Abstract
Purpose:
This study aimed to compare the incidence of fat necrosis after accelerated partial breast irradiation (APBI) vs hypofractionated whole breast irradiation (WBI) in patients with early-stage breast cancer.
Materials and methods:
Data from early-stage breast cancer patients who underwent breast-conserving surgery and adjuvant radiotherapy between 2009 and 2022 were retrospectively collected. Radiation therapy consisted of APBI of 30 Gy in 5 daily fractions (Fx) (delivered in one week, consecutively) to the tumour bed or WBI (42.4 Gy in 16 Fx). Reports on fat necrosis were extracted from yearly mammograms and breast ultrasound imaging. The primary endpoint was the incidence of radiologically detected fat necrosis.
Results:
A total of 536 patients were included among the APBI and WBI cohorts, with 268 and 268 patients respectively. The three-year Kaplan-Meier actuarial rate of fat necrosis was 32.8% (95% CI: 30.0% – 35.6%) for APBI and 22.3% (95% CI: 19.7% – 24.9%) for WBI patients. Univariate Kaplan-Meier survival analysis revealed a Hazard Ratio of 1.6 [95% CI: 1.1 – 2.2; p = 0.0055] for the fat necrosis rate within the APBI group compared to WBI. Multivariate Cox proportional hazard regression confirmed significant associations between fat necrosis and APBI (HR = 2.2 95% CI: 1.2 – 4.0; p = 0.01).
Conclusions:
The occurrence of radiologically diagnosed fat necrosis was higher in the APBI group compared to the WBI. Further investigations aiming to identify a lower-dose schedule with comparable efficacy to 30 Gy in 5 Fx but fewer toxicities, particularly for high-risk patients, are warranted.
Introduction
Partial breast irradiation (PBI) has demonstrated comparable oncological outcomes to whole breast irradiation (WBI) in several randomized phase III trials.1–11 Glynn et al. 12 emphasized that, in terms of cost-effectiveness and expected quality-adjusted life years, PBI compared to WBI was more favorable. International guidelines now endorse PBI as a viable option for early-stage breast cancer (BC) patients.13–15 Among various proposed dose schedules, the RAPID trial-like therapy (38.5 Gy / 10 Fx twice daily) resulted in increased rates of late toxicities7,16 and poorer cosmetic outcomes. Conversely, both 40 Gy in 15 Fx and Accelerated PBI (APBI) (defined with 5 or less fractions) have shown fewer adverse effects.1–11 Nevertheless, Colciago et al. 17 identified an elevated incidence of fat necrosis among patients treated with APBI (30 Gy in 5 daily fractions).
First described in the 1920s by Dr. Lee and Dr. Adair, 18 fat necrosis is a benign, non-suppurative inflammatory process of adipose tissue that mostly occurs after surgery or radiotherapy. While this side effect is often recognized and included in the consent form, it can lead to higher grades of fibrosis, breast pain, and worse cosmesis, causing physical and psychological distress. Additionally, fat necrosis presents a challenge for differential diagnosis, as it may be difficult to distinguish from carcinoma through both clinical examination and imaging. 19
Lövey et al. 20 reported an incidence of fat necrosis ranging from 4% to 52% of cases in PBI, with a higher risk associated with the use of brachytherapy. Dosimetric factors influencing this side effect include the dimension of the planning target volume (PTV) and higher volumes receiving more than 100% of the prescribed dose. 21 External Beam PBI demonstrates lower fat necrosis rates, varying from 3% to 25%. 22 Nevertheless, employing more accelerated treatment schedules may raise concerns about the incidence of fat necrosis, especially when compared to hypofractionated WBI.
This study aims to compare the occurrence of fat necrosis following external beam APBI or hypofractionated WBI in patients with early-stage breast cancer
Material and methods
Patient selection
Data from patients with early-stage breast cancer treated at our institution between 2009 and 2022 were retrospectively collected. All patients underwent breast-conserving surgery and received adjuvant radiotherapy with either an APBI or WBI scheme.
We excluded WBI patients who received a boost to the tumour bed, APBI patients with characteristics amenable for a tumour bed boost (i.e., age < 60), patients who underwent a double-time quadrantectomy, those who underwent chemotherapy, and those lacking post-treatment mammography and/or ultrasonography data. The retrospective analysis of late complications after breast cancer radiotherapy was approved by the Ethics Committee of Fondazione IRCCS Istituto Nazionale dei Tumori (INT 110/22) .
Radiation therapy
Since 2009, every early-stage breast cancer patient who underwent breast-conserving surgery at our institution has been treated with adjuvant hypofractionated WBI. In July 2013, following the results of a pilot study, 23 the "one-week" APBI schedule was introduced. Specific institutional inclusion and exclusion criteria for APBI are detailed in Table 1. Subsequently, although APBI became the preferred option, many patients were still treated with WBI based on physician discretion. As expertise in APBI improved, an increasing number of patients underwent this technique, resulting in a decrease in WBI patients and an increase in APBI patients over time.
Institutional criteria for APBI.

For APBI, the prescribed dose was 30 Gy delivered in 5 daily fractions to the clinical target volume (CTV). The contouring method is outlined in Table 2. Volumetric modulated arc therapy was employed as the delivery technique for this group, with daily image guided radiation therapy using cone-beam computed tomography. Dose constraints for APBI are provided in Table 3.
Contouring.

GTV: gross tumour volume; CTV: clinical tumour volume; PTV: planning target volume.
Dose constraints for APBI.
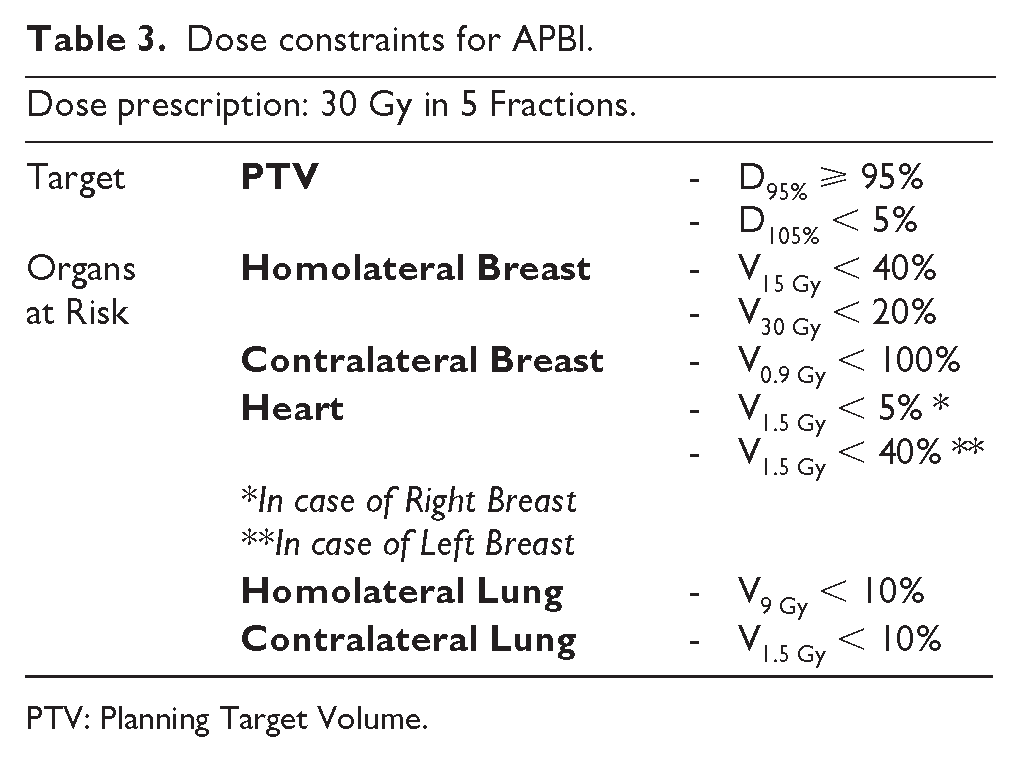
PTV: Planning Target Volume.
WBI patients received a total dose of 42.40 Gy administered in 16 daily fractions to the entire breast, delineated according to the specifications outlined in Table 2. The irradiation technique utilized was three-dimensional conformal radiation therapy, with treatment accuracy monitored using offline portal images.
For both groups, photon plans were generated using energy levels of 6 and/or 15 MV through the Eclipse treatment planning system version 15.6 (Varian Medical Systems, Palo Alto, California). These plans were calculated employing the Analytical Anisotropic Algorithm, with a dose grid resolution of 2.5 mm.
Follow-up
Follow-up consisted of a surgical visit every six months for the first three years, and then annually, along with an annual radiotherapy visit. Annual imaging included bilateral mammography and breast ultrasound.
Endpoint
The primary endpoint of the study was the incidence of radiologically diagnosed fat necrosis. Fat necrosis refers to areas of mammary lipid cyst with associated dystrophic calcification. As shown in Figure 1, mammographic features are radiolucent round/oval lesions with small areas of dystrophic calcifications which are often spherical and related to areas of previous breast trauma or surgery. Imaging was performed annually during the follow-up of each patient. One researcher (R.R.C) retrospectively retrieved data on fat necrosis from radiologists’ reports. The onset date of fat necrosis was determined based on the first radiological examination confirming its presence. These radiological examinations were conducted by proficient radiologists at the institution. Radiologists were not blinded to the choice of radiation therapy.
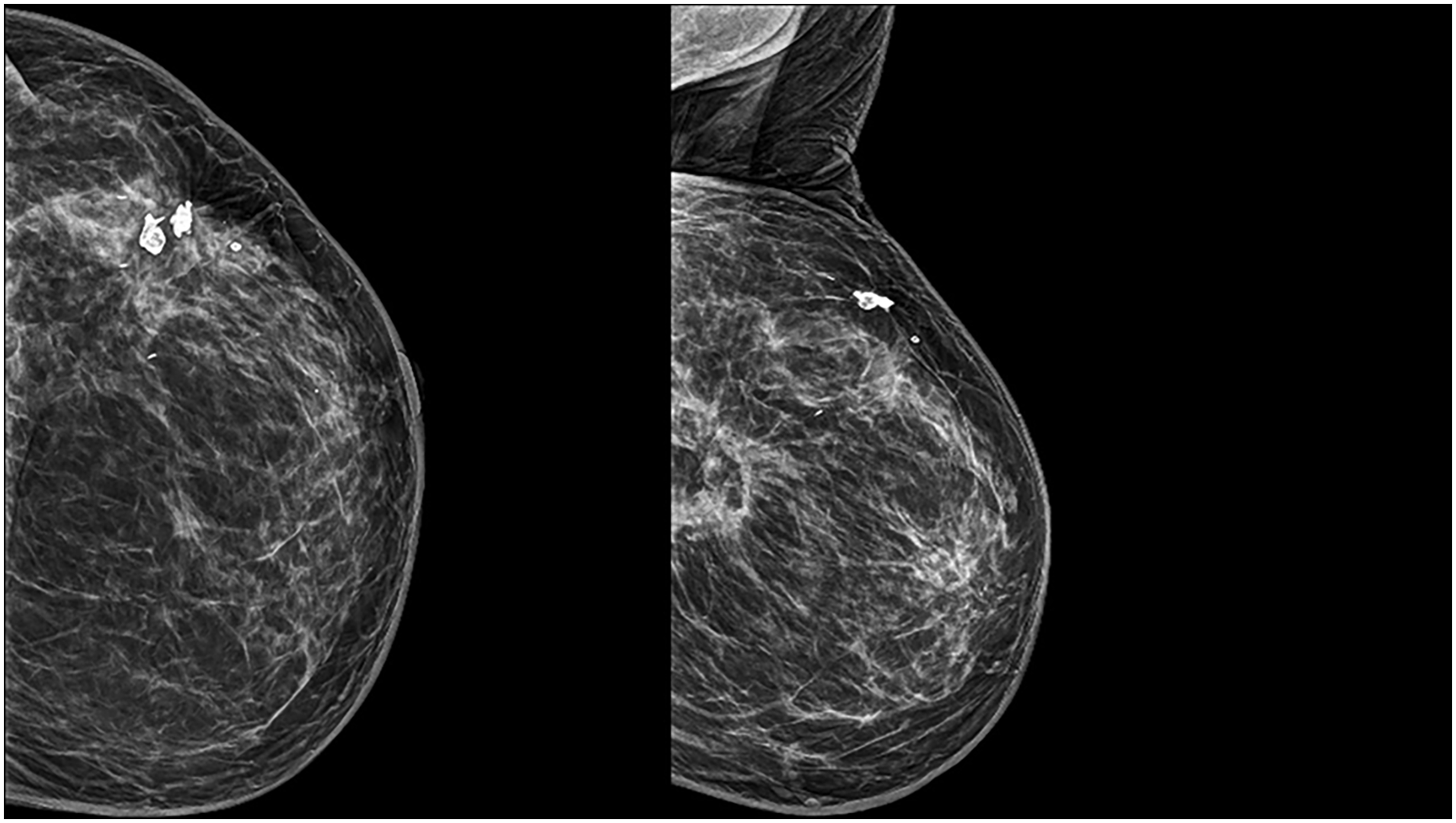
Mammographic evidence of fat necrosis.
If fat necrosis was found to be more prevalent in the APBI group, we planned to conduct physician and patients-assessed cosmetic analyses specifically for these patients, according to the Harvard scale, 24 detected at the last follow-up visit.
Statistical analysis
With a case-control methodology, we matched patients based on age, pathological tumour stage (T-stage), and diabetes status. Variables were selected based on both the existing literature and clinical expertise. Notably, diabetes revealed an association with a higher incidence of fat necrosis. 17 We used the match Controls function from the e1071 package in R (Version 4.0.4, R Core Team (2021); www.R-project.org/.software) to select cases and controls.
We tested the case-control groups for significant differences in the matching features and in variables not used for matching, using the T test for continuous features and the chi-squared test for categorical variables. We used Kaplan-Meier survival curves to calculate the fat necrosis’s actuarial rates for cases (APBI patients) and controls (WBI patients). Univariate Cox proportional-hazards regression survival analysis was performed to detect possible associations between the primary endpoint and patient-specific or treatment-specific variables. Factors with p ⩽ 0.20 in the univariate analyses were included in the multivariate Cox regression. Of note, with a study cohort of 536 patients and 151 events of fat necrosis, we could detect Hazard Ratios (HRs) above 1.6 as statistically significant (p ⩽ 0.05, β = 0.80). We employed MedCalc v22.016 (MedCalc Software Ltd, Ostend, Belgium; https://www.medcalc.org; 2022) for all the statistical analysis. Statistical significance was set at a p-value of 0.05.
Results
Patients, tumour and treatment characteristics
Figure 2 depicts the Consort diagram, beginning with 1395 consecutive patients retrieved from the Institutional database and concluding with the inclusion of 736 patients in the matched case-control selection.
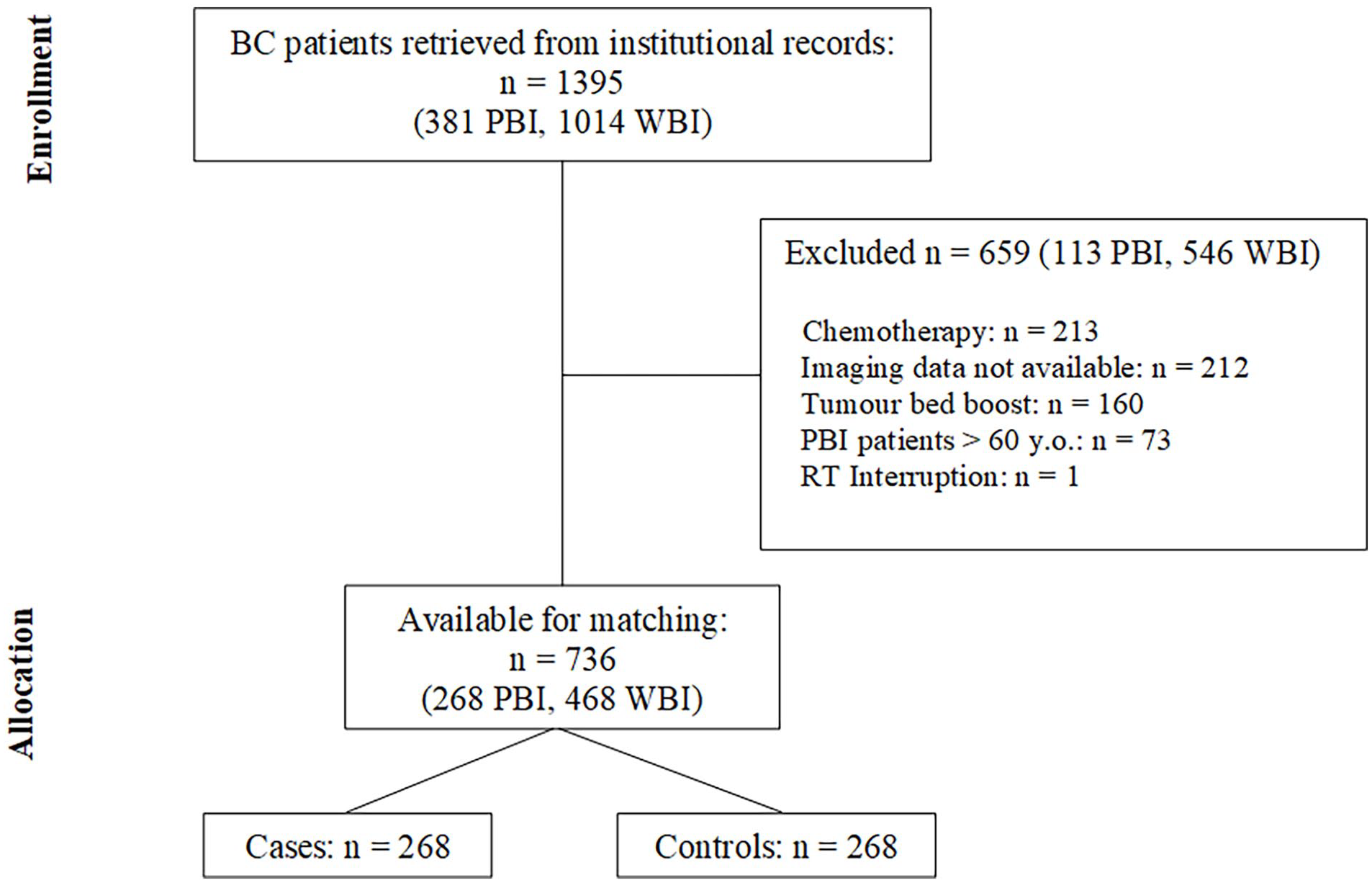
Consort diagram.
Matching resulted in 268 cases and 268 controls, with a median follow-up of 98 months (range: 45 – 160) for the WBI group and 35.5 months (range: 6 – 99) for APBI patients (p < 0.0001).
Patients’ characteristics are comprehensively detailed in Table 4. Within the APBI group the median age was 72 years (range: 60 – 87), in the WBI group it was also 72 years (range: 57 – 89). The median breast volume was 742.2 cc (range: 163.3 – 2835.2) for the APBI group and 669.6 cc (range: 167.8 – 2776.6) for the WBI group (p = 0.73). Nearly all patients (99.6%) had T1 stage tumours in both groups.
Patients’ characteristics.

APBI: accelerated partial breast irradiation; WBI: whole breast irradiation; RT: radiation therapy; VMAT: volumetric modulated arc therapy; AI: aromatase inhibitors; TAM: tamoxifen.
In the APBI group, the median PTV total volume was 135.1 cc (range: 32.7 – 445.6); conversely in the WBI arm the PTV was equal to the breast volume. Dose distribution was very homogeneous, with a median volume of the PTV receiving 95% of the dose of 99.9% (range: 94.7 – 100) and a median volume of the PTV receiving 105% of the dose of 0.14% (range: 0 – 8.75). Median overall treatment time (OTT) was five days (range: 5 – 9).
A total of 217 cases (80.9%) received endocrine therapy (191 aromatase inhibitors, 26 tamoxifen), comparable to 215 controls (80.5%) (189 aromatase inhibitors, 26 tamoxifen; p=0.99). No statistically significant differences were observed in matching characteristics (p-values: 0.31 for age, 0.35 for diabetes, and 1 for T-stage). However, hypertension was more prevalent in the APBI group, affecting 143 patients (53.3%), compared to 114 (42.7%) in the WBI group (p = 0.01).
Fat necrosis
Radiologically detected fat necrosis occurred in a total of 151 patients, with a median onset time of 17.5 months (range: 2 – 107), all developing inside the tumoral bed. Among these patients, the median age was 71 years (range: 57 – 86), the median breast volume was 769 cc (range: 219 – 2835), 18 (11.9%) had diabetes, and 81 (53.6%) had hypertension.
Seventy-eight were treated with APBI (crude rate 51.6%), compared to 73 (48.4%) in the WBI group. However, the median onset time of fat necrosis differed between the two groups: nine months (range: 2 – 67) for the APBI group versus 22 months (range: 4 – 107) for the WBI group. The three-year actuarial rate of fat necrosis was 32.8% (95% CI: 30.0% – 35.6%) for the APBI group and 22.3% (95% CI: 19.7% – 24.9%) for the WBI group (Kaplan-Meier curves in Figure 3).
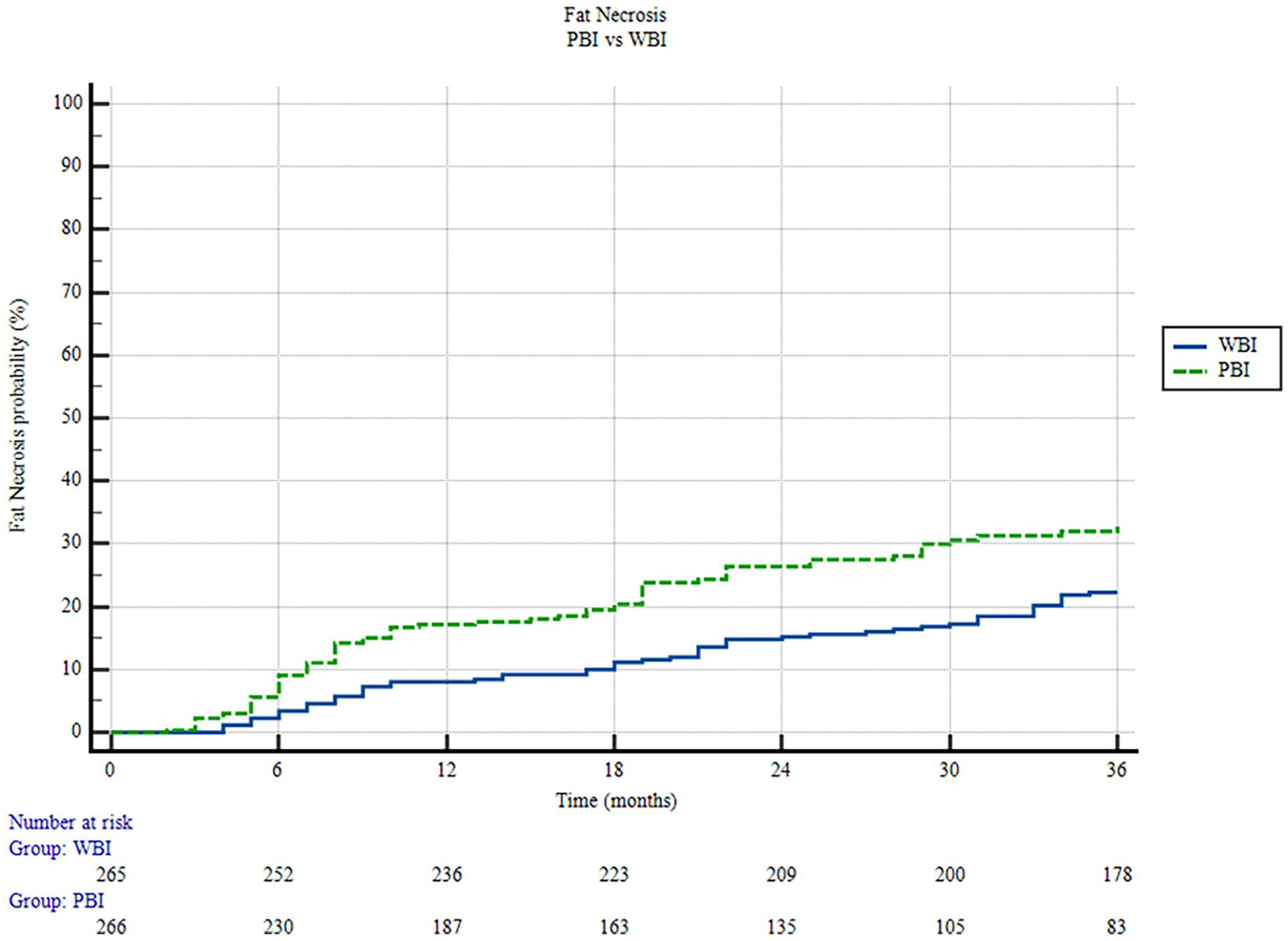
Kaplan Meyer for fat necrosis – comparison between APBI and WBI groups.
Univariate analysis
A statistically significant correlation was detected at Cox univariate analysis within the APBI group compared to WBI, with a HR of 1.6 (95% CI: 1.1 – 2.2; p = 0.006) for the fat necrosis rate. Hypertension (HR = 1.39, 95% CI: 1.01 – 1.92; p = 0.04) and total breast volume (HR = 1.0006, 95% CI: 1.0001 – 1.0011; p = 0.01) were both significantly associated with fat necrosis at univariate analysis, whereas diabetes emerged as a risk factor, although not statistically significant (HR = 3.3, p = 0.07). Age was the only variable identified as protective against fat necrosis, albeit not statistically significant (HR = 0.98 for a 1-year increase, p = 0.13).
Among patients receiving APBI, neither PTV volume nor dose distribution parameters showed a significant correlation with increased rates of fat necrosis.
Multivariate analysis
Specifically, for APBI, the multivariate analysis revealed a HR of 2.2 (95% CI: 1.2 – 4.0), demonstrating strong statistical significance (p = 0.01) compared to WBI.
Multivariate Cox proportional hazard regression identified significant associations between fat necrosis and diabetes (HR = 1.9, 95% CI: 1.1 – 3.3; p = 0.02). Conversely, older age appeared to be protective against fat necrosis (HR = 0.96 for a 1-year increase, 95% CI: 0.93 – 0.99; p = 0.005). The receiver operating characteristic curve indicated that an age of ⩽ 65 years may be associated with a higher incidence of fat necrosis; however, this was not statistically significant (p = 0.16).
Table 5 summarizes the correlation between fat necrosis and all variables, along with their respective p-values.
Univariate and multivariate Cox regressions for fat necrosis.

APBI: accelerated partial breast irradiation; AI: aromatase inhibitors.
Cosmetic outcomes
None of the cases of fat necrosis required surgical intervention. Cosmetic evaluation was available for 192 APBI patients (71.6%). Physicians rated 185 patients (96.3%) among them with excellent or good outcomes. Only six patients (4.4%) received fair scores, and one (0.3%) was rated as poor. We observed no significant association between fat necrosis and cosmetic outcomes worse than "good" (chi-squared test, p = 0.13).
Two hundred and five patients reported self-perceived cosmetic outcomes. One hundred eighty-two (88.7%) rated their outcomes as at least “good”. In the group with outcomes worse than “good”, poor cosmesis was reported in 12 cases, and fat necrosis was diagnosed in nine patients. We observed no significant association between fat necrosis and cosmetic outcomes worse than “good” (chi-squared test, p = 0.28).
Discussion
Partial breast irradiation has emerged as a viable treatment for early-stage breast cancer patients, demonstrating efficacy and safety comparable to WBI.13–15,25,26 However, uncertainties persist regarding the optimal schedule for this irradiation technique.13–15 Some trials proposed an hypofractionated (10 to 15 fx) regimen while others used an accelerated dose schedule with only 5 fractions.
At our institution, we adopted an APBI approach—delivering 30 Gy in 5 daily fractions—based on pilot study results 23 showcasing the safety and feasibility of this regimen. Nevertheless, literature indicates that while this schedule demonstrates acceptable acute and mid-term safety outcomes,17,27 it is associated with elevated fat necrosis rates. Colciago et al., 17 analyzing 344 patients treated with APBI with a median follow-up of 34 months, reported a 30.5% incidence of radiologically diagnosed fat necrosis.
Although this side effect is often mild and clinically non-relevant, it can lead to higher grades of fibrosis, breast pain, and worse cosmesis, impairing patients physically and psychologically. Additionally, fat necrosis could pose a challenge for differential diagnosis: distinguishing it from recurrent disease could be difficult both by clinical examination and imaging. 19
The pathophysiology of fat necrosis is largely unknown. In the very first report that described fat necrosis, as shown by Lee and Adair, Dr. James Ewing conducted pathological evaluation and noted endarteritis obliterans. 18 Notably, radiation therapy induces endarteritis obliterans, as it is a cardinal characteristic of radiation injury. This effect is defined by inflammation of the arteries and degeneration of their walls, leading to hypoxia, submucosal fibrosis, and necrosis. In 1987, Rostom and el-Sayed 28 were the first to suggest that radiation-induced endarteritis obliterans was a fundamental factor in fat necrosis. In 1988, Boyages et al. 29 hypothesized that the cause of ischemia leading to necrosis could be found in the association between an aberrant local reaction to surgical material, together with post-radiation endarteritis obliterans. Indeed, surgery alone could cause fat necrosis even without the adjuvant use of radiation therapy. Lumpectomy alone could lead to an incidence of this adverse event ranging from 1% to 9% of cases. 30 Moreover, oncoplastic surgery is even more associated with fat necrosis. Nakada et al. 31 analysed 1476 patients who underwent breast surgery. Among those who received a pedicled fat flap or a free dermal fat graft, the incidence of fat necrosis was 16% and 100%, respectively.
Unfortunately, our study lacks data on the influence of surgery on fat necrosis. We did not perform imaging between surgery and radiation therapy to assess this. Moreover, another limitation is that we do not have data about oncoplastic reconstruction methods. Nevertheless, in the APBI group, it is rare for patients to undergo reconstructive procedures, as these techniques would entail major distortion of the tumour bed. Conversely, our findings suggest a significant correlation between diabetes and fat necrosis, thus aligning with the microvascular injury theory, as hyperglycemia implies significant vessel damage. 32 It is difficult to explain how age demonstrated to be a protective factor. However, in the literature, age has a debated role in the onset of adverse effects. 33
About radiation therapy prescription in particular, literature examining fat necrosis incidence after APBI indicates a wide range of occurrences from 1% to 52%,20,30 combining radiological and clinical data. Studies employing brachytherapy report the highest rates of fat necrosis: 22.2%, 34 27%, 35 and 36.8%, 36 with median follow-ups of 84, 24, and 48 months, respectively. The reason for these elevated rates of adverse events may be attributed to the micro-damage caused by catheterization and the presence of excessive volume receiving more than 100% of the prescribed dose. 21
External beam PBI typically demonstrates lower fat necrosis rates, ranging from 3% to 25%, especially when volumetric delivery techniques are employed. 22 However, concerns arise when accelerated schedules are employed. Our findings confirm a higher incidence of fat necrosis with one-week APBI in both univariate and multivariate analyses compared to hypofractionated WBI. Korzets et al., 37 in a meta-analysis involving 14,514 patients across nine trials comparing PBI to WBI for early-stage breast cancer, found a higher association of fat necrosis with PBI: HR = 1.72 (95% CI: 1.21 – 2.43; p = 0.002).
Despite the higher incidence of radiologically diagnosed fat necrosis detected in our cohort of APBI patients, the clinical impact was minimal, as none of the patients required surgical intervention. Furthermore, cosmetic outcomes were comparable to existing APBI literature.10,38
Remarkably, the technique of irradiation delivery that we used cannot be held responsible, as we utilized VMAT, ensuring a more homogeneous dose distribution compared to the 3D technique employed for WBI. 39 Indeed, dosimetric parameters did not show any relation to the outcome. Not even the volume of the PTV showed an impact on adverse effects, contrary to what is shown in literature. 40 Moreover, the two cohorts (APBI and WBI) were well-balanced, except for hypertension, which did not prove significant in the onset of fat necrosis at multivariate analysis. Therefore, the increased incidence of fat necrosis in one-week APBI might be attributed to its biologically effective dose (BED) for late effects on fat tissue. Using the Fowler formula, 41 assuming an α/β = 3 Gy for late effects on fat tissue, the BED for one-week APBI amounts to 90 Gy versus 79.85 Gy for the WBI schedule utilized in this study. This difference of 10.15 Gy could further escalate if the time-adjusted BED (taBED) 45 is considered. The delivery of APBI in only five days (instead of 22 for WBI) results in a delta taBED of 22.3 Gy (difference between 87 Gy taBED for APBI and 64.7 Gy taBED for WBI).
In literature, higher doses are indeed associated with a higher incidence of fat necrosis. For example, Bourgier et al. 42 administered 40 Gy in 10 fractions twice daily (BED = 93.33 Gy, taBED = 90.33), resulting in a high and early fat necrosis incidence of 16% in 12 months. However, when reducing the dose schedule slightly, results are more optimistic. Chen et al. 43 and Hepel et al. 44 prescribed PBI with RAPID-like schedules (BED = 87 Gy; taBED = 84 Gy) to 60 and 94 patients with median follow-ups of 48 and 15 months, respectively, demonstrating fat necrosis incidences of 24.1% and 13%. Moving towards a reduction in BED, the ACCEL trial treated 219 women with 27 Gy in 5 fractions (BED = 75.6 Gy, taBED = 72.6 Gy vs. 90 Gy and 87 Gy of our schedule) daily fractions of APBI, reporting low rates of adverse events at the four-year mark. 45 However, specific data regarding fat necrosis were not provided. Lastly, Lövey et al. 20 treated 40 patients with external beam PBI of 50 Gy in 25 fractions (BED = 83.3; taBED = 63.5 Gy) and compared them to 130 conventional WBI patients. The use of a more diluted dose schedule resulted in only 17.7% fat necrosis over four years of follow-up and a significant reduction compared to WBI (p = 0.025).
Our study is not without limitations. Its retrospective nature introduces biases (selection, performance, measurement), limiting the inference of robust evidence. Moreover, one of the most apparent limitations of the study is the significant difference in follow-up duration between the two groups. This discrepancy arose because APBI was introduced as a new irradiation technique more recently, and most patients underwent treatment in the last few years. A subset of patients in the WBI group lacked retrievable breast volume data, impacting the selection of this variable for matching, despite literature indicating breast volume as a potential factor for higher side-effects rates.40,46 Additionally, the WBI group lacked cosmetic evaluations, resulting in no comparison between cases and controls regarding cosmetic outcomes. Despite these limitations, our analysis is the first to compare fat necrosis outcomes between hypofractionated WBI and 5-fraction external beam APBI directly, using a case-control matching approach.
Conclusions
Despite efforts to reduce volumes and improve dose homogeneity in APBI, the incidence of radiologically diagnosed fat necrosis was higher than in WBI. However, this side effect had minimal clinical and cosmetic implications for patients. Further investigations are needed to identify a lower-dose schedule with comparable efficacy to 30 Gy in 5 daily fractions but fewer toxicities, especially for patients with risk factors for developing fat necrosis.
Footnotes
Author Contributions
RRC conceptualization, data curation, formal analysis and writing original draft; ELR conceptualization, data curation, formal analysis; CG, MGC, GVB, IM, CD, SM, AC, EP and LL investigation; TR conceptualization, data curation, formal analysis; MCDS conceptualization, supervision and validation. Each author contributed to the reviewing process of the original article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
The study was approved by the IRB of the participating centre.
Informed consent was obtained from all individual participants involved in the study.
