Abstract
Pleuropulmonary blastoma (PPB) is a rare biphasic tumor of children formed by mixed epithelial-and-mesenchymal elements. In this article, the authors report a pulmonary mass in an equine fetus with characteristics of PPB. A soft multicystic broad-based pleural mass was identified in the right caudal lung lobe. The mass comprised solid areas of loose mesenchyme, fenestrated by small ducts or large cystic areas lined by cuboidal epithelium. Mesenchymal elements had moderate anisocytosis, anisokaryosis, and cellular pleomorphism and were immunoreactive for vimentin. Epithelial cells lining ducts and cystic lumina were nonciliated and cuboidal with central round nuclei, minimal cellular pleomorphism, and strong immunoreactivity for cytokeratin. Pertinent characteristics in common with human PPB were the pleural-based location, the dual solid or delicate multiloculated cystic structure, the primitive mesenchymatous stroma fenestrated by well-differentiated cuboidal epithelial-lined lumina, and the occurrence during gestation.
Pleuropulmonary blastoma (PPB) and pulmonary blastoma (PB) constitute two rare but clinically and pathologically distinct pulmonary tumors characterized by embryonic structure. PB is an aggressive biphasic tumor formed by mixed malignant epithelial and mesenchymal elements that mimic embryonic structure. 1 These tumors are described in humans 1,9 and adult cattle, 8 and single-case reports exist for a 620-day-old rat, 2 a 4-year-old Lhasa Apso dog, 17 and an adult horse. 12 In humans, PB has a predilection for individuals between 30 and 50 years of age and is reportedly the only dysembryonic or dysontogenetic neoplasm to develop in adulthood consistently. 9 PPB is a related but distinct dysembryonic pulmonary neoplasm observed in children. PPBs are aggressive, often pleural-based masses, and they are distinguished from the PB of adults by occurrence in children, variable anatomic location (intrapulmonary, mediastinal, or pleural), and the mixture of mesenchymal and primitive small cell elements and bland (nonmalignant) epithelial components. 3,10,13 A distinctive feature of PPB is that these tumors often occur in a familial setting of dysplastic or neoplastic conditions 14 and may be a marker of childhood neoplasms in family members. 3 Human familial PPB has recently been associated with mutational loss of DICER1 from the epithelium of developing lung, leading to altered regulation of factors that promote mesenchymal proliferation. 4 Here we report a pulmonary mass in an equine fetus with characteristics of PPB.
History and Macroscopic Findings
A male Thoroughbred fetus was aborted at approximately 240 days' gestation, following a 3-week period of progressive weight loss and illness in the 10-year-old mare. The mare was diagnosed with severe peritonitis and euthanized 2 days following abortion. Postmortem examination was conducted on the fetus only.
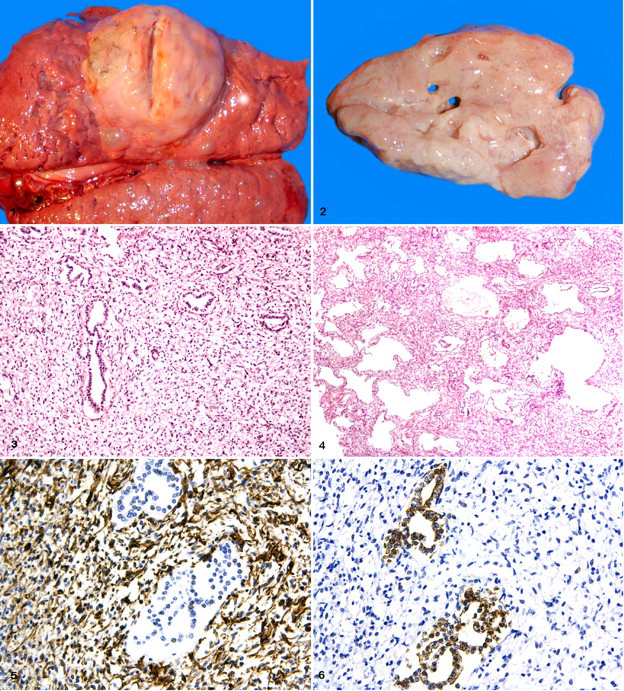
Grossly, the fetus had marked subcutaneous emphysema and was malodorous. Opening of the thoracic cavity revealed a pale pink, soft, welldemarcated, broad-based, ovoid mass measuring 6 cm × 5 cm × 3 cmcontiguous with the pleural surface of the right caudal lung lobe (Fig. 1). The cut surface of the mass bulged on sectioning and was fenestrated with multiple cystic lumina up to 15 mm in diameter (Fig. 2). No other lesions were noted in the thoraciccavity or elsewhere in the fetus. The allantochorion was complete; the cervical star displayed a 5 mm × 30 mm irregular focus of white to brown, gritty, nodularconcretions. The amnion contained meconium.
Histologic Examination
Tissue samples from the pleural mass and adjacent pulmonary parenchyma were fixed in 10% neutral buffered formalin, processed routinely for paraffin embedding, sectioned at 4 to 5 μm, and stained with hematoxylin and eosin. Immunohistochemistry was performed on sections of the mass, with appropriate controls and standard indirect peroxidase-based methods for the detection of cytokeratin (clone MNF116, Dako, Carpinteria, CA; anti-cytokeratins 5, 6, 8, and 17), vimentin, S-100 protein, smooth muscle actin, desmin, and chromogranin A.
Histologically, the pleural mass was composed of two patterns. Some areas comprised solid, loosely arranged blastematous mesenchyme, fenestrated by empty lumina lined by a single layer of nonciliated cuboidal or attenuated epithelium (Fig. 3). In the second pattern, neoplastic tissue consisted of multiple variable-diameter cystic lumina, formed by trabeculae of loose mesenchyme and lined by nonciliated cuboidal or attenuated epithelium (Fig. 4).
Mesenchymal cells were spindle to stellate, with distinct cell borders, scant eosinophilic cytoplasm, and central ovoid to elongate vesicular nuclei. Anisocytosis and anisokaryosis were moderate; mitotic figures were rare. Epithelial cells were cuboidal with distinct cell borders, a moderate amount of eosinophilic cytoplasm, variable perinuclear clearing, and round euchromatic nuclei with mild anisocytosis and mild anisokaryosis. The mass was vascularized by a delicate capillary network and small- to medium-sized arteries and veins. Mesenchymal elements were strongly immunoreactive for vimentin (Fig. 5) and negative for cytokeratin. Epithelial cells were strongly immunoreactive for cytokeratin (Fig. 6) and negative for vimentin. Neither mesenchymal nor epithelial elements were immunoreactive for S-100 protein, smooth muscle actin, desmin, or chromogranin A.
Clinical, pathological, and immunohistochemical findings were most consistent with PPB. The epithelial cells that lined ducts and cystic lumina were cuboidal or attenuated, with minimal pleomorphism and strong immunoreactivity for cytokeratin. Mesenchymal elements were blastematous, with moderate anisocytosis, anisokaryosis, and pleomorphism and strong diffuse immunoreactivity for vimentin. Coexpression of vimentin and cytokeratin was not observed in neoplastic cells. No evidence of myogenic, neural, or neuroendocrine differentiation was detected.
Discussion
Human PPBs are categorized on the basis of gross and microscopic appearance: 3 Type I PPB is exclusively cystic without a grossly detectable solid component; type III PPB is a solid tumor without epithelial-lined cystic spaces; and type II PPB is partially cystic and solid, bearing the microscopic features of both type I and type III PPB. The mixed solid-and-cystic gross and histologic features of this equine fetal mass are most similar to those of type II PPB.
The biphasic epithelial and mesenchymal appearance of this mass prompted consideration of PB as an alternative diagnosis. Incongruent with a diagnosis of PB, however, were the exclusively pleural-based location and the bland nature of epithelial components in contrast to the typically carcinomatous appearance of PB. In addition, the development of this tumor during gestation is inconsistent with the reportedly late development of PB in human beings and other species, including the recently reported case in an adult horse. 9,12 Other entities in the differential diagnoses for tumors with mixed epithelial and mesenchymal elements include pulmonary hamartoma, teratoma, and mesothelioma. Pulmonary hamartoma has been reported in foals 6 and in newborns or fetuses of other species. 7,11,15,16 Pulmonary hamartoma, also known as polyalveolar lobe, is characterized by an excessive number of alveoli, or “gigantism” of the alveolar region. 5 Polyalveolar lobe is thought to arise from excessive multiplication of alveoli, leading to a higher ratio of alveolus to muscular artery than in normal lungs at comparable age; the structure and size of the alveoli, airways, and blood vessels are normal. 6 These features were not found in the present case.
In conclusion, we report a mass with features of PPB in a nonhuman species. Salient diagnostic characteristics of this mass were as follows: the pleural-based location, the delicate multiloculated cystic structure, the primitive mesenchymatous stroma fenestrated by well-differentiated cuboidal epithelial-lined lumina, and the occurrence during gestation—consistent with the early development of human PPB.
Footnotes
Acknowledgements
We thank the histology and immunohistochemistry technicians of the Animal Health Trust for skillful production of the slides. Lucy Woolford is the recipient of a Senior Clinical Training Scholarship, funded by the generous support of the Horse Trust.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
