Abstract
Aims:
Low skeletal muscle mass index (SMI) has recently emerged as an independent prognostic factor in oncological patients and it is linked with poor survival and higher treatment toxicity. The present study aims to determine the possible impact of low SMI on survival and acute toxicity in oropharyngeal patients.
Methods:
Seventy-six patients with locally advanced oropharyngeal squamous cell carcinoma (stage III-IVC) were treated in our institution with Helical TomoTherapy® (HT - Accuray, Maddison, WI, USA) between 2005 and 2021. All patients received concomitant platinum-based chemotherapy (CT) (at least 200 mg/m2). The SMI was determined using the calculation of cross-sectional area at C3. Twenty patients (26%) presented pre-treatment low SMI, according to Chargi definitions.
Results:
All patients concluded the treatment. Thirteen patients with low SMI (65%) and 22 patients with normal SMI (39%) presented acute toxicity greater than or equal to grade 3, but this difference was not statistically significant (p-value = 0.25). Overall survival was analyzed in 65 patients, excluding those who finished CT-RT less than six months before the analysis. Overall survival was significantly lower in low SMI versus normal SMI patients (p-value = 0.035). Same difference was observed in N0-N2a patients, suggesting an important role of SMI also in lower nodal burden and putatively better prognosis.
Conclusions:
Although the results are limited to a small population, our case series has the advantage to be very homogeneous in patients and treatment characteristics. In our setting, SMI demonstrated a crucial impact on overall survival. Further investigation with larger samples is necessary to confirm our results to improve patient outcomes.
Introduction
Cancers are among the leading causes of morbidity and mortality worldwide, and the number of new cases is expected to rise significantly over the next decades. At the same time, many cancers may be cured or converted to chronic diseases, thanks to the refining of all types of cancer treatment (surgery, radiation therapy, and pharmacological therapies). All of these treatments, however, could be hindered or precluded by the frequent development of malnutrition and metabolic derangements in cancer patients, induced by the tumor or by its treatment. 1
Malnutrition is a well-defined condition in which at least one phenotypic criterion (involuntary weight loss, low body mass index or reduced muscle mass) and one etiologic criterion (reduced food intake or assimilation and inflammation or disease burden) is present. 2
Differently from simple malnutrition, the negative energy balance and skeletal muscle loss observed in cancer patients is driven by a combination of reduced food intake and metabolic changes (e.g. elevated resting metabolic rate, insulin resistance, lipolysis, and proteolysis which aggravate weight loss, systemic inflammation and catabolic factors) which may be host- or tumor-derived. 1
Head and neck cancer patients in particular present high risk of malnutrition due to their lifestyle habits, such as smoking and alcohol abuse, and to the anatomical location of the tumor, which can make feeding difficult.3,4 At diagnosis, up to 46-49% of head and neck cancer patients present signs of malnutrition, such as weight loss and vitamin deficiencies. 5 Furthermore, radiotherapy and chemotherapy could also lead to skeletal muscle loss and to the onset of both acute and late toxicity, which can result in inadequate oral feeding.4,6 Malnutrition also leads to a higher risk of infections and hospitalization, muscular weakness and depression, and reduces treatment compliance. 7
More specifically than malnutrition, sarcopenia reflects the poor general conditions frequently experienced by patients affected by locally-advanced cancer. According to the definition of the European Working Group on Sarcopenia in Older People (EWGSOP), 8 sarcopenia is a condition characterized by low muscle strength, low muscle quantity or quality and low physical performance. Sarcopenia is both a nutrition disorder and a nutrition related condition 1 and seems to be associated with a worse prognosis,9,10 lower tolerance to the treatments11–13 and enhanced inflammatory status.14–16 It does not simply reflect the physical condition, but also acts as an endocrine organ that secretes several specific cytokines also called myokine, able to influence tissue metabolism and organ inflammatory status. The immune suppression observed in malnourished patients has been shown to be related with unimpeded tumor growth.
There are several techniques for the measurement of skeletal muscle mass (SMM), for example dual-energy X-ray absorptiometry (DXA) or bioelectric impedance analysis (BIA), but the most commonly used technique is based on computed tomography (CT) that is part of routine cancer patients’ cure and it has a proven high accuracy in measuring SMM. 17 Most studies quantify SMM using CT scans of the third lumbar (L3) vertebra, although other levels have also been proposed. 18 The cross-sectional area (CSA) of skeletal muscle mass is measured on a single cross-sectional image and normalized for height to obtain the skeletal muscle index (SMI). The SMI thus obtained correlates strongly with total-body skeletal muscle mass.17,19
Although there are many studies that analyze the predictive value of low SMI, they often involve large but non-homogeneous populations and they aim to investigate whether this predictive value is universally present across cancer types or cancer treatments. Our study, by contrast, starts from a different assumption: starting from a population as homogeneous as possible, we aim to retrospectively demonstrate the independent role of low SMM in predicting outcome and toxicity.
Materials and methods
Patient and treatment characteristics
In this retrospective mono-institutional analysis, all patients affected by stage III-IVC oropharyngeal cancer and treated in our institution from July 2009 to December 2020 were included. All patients were treated with helical TomoTherapy® (HT- Accuray, Maddison, WI, USA).
A 18Fluorodeoxyglucose computed tomography (CT) positron emission tomography (PET) was performed in order to identify the biological target volume (BTV). In 95% of patients a hypofractionated schedule with a simultaneous integrated boost (54 Gy in 30 fractions on bilateral neck nodes and 66 Gy on tumor and high risk/PET positive nodes) was used. In four patients with large tumor or worse performance status, a conventional fractionation was preferred (50 Gy in 25 fractions on bilateral neck nodes and a sequential boost of 20 Gy in 10 fractions on tumor and high risk/PET positive nodes). A cisplatinum-based chemotherapy (at least 200 mg/m2) was prescribed. No patients were surgically resected before concomitant treatment.
Daily megavoltage CT image guidance was applied for all patients using the system integrated in the TomoTherapy machine. A two-step matching strategy was used: a fully automatic registration based on bony anatomy followed by ‘fine’ manual adjustment matching the target volumes.
During the treatment and after its completion, clinical and instrumental evaluation were performed in order to register acute and late side effects (according to Common Terminology Criteria of Adverse Events-CTCAE- version 4.1 scale) 20 and clinical outcome.
CT image analysis
For all patients, the skeletal muscle mass was assessed on pre-treatment CT scan (without contrast enhancement) by a small pool of Radiation Oncologists who are dedicated to head and neck disease, with the method previously published by Chargi et al. 21
Muscle tissue was characterized by Hounsfield Unit (HU) range settings from −29 to +150 HU. Muscle tissue was delineated at the level of the third cervical vertebra (C3), selecting the first slide showing both transverse processes and the entire vertebral arc when scrolling from caudal to cranial direction. The contours of the paravertebral muscles and both sternocleidomastoid muscles were manually traced. The skeletal muscle area (SMA) at the level of C3 was calculated as the sum of the areas of paravertebral muscle and both sternocleidomastoid muscles. If evident lymph node metastasis hindered accurate delineation of one sternocleidomastoid muscle, the SMA of the contralateral sternocleidomastoid muscle was used as an estimation of the SMA of the affected sternocleidomastoid muscle. 21 The cervical SMI (CSMI) was calculated by dividing the SMA at the level of C3 by the squared height of the patient.
Gender and BMI specific cut-off values were calculated based on mean − 2SD as suggested by the EWGSOP28 and Chargi et al. 21 For male patients with a BMI <25 kg/m2, a CSMI ⩽6.8 cm2/m2 was defined and with a BMI ⩾25 kg/m2 a CSMI ⩽8.5 cm2/m2 was defined for low SMM. For female patients with a BMI <25 kg/m2, a CSMI ⩽5.3 cm2/m2 was defined and with a BMI ⩾25 kg/m2 a CSMI ⩽6.4 cm2/m2 was defined for low SMM.
Based on this value, the study population was divided into Low SMI (LSMI) and Normal SMI (NSMI).
Statistical methods
Descriptive statistics were calculated for demographic and clinical variables across strata of interest. Chi-square or t tests were used to identify statistically significant differences in proportions or means across strata of interest. Kaplan-Meier estimates of survival proportions over time were calculated for subjects with or without low SMI. A stratified survival analysis was performed by stage (N0-N2a versus more than N2b).
Multivariable Cox proportional hazard model was used to determine the independent prognostic role of low SMI with cancer progression or death, adjusting for other prognostic factors and confounders.
All statistical tests were 2-sided and used a p-value <.05 as the criterion for significance. MedCalc software version 11.1.1.0 (MedCalc Software Ltd, Ostend, Belgium) was used for all analysis.
Results
A total of 76 patients were included in our study. A summary of the patients’ characteristics is reported in Table 1.
Patient characteristics.

According to CSA method, 20 out 76 patients were found to have low SMI (15.2%), only three of whom were also underweight.
All patients concluded chemo-radiotherapy, with a cumulative dose of concomitant cisplatinum ⩾200 mg/m2.
After a median follow up of 41 months (range 3.4-126.1), five patients experienced a local relapse (with or without distant metastases), while seven had a systemic progression. Seven patients died, four of whom from cancer progression.
The main frequent late effects (mucositis, dermatitis, dysphagia and xerostomia) were considered, during and after chemo-radiotherapy and reported according to CTCAE version 4.1.
In eight patients the treatment was discontinued due to side effects, regardless of the skeletal muscle mass condition. No significant difference between LSMI-group and NSMI-group emerged, even if a negative trend in LSMI-group was hypothesized, as 65% in LSMI-group and 39% in NSMI-group presented an acute toxicity greater than or equal to grade 3 (p value = 0.25).
Excluding patients who finished chemoradiotherapy less than six months before our statistical analysis, overall survival was analyzed in 65 patients (47 NSMI and 18 LSMI). Overall survival was significantly higher in NSMI compared to LSMI group (median 46.2 vs 40.3 months, HR 0.27, 95% CI 0.08-0.92, p-value 0.035, Figure 1). The same difference was confirmed in patients with a lower nodal burden (N0-N2a) and theoretically better prognosis (median 46 vs 43 months, HR 0.17, 95% CI 0.04-0.79, p-value 0.02, Figure 2)
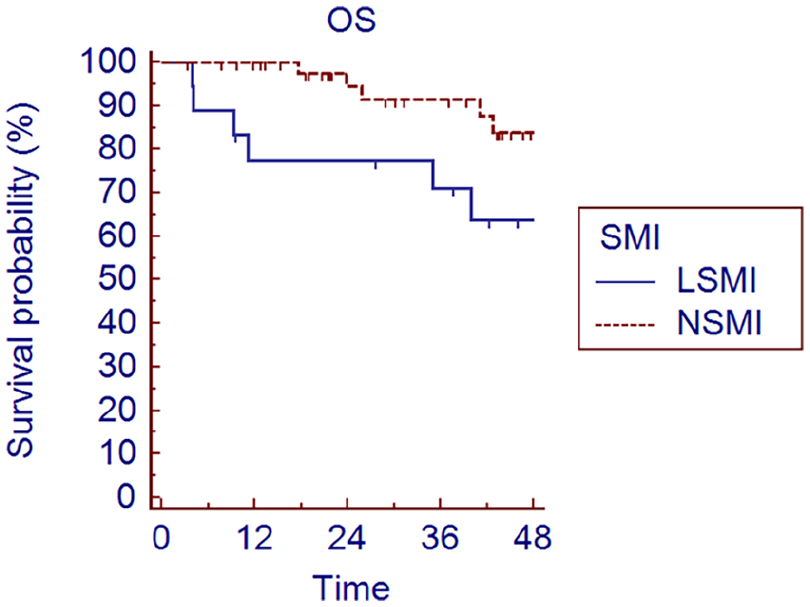
Overall survival (OS) stratified by SMI, according to Chargi’s definition (p-value 0.035).
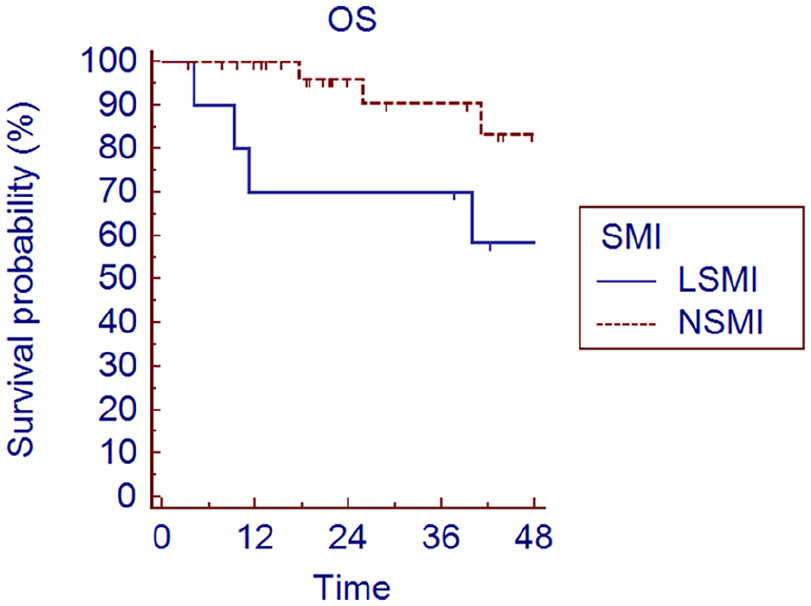
Overall survival in stage N0-N2a patients, stratified by SMI, according to Chargi’s definition (p-value 0.02).
Gender, age at diagnosis, T-stage, N-stage and low versus normal SMI were considered clinically relevant and entered into the multivariate analysis. The sole independent predictor of mortality was found to be the low skeletal muscle index (OR 3.64 95%CI 1.05-12.59 p=0.04).
Discussion
Sarcopenia is a syndrome characterized by progressive and generalized loss of skeletal muscle mass and strength. The EWGSOP 22 recommends using the presence of both low muscle mass and low muscle function (strength or performance) for the diagnosis of sarcopenia. However, not all studies concerning cancer patients applied this strict definition, using sarcopenia as synonymous with low SMI.
There has been growing interest in sarcopenia and its role in predicting recurrence probability and survival. Different studies have been conducted specifically for head and neck cancer patients, considering the severe malnutrition that is often reported, due to neoplasia itself and the therapies prescribed.6,23 In our work, the impact of low CSMI was assessed in a specific population of oropharyngeal cancer patients. To the best of our knowledge, this is the first study focusing on an extremely homogeneous sample of oropharyngeal cancer patient, all treated with radical intent and the same chemo-radiotherapy schedule. In our opinion, this choice allows to reduce the variables related to the tumor site and type of treatment and probably highlights with greater emphasis the real impact of low SMI on prognosis. In our cohort, low CSMI seems to be associated with worse OS, while no correlation with toxicity during and after treatment was found.
The mechanism behind how sarcopenia reduces OS is unknown. Probably sarcopenia could be a surrogate for nutritional status, better than other indicators (weight loss for example): malnourished patients have greater drug toxicity (resulting in under-treatment), increased postoperative complications, decreased quality of life and increased mortality. Sarcopenia is thought to be a sensitive marker for the pro-inflammatory state of the cancer. 24 This marker is thought to be related to the wasting syndrome of cancer cachexia, but the mechanism is not fully known 25 and has been identified as an independent prognostic factor for treatment-related toxicity, overall survival, and progression-free survival within many solid tumors, including head and neck cancer.25–28 Morse et al. 29 hypothesized that a potential mechanism could be related to frequent treatment breaks in radiation, which can worsen outcomes in head and neck cancer due to the accelerated repopulation of cancer cells and increased radioresistance. 30 Sarcopenia may be clinically distinct from “frailty syndrome” that is characterized by declines in physiologic reserve and ability to manage acute stressors. 31 Frail patients may be less suited to tolerate the significant toxicities that accompany head and neck cancer treatments.
Despite quite a large number of studies existing, few of them analyzed the prognostic impact of radiologically defined sarcopenia on outcome in patients affected by head and neck cancer. Wong et al. 32 collected data from 10 studies from 2016 and 2019, in which sarcopenia was radiologically defined (6 at L3 level, 3 at C3 level and one at both) and was found to be associated with worse OS (HR 1.98; 95% CI: 1.64-2.39, p-value<0.00001). Ganju and colleagues 33 had taken into consideration a large cohort of patients treated for head and neck cancer (any site and any treatment), showing the impact of sarcopenia in Overall Survival (HR 1.83 p-value=0.03) and Progression free survival (HR 1.65 p-value=0.03). In addition, Van Rijn-Dekker and colleagues 23 performed an interesting analysis on 750 patients affected by head and neck cancer (any location), treated with curative radiotherapy associated or not with chemotherapy. They showed a correlation between sarcopenia and worse OS (HR 0.72 p-value=0.0012), worse disease free survival (HR 0.67 p-value=0.001) and increased toxicity (dysphagia and xerostomia). Tamaki and colleagues 34 , while focusing only on oropharyngeal cancer, considered different treatment modality (surgery, chemotherapy and radiotherapy, alone or in combination). In 2020, Findlay et al. 35 published a systematic review regarding radiological sarcopenia in head and neck cancer patients and its impact on oncological outcomes: they concluded that sarcopenia is an independent prognostic factor for OS and treatment completion and further research is essential to improve knowledge and to enable future personalized nutritional support.
Likely, the impact of sarcopenia on treatment-related toxicity is unclear. In a systematic review 35 (11 studies between January 2004 and June 2019), pre-treatment sarcopenia was independently associated with prolonged radiotherapy breaks and chemotherapy-related toxicity in 3461 head and neck patients that underwent curative radiotherapy with or without other concomitant therapies. Also, Karavolia et al. 36 demonstrated, in a large cohort of 977 head and neck patients, that sarcopenia was an independent prognostic factor for the development of physician-rated acute grade ⩾ 3 dysphagia and late grade ⩾ 2 and grade ⩾ 3 dysphagia, probably for its impact on the swallowing glands. On the contrary, Huang et al. 37 and, very recently, Bergamaschi et al. 38 could not find an association between sarcopenia and radiotherapy toxicity in 82 nasopharyngeal cancer patients and 116 oropharingeal cancer patients respectively. In our series the acute toxicity profile was not significantly higher in low SMI patients, even if a tendency to a reduced tolerance to radiotherapy could be found.
There are several methods for defining CSMI, but recently its evaluation on a radiological basis has taken hold. The radiologically defined skeletal muscle mass (using CT or magnetic resonance imaging-MRI) relies on a more objective and less variable way of definition. Furthermore, CT imaging is always available in cancer patients, for staging or for radiotherapy planning. At the third lumbar vertebra, the radiologically measured cross sectional area (CSA) has a linear relationship with muscle mass and then with sarcopenia. 19 Swartz et al. 18 identified a strong correlation in skeletal muscle CSA at C3 and at L3. Assessing skeletal muscle mass on a head and neck CT-scan at the C3 level appears a good alternative to abdominal CT-scans and it allows researchers to study the clinical effects of sarcopenia in these patients using already available clinical data, without additional costs or discomfort for the patient. As of today, there is no consensus regarding the methodology for defining low CSMI. Among the various proposed in literature, Chargi et al. 21 cut offs were chosen in our analysis because they are the first gender- and BMI-specific cut-off values of the mean quantity of SMM (SMA and skeletal muscle mass index) derived specifically from cervical muscle mass evaluation. The heterogeneity of sarcopenia cut-off definition makes the comparison of various series difficult.
Cancer related fatigue (CRF) is a complex scenario characterized by significant physical, emotional, and/or cognitive exhaustion, which is disproportionate to the activity level and interferes with usual functioning in patients with cancer. 39 Cancer-related fatigue is one of the most prevalent symptoms experienced by patients with cancer, during and after treatment 40 but the etiology is not yet fully understood. The pathophysiology of CRF is determined by central and peripheral aspects of fatigue. Pro-inflammatory effects of cancer lead to direct changes in the muscle metabolism, with a reduced adenosine triphosphate levels, reduced protein synthesis, and localized electrolyte imbalances causing peripheral CRF. 41 Central effects of elevated cytokine levels (among which interleukin-2, interleukin-6, and tumor necrosis factor alpha) cause changes in activity of the hypothalamus-pituitary-adrenal axis, leading to a sensation of reduced capacity to perform physical work, but also to a reduced androgen expression. 41 Both pathways could result in a loss of muscle mass, so CRF and sarcopenia are inextricably linked.
Given the uncertainty surrounding the identification of patients prone to developing sarcopenia during chemo-radiotherapy, the inclusion of pre-treatment radiologically determined CSMI as a standard practice could serve as a valuable strategy for discerning individuals with a potentially unfavorable prognosis. Moreover, it could aid in recognizing those requiring prompt initiation of nutritional support. 42 Intense nutritional interventions have been demonstrated to minimize weight loss and improve treatment tolerance. 43 The ability of amino acids to stimulate protein synthesis in cancer patients is reduced. 44 This anabolic resistance could be in part counteracted by nutritional intervention, increasing protein intake or giving specific amino acids. In particular, the addition of leucine could increase substrate availability and directly modulate the anabolic signal pathway. 45 Few studies showed the possibility of increasing muscle protein synthesis by specific nutriments and/or by increasing amino acids or protein administration. Deutz et al. 46 , in a randomized trial with a small cohort, demonstrated that, in cancer patients, conventional nutritional supplementation is ineffective in stimulating muscle protein synthesis and that the anabolic resistance can be overcome with a specially formulated nutritional supplement. 46 A recent study from the Trans-Tasman Radiation Oncology Group (TROG) showed the correlation between psychological interventions and nutritional status: patients receiving motivational interviewing and cognitive behavioral therapy were more well-nourished, lost a smaller percentage of weight, and had a higher quality of life. 47
There are current trials assessing the impact of a muscle training (seven conventional resistance training exercises targeting the large muscle groups of the body), during and after chemo-radiotherapy, on body composition, muscle strength, and functional performance. 48 Grote et al. 49 demonstrated that progressive resistance training during RT for cachectic head and neck cancer patients is feasible, well tolerated and safe, with better outcomes in term of general fatigue and quality of life. In Mustian et al.’s meta-analysis, 50 exercise and psychological interventions are significantly more effective for improving cancer related fatigue compared with pharmaceutical interventions both in metastatic and nonmetastatic stages. However, the last Cochrane meta-analysis specifically focusing on the effect of physical activity on cancer and cachectic patients concluded that there was insufficient data to determine its safety and efficacy in this context. 51
In sarcopenic cancer patients (head and neck, prostate, lymphoma), or probably sarcopenic but not cachectic, regular physical activity during treatment improves muscle mass and strength, fatigue, and quality of life.52–55
Even if many studies provide evidence of a benefit of adapted physical activity (APA) in advanced cancer patients, it is difficult to specify the most appropriate type of exercise, and the optimum rhythm and intensity. In a survey of advanced cancer patients, all patients felt physically able to follow an APA program, but only two-thirds had undertaken to do so. 56 In the study by Chasen et al. 57 , the only independent predictor of good adherence to an APA program in patients with advanced cancer was low cancer related fatigue, suggesting the lack of interest in APA in patients with severe or refractory cachexia.
Moreover, the benefits of physical activities and of protein support seem greater when it is started at the precachexia stage rather than at the cachexia stage, and their benefits are limited or nonexistent at the stage of refractory cachexia. 58
Given the pre-treatment identification of sarcopenia, it could become feasible to anticipate individuals in greater need of assistance, thereby enabling clinicians to recommend heightened nutritional augmentation and physical rehabilitation strategies, potentially enhancing overall prognoses. 59 A prospective study evaluating the effects of these interventions exclusively among patients manifesting sarcopenia at the time of simulation would yield elucidating insights.
There are some limitations of our study to discuss. First, the retrospective nature and the small sample size may have created bias during the result analysis. Then, to date, no consensus regarding thresholds for sarcopenia has been reached in literature, which makes it more difficult to compare our results to those obtained in other centers. Finally, due to the retrospective nature of the research, some relevant parameters (such as HPV status) may not have been reported on in a consistent manner. The homogeneous sample size and the long follow up represent the main strengths of the present analysis.
Conclusions
In conclusion, our study confirms the relationship between radiologically measured low skeletal muscle mass and worse OS, in a homogeneous series in term of tumor and treatment characteristics. Our results show the potential importance of routine estimation of SMM using pre-treatment CT scan in order to determine which patient could be more susceptible to a worse prognosis, due to their low muscle mass. Validation of our findings in a larger set of patients with prospective data collection is necessary to better explain the relationship between low SMM and oncological outcomes in head and neck patients.
Footnotes
Author contribution
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by CLD, AM, ID’O and AC. The first draft of the manuscript was written by CLD and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
