Abstract
Introduction:
Individuals with Familial Adenomatous Polyposis (FAP) or APC-associated polyposis, an autosomal dominant inherited condition, develop multiple adenomatous polyps and have an increased colorectal cancer (CRC) risk. A change in diet can help reduce cancer risk, and several dietary components have an antitumor effect. We aimed to evaluate the potential of the anti-inflammatory and anticancer substances quercetin (QER), epigallocatechin gallate (EGG) and fisetin (FIS) in decreasing the risk of CRC by reducing the growth of polyps in an organoid model.
Methods:
Patient-derived organoid (PDO) lines were generated from polyps obtained from patients with FAP undergoing prophylactic colectomy. PDOs were treated with QER, EGG, or FIS to determine their effect on cell growth. Changes in caspase 3/7 activity and expression of inflammation and apoptosis mediators were assessed by luminescent and colorimetric assays.
Results:
Three PDO lines with different inactivating pathogenic variants in the APC gene were developed using a combinatorial approach. FIS was the most active of the three substances tested, presenting the lowest IC50 in all PDO lines (range: 42.6-9.2 uM). The IC50 was defined as the concentration required to halve the number of cells after 72 hours. All molecules tested induced apoptosis through activation of caspases 3/7.
Conclusions:
QER, EGG, and FIS can be easily taken from foods or dietary supplements, show toxicity on PDOs derived from adenomatous polyps, while they are known to be harmless on normal cells. Diets enriched with these substances could be potential supplemental treatments to reduce the risk of CRC in individuals with FAP.
Introduction
Familial Adenomatous Polyposis (FAP) or APC-associated polyposis, 1 is a hereditary autosomal dominant condition caused by germline pathogenic variants in the Adenomatous Polyposis Coli gene (APC). Individuals with FAP develop multiple adenomatous polyps in the large bowel that, if not treated, progress to colorectal cancer (CRC). 2 Highly invasive prophylactic surgery, such as proctocolectomy with ileal pouch anal anastomosis (PC/IPAA) or total colectomy with ileorectal anastomosis (TC/IRA), is currently the only primary prevention option to reduce the occurrence of CRC in subjects with FAP. 3 After surgery, life-long endoscopic surveillance is mandatory to limit the risk of cancer in the transition zone (for IPAA), or rectal stump (for IRA).2,4
The risk of developing CRC in subjects with FAP is strongly related to the genotype, 4 and chronic intestinal inflammation and the local microenvironment may influence the formation and growth of intestinal adenomas and their progression to cancer. 5 Diet influences intestinal and systemic inflammation, and affects the risk of adenoma/colorectal cancer onset. 6 Indeed, the use of different diets and dietary components for cancer prevention and treatment has been proposed.7-9 Diet is also a major concern for subjects with FAP, especially after prophylactic colectomy, as they suffer from many bowel disorders. 10 Still, there are no specific dietary treatments or preventive recommendations for subjects with FAP. In a non-randomized pilot study on subjects with FAP, we demonstrated that a three-month dietary intervention based on principles and recipes of the Mediterranean diet is effective in improving markers of intestinal (fecal calprotectin) and systemic (serum calprotectin and neutrophil-lymphocyte ratio) inflammation, and in regulating the expression of miRNAs and genes with inflammatory, oncogenic or tumour-suppressor activities.11-13 Bioactive molecules found in food may exert an anticancer or anti-inflammatory effect by interacting directly with cancer or tumor-associated cells, but also by influencing the tumour immune microenvironment or microbiota composition.14,15
Flavonoids are well-known plant derivatives, many of which have therapeutical activity against cancer and inflammatory disorders.16,17 Among them, Quercetin (QER), Epigallocatechin gallate (EGG) and Fisetin (FIS) can be found in foods, and have been shown to be promising antitumoral chemopreventive agents while being considered non-toxic for healthy cells.18-20
We aimed to determine whether QER, EGG and FIS can contribute to preventing the progression of adenomatous polyps to CRC in subjects with FAP. For this, we evaluated their antitumor activity on patient-derived organoids (PDO) obtained from adenomatous polyps of colectomized or proctectomized individuals with FAP. PDOs are self-organized three-dimensional tissue cultures derived from cancer stem cells. PDOs retain the genetic and phenotypic characteristics of the original tissue and can be used as in vitro surrogates for anticancer drug testing. 21
Methods
Development and characterization of FAP-derived PDOs
The biological samples were collected in adherence with the guidelines of the IRCCS Foundation, Istituto Nazionale dei Tumori di Milano, review board protocol (INT 103/19, INT 90/21). The study complied with the Declaration of Helsinki and was approved by the Ethics Committee of the Fondazione IRCCS Istituto Nazionale dei Tumori of Milan, Italy. Written informed consent from each patient was obtained. PDO lines were generated from adenomatous polyps obtained from patients with FAP undergoing colectomy or proctectomy (FAP-PDO; Table 1), and maintained by following the basic protocols described by Pleguezuelos-Manzano et al. with few modifications. 22 Briefly, three to four polyps were cut into pieces around 1 mm in volume, incubated with 10mL of DMEM-F12 (Merck) supplemented with Normocure (Invivogen) for 20 minutes, and washed three times with PBS supplemented with Normocure and the Rho-kinase inhibitor Y-27632 (MedChem Express). Cells were then separated from the tissue by digesting the sample with collagenase type II (Sigma-Aldrich), and washed three more times with PBS supplemented with Normocure and Y-27632. Cells were seeded on Matrigel (Corning) and grown in a cell growth factor-containing culture medium (FAP-PDO basal culture medium, detailed in Online Supplementary Table 1) as described in previous works.23,24
Germline pathogenic variants and clinical characteristics of the FAP patients.
FAP-PDOs were characterized by immunohistochemistry. Briefly, FAP-PDOs in exponential growth phase were recovered and fixed in 4% buffered formaldehyde for 30 minutes. After fixation FAP-PDOs were resuspended in Bio-Agar, processed and paraffin embedded. 23 Immunophenotype characterization was performed on 3 µm sections. Antigen retrieval was performed on a Dako PT-Link pre-treatment module according to the manufacturer instructions (Dako, Agilent Technologies). Immunostaining was performed on a Dako Autostainer link 48 (Dako, Agilent Technologies) using the following primary antibodies and relative dilutions: anti-CK AE1/AE3 (Dako) 1:100, anti-CK20 (Dako) 1:500, anti-CK19 (Sigma Aldrich) 1:1000, anti-CDX2 (Dako) 1:50, anti-Mib1 (Dako) 1:400, anti-LGR5 (Origene) 1:200. The antibody binding was detected by deaminobenzidine chromogen reaction and counterstained with Harris' Hematoxylin. Concordance of FAP-PDOs with their tumor of origin was evaluated as described in Varinelli et al., 2024. 24
FAP-PDOs treatment with anti-inflammatory substances
Three lines of organoids derived from polyps from FAP patients, FAP-PDO4, FAP-PDO5 and FAP-PDO6, were used to evaluate the flavonoids bioactivity. Table 1 summarized the characteristics of the patients from whom they were derived. QER, EGG (Sigma Aldrich, codes Q4951 and PHR1333) and FIS (ThermoFisher Scientific, code 119150010), were diluted in DMSO (QER, FIS) or water (EGG) to prepare 10mM stock solutions. For the viability assay, 6x103 PDOs were suspended in culture medium and seeded in 96-well plates (100 µl/well, Costar 3904; Corning, USA) previously coated with 40 µl of Matrigel (~ 60 PDO/well). PDOs were then incubated at 37C for 24h in normal conditions, and afterwards incubated for 72h with FAP-PDO basal culture medium supplemented with QER, EGG and FIS at concentrations ranging from 12 to 200uM.
Analysis of the effects of the anti-inflammatory substances
Cell viability was determined with CellTiterGlo 3D Cell Viability Assay kit (Promega, USA), following the provider instructions. To determine the mechanisms of cytotoxicity, PDOs were seeded and incubated for 24h under normal conditions, then incubated for 24, 48 or 72h with QER, EGG and FIS at the corresponding IC50, defined as the concentration needed to halve cell viability after 72h compared to untreated controls. Caspase 3/7 activation, cell viability and cytotoxicity were measured on a TECAN spark microplate reader (Tecan Trading AG, Switzerland), using the ApoTox-Glo Triplex Assay (Promega) according to the manufacturer’s instructions.
To confirm Caspase 3/7 activation and evaluate the increase in Caspase 3/7 activity, PDOs were incubated with FAP-PDO basal culture medium supplemented with QER, EGG and FIS at their corresponding IC50, recovered after 48h with Cell recovery solution (Corning), and washed with PBS to eliminate Matrigel residues. Recovered cells were lysated and the caspase activity measured using Caspase 3/7 Activity Assay kit (Elabscience), by following the manufacturer instructions. Caspase activity was adjusted according to the protein content of each sample, which was determined with a Bradford assay. All colorimetric determinations were performed on a on a TECAN spark microplate reader (Tecan Trading AG, Switzerland). All data were normalized to the mean of the untreated group (PDO grown in FAP-PDO basal culture medium).
Statistical analysis
All experiments and analyses were performed in triplicate. Statistical differences in caspase activity between treatment and control groups were assessed using an unpaired two-tailed t-test, calculated using GraphPad Prism 9.5.1 (Online Supplementary Table 2). Values below 0.05 were considered statistically significant.
Results
Development of FAP-derived organoids
We generated six PDO lines (FAP-PDO1-6) from adenomatous polyps obtained from patients with FAP undergoing colectomy or proctectomy, following established protocols, as detailed in the Materials and Methods section. 22 Characteristics of the patients and the germline pathogenic variant associated to their diagnosis are reported in Table 1.
FAP-PDOs presented rapid growth, reaching confluence in four to five days of culture after splitting. FAP-derived organoids retained the main characteristics of their tissues of origin, expressing colorectal-specific markers in the same percentage of cells as the tissue from which they originated. Specifically: Cytokeratins (CK) AE1/AE3, which identify glandular/epithelial differentiation of cells, CK19 and CK20, which are specific to the large intestinal tract; and Caudal Type Homeobox 2 (CDX2), which is specific to intestinal tract cells located downstream of the diaphragm area (Figure 1). FAP-PDO1 and 6 lines exhibited glandular structures lined by pseudo-stratified columnar cells, frequently observed in colonic epithelium, with central fibrinoid core and dirty necrosis along with roundish solid aggregates of unaligned epithelial cells. These features were also partially present in FAP-PDO 3 and 5 lines, in which the glandular structures showed a more complex necrosis structure. FAP-PDO3, 4, 5 and 6 lines showed high positivity to all CKs and other markers. According to the observed rapid growth, all PDOs had a very high Ki67 staining (Mib1). Staining for LGR5, which is a strongly recognized marker of colorectal stem cells, showed different patterns among the PDO cultures: FAP-PDO1, 4, 5 and 6 displayed diffuse immunoreactivity in most cells, while FAP-PDO2 and 3 showed less widespread LGR5 immunoreactivity, corresponding to a smaller component of stem cells within them.
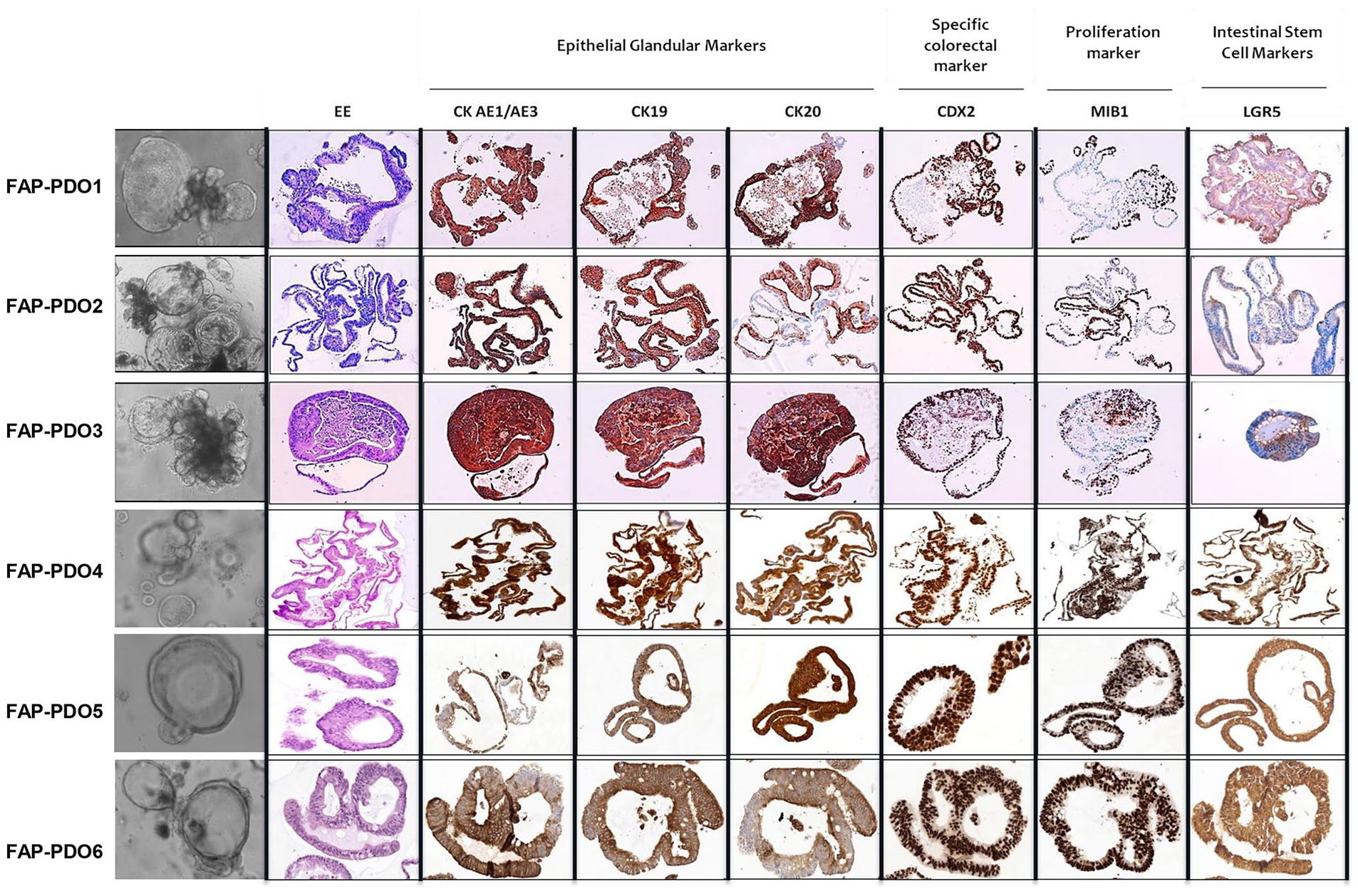
Immunophenotypical characterization of FAP-PDOs. Immunohistochemical staining of formalin-fixed paraffin-embedded FAP-PDO sections evidencing the presence of glandular epithelial (CK AE1/AE3, CK19, CK20), colorectal-specific (CDX2), proliferation (Mib-1) and stem cells (LGR5) markers.
The genetic characterization of three of them (FAP-PDO1, 2 and 3) confirmed that PDOs retained the APC pathogenic variant of the tumor of origin. In the case of FAP1, a second truncating pathogenic variant in APC was found (Table 2).
Genetic characterization of FAP-PDO1, FAP-PDO2 and FAP-PDO3. APC pathogenic variant and protein alterations in the FAP-PDOs and their tissue of origin.
The bioactive molecules induce apoptosis in FAP-PDOs
The FAP-PDO4, FAP-PDO5 and FAP-PDO6 lines were treated with the bioactive molecules QER, EGG, and FIS, three flavonoids that were chosen for testing as they are found in food, have shown to exhibit antitumor activity at relatively low doses (less than 100 uM) on tumor cell cultures,18-20 and have demonstrated activity when administered orally.25-27
Dose-response curves indicate that the three flavonoids can decrease the viability of the three FAP-PDO lines after 72 h of treatment (Figure 2A). FIS presented the lowest IC50 values, 34.7, 42.6 and 9.2 uM for lines FAP-PDO4, FAP-PDO5 and FAP-PDO6 respectively (Table 3). While EGG presented IC50 values of 37.6, 114.4 and 29.5 uM, and QER of 47.9, 58.6 and 19.9 uM for lines FAP-PDO4, FAP-PDO5 and FAP-PDO6 respectively (Table 3). Changes in caspase 3/7 activity (Figure 2B and C), and in cell viability and cytotoxicity over time (Online Supplementary Figure 1) were similar in all PDO lines when treated with all three substances at their corresponding IC50 values. At these concentrations, the three substances induced the apoptotic death in FAP-PDO, as indicated by the increase in the levels of cytotoxicity and caspase 3/7 activity, and the decrease in cell viability.
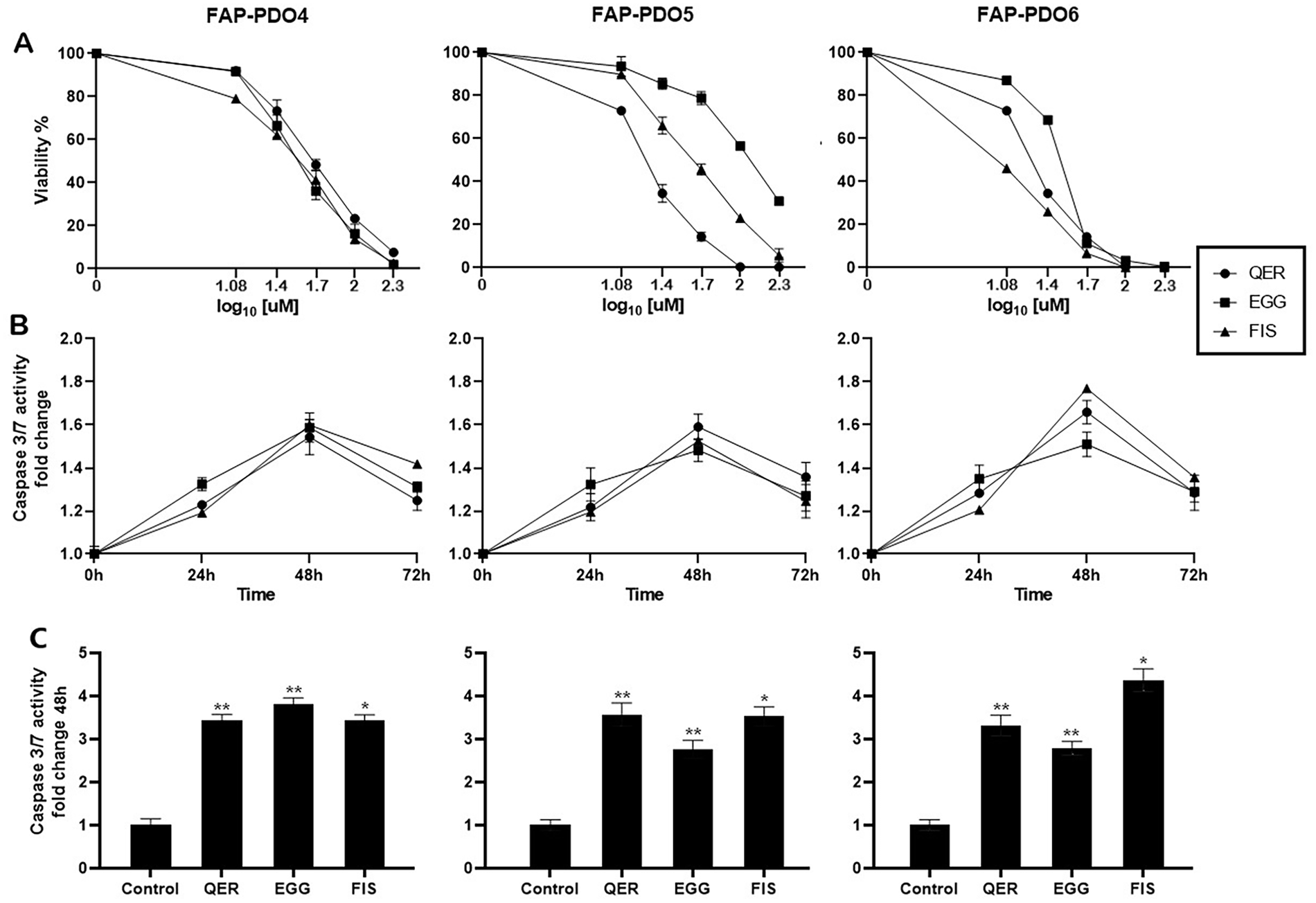
Proapoptotic effect of flavonoids Quercetin (QER), Epigallocatechin gallate (EGG) and Fisetin (FIS) on FAP-PDO. A) Viability measured after 72h of treatment. Concentrations of QER, EGG and FIS are represented as log10 of 0, 12, 25, 50, 100 and 200uM. B) Fold change of caspase 3/7 activity, after treatment with QER, EGG and FIS at the corresponding IC50 for 24, 48 and 72h, measured ApoTox-Glo Triplex Assay (Promega). Error bars shorter than the symbol are not shown. C) Fold change in Caspase 3/7 activity after 48h treatment with QER, EGG and FIS at their corresponding IC50, measured using Caspase 3/7 Activity Assay kit (Elabscience). Statistically significant differences when comparing treated FAP-PDO with untreated controls are indicated with ** = p<0.01, * = p<0.05.
IC50, defined as the concentration needed to halve cell viability after 72h of treatment compared to untreated FAP-PDOs. All values in uM.

Discussion
Subjects with FAP present a number of digestive symptoms after colectomy that directly affects their quality of life. This, coupled with their increased risk of developing CRC, which remains even after prophylactic colectomy or proctocolectomy, leads them to frequently ask their physicians for dietary recommendations. Unfortunately, no food or diet recommendations are available for their condition. We previously demonstrated that an intervention with a low-inflammation diet can reduce inflammatory markers and induce changes in miRNAs and genes expression that may help hinder tumor development.11,12 Here, we demonstrate that food-derived flavonoids whose consumption was enriched in the same low-inflammatory diet, such as QER, EGG and FIS, interfere with the growth of PDO derived from adenomatous polyps of subjects with FAP. Also, all three substances induce apoptosis when treating the FAP-PDO with the corresponding IC50.
QER, EGG and FIS are generally regarded as non-toxic for normal cells. They are found in high concentrations in some easily accessible foods such as onions, green bell peppers and snap beans (QER), green tea (EGG), and strawberries, apples and persimmon (FIS).19,28 All these products were included in the low-inflammatory dietary intervention we conducted, that reduced markers of systemic inflammation and helped improve quality of life markers of colectomized subjects with FAP.11,12 Our results suggest that diets which include foods rich in QER, EGG and FIS may help hinder the development of adenomatous polyps cells in colectomized FAP patients.
FIS presented the lowest IC50 values for all lines, even falling below 10uM for FAP-PDO6. However, the intestinal mucosal concentrations of the three substances obtained from the low-inflammation dietary intervention were probably lower than the levels that showed efficacy in inducing apoptosis in vitro, due to the low abundance of these flavonoids in most foods. Nevertheless, given their safety, QER, EGG and FIS are available as freely-accessible dietary supplements that could be used to reach concentrations similar to those tested.
Also, foods containing QER, EGG and FIS were consumed together in our dietary intervention and colon tissue may have been exposed to combinations of the three substances, and also to other bioactive compounds. Considering that the three molecules have been reported to have different molecular targets that mediate their activity,29-31 it would be interesting to evaluate combinations of QER, EGG, FIS and other compounds with anti-inflammatory or antitumoral activity, in concentrations attainable through diet and over a prolonged period of time, in order to reproduce the conditions generated in the gut by a diet. This would help us evaluate the preventive potential of the bioactive compounds and of the diet as a whole, in subjects with FAP. A limitation of the study is that the FAP-PDO lines analysed were generated from patients that did not participate in the dietary intervention. In fact, the FAP-PDO lines were generated from tissue obtained during colectomy, and one of the inclusion criteria for subjects recruited for the dietary intervention was that they had undergone prophylactic PC/IPAA or TC/IRA prior to the start of the study. 13 Still, considering their security, availability and activity, the three substances tested may have great potential as part of a dietary-focused therapeutic or preventive approach for subjects with FAP. Further studies will evaluate combinations of different bioactive compounds, also for prolonged times, and focus on the comprehension of the molecular mechanisms modulating their pro-apoptotic activity and their ability to target the stem cell component of PDO. This will enable us to evaluate their possible application on other inflammatory conditions and diseases, complementary to the standard therapy.
Supplemental Material
sj-pdf-1-tmj-10.1177_03008916241291301 – Supplemental material for Effect of anti-inflammatory molecules from food on organoids derived from adenomatous polyps of FAP subjects
Supplemental material, sj-pdf-1-tmj-10.1177_03008916241291301 for Effect of anti-inflammatory molecules from food on organoids derived from adenomatous polyps of FAP subjects by Oscar Illescas, Antonino Belfiore, Luca Varinelli, Davide Battistessa, Susanna Zanutto, Clorinda Brignola, Francesco Segrado, Irene Cafferati, Maria Teresa Ricci, Giovanna Sabella, Massimo Milione, Vito Ladisa, Stefano Signoroni, Marco Vitellaro, Patrizia Pasanisi and Manuela Gariboldi in Tumori Journal
Footnotes
Acknowledgements
The authors wish to thank all subjects that participated to the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Ministry of Health Ricerca Corrente and 5xmille funds for healthcare research. MG is funded by the European Union's Horizon 2020 research and innovation program, grant agreement No 825410 (ONCOBIOME project). OI is recipient of the Fondazione Umberto Veronesi (FUV) Post-doctoral fellowship from 2021 to 2024.
Ethical considerations
Biological samples were collected in adherence with the IRCCS Foundation Istituto Nazionale dei Tumori di Milano guidelines, review board protocol (INT 103/19, INT 90/21). The study complied with the Declaration of Helsinki and was approved by the Ethics Committee of the IRCCS Foundation Istituto Nazionale dei Tumori of Milan, Italy.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
