (100 IU/ml), and streptomycin (100 μg/ml), and 1 × 107 cells per milliliter were seeded into 24-well plates. Nonadherent cells were gently washed off after 18 hours; medium was changed every 48 hours; and wells achieved 95% confluence by 10 days after seeding. For LPS stimulation of macrophages, macrophage monolayers were washed with PBS; then, LPS (100 ng/ml) in serum-free RPMI medium was added (or RPMI medium alone as a control), and cells were incubated for 24 hours. Conditioned medium from 4 to 6 wells was pooled, centrifuged (400 × g for 5 minutes) to remove cells, and used immediately or stored at –80°C for up to 1 week.
Neutrophil Chemotaxis and Oxidative Burst Assays
Neutrophil chemotaxis to conditioned medium was assessed with a previously described fluorescence-based chemotaxis assay. 7 Briefly, neutrophils were incubated in 5 μg/ml calcein AM (Molecular Probes, Eugene, OR) before loading on the upper aspect of a membrane containing 5-μm diameter pores (ChemoTx, NeuroProbe, Gaithersburg, MD). This membrane was affixed over a 96-well plate containing quadruplicate wells filled with RPMI (negative control), zymosan-activated serum (positive control), conditioned medium from macrophages, rbOBP, or conditioned medium containing rbOBP. Four lower wells contained labeled neutrophils and did not have cells placed above the membrane; these wells were used to define 100% migration. Plates were incubated for 1 hour at 37°C, washed, and centrifuged; then, fluorescence was measured with a Typhoon variable mode imager (GE Healthcare) based on excitation and emission wavelengths of 485 and 538 nm, respectively. Percentage chemotaxis was calculated with the fluorescence of wells containing labeled cells as 100% and with migration to RPMI medium as 0%.
To determine if OBP opsonized bacteria to enhance neutrophil responses, M haemolytica or E coli bacteria were used nonopsonized or opsonized with bovine serum and concurrently incubated with or without 3 × 10–6 M rbOBP for 30 minutes. Bacteria were washed 3 times in PBS by centrifugation at 6,000 × g for 10 minutes, then incubated with bovine neutrophils for 30 minutes at 37°C. Neutrophil oxidative burst was measured with flow cytometry as previously described. 16 Bacteria opsonized with bovine serum served as the positive control.
Statistical Analysis
Data are presented as mean ± standard error of the mean. Differences in normalized spot volumes were analyzed by Student’s t test. Oxidative burst and chemotaxis data were analyzed by one-way analysis of variance and post hoc Tukey’s multiple comparison test. Statistical analyses were performed with GraphPad Prism 4.03 (GraphPad Software, San Diego, CA).
Results
Localization of Bovine OBP Expression
To determine the histologic location of OBP expression, immunohistochemistry was performed on multiple sections of lung with an OBP-specific rabbit antibody. Intense and specific labeling was found in nasal mucosal glands, in which high levels of OBP expression had already been documented (Figs. 1, 2).2,21 Comparably high OBP expression was identified in tracheal and bronchial mucosal glands with occasional faint labeling of ciliated epithelial cells at the surface of nasal and tracheal mucosa (Figs. 3, 4). No labeling was detected in alveoli or bronchioles. Further evidence for OBP expression by airway surface epithelium was determined through real-time reverse transcription polymerase chain reaction on cultured and differentiated tracheal epithelial cells. Specific OBP mRNA could be detected in these cells; however, initial amplicon numbers were approximately 500- to 1000-fold lower in cultured cells compared to those detected in bronchial biopsies (data not shown).
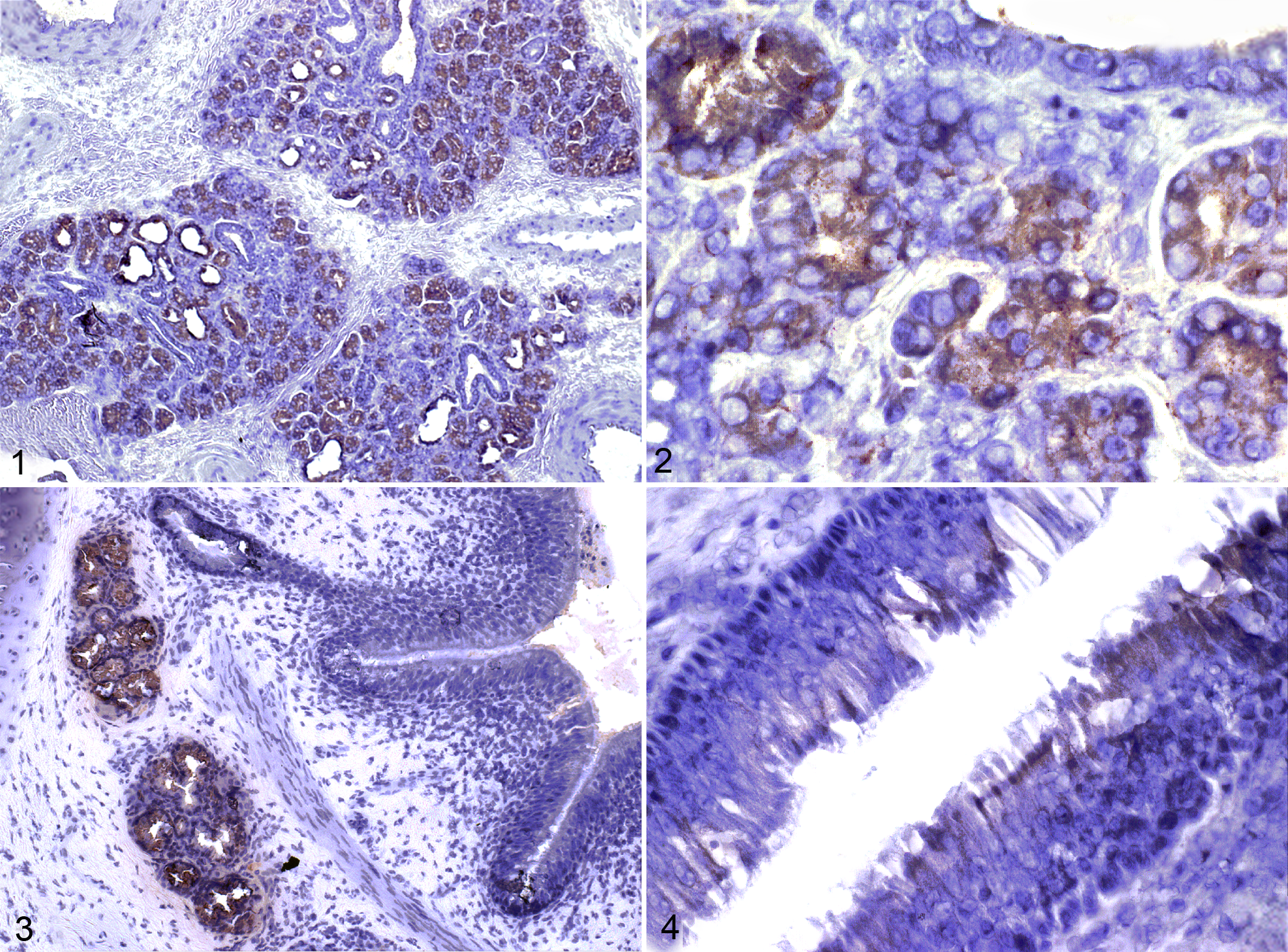
Immunohistochemical detection of odorant-binding protein in sections of bovine nasal mucosa, trachea, and bronchi. Immunolabeling is prominent in mucosal glands of the nasal mucosa (Figs. 1, 2), tracheal mucosa (not shown), and bronchial mucosa (Fig. 3), with scant cytoplasmic labeling of tracheal surface epithelial cells (Fig. 4).
Effect of Inflammation on OBP Expression in BALF
To examine the expression of OBP during pulmonary inflammation, bronchoalveolar lavage was performed before and 24 hours after the intrapulmonary infusion of LPS, and OBP in BALF was resolved by 2-DE (Fig. 5A ). Identification of these spots as OBP was previously confirmed by mass spectrometry and Western blotting.18,19 Densitometry of these spots revealed that the overall concentration of OBP was significantly greater in pooled samples from calves before LPS administration, compared to pooled samples after LPS administration (Fig. 5B). Although both isoforms were significantly reduced, there was a more marked reduction of the lower pI isoform (OBPa) compared to OBPb (Fig. 5C).
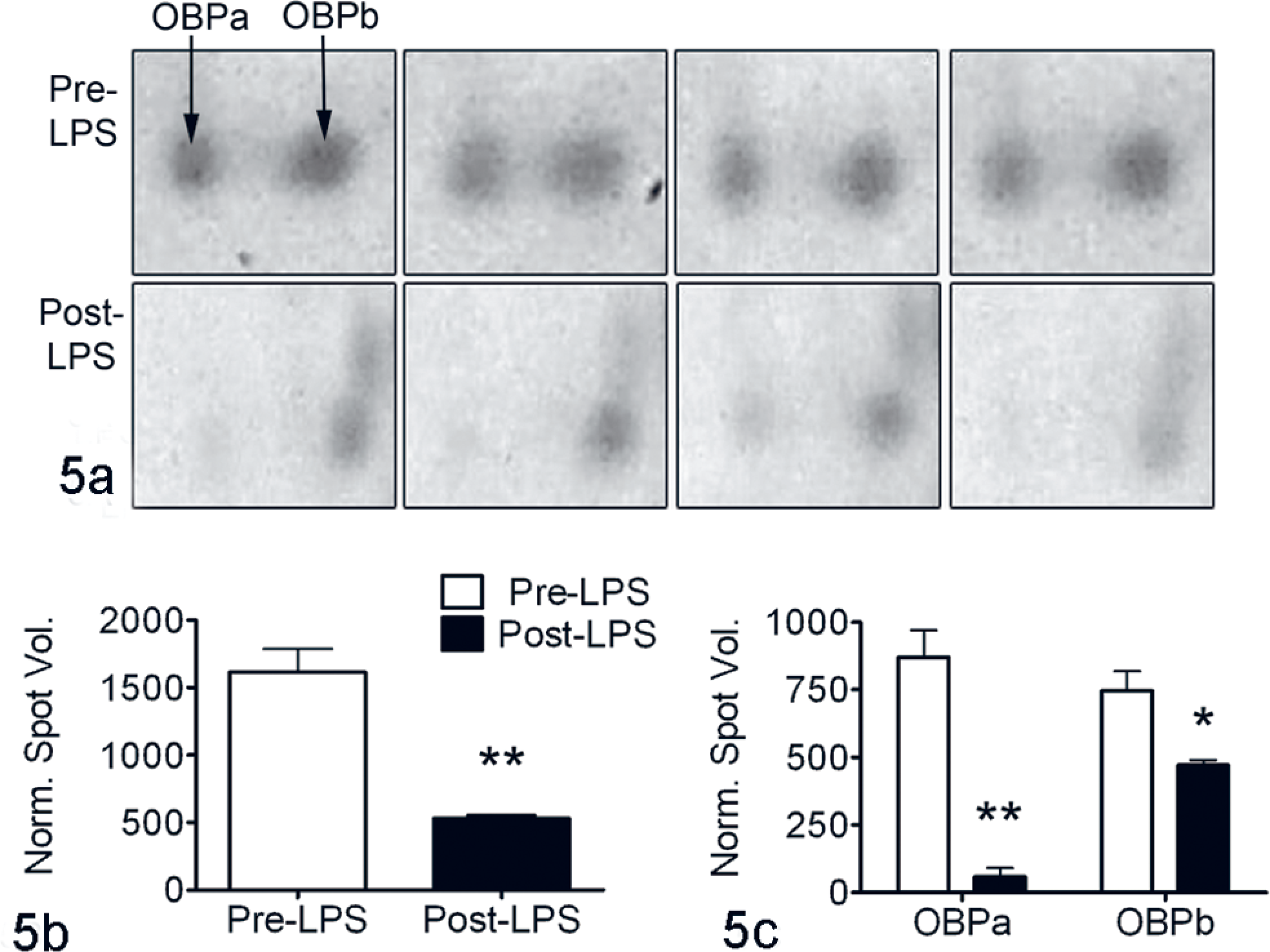
Lipopolysaccharide (LPS) alters the levels of odorant-binding protein (OBP) isoforms. A, 2-dimensional gel electrophoresis. Quadruplicate replicates of pooled bronchoalveolar lavage fluid from 8 calves before (pre-LPS) and 24 hours after (post-LPS) the intrapulmonary infusion of LPS. Mean (± standard error of the mean) normalized spot volumes for combined (B) or individual OBP isoforms (C) demonstrate lower amount of total OBP in pooled bronchoalveolar lavage fluid after LPS treatment, which is predominantly due to a loss of the lower pI isoform, designated OBPa. *P < .05. ** P < .01.
Mass Spectrometric Evaluation of OBP Isoforms
MALDI-TOF mass spectrometry was performed to determine if differences in peptide masses could explain the differences in isoelectric point and regulation by LPS of the 2 isoforms OBPa and OBPb. BALF samples collected before and 24 hours after LPS infusion were separated by 2-DE. Each OBP isoform spot was excised and trypsin digested. Comparing the resulting mass spectra revealed no clear differences between the 2 OBP isoforms (Fig. 6).
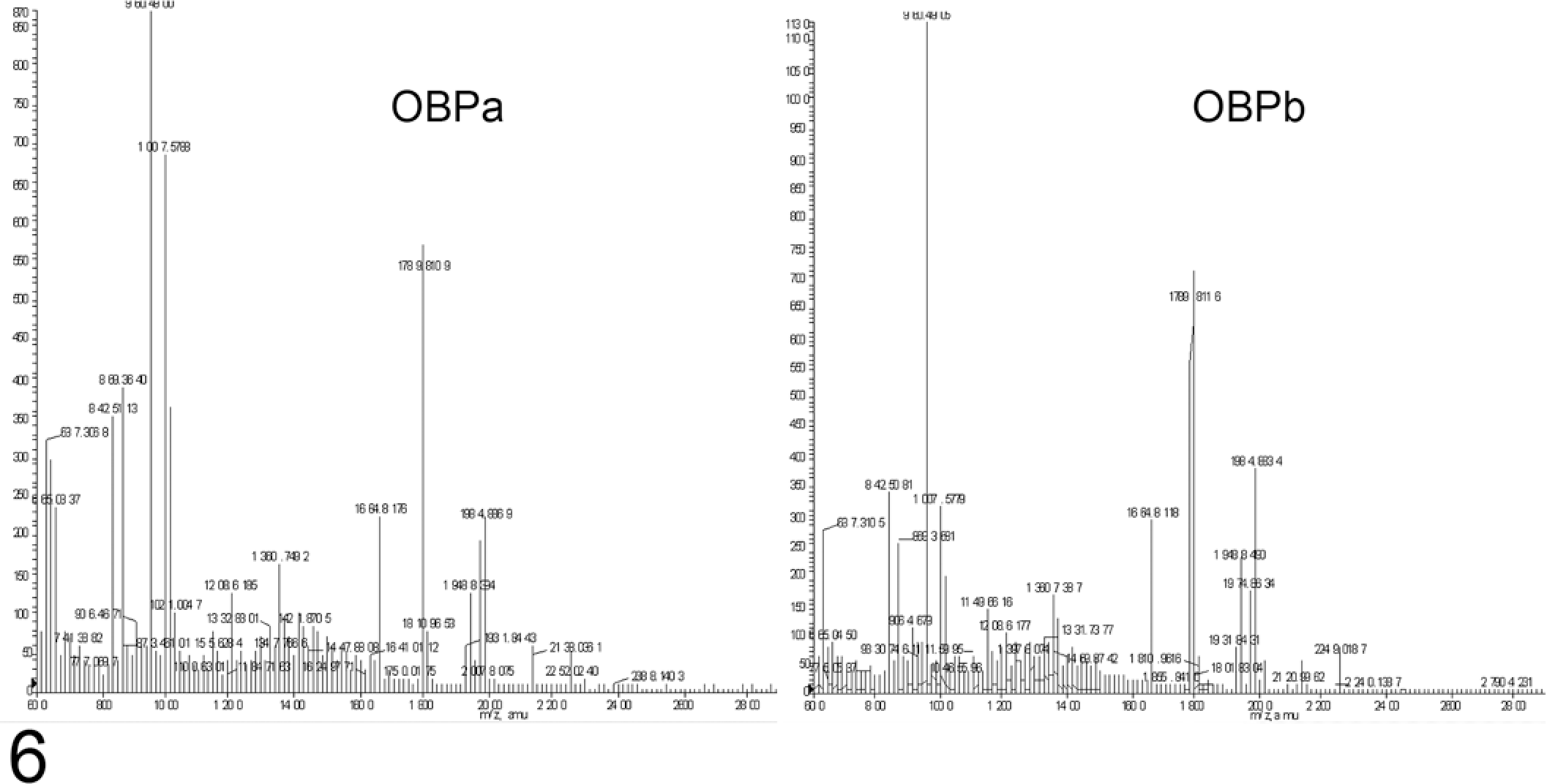
Matrix-assisted laser desorption/ionization–time of flight mass spectra of the 2 odorant-binding protein isoforms (OBPa and OBPb) show no significant shifts in the masses of peptides attributed to odorant-binding protein.
Effect of OBP on Survival and Opsonization of Bacteria
To determine if OBP possesses antimicrobial properties, bacteria were incubated in the presence or absence of rbOBP. No inhibition of E coli or M haemolytica growth occurred at rbOBP concentrations ranging from 3 × 10–5 to 3 × 10–8 M when incubated for 2 or 5 hours in iron-replete conditions or for 24 hours in iron-depleted conditions (data not shown).
To determine if OBP opsonized bacteria to enhance neutrophil oxidative burst, bacteria were incubated with rbOBP before the addition of bovine neutrophils. E coli or M haemolytica bacteria opsonized with bovine serum induced an oxidative burst in neutrophils, but the addition of rbOBP to nonopsonized or serum-opsonized bacteria did not alter the magnitude of this response (data not shown).
Effect of OBP on Neutrophil Chemotactic Activity of LPS-Conditioned Macrophages
The hypothesis that OBP modulates pulmonary inflammatory responses was addressed with in vitro assays of neutrophil chemotaxis, based on conditioned medium from LPS-stimulated monocyte-derived bovine macrophages as the neutrophil agonist. Neutrophils did not undergo chemotaxis in response to rbOBP alone. The percentage migration of bovine neutrophils toward the supernatant of LPS-stimulated macrophages was significantly greater (P < .05) than that toward the supernatant of unstimulated macrophages (Fig. 7). When rbOBP was added to the supernatant from LPS-stimulated macrophages 30 minutes before the chemotaxis assay, the increase in neutrophil migration was significantly reduced (Fig. 7). This experiment was repeated with neutrophils from 4 different animals, with similar results each time.
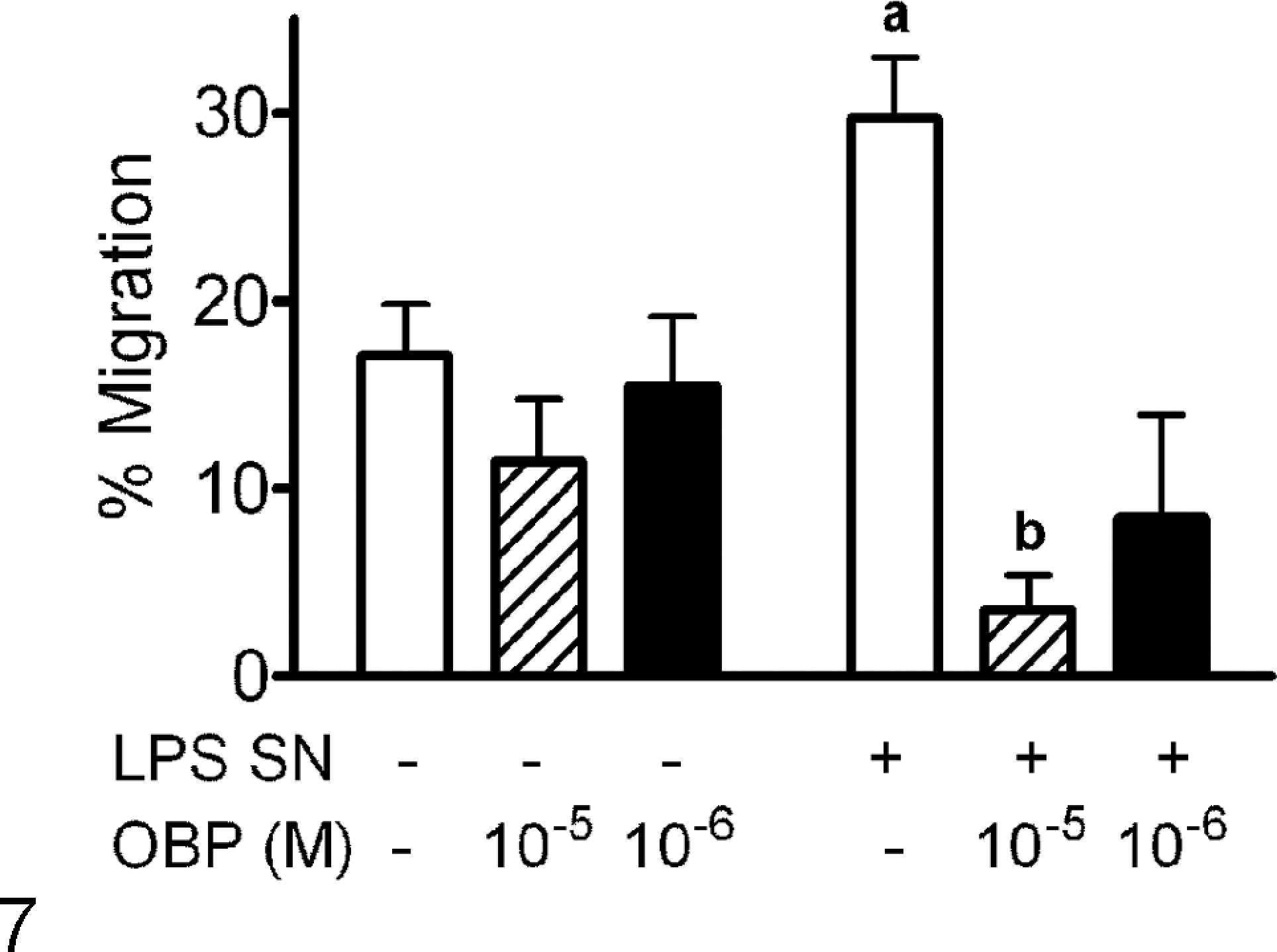
Odorant-binding protein (OBP) inhibits neutrophil migration to culture supernatants derived from lipopolysaccharide-stimulated macrophages. Supernatants from macrophages cultured in the presence or absence of lipopolysaccharide (LPS SN + and LPS SN –, respectively) were added to different concentrations of recombinant bovine OBP (0, 10–5, 10–6 M) 30 minutes before neutrophil chemotaxis was assessed. Data are expressed as mean (± standard error of the mean) percentage migration relative to positive and negative controls included in the assay (neutrophils in the bottom chamber and RPMI medium, respectively). A, P < .05 versus untreated supernatant (LPS SN –) containing no recombinant bovine OBP; B, P < .05 versus treated supernatant (LPS SN +) containing no recombinant bovine OBP.
Discussion
The epithelial lining fluid of the lung forms a critical interface between the pulmonary epithelium and the particulate, gaseous, and microbiological components of inhaled air. A diverse array of proteins and small peptides accumulate in this fluid and function to maintain homeostasis.5,20,30 We investigated the expression and function of OBP, confirming pulmonary expression and demonstrating a potential anti-inflammatory role by blocking macrophage-induced neutrophil migration.
Expression of OBP in bovine nasal glands is well documented;2,3,21 however, the presence and function of OBP in the lung has not been characterized. High binding activity of an OBP ligand has been found in nasal mucosal homogenates and minimal but variable binding activity in lung homogenates, implying that some may be present in the lung. 3 Although we found strong immunolabeling of OBP in tracheal and bronchial mucosal glands, we detected none in the bronchioles or alveoli. Our finding of scant expression in the airway surface epithelium has been noted by others and speculated to be artifact;2,21 however, our finding of OBP mRNA in tracheal epithelial cultures confirms that low-level expression occurs. The physiologic significance of the surface epithelial cell contribution to OBP expression is probably minimal in relation to the abundant glandular production; however, epithelial cell expression could be upregulated in some circumstances.
Given the putative transcription factor binding sites found in the 5′ flanking region of the OBP gene, 18 we considered that OBP expression may be driven by proinflammatory mediators. Although infusion of LPS did cause pulmonary inflammation as evidenced by an increased number of neutrophils in BALF, 18 we found that OBP levels in BALF were decreased (Fig. 5). Although a dilutional effect from higher concentrations of other proteins in BALF following LPS administration may have contributed to this effect, the selective reduction in OBPa levels and the lack of obvious reductions in other BALF proteins suggests that this effect was minimal. Confirmation of this decrease at the level of OBP mRNA would partially clarify whether the observed reduction in OBP protein is due to lower production or increased degradation or removal. 30
Interestingly, it has been suggested that the amount of OBP present in nasal mucosal extracts is highest in the winter, representing approximately 1% (w/w) of the crude extract. 4 Analysis of our 2-DE gels of BALF revealed that the density of OBP spots in relation to the density of all resolved spots consistently fell within the range of 1% to 3%; however, this may be a modest overestimate because not all proteins in BALF are resolved by 2-DE. Seasonal variation in pulmonary OBP expression has not been investigated, and all our studies were performed during the winter months.
OBPs have been identified in a variety of mammalian species, and in all cases except cattle, OBPs exist primarily as monomers. 26 Bovine OBP is unique because it exists as a homodimer in which the 2 C-terminal α domains are swapped. 23 Based on the molecular weights being half the native dimer in 2-DE, the 2 isoforms we observe in bovine BALF likely represent 2 monomeric forms. Why they differ in isoelectric point is not clear despite an investigation using MALDI-TOF mass spectrometry. We found no evidence of differential phosphorylation between the 2 forms, which could account for the isoelectric difference. Their differential expression following LPS infusion suggests some difference between the 2 OBP monomers does exist.
The addition of rbOBP to the supernatant of LPS-stimulated macrophages blocked the ability of this supernatant to induce neutrophil chemotaxis. Because rbOBP alone did not directly influence neutrophil chemotaxis, it probably acts by preventing stimulatory signals from affecting neutrophils. Numerous chemokines (eg, interleukin 8), cytokines (eg, tumor necrosis factor α, interleukin 1β), and other proinflammatory mediators (eg, leukotrienes) are released into the supernatant of LPS-stimulated macrophages. 27 Which mediators are being bound by OBP or otherwise prevented from acting is currently not known and the ability of OBP to bind a range of small hydrophobic molecules 26 implicates a myriad of possibilities. Similarly, whether native OBP exhibits the same function found with rbOBP remains to be determined.
The pulmonary function of OBP is becoming clearer and likely involves protecting airways from lipid peroxidation. 11 Relating to host defense, we found that OBP did not alter bacterial growth or bacterial stimulation of neutrophils, nor did it directly affect neutrophil function. However, OBP does appear to bind compounds that are chemotactic to neutrophils. The reduced levels of OBP during inflammation and the indirect inhibitory effects of OBP on neutrophil function suggest that reduced OBP levels may be an additional mechanism whereby LPS or LPS-induced inflammatory mediators stimulate neutrophil recruitment and oxidative burst in the lung. Conversely, the increased levels of OBP in the lung of dexamethasone-treated calves 18 suggests that this may be one mechanism by which corticosteroids modulate pulmonary inflammatory responses, which implicates OBP as a moderator of inflammation that may influence the outcome of many pulmonary disease conditions.
Footnotes
Acknowledgments
We thank Prof Roberto Ramoni, University of Parma, Italy, for generously providing recombinant bovine odorant binding protein.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was supported by the Natural Sciences and Engineering Research Council of Canada and the Ontario Cattlemen’s Association. Dr Mitchell was supported by a fellowship from the Canadian Institutes of Health Research.
