Abstract
The use of Digital Healthcare Products is leading to significant improvements in clinical practice. Herein, we discuss the development of PROACT 2.0 (Patient Reported Opinions About Clinical Tolerability v2.0), a novel open-source mobile and web application developed at Fondazione IRCCS Istituto Nazionale Tumori in Milan. It was developed in collaboration with The Christie, Manchester, in the context of work package 2 of the UpSMART Accelerator project, involving a consortium of referral cancer centers from the UK, Spain and Italy. PROACT 2.0 enhances communication between patients and healthcare providers in cancer clinical trials, allowing patients to report adverse events and side effects, and healthcare teams to collect valuable patient-reported outcome measures for treatment management. PROACT 2.0 supports text, audio, and video messaging, offering a secure, non-urgent communication channel that integrates with, or replaces, traditional methods. Its user-friendly and multilingual interface provides a new route for patient engagement and streamlines the handling of logistical information. Positive feedback from initial testing warrants future enhancements for broader applicability in cancer research and treatment.
Introduction
Digital Healthcare Products (DHPs) offer the potential for rapid and spontaneous reporting of symptoms, facilitating remote health monitoring and communication between patients and healthcare providers (HCPs). In the clinical research field DHPs are considered valid tools for improving daily communication between patients and their medical team both for clinical care and for logistical information, which may facilitate patients’ participation in clinical trials.1,2 In particular, since the main purpose of early-phase clinical trials is to evaluate the safety and tolerability of a new drug, DHPs can be used for the efficient and real-time collection of adverse events (AEs), in addition to monitoring the patient experience during the execution of the trial. 3 Nowadays, the digitization of clinical trials is considered by National Institutes of Health (NIH) as an important tool to accelerate the pace at which evidence is generated by clinical trials. 4
DHPs may therefore enhance patients’ engagement in clinical trial participation through the use of mobile apps, aimed at facilitating the communication with HCPs, improving patients’ awareness and involvement in clinical research studies, reporting adverse events and side effects to the medical team through a prompt, easy-to-use communication route. Indeed, it is now commonly accepted that Patient Reported Outcome Measures (PROMs) represent essential information in the management of oncology patients and development of new treatment approaches.5-10
The development of the mobile app PROACT 2.0 at the Fondazione IRCCS Istituto Nazionale Tumori in Milan (INT) represents, to our knowledge, the first time in which a digital tool for patient-to-doctor communication in clinical trial presents all the features of being fully realized in the context of the Italian National Healthcare System, released in open-source format and under public license, and supported by public resources.
PROACT2.0 was a redevelopment project of PROACT, originally developed by AstraZeneca, subcontracted to Cancer Research UK, Manchester Institute, moving over to the UpSMART Consortium in 2020. The UpSMART consortium, a collaboration among the UK, Spain, and Italy, comprises 23 participating centers that include Experimental Cancer Medicine Centres in the UK and Early Drug Development Units in Spain and Italy, collectively referred to as Experimental Cancer Centers. This network of European centers aims to enhance their digital capabilities through collaboration and expertise sharing, focusing on improving data acquisition and analysis to ultimately benefit patients. The consortium's funding comes from an Accelerator Award, a joint effort of Cancer Research UK (CRUK), the Italian Association for Cancer Research (Associazione Italiana per la Ricerca Contro il Cancro, AIRC), and the Spanish Association Against Cancer's Scientific Foundation (Fundación Científica de la Asociación Española Contra el Cáncer, AECC).
In 2015, the original PROACT App, in a beta-version format that is no longer available, was tested via smart phone or tablet by 16 patients at a drug development unit in London. 11 The app was used for sending audio or video messages, available in real-time to the clinical team but analyzed after a 32-day delay by a research scientist: the analysis focused on identifying key words and topics, to categorize how patients felt during the trial treatment. Although some limitations were reported (particularly concerning the low number of patients recruited, the early version of the application trialed, and the low uptake rate due to users’ digital divide), the outputs of the pilot study showed PROACT to be a valid approach for engaging and empowering patients in early oncology trials. Furthermore, it allowed the identification of several areas of improvement and the possibility for considering additional functionalities.
On moving into UpSMART, several areas of improvement were identified and in 2020, the app was further developed as PROACT2.0 within the UpSMART Accelerator Project and INT was given a primary role in the app’s development. In 2021, a team from the Medical Oncology Department at INT globally redefined the architecture, the functions, and the interface, creating a new version of the app named PROACT 2.0. This new tool was developed with the primary aim of improving communication between patients enrolled on clinical trials and doctors, nurses and researchers. Herein, we aimed at describing the main features and future perspective of the usage of PROACT 2.0 in the context of national and international cancer referral centers.
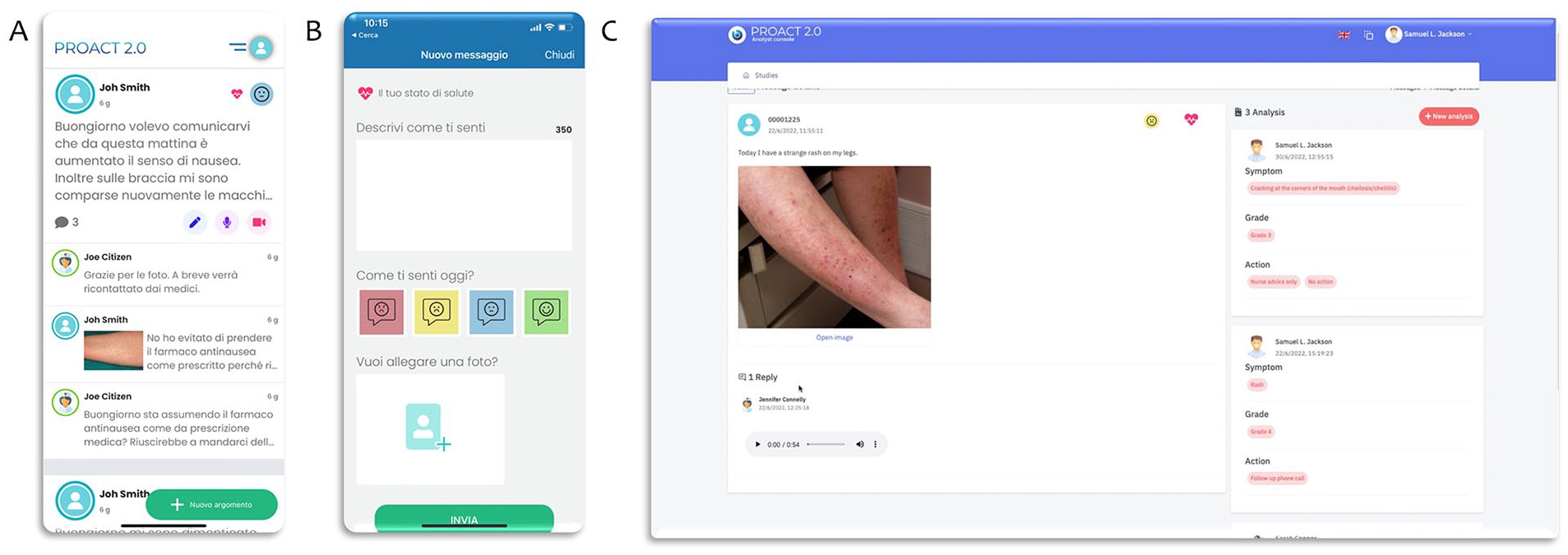
PROACT 2.0 is a mobile phone and web application, provided with a simplified authentication system, which provides an additional way of securely communicating between patients and HCPs. The application was originally aimed at supporting patients, doctors, nurses, and researchers involved in oncological studies for early-access programs for drugs, according to the main purpose of the UpSMART project that was focused on digitalization of early-phase clinical trials. The app provides access to an intuitive platform and enables non-urgent information exchange in a secure and organized manner through text, audio, and video messages. Thus, it simplifies the communication of adverse events and side effects to treatments thanks to additional tools, with respect to standard communication channels. Furthermore, PROACT 2.0 allows the submission of questionnaires and validated surveys, which can be sent by the medical team, in order to collect data and conduct analyses (by means of a system named Analyst Console) for research purposes at set timepoints.
Overview of app features
Figure 1
summarizes the overall study structure and the connection between all the stakeholders involved in the clinical study. Currently PROACT 2.0 can be considered as a platform composed of: (i) user interfaces, both for the mobile app (available for iOS and Android,
Figure 2A
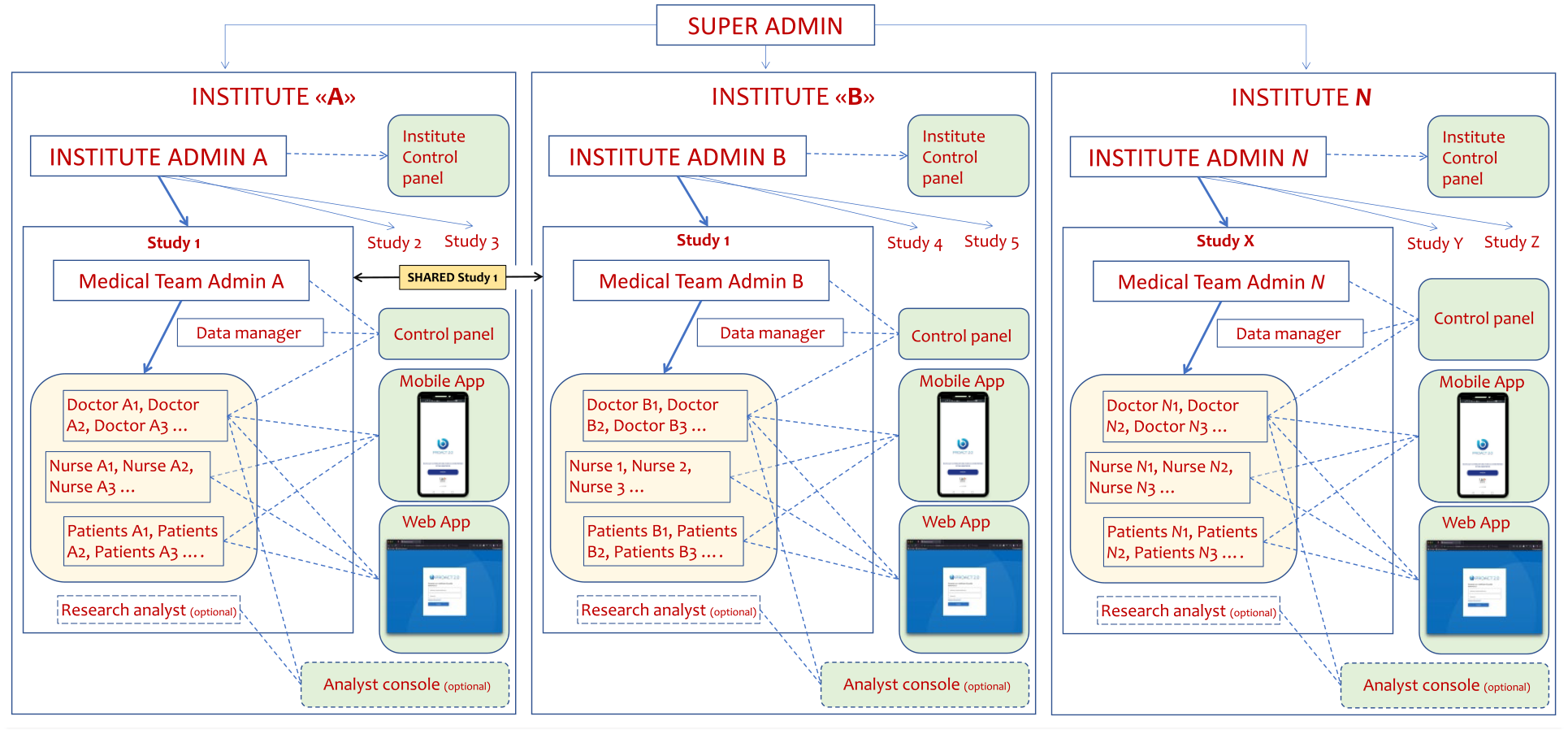
and
PROACT 2.0 functional architecture in the context of clinical studies. The architecture describes the parallel use of PROACT 2.0 in different institutions, among which studies can be shared when necessary (exemplarly, Study 1).
Briefly, to gain access to the system, patients, doctors, nurses and researchers need to be registered by a Medical Team Administrator (MTA) onto the platform. The MTA is created by an Institute Administrator (IA), who is likely elected by the projet team and is in charge of managing all the technical aspects of the studies. The app supports simultaneous instances in different institutions, different studies, and different medical teams. Importantly, studies can be shared, if needed, among different institutions, each one retaining the ownership and the reserved access to proprietary data (unless differently agreed by the involved subjects).
In detail:
The IA account is responsible both for the general settings of the PROACT 2.0 platform in the Research Institute and for the activation of each clinical study. The IA enters the users starting with the MTA. Depending on the specific requirements of any Team, the system allows the IA to customize the functions of the mobile/web app for each study, such as enabling/disabling the messaging functions, enabling/disabling the Analyst Console, etc.
The MTA can customize the features of PROACT 2.0 in relation to each study and in relation to the needs of the medical team associated with a group of patients. The MTA Control Panel allows the customization of PROACT 2.0 in several aspects, e.g.: (i) including a description of the protocol in pdf format and the specific contacts (e.g. telephone numbers, e-mail, helpdesk, etc.) for each medical-nursing team; (ii) setting a text that can be sent to patients immediately after the account creation/suspension/deactivation; (iii) registering doctors, nurses, patients, researchers, data managers; and (iv) compiling, organizing and sending questionnaires.
The Doctor users are members of the medical team who, through the control panel (a simplified version of the MTA control panel), have the option to create and send additional questionnaires, as well as view and export individual and aggregated data related to the answered surveys when allowed by the study.
The Analyst Console (AC) can be accessed by the Researcher, Doctor and Nurse users, i.e. those who have the option of reading and viewing text, audio and video messages between the medical team and patients in the context of each individual study for research purposes. The Researcher user is allowed to access data visualization but does not participate in the study. The AC is optionally enabled by the IA for each study.
PROACT 2.0 is not intended for emergency communication that requires an urgent response and in those circumstances patients are referred to country-specific emergency numbers (this is clearly highlighted to the patients both in user manual and at the time of first login).
PROACT 2.0 can efficiently integrate or completely replace traditional contact channels (such as emails and phone calls) in the routine of non-urgent communication between patients and their medical team. The application is available in six different languages, namely Italian, English, German, French, Spanish and Dutch.
Technical overview
PROACT 2.0 was designed in .Net6 Framework and with C# Language. Specifically, .Net6 Framework has been used for Web API Services; while Xamarine Framework, JavaScript and SCSS Forms for the mobile application. In addition, the web app was developed in NetMVC with C# Language and the Control Panel Interface in React.js. Finally, the Application Programming Interface (API) was designed in .Net6 Framework. PROACT 2.0 is hosted in a secure cloud environment, provided by MS Azure, located in Europe and owned by INT, which has bought the license until February 2025.
The PROACT 2.0 system does not store users' information, that could make them identifiable beyond those voluntarily contributed by the patient and health care providers themselves (e.g., in the form of the text/audio/video messages or profile pictures).
Additional details about the app architecture, the installation and the usage procedures and the maintenance are provided by the authors upon request. Public documentation is available at https://proact.gitbook.io/introduction/. The full code is available at the public repository https://github.com/Proact2 under Mozilla Public License 2.0.
The advantages in the context of clinical studies include: security, portability, user friendly set-up and usage.
Both the app and web systems are browsable by any mobile device or via the web under protected access, granted by the healthcare providers involved in the project at the time of admittance of the patient to a clinical study. User authentication and data storage in MS Azure databases are secured by encryption.
After the acquisition of informed consent, the medical team administrator will assign a unique identifier code (ID) to the patient. The administrator will access a secured web-based platform (PROACT 2.0 dashboard) where the patient will be registered using their name and surname's initials, an identification code and an email address provided by the subject. Upon activation of the account, patients will receive a temporary password from the medical team administrator and an e-mail invitation to download the app on their mobile. The first log in will require inserting the email address and a temporary password, which the users will be invited to change promptly with a self-chosen one.
The medical team administrator will also register the accounts of physicians and research nurses participating in the selected study into the system . All those accounts will be activated using a temporary password as previously explained.
State of the art
In 2023, PROACT 2.0 was tested in a feasibility study with 15 patients, enrolled in a phase I clinical trial for the treatment of advanced solid tumors at the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan. The study aimed to assess the app’s functionality and patient’s compliance in a limited cohort of cases. Patients showed a high level of satisfaction in using the app, appreciating the ways offered to communicate with the medical team and the timeliness in receiving feedback on their health status.
In addition, the medical team stated that the use of PROACT 2.0 facilitated the management of logistical information, and they appreciated the immediacy and effectiveness of communication with enrolled patients.
In 2024, PROACT 2.0 will be used in a multicentric trial in the context of the project CCE-DART, funded by Horizon 2020, which will involve referral cancer centers from UK (Cambridge University Hospital), Spain (Vall d’Hebron Institute of Oncology, Barcelona), France (Gustave Roussy Institute, Paris), Germany (National Center for Tumor Diseases, DKFZ, Heidelberg) and the Netherlands (Netherlands Cancer Institute, Amsterdam) inside the Cancer Core Europe (CCE) Consortium.
In addition to enabling future portability and sustainable maintenance of the app, current efforts are aimed at re-coding the app to convert it into a Progressive Web App (PWA) structure, which will allow it to gain independence from android and app store repositories. Primary release, with additional functionalities to improve the management and the quality of care of the patients enrolled, are expected within Q2 in 2024 (no interruptions in the app services are foreseen). Future development plans include conditional branching in surveys design; an electronic dosing diary; improved push-notifications (i.e. the introduction of notification prioritization system by text recognition methods); and improved data export through data aggregation and development of visualization functionality for research purposes.
For these last aims, INT is taking advantage of the experience of a software company for PWA development, warranting maintenance and help desk services for stakeholders. In particular, the app maintenance will be guaranteed by this software company until the end of the CCE-DART project.
Conclusion
DHPs, exemplified by PROACT 2.0, have huge potential in early-phase clinical trials and beyond, enabling prompt and efficient reporting of adverse events and enhancing patient-clinician communication, significantly contributing to the optimization of trial processes and patient care. The integration of DHPs into clinical research is aimed at enhancing patient engagement, facilitating accurate data collection, and improving the overall management of oncology patients. As such, the adaptability and scalability of digital tools like PROACT 2.0 represent a fundamental step towards broader applications in cancer research.
Supplemental Material
sj-pdf-1-tmj-10.1177_03008916241248007 – Supplemental material for PROACT 2.0: A new open-source tool to improve patient-doctor communication in clinical trials
Supplemental material, sj-pdf-1-tmj-10.1177_03008916241248007 for PROACT 2.0: A new open-source tool to improve patient-doctor communication in clinical trials by Luca Agnelli, Andrea Villa, Fouziah Butt, Matteo Duca, Alessandro Guidi, Marcello Carapezza, Michele Addante, Gaetano Lenoci, Paul O’Regan, Laura Russo, Sara Cresta, Alessandra Castano, Elisabella Ebrahem, Sara Alfieri, Akshita Patil, Louise Carter, Caroline Dive, Filippo Guglielmo De Braud and Silvia Damian in Tumori Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Associazione Italiana per la Ricerca Contro il Cancro (AIRC) IG #24286; by Cancer Research UK via Core funding to the National Biomarker Centre and the Accelerator Award [A29374] funding to UpSMART; and by Ministero della Salute (fondi per la Ricerca Corrente). Support was received from the Manchester Experimental Cancer Medicine Centre and the NIHR Manchester Biomedical Research Centre.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
