Abstract
Purpose and design:
BreathBC is a multicenter prospective observational cohort study aimed at comparing metabolic profiles from exhaled breath of patients with breast cancer (BC) and malignancy-free controls. The study accounts for the novelty and complexity of breath analysis, with a particular emphasis on the standardization of each step in the process.
Participants:
Women with primary BC without distant metastasis, women carriers of germline BRCA1/2 pathogenic variants and controls were consecutively recruited in two clinical independent cohorts and two technical validation cohorts. Breath samples were collected and linked to clinical breast status, personal, medical and lifestyle data that were retrieved using a questionnaire focused on factors potentially affecting breath analysis.
Results:
Among 1010 participants, a group of 846 subjects, compliant with criteria of recruitment and sampling of the study, were preliminarily characterized. The mean age was 61 years for patients with BC and 58 years for controls, with 71% of women in post-menopause. The control group included 48% of participants with benign disease. Hypertension was the main age-related morbidity observed in 28% of participants and 10% were smokers. Among patients with BC, 15% had in situ disease and 85% an invasive cancer whose sub-typing presented a high prevalence of luminal subtypes, in agreement with the consecutive recruitment.
Future activities will be focused on data analysis of breathomics data and on technical enhancement of prototypes used for sampling and instrumental analysis.
Introduction
Breast cancer (BC) is a heterogeneous disease and is the leading cause of cancer-related mortality in women worldwide. 1 BC screening in women without signs of disease leads to identification of cancer at an earlier stage than in symptomatic patients, ensuring a more treatable disease. Specifically, mammography screening was found to be significantly associated with reduced BC mortality and to provide prognostic information beyond stage migration for patients with invasive BC.2,3 The benefits versus risks vary based on the individual’s age, risk factors, and frequency of screening. 4 Despite the efficacy of imaging modalities in detecting the disease, its efficacy is suboptimal in detecting cancer in dense breasts with a prevalence of overdiagnosis and false positives especially among younger women, including high-risk young women.4,5 Histopathological examination establishes a definitive diagnosis of BC through tissue biopsy obtained by ultrasound or stereotactic guidance. Biopsy is mandatory before any type of treatment to ensure diagnosis of invasive disease, especially to assess the pattern of druggable targets if pre-surgical treatment is planned. 6 However, biopsy is inherently subject to limitations due to the invasiveness of the procedure and the potential promotion of pro-metastatic tissue changes and systemic diffusion of cancer cells in the case of a delay in surgery after diagnosis. 7
For these reasons, there is a growing need to develop non-invasive strategies that can support imaging techniques in early detection of BC. This has promoted the identification of novel diagnostic markers by searching for pathological cells and cancer-associated molecules, including metabolites, in blood or other non-invasive fluids such exhaled breath. 8 Breath analysis offers a tremendous untapped potential for clinical diagnosis of cancer due to its non-invasiveness, painlessness, safety, ready acceptance by patients, and nearly unlimited access to samples. 9 Mass spectrometry (MS) technologies have been developed for the detection of volatile organic compounds (VOCs) in exhaled breath with gas chromatography coupled to mass spectrometry (GC-MS) being a widely used technique for VOC detection in breath samples. 10 Recently, alternative techniques such as secondary electrospray ionization (SESI) coupled to mass spectrometry have emerged, 11 offering rapid, real-time analysis without the need for sample preparation, thus making it well-suited for diagnostic applications in clinical practice.12-14
The application of breath analysis to clinical research requires the global breath profiling of a large number of healthy and diseased subjects. This has transformed breath analysis into an “omics” technology, i.e. breathomics, a field focused on the metabolomics of exhaled human breath.13,15 Despite its potential, the diagnostic clinical development of breath analysis is a lengthy process and the majority of reported breath studies on disease-associated VOCs are currently small in size, single-centered, and generally without validation. The successful implementation of a breathomics clinical study requires large and well-characterized cohorts, a proper study design, and a robust technology. Furthermore, the integrity and quality of experimental and clinical data, and the implementation of homogenous practices, including standardized operating procedures (SOPs) for breath sample collection, storage, and analysis9,13,15,16 are the foundation of a reliable breathomics study.
Herein, we present a standardized clinical study of breath analysis aimed at identifying a VOC signature that can discriminate breath samples from patients with BC from those of malignancy-free controls. Sample collection protocols, instrumental parameters, QC procedures and cohort composition are detailed whereas data analysis is not presented here. The multicenter study recruited more than 1000 participants and was designed to minimize the pre-analytical variability and consider confounding factors potentially affecting breath analysis.
Cohort description and methods
Study design
The prospective observational study BreathBC, conducted in accordance with principles for Good Clinical Practice (GCP) founded in the Declaration of Helsinki, 17 has been designed to collect the exhaled breath of a large cohort of women including patients with BC and malignancy-free controls, profile breath samples and compare the breath profiles to identify a cancer-associated signature that can detect BC in a non-invasive manner. The core objective is the development of a breath test tailored for clinical practice in the perspective of future applications in diagnosis of cancer.
In the present study, independent cohorts have been enrolled within three centers: Fondazione IRCCS Istituto Nazionale dei Tumori (INT) that was the coordinator center (center 1); Cremona hospital (ASST-Cremona, center 2) and Desio hospital (ASST-Brianza, center 3). A series of women carriers of germline BRCA1/2 pathogenic variants was included in the study due to their high risk of developing BC or ovarian cancer during their lifetime. 18 Blood samples from patients and carriers of germline BRCA1/2 pathogenic variants were collected on the same day of the breath sampling for further metabolic-based analysis.
The exhaled breath was collected and analyzed following the standard breath analysis steps: sampling, profiling of samples and data analysis, as shown in
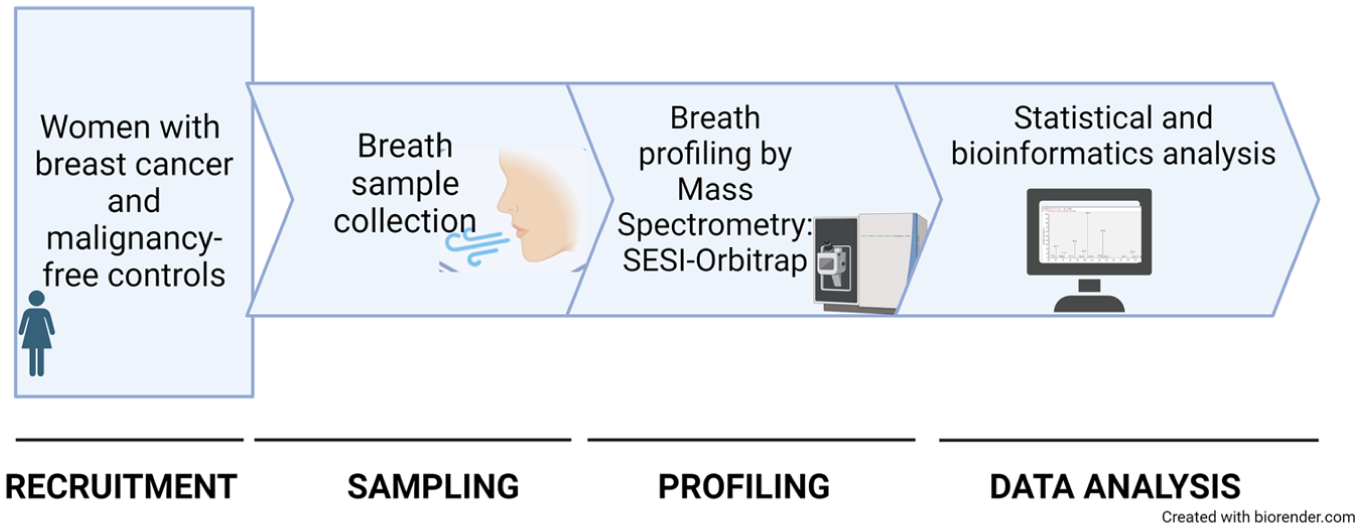
Schematic overview of steps in breath analysis. The process of breath analysis follows a sequential order including breath sampling, mass spectrometry analysis and data processing. In the present study samples were collected in a dedicated room equipped with a HEPA-filtered air purification system, adhering to safety protocols. Within two hours, the collected samples were analyzed by an LTQ Orbitrap Elite (Thermo Fisher Scientific) equipped with a SuperSESI ionization source (Fossiliontech) and data were addressed to computational analysis. All steps, from sampling to final data interpretation and storage, were completed on the same day.
The primary outcome of this study will be the classification of exhaled breath from patients with BC and malignancy-free controls, based on a comparison of global VOC profiles of the study participants. The predictive result will be compared with histopathological or imaging-based diagnoses for patients with BC and control subjects, respectively. The secondary outcome will explore a potential diversity of exhaled breath from controls without any breast involvement and those with benign disease.
A preliminary and explorative comparison will be performed on exhaled breath collected by women carriers of germline BRCA1/2 pathogenic variants and matched non-BRCA women to define characteristics of BRCA-related volatome that are potentially useful for early detection of hereditary BC.
Sampling and profiling of breath samples were performed under the same conditions at INT, whereas the sampling procedure or the instrumental analysis were respectively tested in the multicenter technical validations. To evaluate the potential sources of variability related to a future application of our breath test in different centers, an independent recruitment and sampling was performed at Cremona Hospital, while an independent MS instrumental analysis validation was performed using the Desio Hospital instrumentation.
Cremona Hospital independently recruited participants compliant with the inclusion/exclusion criteria of the study that were sampled according to INT protocol. Breath samples were transferred to INT and analyzed using an LTQ Orbitrap Elite (Themo Fisher Scientific) coupled to SuperSESI (Fossiliontech) following protocols described in Online Supplementary File 2.
Desio Hospital analyzed breath samples from participants that were recruited and sampled in INT following the described protocols. Breath samples were transferred to Desio hospital and MS analysis was performed using an Orbitrap Exploris 120 (Themo Fisher Scientific) coupled to Super SESI-X (SSX, Fossiliontech) following INT protocols.
The technical validations will provide new insights for the standardization of all the steps involved in pre-analytical and analytical breath analysis. The technical validations meet the need to establish new clinical and technical standards tailored for sensitive analytical procedures and understand bias related to different batches and origin of breathomics data.
Participants
The study exclusively recruited females with and without BC who were prospectively and consecutively recruited at Fondazione IRCCS Istituto Nazionale dei Tumori (INT) and at Cremona Hospital for the technical validation between June 2021 and October 2023, following the inclusion and exclusion criteria outlined in Figure 2. The investigator or authorized designee explained to each participant the objectives of the study and the clinical significance of the breath test, specifying the explorative nature of the present research. Participants were informed that they were free to refuse to participate and may withdraw their consent at any time for any reason. Participants were enrolled in two clinical cohorts (June 2021 to July 2023; January 2023 to July 2023), and in two technical validation cohorts (June 2023 to October 2023) and exhaled breath samples were collected after signing informed consent and filling the questionnaire, in accordance with the sampling protocol provided in Online Supplementary File 1. Patients with BC were selected among those undergoing breast surgeries on the day of pre-operatory testing, whereas malignancy-free controls were enrolled during breast screening and women with negative mammography and/or ultrasound were included in the control group. Patients with BC had a primary BC without distant metastasis and were sampled at the baseline, before surgical or pharmacological treatment. A significant portion of the women carriers of germline BRCA1/2 pathogenic variants were part of a prospective trial cohort and were under follow-up. 18
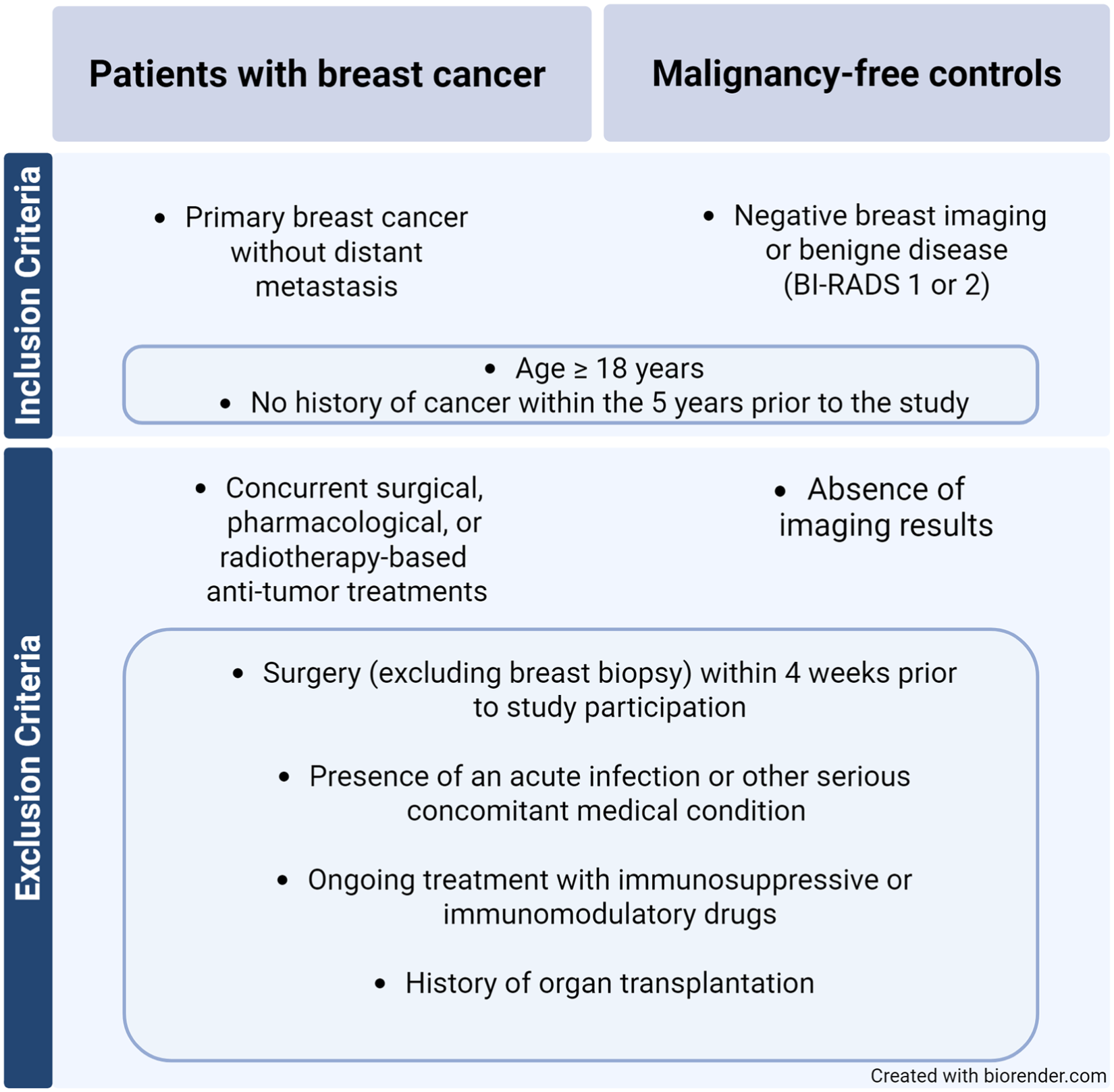
Inclusion and exclusion criteria used in the breath analysis clinical study. The study was designed with the aim of recruiting groups of patients reflecting the complexity of breast cancer and minimizing the interference of factors that could potentially affect breath analysis such as clinical treatments and inflammatory status. Patients with the presence of a primary breast cancer were recruited but without distant metastasis to prevent advanced disease.
Breath sample collection and analysis
Participants were required to refrain from eating or drinking sugar-based beverages for the two hours prior to sampling to avoid uncontrolled metabolic changes. They were also advised not to use products that could potentially interfere with MS measurements such as body lotions or menthol-based toothpaste for two hours before testing. Medications taken on the day of sampling were reported in the questionnaire administered before sampling.
Breath samples were collected in a dedicated sampling room that was equipped with a HEPA-filtered air purification system which reduces gaseous residues of detergents, deodorants, or other factors interfering with breath analysis. Only authorized personnel could access the sampling room. Each participant remained in the breath analysis room for at least 15 min before breath sampling, using this time to sign informed consent and fill in the questionnaire with the operator’s help. This time enabled participants to breathe purified air and if necessary, to calm down, minimizing some of the environmental and individual factors interfering with metabolic measurements, in a context of standardization of all breath analysis procedures. A sterilized sampling system, 14 implemented with an electrostatic bacterial and viral filter, ensured germ-free content of the storage bags (Online Supplementary File 1). The breath sample collection protocol required that study participants take four normal breaths and then inflate the sampling bag with a single deep exhalation. Each subject produced two replicate samples in rapid succession for a total of two samples per subject, which were compared to verify their similarity as part of quality control of sampling and profiling steps. A sample of environmental air was collected during each sampling session for further comparative analysis with the profiles of study participants.
Breath samples are subjected to contamination and degradation phenomena, which increase over time as described by Ghimenti et al. 19 In this study, breath sampling storage and transport to the laboratory were restricted to a maximum of two hours after their collection.
Sample profiles were obtained in INT by a SuperSESI ionization source (Fossiliontech) coupled to an LTQ Orbitrap Elite MS (Thermo Fisher Scientific). Samples were analyzed in positive ionization mode and full scan acquisition. Electrospray was generated using a double-capillary ESI system and optimized to maximize total ion current. The ESI solution consisted of 0.1% v/v formic acid in bidistilled water. Calibration was performed every two weeks using a standard ESI calibration solution. Further analytical details and instrument parameters are available in the Online Supplementary File 2.
Safety procedures
Breath sampling was carried out by adopting a risk-reduction approach in every step of the sampling process in order to avoid the spread of SARS-CoV-2 or other infection agents, with the aim of protecting both participants and operators and taking into account the fragility of oncology patients (Online Supplementary File 1). Specifically, this included risk-reduction in terms of personal protective measures, safety of breath sampling equipment, safety procedures for transport and analysis of collected samples, and disposal of all potentially contaminated samples and materials, according to Pleil and co-workers. 20
The present study did not recruit during peaks in the SARS-CoV-2 pandemic and all participants were free of acute respiratory tract infections and vaccinated against SARS-CoV-2. However, the safety measures of our sampling protocol designed concurrently with SARS-CoV-2 pandemics, include general procedures useful not only during disease outbreaks, but even prospectively for sampling subjects in the acute phase of a communicable disease for studies associating breath VOCs with microbial and viral infections.
Standardization of pre-analytical procedures and quality controls
The study was conducted following principles for Good Laboratory Practice,21,22 REMARK recommendations 23 and STROBE statements for reporting. 24 To minimize the experimental variability intrinsically related to the complex procedures of breath analysis, SOPs were designed for sampling and instrumental analysis, properly tailored for oncology clinical practice (Online Supplementary Files 1 and 2). Adequate quality assurance procedures were established to assess the overall quality of the experimental procedure and its repeatability and reproducibility at the beginning and during the study by qualitative evaluation of the final output of instrumentation, i.e. the individual breath profile according to the technical requirement for high-quality data.
Quality assurance steps include i) the operator’s inspection of the background’s and samples’ spectra, ii) a software-assisted evaluation on spectra using a quality scoring designed by our group iii) quantitative comparison of replicates.
Operator’s inspection of spectra includes a stability check of the background spray. The presence of interfering peaks in the background spectrum indicates the presence of contaminant species which are removed for instance by substituting either the ESI solution, or the capillaries. The operator monitors the ionization trend while acquiring the samples and in case of spray instability adjusts the instrument parameters to stabilize the sample ionization profile.
Our software-assisted spectra evaluation estimates several parameters like the breathprint profile, signal to noise ratio etc., and assigns a quality score to discriminate and exclude low quality samples. Sample quality is assessed right after the acquisition. The software is currently under patent filing process.
The quantitative comparison of the two replicates obtained for each study participant was also used to control the repeatability of sampling and instrumental analysis. It is performed post mass spectrometry analysis using Lin’s concordance correlation coefficient (CCC) as described by Martinez-Lozano and coworkers. 14 Residual experimental variability was then corrected by statistical procedures embedded in data pre-processing of our computational pipeline. 14 Non-compliant samples were reported and data were not included in the analysis and were separately archived. Since samples are analyzed within two hours, where possible the subjects whose breath samples were non-compliant were asked to perform the breath sampling again.
Part of our quality procedures is the training of personnel involved in all steps of breath analysis and the use of a dedicated sampling room, as described above.
Confounding factors potentially affecting breath analysis
To minimize heterogeneity of the cohort only Caucasian women were recruited including a homogeneous and well characterized group of women carriers of germline BRCA1/2 pathogenic variants. 18 To retrieve known variables that could influence systemic metabolic status and, consequently, breath measurements, the questionnaire provided in Online Supplementary File 3 was administered to study participants. As summarized in Table 1, the questionnaire collected personal and anthropometric data, lifestyle factors (including smoking and dietary habits), and medical history, with a particular focus on chronic disease such as asthma, hypertension, metabolic disorders such as diabetes, as well as the chronic use of medications.
Summary of data collected with the questionnaire before breath sampling.

Self-reported information was verified and implemented when available using the institutional clinical databases.
Self-reported smoking habit was used to assess tobacco smoke exposure. Smokers were asked about the average daily number of cigarettes smoked and the total number of smoking years, which were used to calculate the number of pack-years as follows: number of cigarettes smoked per day/20 × number of smoking years. Never smokers or subjects who stopped smoking for at least one year were considered non-smokers, whereas all others, independently of smoking intensity, were considered active smokers. E-cig or heated tobacco products were not used by smokers or former smokers.
Clinical data collection and management
All data were collected and processed in accordance with the General Data Protection Regulation (EU) 2016/679 (GDPR). 25 The clinicopathological data of in situ or invasive BC were retrieved from the institutional clinical database, including ER, PGR, HER2 and Ki-67 status. The subtyping of invasive BC was assessed according to immunohistochemical (IHC) determination of hormone receptors, HER2, and Ki-67 as follows: Luminal A (ER + and/or PR +, HER2 −, Ki-67 < 20%); Luminal B (ER + and/or PR +, HER2 −, Ki-67 ⩾ 20%); Luminal HER2 (ER + and/or PR +, HER2; +, any Ki-67); HER2 + (ER - and/or PR -, HER2+, any Ki-67); triple negative BC (TN) (ER −, PR −, HER2 −, any Ki-67). 6 Imaging diagnosis of controls was referred to the Breast Imaging-Reporting and Data System (BI-RADS) and was reported as 1 (negative) or 2 (benign with 0 % probability of malignancy). 26
Clinical data were collected in an integrated database dedicated to the present study (BreathBC database) that includes all breath profiles associated with technical information and personal/clinical data. All data were anonymized to prevent patient identification. Data related to study participants encompasses personal information, imaging data, surgery, histology, and BC data retrieved by institutional database and clinical records, whereas data on style of life, chronic disease and medication usage were recovered by the questionnaire administered to all study participants (Online Supplementary File 3). Database generation included manual checks of data and their integrity.
Statistical analysis
Clinical data were evaluated using standard descriptive statistics to summarize cohort baseline characteristics between BC patients and malignancy-free controls. For continuous variables, summary statistics were calculated using (mean), (median), (range [min-max]) and (SD), whereas for categorical variables frequency count (N) or percentage (%) was used as appropriate.
Correction of pre-analytical and analytical variability, missing data management, quality control steps and supervised analysis for the classification of breath profiles from BC patients and malignancy-free controls will be performed essentially as previously described. 14
Sample size adequacy is detailed in Online Supplementary File 4.
Statistical analyses were carried out using R software, version 4.3.2 (www.r-project.org/). Test results were considered significant at p-values <0.05.
Results
A total of 1010 women with or without BC were consecutively recruited and their exhaled breath was collected and analyzed (Figure 3). The generous compliance of women participating in the study was noteworthy, mainly because they appreciated the non-invasiveness of the sampling.
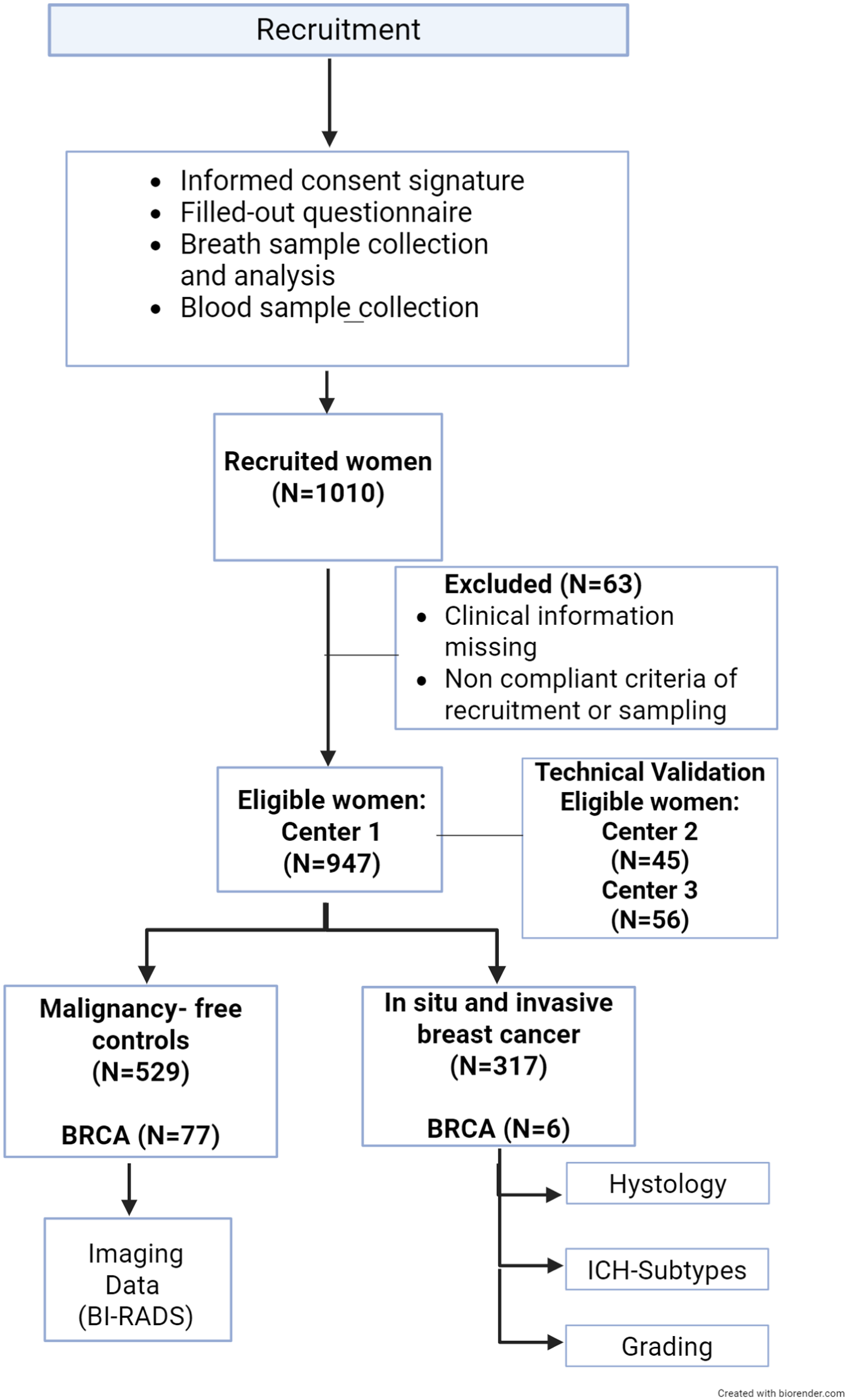
Study flow chart outlying the breath analysis path from recruitment of patients to data collection. The figure reports the sampling steps and the number of recruited, excluded and eligible participants, including the number of subjects recruited in the technical validation cohorts. Study participants provided informed consent, completed a questionnaire including personal and demographic data, medical history and information on lifestyle, and donated a breath sample, with patients with breast cancer and women carriers of germline BRCA1/2 pathogenic variants also contributing a blood sample. Participants were recruited in three centers and categorized into two groups: malignancy-free controls with negative breast imaging (BI-RADS 1 or 2), and patients with breast cancer, for whom clinical data including histology, IHC subtype, and grading were collected. Women carriers of germline BRCA1/2 pathogenic variants were recruited in center 1. Six of them had breast cancer at the time of recruitment, whereas the remaining were malignancy-free controls.
As shown in Figure 3, data extracted by the BreathBC database indicated that 947 participants in the study were adherent to the recruitment criteria described in Figure 2. The INT cohort (center 1) comprised 846 participants with 317 BC patients and 529 malignancy-free controls. Controls included 48% of subjects with benign disease. Eighty-three participants of INT cohort were carriers of germline BRCA1/2 pathogenic variants. Six of them had a BC at the time of recruitment, they were sampled before surgery and included in the BC group. The remaining 77 were considered malignancy-free controls as were without signs of disease at recruitment and without history of cancer within the five years before the study, in agreement with the inclusion criteria of the study.
To test the variability of sampling within the technical validation, center 2 recruited and independently sampled 45 participants compliant with the inclusion/exclusion criteria of the study, including eight BC patients and 37 malignancy-free controls. Breath samples of center 2 were analyzed by INT by SESI-HRMS, as the majority of samples of the study. The breath samples from 56 participants (19 BC patients and 37 malignancy-free controls including 26 subjects with benign disease) recruited and sampled at INT were instead analyzed by the center 3 using the INT experimental conditions to explore the impact of instrumentation characteristics on breath profiles (Figure 3).
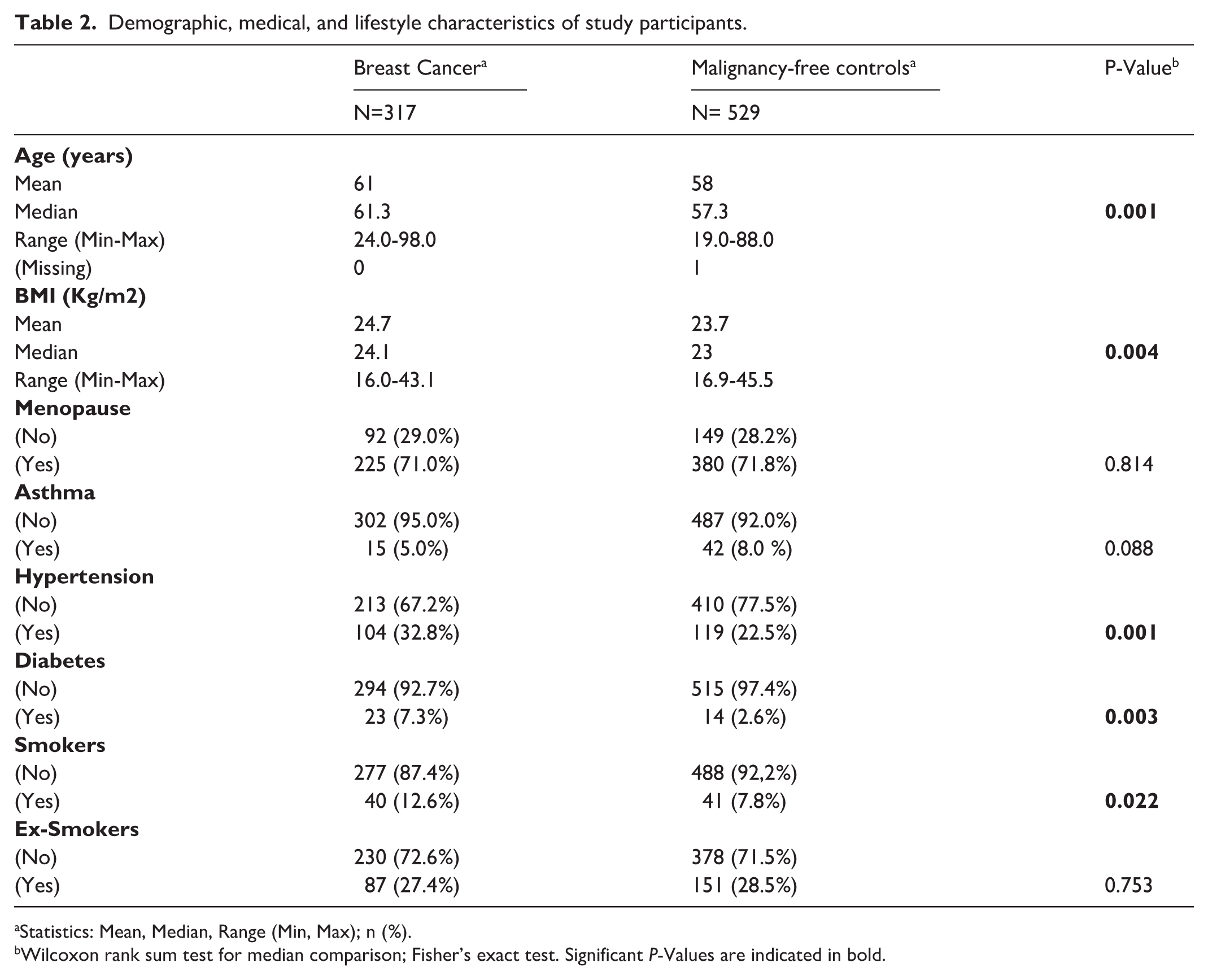
Table 2 details the data collected from the 846 participants of center 1 using the questionnaire reported in Online Supplementary File 3. The age of study participants ranged from 19 to 98 years with a mean of 61.0 years for patient with BC and 58.0 years for controls, with most of the women in post-menopause (71-71.8%). BMI ranged from 16.0 to 45.5 kg/m2 with means ranging between 23.7 and 24.7 kg/m2. The large majority of participants were omnivorous, with only 3% vegetarians or vegans. The series was characterized by a low proportion of smokers (7.8-12.6%) compared to a smoking prevalence of 18-23% among women in Italy. 27 There were 81 smokers and 238 former smokers who were considered non-smokers. Mean smoking history was 15.6 and 11.8 pack/years for smokers and ex-smokers, respectively; ex-smokers stopped smoking from 1 to 50 years before breath sampling. Less than 8% of women had asthma or diabetes, whereas hypertension and use of antihypertensive drugs were registered in the 22.5-32,8% of participants without or with BC, and depend on age. 28
Demographic, medical, and lifestyle characteristics of study participants.
Statistics: Mean, Median, Range (Min, Max); n (%).
Wilcoxon rank sum test for median comparison; Fisher’s exact test. Significant P-Values are indicated in bold.
The significant difference of age, BMI, number of smokers and subjects with hypertension or diabetes between BC and malignancy-free control groups reported in Table 2 suggests a proper balancing of baseline characteristics between the two groups during the computational analysis and classification of breath profiles.29,30
In all, 315 BC patients had information on tumor characteristics (15% in situ and 85% invasive BC) and the classification in IHC subtype shown in Table 3 agrees with our previous data, 31 except for the under-representation of the HER2 and TN subtypes that reflects the modality of patient recruitment and the current treatment of estrogen negative diseases. HER2 and TN cancers are less common than luminal ones and our selection of women with BC among those undergoing breast surgery on the day of pre-operatory testing sometimes excluded women with HER2 and TN cancers that were treated with neoadjuvant therapy and therefore at surgery did not meet the inclusion criteria of the present study requiring absence of treatment.
Prevalence of in situ and invasive disease in participants with breast cancer.
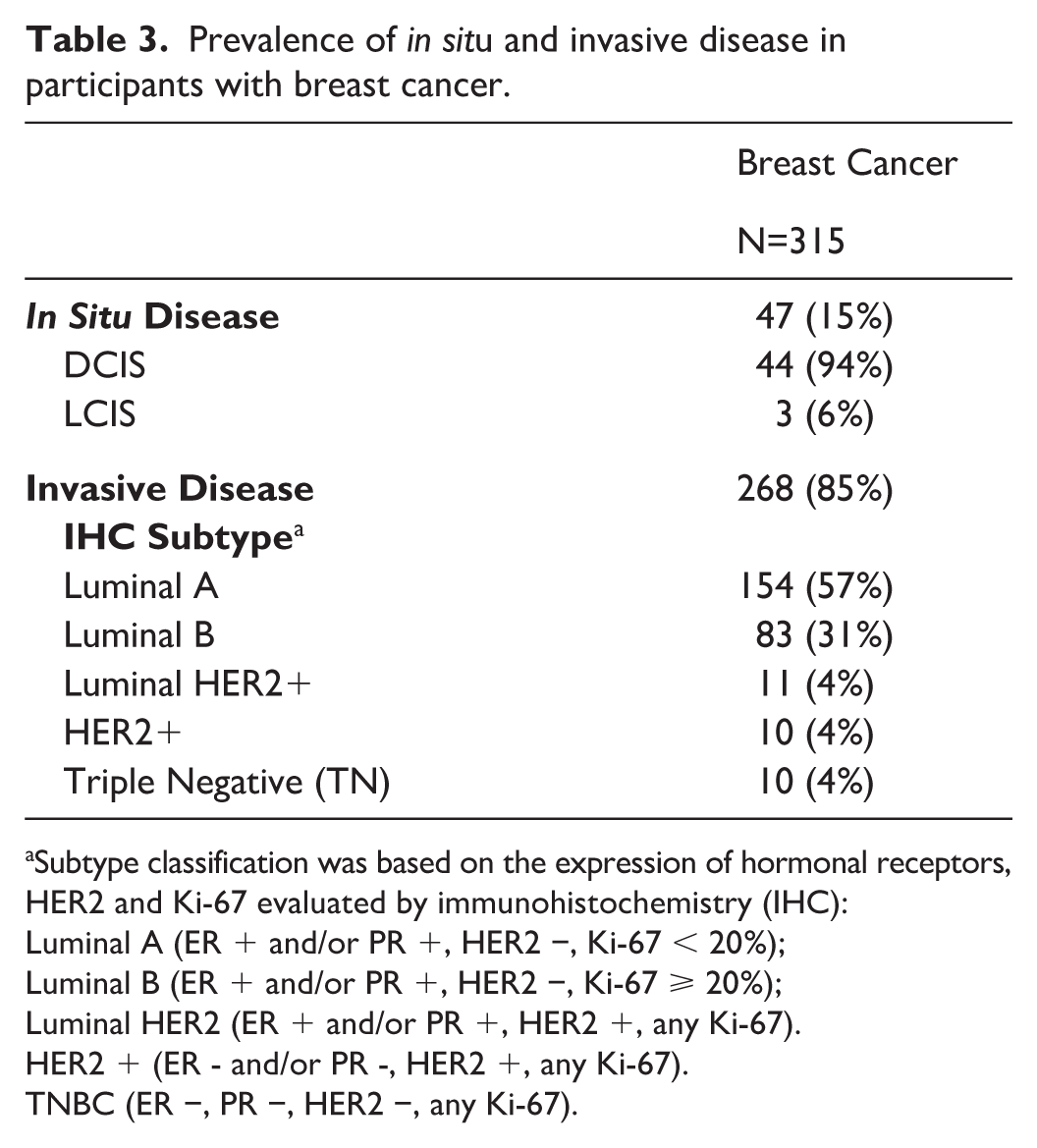
Subtype classification was based on the expression of hormonal receptors,
HER2 and Ki-67 evaluated by immunohistochemistry (IHC):
Luminal A (ER + and/or PR +, HER2 −, Ki-67 < 20%);
Luminal B (ER + and/or PR +, HER2 −, Ki-67 ⩾ 20%);
Luminal HER2 (ER + and/or PR +, HER2 +, any Ki-67).
HER2 + (ER - and/or PR -, HER2 +, any Ki-67).
TNBC (ER −, PR −, HER2 −, any Ki-67).
The patient cohort is skewed towards early disease, with advanced disease minimally represented. The recruitment included only patients with primary BC without distant metastasis (Figure 2). In situ cancers, defined as Stage 0 disease, were 15% (Table 3). A representative set of 155 patients with invasive disease showed mainly Grade I-II BC tumors (76%) and without involvement of lymph-nodes (72%). BC tumors had a size range of 0.2-4.5 cm with an average of 1.6 cm and 74% of tumors were < 2 cm.
Discussion
We present a clinical study aimed at identifying a VOC signature that can discriminate breath samples of patients with BC from those of malignancy-free controls, with the aim to exploit the potentiality of breath analysis in clinical research and diagnosis. The clinical study, based on our previous work, 14 considers the complexity and heterogeneity of BC, the need for validation settings in breath test development, and the standardization of each step of breath analysis including sampling, instrumental analysis, and clinical data management, along with omics data mining and analysis. This attempt requires a large consecutive cohort representative of BC and rigorous compliance with pre-analytical SOPs to achieve high–quality breath data. Our hypothesis is that a fully standardized breath analysis may provide high-quality data for computational procedures and promote the development of non-invasive diagnostic tools for early identification of BC. A series of women carriers of germline BRCA1/2 pathogenic variants is included in the present study because they are subjected to continuous and frequent imaging screening starting at a young age, undergoing numerous biopsies, and would particularly benefit from early non-invasive detection of BC.
Breath sampling is a challenging analytical process which encompasses the modality of breath release, the sampling system, the influence of environmental air, and the interference of volatile contaminants, etc. To reduce the experimental variability, the described analytical pipeline includes the collection of all samples under the same conditions and apply quality checks immediately after sampling and during data pre-processing. Thus, the major sources of variability remain mostly unknown sources of biological variability, as metabolic variations not managed by fasting or other measures were applied.
Breath analysis is an innovative technology currently applied in clinical diagnosis for the detection of a single volatile molecule, as in the case of exhaled nitric oxide for respiratory diseases 32 and Helicobacter pylori (H. pylori) infection in gastroduodenal disease 33 but its application in cancer research requires to scale up to global profiling of volatile metabolites of exhaled breath. There is need to establish new clinical and technical standards tailored for sensitive analytical procedures and understand bias related to different batches and origin of breathomics data.
A recent study has highlighted the significant impact of some factors such as sex, age, BMI, and smoking on the composition of exhaled breath using a large cohort of volunteers subjected to breath analysis. 34 So far little is known about the effect of chronic inflammatory disease and on the use of medications on the composition of exhaled breath and the questionnaire we proposed to all participants meets the need of exploring known and new factors potentially interfering with breath analysis.
The main limitation of the present study is related to the novelty of breath analysis and is represented by the lack of commercial tools designed especially for large clinical studies of breath analysis to minimize experimental variability. Therefore, the consequent development and validation of prototypes for sampling, instrumental and computational analysis was required.
Future activities will be focused on data analysis of breathomics data, based on design and characteristics of the cohorts described herein. In particular, the optimization of data pre-processing will exploit data from questionnaires and technical validations and will produce robust data for the generation of predictive models discriminating the breath profiles of women with BC. The prototypes for sampling and the dedicated computational tools that represent the technical deliverables of this study will be further enhanced from the perspective of a possible industrial development.
Supplemental Material
sj-docx-1-tmj-10.1177_03008916261431054 – Supplemental material for Breath analysis for non-invasive detection of breast cancer: A prospective observational cohort study
Supplemental material, sj-docx-1-tmj-10.1177_03008916261431054 for Breath analysis for non-invasive detection of breast cancer: A prospective observational cohort study by Michela Bianchi, Francesco Segrado, Alessio Polymeropoulos, Andreina Oliverio, Paolo Baili, Luisa Emma Morandi, Roberto Agresti, Gianfranco Scaperrotta, Siranoush Manoukian, Manuela Milani, Valerio Leoni, Rosalba Miceli, Patrizia Pasanisi and Rosaria Orlandi in Tumori Journal
Supplemental Material
sj-docx-2-tmj-10.1177_03008916261431054 – Supplemental material for Breath analysis for non-invasive detection of breast cancer: A prospective observational cohort study
Supplemental material, sj-docx-2-tmj-10.1177_03008916261431054 for Breath analysis for non-invasive detection of breast cancer: A prospective observational cohort study by Michela Bianchi, Francesco Segrado, Alessio Polymeropoulos, Andreina Oliverio, Paolo Baili, Luisa Emma Morandi, Roberto Agresti, Gianfranco Scaperrotta, Siranoush Manoukian, Manuela Milani, Valerio Leoni, Rosalba Miceli, Patrizia Pasanisi and Rosaria Orlandi in Tumori Journal
Supplemental Material
sj-docx-3-tmj-10.1177_03008916261431054 – Supplemental material for Breath analysis for non-invasive detection of breast cancer: A prospective observational cohort study
Supplemental material, sj-docx-3-tmj-10.1177_03008916261431054 for Breath analysis for non-invasive detection of breast cancer: A prospective observational cohort study by Michela Bianchi, Francesco Segrado, Alessio Polymeropoulos, Andreina Oliverio, Paolo Baili, Luisa Emma Morandi, Roberto Agresti, Gianfranco Scaperrotta, Siranoush Manoukian, Manuela Milani, Valerio Leoni, Rosalba Miceli, Patrizia Pasanisi and Rosaria Orlandi in Tumori Journal
Supplemental Material
sj-docx-4-tmj-10.1177_03008916261431054 – Supplemental material for Breath analysis for non-invasive detection of breast cancer: A prospective observational cohort study
Supplemental material, sj-docx-4-tmj-10.1177_03008916261431054 for Breath analysis for non-invasive detection of breast cancer: A prospective observational cohort study by Michela Bianchi, Francesco Segrado, Alessio Polymeropoulos, Andreina Oliverio, Paolo Baili, Luisa Emma Morandi, Roberto Agresti, Gianfranco Scaperrotta, Siranoush Manoukian, Manuela Milani, Valerio Leoni, Rosalba Miceli, Patrizia Pasanisi and Rosaria Orlandi in Tumori Journal
Footnotes
Acknowledgements
We thank the Nutritional and Metabolomics laboratory (NuMeLab) for technical support, the INT Central Sterile Facility for sterilization of sampling systems and Patrick Moore for language revision of the manuscript.
We thank Lega Italiana per la Lotta contro i Tumori (Lilt) and Salute Donna that harmonized our research activities within the clinical journey of study participants and Lilt volunteers that supported the experimental activities during preparation of the home-made sampling system.
We also thank all volunteers who participated in the study during their pre-operatory testing and BC screening.
Author contributions
Conceptualization: R.O., R.M. Data curation: A.P., M.B., A.O., P.B., L.E.M., S.M. Formal analysis: A.P. Funding acquisition: R.O. Investigation: F.S., M.B. Methodology: F.S., A.P., M.B. Project administration: R.O. Resources: R.A., G.S., P.P., M.M. Supervision: R.O. Validation: M.M., V.L. Writing – original draft: R.O. Writing – review and editing R.O., M.B., F.S., P.P., A.P., R.M.
Data availability
The clinical data that support the findings of the present study are available on reasonable request and according to institutional rules. For more information contact the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was granted by the Italian Ministry of Health (Grant No. RF 2016-02361759 to R. Orlandi, “Ricerca Corrente” funds and “5 per 1000” funds - financial support for healthcare research).
Ethical approval
The clinical study was approved by the Ethics Committee of Fondazione IRCCS Istituto Nazionale dei Tumori (INT 69/18) in accordance with the General Data Protection Regulation (EU) 2016/679 (GDPR).
Informed consent
Written informed consent was obtained from all participants to the study.
Trial registration
Trial registration number NCT06499077 (ClinicalTrials.gov ID).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
