Abstract
Introduction
The synthesis of the periprosthetic capsule during implant-based breast reconstruction is the result of a coordinate cascade of inflammatory events ending in a fibrous tissue deposition around the expander or implant. Although the development of small volumes of fluid is one of the complications of prosthetic-based breast reconstruction, the characterization of the periprosthetic effusions coupled with the micro-textured devices, that have been recently introduced after the recall of macro-textured ones, is still lacking. The investigation of these periprosthetic effusions and paired capsules in terms of immunological content were the primary and secondary aims of the present study, respectively.
Methods
For this, 68 women, 41 of whom had periprosthetic effusions at the time of expander replacement with implant, were recruited. For each case, capsule and healthy dermal tissues were taken and for women with periprosthetic effusion, peripheral blood was also collected. Periprosthetic effusions and peripheral blood were characterized by cytometry while capsules and dermal tissues by immunohistochemistry and Nanostring analysis.
Results
The results showed an increase of Th1, Th2 lymphocytes and a HLA-DR+bright CD16+ cells (likely representing monocytes-derived macrophages) in periprosthetic effusions in respect to peripheral blood. These pro-inflammatory cells were counterbalanced by the gain of suppressive CD4 Treg cells. In the corresponding capsules, immunohistochemistry revealed the absence of Th1 cells and the presence of tissutal FOXP3 Treg. No significant difference in expression of inflammatory-related genes between capsules and dermal tissues was present.
Conclusions
These results suggest the presence of a Treg-controlled inflammation in both periprosthetic effusions and capsules.
Introduction
Implant-based breast reconstruction (IBR) is the most common surgical procedure in women undergoing mastectomy and it can be performed as one-phase reconstruction (direct-to-implant) or as two-stage reconstruction (the reconstruction with the implant is preceded by the positioning of an expander, which allows a better healing of tissues and a better adaptability to the prosthesis). 1 Both expander and implant are silicon-made and have a textured surface that facilitates their firmness to the body thanks to the periprosthetic capsule (PPC) development. 1 PPC is deposited after a multi-stage physiological process that begins immediately after device implantation, first with acute then with chronic inflammation and ends with a wound healing phase. 2 The acute inflammatory phase is characterized by the presence of neutrophils and eosinophils which are than replaced by lymphocytes and monocytes/macrophages cells (M/M) in chronic phase. 3 During chronic inflammation, CD4 T lymphocytes and M/M cells produce cytokines able to induce PPC development through the recruitment and stimulation of fibroblast, endothelial and adipose cells. 3 CD4 lymphocytes include at least three major cellular subsets: i) pro-inflammatory Th1 CD4+ producing IFN-γ and IL-2, ii) pro-tissue repair Th2 CD4+ secreting IL-4 and iii) an additional subset of T CD4+ CD127- CD25+ cells with regulatory functions (the so called Treg). 4 Similarly, M/M cells can also be distinguished into HLA-DR positive pro-inflammatory (M/M1) cells producing TNF-α, IL-6, IL-12 as well as CD163 positive pro-tissue repair (M/M2) cells secreting IL-10, IL-23, VEGF and TGF-B. 5
The most common complication within IBR is capsular contracture (CC), a massive deposit of fibrosis around the implant that has been reported in 10.6% of cases and postulated as a sequela of inflammatory processes. 6 In addition to CC, the accumulation of small volumes (a few milliliters) of periprosthetic effusion (PPE) containing lymphocytes and M/M can be observed with an estimated incidence of 5.4%. 6 The origins of PPEs are not fully understood, however, studies on mastectomy without reconstruction suggested irregularity of the axilla and chest wall movements with potential spaces for fluid accumulation, lymphatic disruption and ongoing inflamm-ation leading to persistent exudation 7 as potential causes of its formation. Finally, a late consequence of IBR, is the increased risk of a breast implant-associated anap-lastic large cell lymphoma (BIA-ALCL) development. 8 Although the lifetime prevalence of BIA-ALCL was 33 per 1 million persons with textured breast implants, 9 FDA and Allergan (Inc., Irvine, California, USA) recently recalled the BIOCELL® textured implants 10 and, as a result, micro-textured expander/implants (POLYMSiltex, Mentor®, California, USA) have been introduced into our clinical practice as decided by our National Healthcare System. The effects of textured implants on capsule deposition during breast reconstruction are still to be assessed and, since we noticed the formation of PPEs during the replacement of the Mentor expanders, we hypothesized that they could be the result of incorrect PPCs development and therefore could cause an inflammatory reaction potentially leading to IBR complications. For these reasons, we conducted a study whose primary endpoint was PPEs characterization in terms of immune cells content (CD4 Treg, Th1 and Th2) compared to paired peripheral blood (PB) samples. In addition, as a secondary endpoint, a comprehensive analysis of PPCs obtained from patients (pts) with or without PPE was performed using both immunohistochemistry (IHC) and gene expression analysis.
Material and methods
Patients
The prospective observational study was started in the Plastic Reconstructive Surgery Unit at Fondazione IRCCS Istituto Nazionale Tumori Milano in December 2019 and ended in June 2021 (with an interruption due to the COVID-19 pandemic). Women undergoing two-stage breast reconstruction with texturized expander (Mentor, CPXTM 4 breast tissue expander) replacement with implant (CPGTM Implants Cohesive III) were enrolled. All patients signed an informed consent. This study was approved by our institutional ethics Board (Protocol: INT 181/19).
Surgery
Before anesthesia, PB was collected for each patient. All surgeries were performed under general anesthesia by standard techniques. After skin incision, a piece of dermal tissue was harvested and kept for the subsequent biological evaluations. After pectoralis major muscle incision, we collected as much PPE as possible if present around the expander.
All patients underwent capsulectomy and Mentor textured anatomic implants were placed submuscular, after periprosthetic pocket remodeling. One suction drain was placed. The pectoralis major muscle was sutured above the implant before skin closure.
PPEs and PBs samples
PPEs: Macroscopic evaluation
After a preliminary evaluation in terms of volume and erythrocyte contamination (Table 1), the cells and PPEs supernatants were recovered after two successive centrifugations at 1800 rpm for 10 minutes at 4°C. The cells were immediately used for flow cytometry and T cell activation while supernatants were stored at -80°C for subsequent uses.
Study enrolment timeline and PPEs macroscopic features.
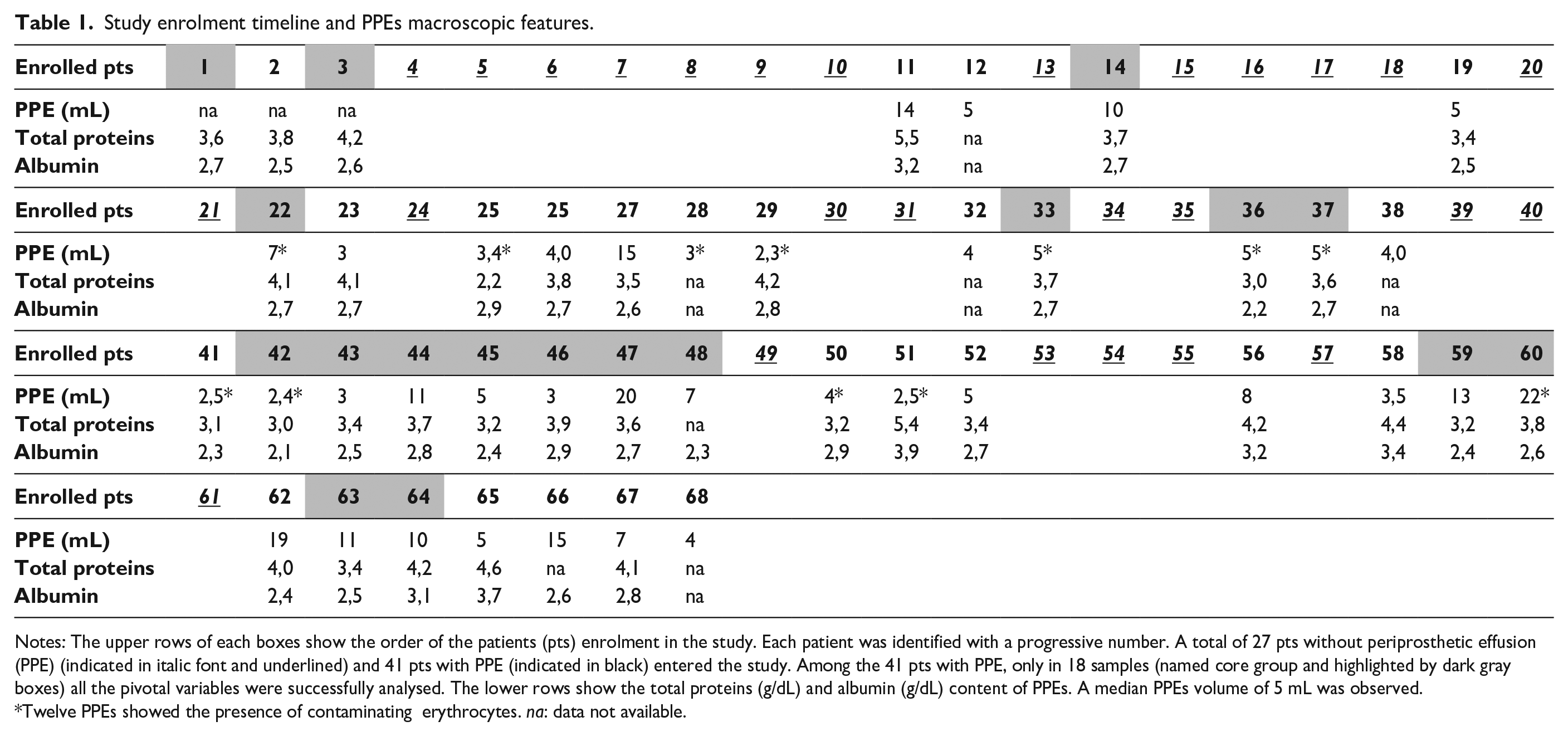
Notes: The upper rows of each boxes show the order of the patients (pts) enrolment in the study. Each patient was identified with a progressive number. A total of 27 pts without periprosthetic effusion (PPE) (indicated in italic font and underlined) and 41 pts with PPE (indicated in black) entered the study. Among the 41 pts with PPE, only in 18 samples (named core group and highlighted by dark gray boxes) all the pivotal variables were successfully analysed. The lower rows show the total proteins (g/dL) and albumin (g/dL) content of PPEs. A median PPEs volume of 5 mL was observed. *Twelve PPEs showed the presence of contaminating erythrocytes. na: data not available.
PPEs: Total proteins and albumin content
After supernatants thawing, their total proteins and albumin content (Table 1) was measured by Cobas pro equipped with C305 module (Roche Diagnostic) according to manufacture instructions and using Total protein gen.2 (cat: 08058652190, Roche) and Albumin gen 2 (cat: 08056692190, Roche).
Flow cytometry (FC)
Cells were labelled using the following antibodies: CD3, CD4, CD8, CD14, CD16, CD25, CD45, CD64, CD127 and HLA-DR. See online supplementary material and methods for further details.
T cell activation
Production of IFN-γ and IL-4 was measured in PPEs and matched PBs samples (online supplementary material and methods).
PPCs and dermal tissues
After a macroscopic evaluation, all the PPCs were split into two similar halves, one half was formalin fixed and paraffin embedded (FFPE) while the remaining half and the dermal tissue were quickly frozen.
IHC
Representative 2 μm sections of FFPE samples were selected and phenotyped. The antibodies and experimental conditions used to detect the expression of CD3, CD4, CD8, FOXP3, TBX21 (T-bet), CD163 and Ki67 are shown in the online supplementary material and methods.
Nanostring human inflammation panel
RNA was extracted from frozen PPCs and matched dermal tissues as detailed in supplementary material and methods. 150 ng of total RNA was used for nCounter Nanostring evaluation (Human inflammation panel V2CSO.12, Diatech labline) according to datasheet instructions (detailed in the online supplementary material and methods).
Statistical consideration
Details about the sample size consideration are reported in the online supplementary material and methods. Standard descriptive statistics (medians and ranges for continuous variables and frequency tables for categorical variables) were used to describe patient cohort and biological data. For the primary endpoint, the Wilcoxon signed-rank test (WSR) was used due to the paired nature of the data. For Nanostring data, raw data were normalized following the guidelines manual (see online supplementary material and methods for further details). Normalized data (in terms of log2 expression levels) of the PPCs relative to the normal ones were then used as pivotal quantity for the subsequent analysis. The nonparametric Wilcoxon test was used to evaluate differences between groups (i.e., PPE-associated vs PPE non-associated and inflammation levels) whereas the WSR was used for paired data. All the statistical analyses were performed in SAS (version 9.4.; SAS Institute, Inc., Cary, NC) by considering a Type I error of 0.05 and properly adjusted for multiple comparison using false discovery rate (FDR) or Bonferroni correction as detailed through the text.
Results
Study cohort
A total of 68 women were enrolled (41 with PPEs and 27 without respectively; Table 1), which was higher than planned (i.e., 29 with and 18 without PPEs). This discrepancy was likely due to the low cell content of some PPEs that caused a low Th1 and Th2 number after stimulation of CD3 with specific beads. Overall, Th1-Th2 frequencies, were obtained in 18 out of 41 PPEs. Accordingly, results of the primary endpoint focused on this group of 18 samples for which the pivotal variables were obtained (core group). The clinical characteristics of all the enrolled women and those of core group are reported in Supplementary Table 1.
Primary endpoint
On the whole, an increase of lymphocytes (Figure 1 panel A) was observed in PPEs in respect to their corresponding PBs in core group and then confirmed in the rest of the PPEs. In line with the increase in lymphocytes, there was a decrease in neutrophils and monocytes in PPEs. Among lymphocytes, the CD3+ CD4+ CD127+bright CD25- or the CD3+ CD8+, CD127+bright CD25- were defined as conventional and hereafter named CD4 and CD8. CD4 Treg were defined as CD3+ CD4+ CD127- and CD25+ (Supplementary Figure 1). 4
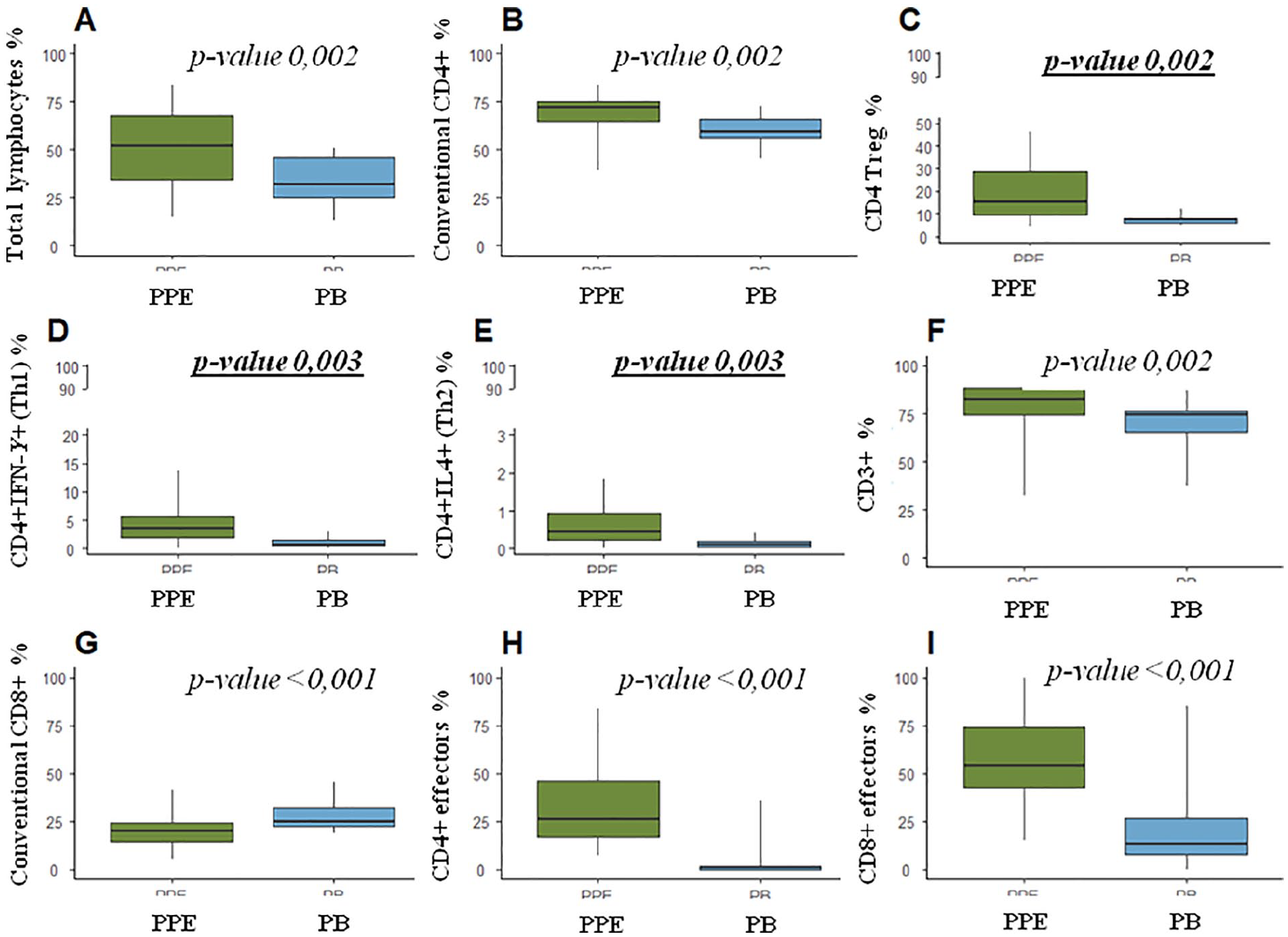
Wilcoxon signed rank test results. Box plots showing the distribution of lymphocytes (A), conventional CD4 (B), CD4Treg (C), Th1 (D), Th2 (E), CD3 (F), CD8 (G), CD4 effectors (H) and CD8 effectors (I) in the periprosthetic effusions (PPEs) and corresponding peripheral blood (PB) samples of core group (n=18). Each box indicates the 25th and 75th centiles. The horizontal line inside the box indicates the median, and the whiskers indicate the extreme measured values. Wilcoxon signed rank (WSR) p-value for the three primary endpoint (CD4 Treg, CD4 Th1 and CD4 Th2) are underlined and reported in bold character.
CD4 Treg, Th1 and Th2 frequencies
The results obtained in the core group showed a significant increase of conventional CD4, CD4Treg, Th1 and Th2 frequencies in PPEs compared to the corresponding PB (Figure 1 panels B, C, D and E and Supplementary Figure 2). Notably, only in two cases a significant percentage of CD4+ IFN-γ (> 10% of CD4+ cells, Figure 1 panel D) was found in PPEs whereas for the other cases, the frequency of Th1 was less than 10%.
CD4 and CD8 polarization
The CD4 increase in PPEs was coupled with that of CD3 while a lower frequency of CD8 was observed (Figure 1 panels F and G). Furthermore, both CD4 and CD8 cells present in PPEs showed a decrease in CD127 expression combined with an increase in that of HLA-DR (Supplementary Figure 3). This modulation was in line with a transition from naïve (TN) cells CD127+ bright HLA-DR- to a T effector (TE) CD127+ HLA-DR+bright in both, CD4 (Supplementary Figure 3) and CD8 (not shown). 11 Notably, although in three cases of the core group, all CD4 and CD8 lymphocytes in PPE consisted of TE, in most of these samples, only a fraction of CD4 or CD8 acquired TE polarization (Figure 1 box plots H and I).
Th1, Th2, Treg CD4 and CD8 polarization and clinical features
Overall, no correlation was observed between the considered clinical characteristics (Supplementary Table 1) and the development of PPEs or the CD4 Treg, Th1 and Th2 frequencies in PPEs (data not shown). Only one association was found between Th1 and comorbidity (Fisher Exact p-value = 0.012). Interestingly, all three patients with comorbidity did not experience any increase in Th1frequencies in PPE compared to the corresponding PB.
M/M cells polarization
A peculiar cellular population likely representing M/M cells were found in PPEs (Figure 2).
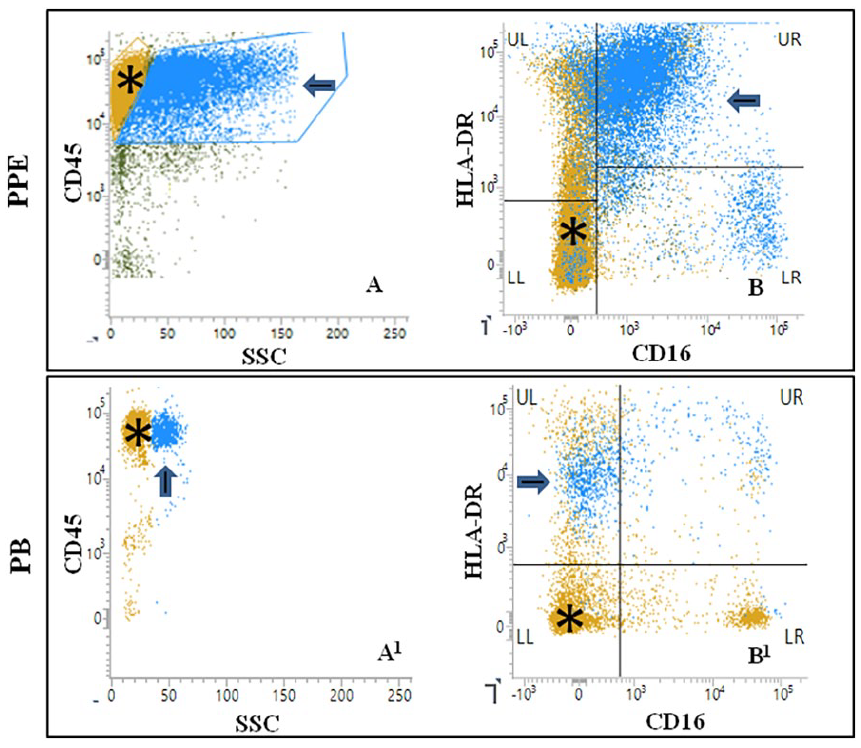
Monocytes/macrophages activation profile in PPEs. Figure showing the peculiar flow cytometric profile of monocytes/macrophages (M/M) cells in a representative (sample n=14, Table 1) periprosthetic effusion (PPE) compared to paired peripheral blood (PB) monocytes. M/M cells in PPE (blue cells indicated by arrow in A) showed greater complexity (side scatter: SSC) than PB monocytes (blue cells indicated by arrow in A1). M/M cells (blue cells indicated by arrow in B) also showed an higher expression of HLA-DR and CD16 than monocytes (blue cells indicated by arrow in B1). Lymphocytes (CD45+brightSSClow coloured in ocher and indicated by asterisk in all plots) are shown for comparison. All the analyses were performed in live cells as shown in supplementary Figure 1.
In respect to PB monocytes, M/M cells showed a significant increase in CD16 expression (WSR p-value: 0.004) and augmented cellular complexity as demonstrated by high side scatter (SSChigh, WSR p-value < 0.001) along with a borderline increase (WSR p-value 0.054) in HLA-DR (HLA-DR+bright) expression. As described for Treg, Th1, Th2 and CD4-CD8 polarization, M/M1 cells also showed no correlation with the clinical features considered in Supplementary Table 1. This phenotype, suggestive of activation and polarization towards a pro-inflammatory M/M1 cells, 5 was observed in both core and rest of the PPEs.
Secondary objective
The CD4, CD8 and M/M cells distribution in PPCs was performed through IHC while the expression of genes related to inflammation was investigated using the human inflammation panel (Nanostring).
Capsule morphological features
The morphological features of the 41 PPEs-matched PPCs were compared with those obtained from 27 women without PPE that was considered as a ‘negative control’ group.
All PPCs were composed by a fibrous extracellular matrix (ECM) that enclosed both round and fibroblast-like spindle cells and were lined by synovial metaplasia (SM) on the side facing the expander in 83% (34/41, PPEs group) and in 67% (18/27, negative group) respectively. Furthermore, both PPEs-matched PPCs and negative control group, showed the presence of a variable quantity of infiltrating lymphocytes (Figure 3) with a trend towards a higher frequency in PPCs with associated PPE with respect to those without.
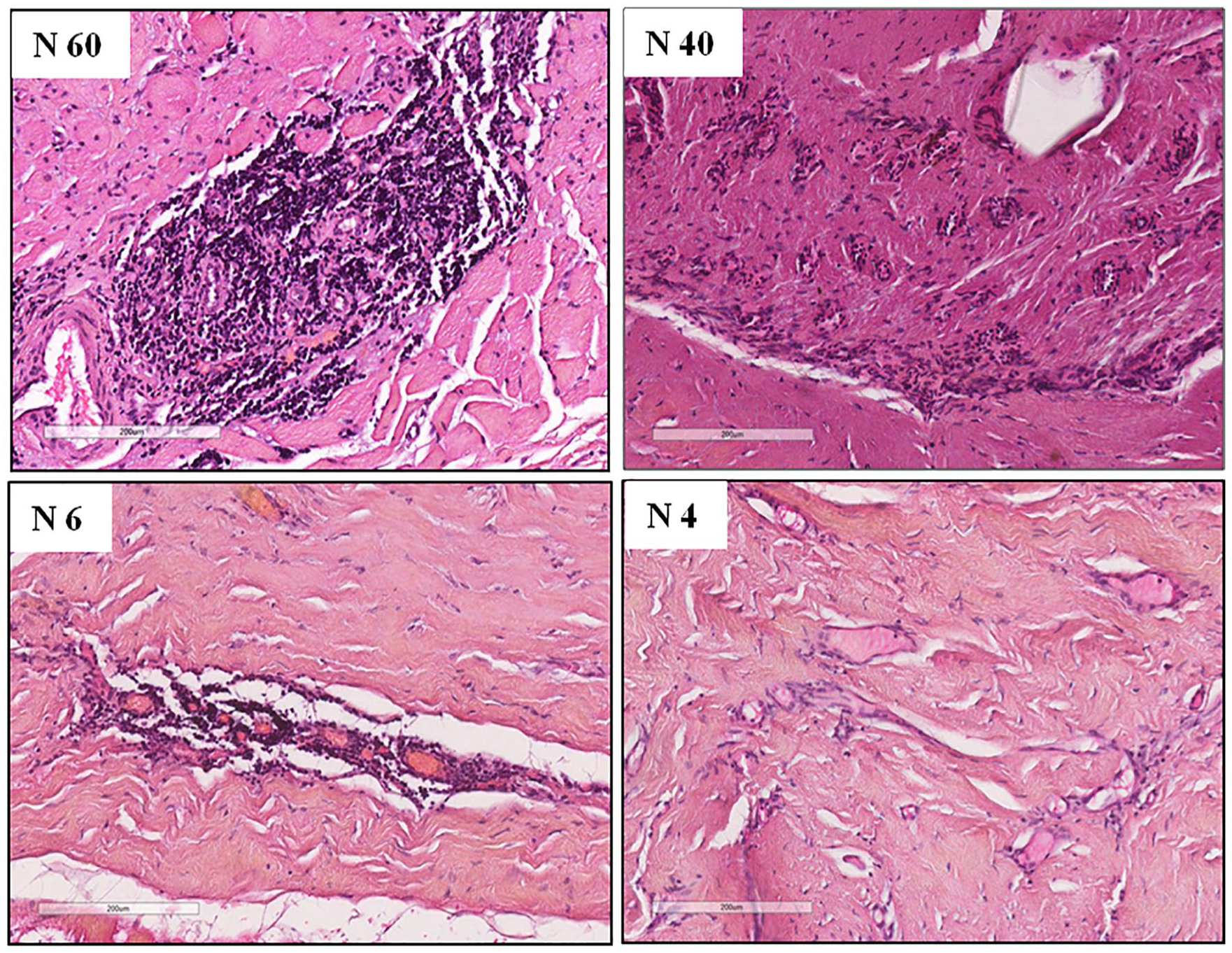
Ranges of lymphocytes infiltration in periprosthetic capsules (PPCs). Figure shows four representative cases of the lymphocytes infiltration spectrum in PPCs ranging from strong (sample n 60, Tables 1 and 2), median (sample n 40, Tables 1 and 2), low (sample n 6, Tables 1 and 2) and focal (sample n 4, Tables 1 and 2). The images were acquired by Aperio Scan Scope (version 12.4.3.5008) (Leyca Bio System) using 4x amplification objective. The Aperio electronic magnification was 14x in all samples.
IHC
The cell lineage was investigated through IHC in 22 PPCs (i.e., 11 PPCs for each group) selected to represent all the spectrum of lymphocytes distribution (Figure 3 and Table 2). Based on the results obtained by FC in PPEs, we focused our analysis on CD4, CD8, CD4 Treg and Th1.
IHC results.
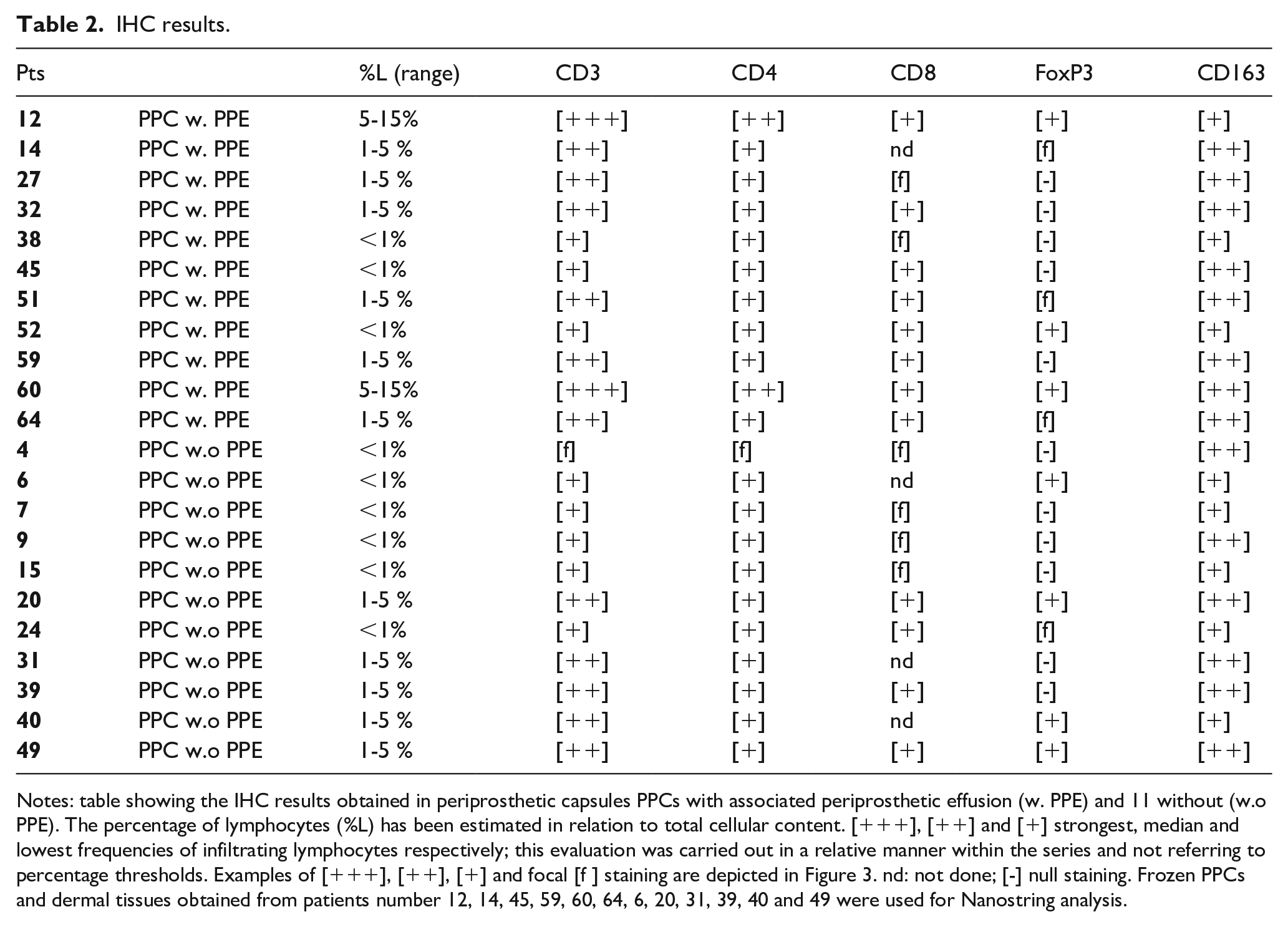
Notes: table showing the IHC results obtained in periprosthetic capsules PPCs with associated periprosthetic effusion (w. PPE) and 11 without (w.o PPE). The percentage of lymphocytes (%L) has been estimated in relation to total cellular content. [+++], [++] and [+] strongest, median and lowest frequencies of infiltrating lymphocytes respectively; this evaluation was carried out in a relative manner within the series and not referring to percentage thresholds. Examples of [+++], [++], [+] and focal [f ] staining are depicted in Figure 3. nd: not done; [-] null staining. Frozen PPCs and dermal tissues obtained from patients number 12, 14, 45, 59, 60, 64, 6, 20, 31, 39, 40 and 49 were used for Nanostring analysis.
In all PPCs, CD3 IHC confirmed the presence of T cells in lymphocytes clusters without significant differences in spatial distribution between groups with or without PPEs (Figure 4).
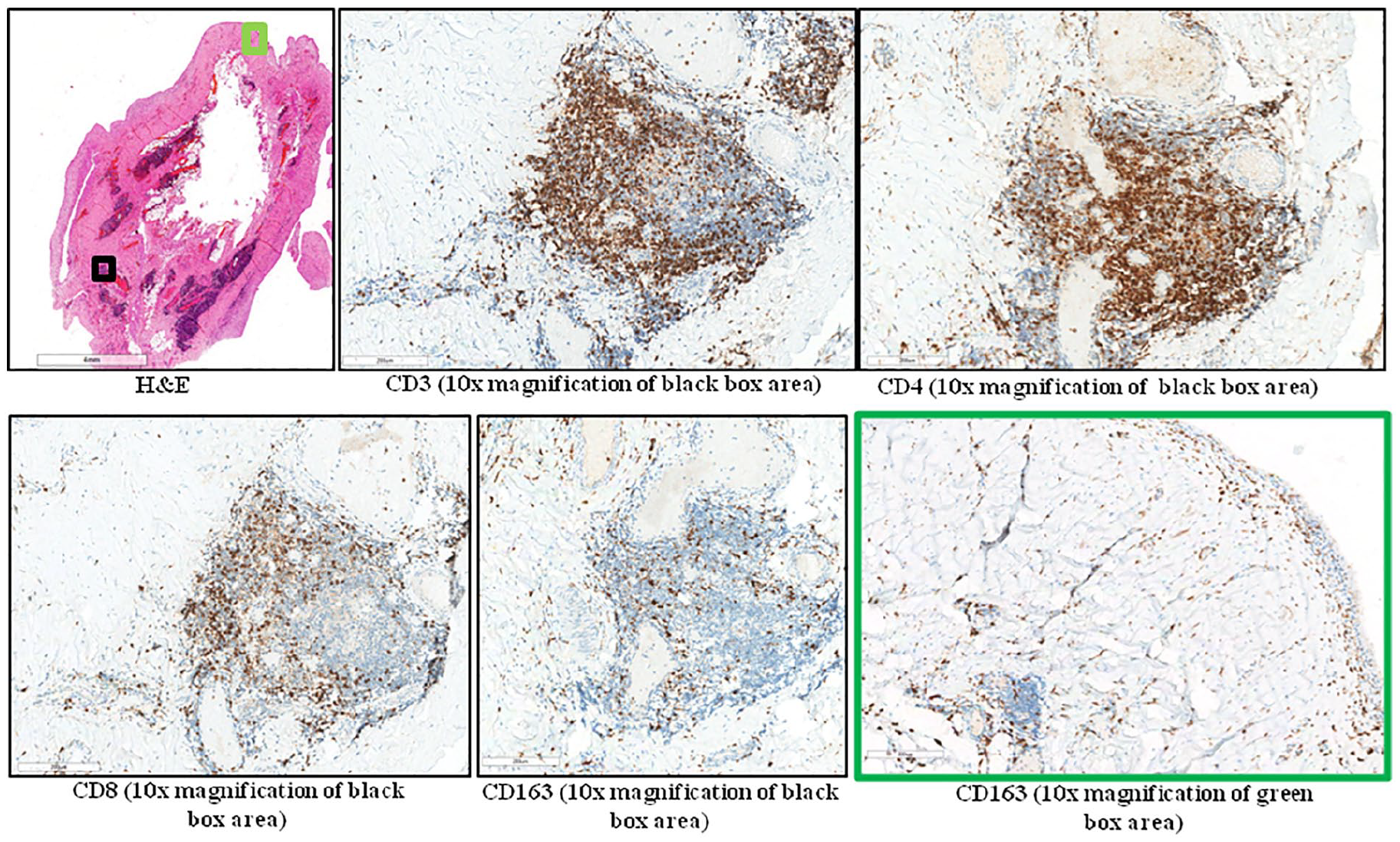
IHC results in a representative periprosthetic capsules (PPC). Figure showing IHC results in a representative case of PPC associated with PPEs (sample n 12, Tables 1 and 2). As can be seen from Hematoxylin & Eosin (H&E), lymphocytes were mainly aggregated near vessels along the boundary between capsule and muscle tissue and were positive for CD3, CD4 and CD8 staining. CD4 also decorated CD3- CD8- CD163- cells placed in the centre of aggregates likely constituted by small tissutal monocytes derived from blood. The same distribution of CD3, CD4 and CD8 staining was observed in PPCs not associated with PPEs. The CD163 positive cells were principally distributed below the capsule border or enclosed in ECM. The images were acquired by Aperio Scan Scope (version 12.4.3.5008) (Leyca Bio System) using 4x amplification objective. The Aperio electronic magnification for all the images is reported as indicated.
In both groups, the CD4/CD8 ratio was similar to those observed in PPEs with an augmented percentage of CD4 respect to CD8 (Table 2, Figure 4 and Supplementary Figure 4). Although two cases showed focal Tbet staining, the rest of the samples showed no reactivity. These results proved the absence of Th1-oriented microenvironment in PPCs. On the contrary, both groups showed FOXP3 positive staining proving the presence of tissutal CD4 Treg (Table 2 and Supplementary Figure 4). Finally, the proliferative index Mib1 (Ki67 nuclear staining) was generally very low (1% of lymphocytes), proving a non-proliferative state in the majority of samples (Supplementary Figure 5).
Given the absence of Th1 cells, we next focused on CD163 that has been proposed as a specific marker for M/M2 with anti-inflammatory phenotype.5,12 Our results showed that CD163 mainly decorated round cells in ECM or under the capsule border (Figure 4). Taken together, the IHC results pointed to a CD163-orchestrated pro-wound healing rather than a Th1 mediated pro-inflammatory microenvironment in all PPCs.
Nanostring analysis
Considering the fact that in addition to lymphocytes and M/M cells, a pro-inflammatory or a pro-wound healing microenvironment is the result of the cross-talk of various cell strains not all identifiable through IHC, we investigated the potential differences between PPE-associated and non-associated PPCs through the human inflammation panel (Nanostring). To this purpose, RNA was extracted from 12 frozen capsules of selected cases (i.e., six cases for each group). As described for IHC, PPCs were selected to represent all the spectrum of the immune infiltration (detailed in Table 2). A total of 23 genes resulted as differentially expressed in the PPE-associated vs PPE non-associated PPCs (Supplementary Figure 6), although none of them retained the statistical significance after FDR and Bonferroni adjustment.
Since for all 12 capsules, RNA was also obtained from their matched dermal tissue, a comparison between capsules and these tissues was also performed. The results showed 45 genes differentially expressed (Supplementary Figure 7) and after FDR adjustment, three genes maintained their significance: Complement C2 (C2) up-regulated in capsules, Cell Division Cycle 42(CDC42) and High Mobility Group Box 1 (HMGN1) all down-regulated in capsules respect to dermal tissues. Interestingly, with respect to control tissues, PPCs showed a TGF-B1 increase and IFN-y decrease. Even if both genes did not achieve statistical significance after FDR adjustment, their modulation supports the presence of a PPCs microenvironment likely similar to those present in normal dermal tissues used as a control.
Discussion
The presence of PPEs during breast reconstruction can cause early and late complications. We designed this study to explore the degree of inflammation in PPEs and their matched PPC obtained during the replacement of the Mentor expanders that have been recently introduced in our clinical practice after the recall of macro-textured ones (Allergan). 10 The results showed that M/M1 (HLA-DR+bright CD16+) and CD4+ IFNy+ Th1 lymphocytes increased in PPEs respect to corresponding PB. However, these pro-inflammatory cells were counterbalanced by the simultaneous gain of CD4+ IL-4+ Th2 and Treg cells. IHC and Nanostring-based comparison revealed no significant differences between capsules and matched dermal tissues. These results suggest the presence of Treg controlled inflammation in both PPEs and PPCs as well as the absence of pathological inflammation in PPCs.
Although the acute and chronic physiological inflammation that follows IBR allows the proper implantation of prosthesis to the body, if it persists for too long it may cause early and late complications. Accordingly, a recent paper confirmed that the presence of hematoma or seroma after breast augmentation increases the risk of CC. 13 A further late consequence of prolonged inflammation is the recently described BIA-ALCL, a rare tumor associated with textured breast implants. 8 The cytokine profile of BIA-ALCL specimens suggested the Th1/Th17 as possible cells of origin but, a Th2-mediated allergic response has also recently been proposed as a possible milieu for its development. 8
Even if the presence of few mL of PPEs was a common finding during the expander substitution with implant, their characterization after the introduction of Mentor expander is not reported in literature. Furthermore, although micro-textured implants should reduce complications compared to macro-textured ones, we also noticed an increase of PPEs volumes and frequency during the replacement of the Mentor expander compared to those encountered previously (i.e., BIOCELL).14-15 However, these increases are not quantifiable because, before the introduction of Mentor, the volumes and frequencies of PPEs were not recorded regularly.
Mentor POLYMsiltex is a non-porous micro-textured surface that has been reported to be inflammatory protective. 16 Biocell-macrotextured expander presents a significant microscopic depression in its architecture and induces rapid and aggressive tissue ingrowth through surface pores, whereas the Siltex-microtextured expander lacks porous features and causes a linear fibrotic patterned capsule. Using macrotextured expander, early initiation of tissue ingrowth and capsule formation in the periprosthetic space may reduce risks of prolonged drain placement and PPCs formation. 17
Since the presence of PPEs around expanders could be a cause of local inflammation, we explored the nature of the cells contained in PPEs, their matched PB and PPCs. Two main cell lineages were highlighted: the CD45+CD3+CD4+ high forward scatter (FSChigh) SSClow lymphocytes and the CD45+ CD14+ CD64+ HLA-DR+bright CD16+ SSChigh cells that most likely derived from circulating monocytes (that we named M/M1). The presence of heterogeneous CD14+ monocytic-like cells in seromas has already been reported,18,19 however to our knowledge, this is the first report in which their phenotype is widely described.
These findings are in line with the scarce presence of neutrophils and monocytes and support the notion that specific adaptive (lymphocytes mediated) and innate (M/M1 mediated) responses are ongoing in PPEs. Our results also showed a significant increase in CD4 with respect to CD8 and, within a subset of samples (i.e., core group), an increase of CD4 Th1 (IFN-γ positive), Th2 (IL-4+) and CD4 Treg. We also observed an increase in both CD127+ HLA-DR+bright CD4 TE and CD8 TE. These results suggest the overall presence of a pro-inflammatory environment in PPEs, sustained mainly by the presence of Th1 and M/M1 cells. Although according to recent literature Th1 are the key actors in defending the host against pathogens, 20 their presence in PPEs may be due to an activation loop between Th1 and M/M1 cells rather than to a bacterial infection. In fact, IFN-γ produced by Th1 can force naive M/M to acquire a M/M1 phenotype that in turn, by releasing IL-12, induces naive CD4 to become Th1 while few neutrophils were found in most PPEs. This circuit can also explain why Th1 showed higher frequencies than Th2 in PPE. An important limitation to this conclusion is that, although the frequency of M/M1, CD4 TE and CD8 TE has been evaluated in all samples, Th1 cells have been successfully detected only in the core group. Another important PPE feature was the increase in CD4+ CD127- CD25+ Treg cells compared to the corresponding PBs. It is widely recognized that Treg suppresses abnormal or excessive immune response to self- and non-self-antigens to maintain immune homeostasis by various mechanisms. 4 Since the frequency of Treg in PPEs was particularly high in most cases, we assumed that the pro-inflammatory environment induced by Th1-M/M1 activation loop is counterbalanced by the suppressive activity of Treg. The fact that the frequencies of Th1 in PPE exceeded 10% in only two cases further supports this hypothesis. These results are different from those reported by Pochert et al. 19 that evidenced a Th2 increase in seromas after a simple breast cancer mastectomy. However, the seromas analyzed by these authors were particularly abundant and recurrent, so it is difficult to compare their results with those obtained in our study.
We wondered if the PPEs environment, characterized by an increase in Th1, M/M1 and Treg cells was coupled with an unbalanced PPC development. To explore this, we chose to compare the PPE-matched PPCs, with a group of capsules obtained from women without PPE (negative control group) when replacing the expander with prosthesis. SM, that is rather common and peculiar form of metaplasia in the inner lining of the PPCs 21 was more frequent in capsules coupled with PPEs rather than without (83% vs 67% respectively). Although this finding could be explained assuming a ‘protective effect’ of SM from inflamed PPE, it has to be considered with caution since only a fraction of capsule has been sampled.
A trend towards increased frequency of CD3 infiltration has been observed in PPCs with associated PPEs. As observed in PPEs through FC, IHC confirmed the prevalence of CD4 over CD8 in PPCs. Although the presence of higher CD3 frequencies in PPCs with associated PPEs suggested a more inflamed microenvironment, this finding was not supported by either IHC staining nor Nanostring analysis. In fact, all samples showed the absence of TBX21 (i.e., the Th1 master transcription factor) 22 coupled with the presence of FOXP3 positive tissutal Treg and CD163 pro-wound healing M/M2 cells in both group of PPCs, supporting the notion that these results mirrored different time-points of capsule deposition rather than a different nature of inflammation. In particular, we might suppose that PPCs with lower CD3 frequencies are in a more advanced healing phase than those with higher CD3 frequencies. In line with this, Nanostring showed no difference between 249 inflammatory-related genes in both, PPEs matched PPCs vs negative control group and PPCs vs corresponding dermal tissues. Notably, although transcription levels of TGF-B1 and INF-y supported the presence of a M/M2 environment in PPCs in respect to dermal tissues, this difference did not retain a statistical significance after FDR adjustment. This might be due to the low number of PPCs (i.e., 12) or, more likely, to the fact that the PPCs chosen for Nanostring represented the entire spectrum of immune infiltration and thus constituted a heterogeneous case study.
These findings differ from those previously reported, where defective FOXP3 cells were unable to suppress a pro-inflammatory Th1/Th17 environment in PPCs. However, the PPCs analysed were obtained from women undergoing implant replacement due to capsular contracture (which justifies the presence of Th1/Th17), implant deflation, or aesthetic reasons. 23
The overall results obtained in the present work support the notion that a Treg controlled Th1-M/M1 inflammation might be ongoing in PPEs collected at the moment of expander replacement, that however is not coupled with pathologic inflammation in corresponding PPCs where, on the contrary, a CD163 mediated pro-wound healing activity can be highlighted. Lending further support to this conclusion is the fact that among women investigated in the present paper, no one reported any clinical sign ascribable to an in-progress CC during follow up after expander replacement.
Supplemental Material
sj-pdf-1-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-1-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Supplemental Material
sj-pdf-2-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-2-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Supplemental Material
sj-pdf-3-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-3-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Supplemental Material
sj-pdf-4-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-4-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Supplemental Material
sj-pdf-5-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-5-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Supplemental Material
sj-pdf-6-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-6-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Supplemental Material
sj-pdf-7-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-7-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Supplemental Material
sj-pdf-8-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-8-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Supplemental Material
sj-pdf-9-tmj-10.1177_03008916231189532 – Supplemental material for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization
Supplemental material, sj-pdf-9-tmj-10.1177_03008916231189532 for Periprosthetic effusions surrounding breast expander: a flow cytometric, immunohistochemical and molecular characterization by Laura Sala, Chiara Maura Ciniselli, Fabio Bozzi, Valeria Summo, Chiara Bonini, Silvia Brich, Alessia Bertolotti, Desiré Viola Trupia, Chiara Costanza Volpi, Sara Pizzamiglio, Biagio Paolini, Antonella Aiello, Giovanni Apolone, Paolo Verderio and Umberto Cortinovis in Tumori Journal
Footnotes
Availability of data
The data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant D126CU, supported by 5 per 1000 Funds – financial support for research 2014 (Ministry of University and Research).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
