Abstract
Introduction:
Evaluation of tumor response according only to dimensional criteria may underestimate treatment benefit in patients treated for metastatic renal cell carcinoma (RCC). In this study we evaluated the role of lesion enhancement modifications and Choi criteria in patients affected by renal cell carcinoma treated with immunotherapy.
Methods:
We collected data of 60 consecutive patients (with a total of 154 measurable lesions) treated with immunotherapy (nivolumab or ipilimumab plus nivolumab) at a single Institution. We evaluated tumour response using both RECIST1.1 criteria and Choi criteria at the first radiological assessment; we subsequently associated response with progression free survival and overall survival.
Results:
Choi criteria found a higher rate of objective response compared to RECIST criteria (38.3% vs 18.3%). An objective response according to both criteria was associated with longer progression free survival and overall survival. Response rate for Choi did not vary according to lesion site.
Conclusion:
Choi criteria seemed to be able to predict clinical benefit in a higher proportion of patients with renal cell carcinoma treated with immunotherapy than RECIST criteria. Partial response according to RECIST was confirmed as a predictor of longer progression-free survival and overall survival.
Introduction
In recent years many different therapies have been developed for treating metastatic renal cell carcinoma (mRCC). More specifically, tyrosine kinase inhibitors (TKIs) such as sunitinib, 1 pazopanib 2 or cabozantinib 3 and immunotherapies in the form of check-point inhibitors (CPIs) targeting the Programmed Death receptor 1 (PD-1), Programmed Death receptor 1 Ligand (PD-L1), or CTLA-4 (Cytotoxic T-Lymphocyte Antigen 4). Of these, nivolumab (an anti-PD-1) has been approved by regulatory authorities for the treatment of mRCC in second- or third-line (after a TKI) 4 or in first-line in combination with ipilimumab (anti-CTLA-4). 5 Moreover, the combination of TKI and CPIs (nivolumab, pembrolizumab, avelumab) in clinical trials recently demonstrated a major benefit in overall survival (OS)6,7 and progression free survival (PFS)6 -8 versus TKI monotherapy.
The introduction of CPIs in clinical practise brought new challenges in the tumor response evaluation, because the traditional Response Evaluation Criteria in Solid Tumors (RECIST) 9 seemed to be insufficient for a precise assessment, due to unusual response patterns observed in clinical trials such as pseudoprogression, hyperprogression or dissociated responses. 10 For this reason, new criteria have been studied, such as the immuno-related response criteria irRC 11 and in 2019 the RECIST working group published new guidelines about the immuno-RECIST (iRECIST) 12 (Table 1).
Characteristic of principal radiological criteria of response assessment (RECIST, immune-RECIST and Choi).
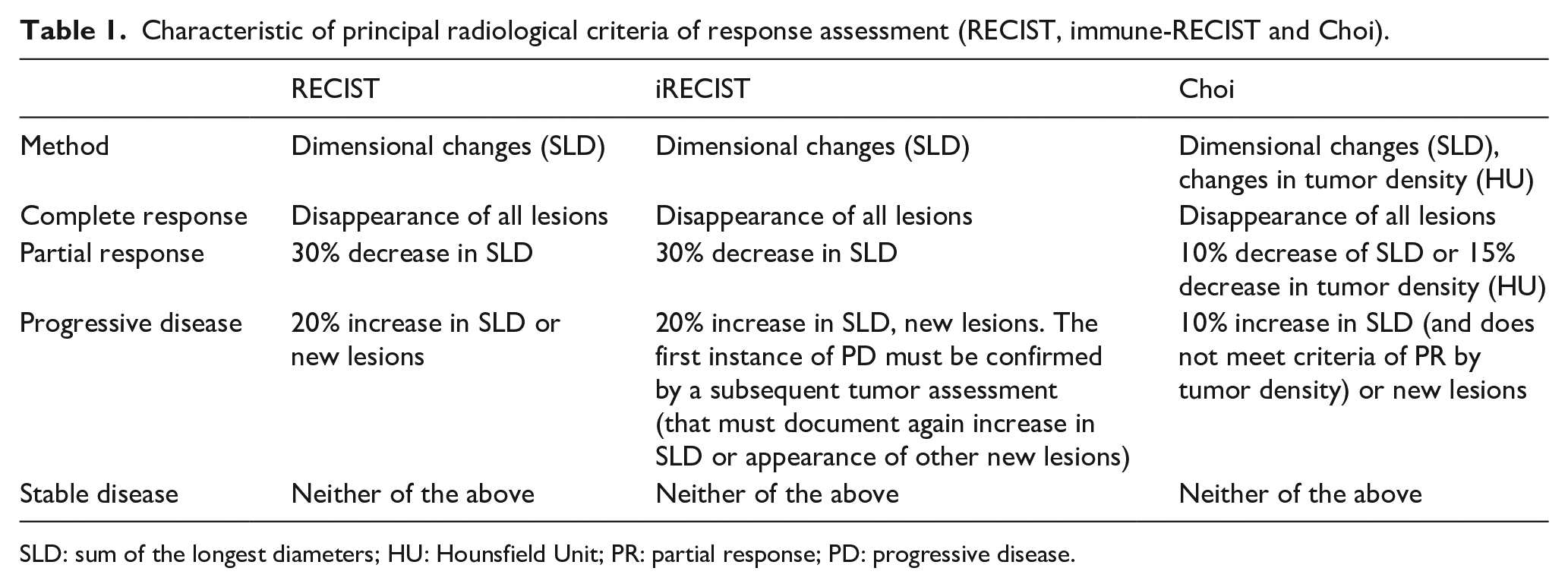
SLD: sum of the longest diameters; HU: Hounsfield Unit; PR: partial response; PD: progressive disease.
However, both the RECIST and iRECIST consider only a dimensions of target lesions assessed with computerized tomography (CT) or magnetic resonance (MR): only the maximum diameter of the target lesions is considered, together with the presence or absence of non-measurable lesions. None of these criteria consider any non-dimensional assessment. In 2007, Choi et al. demonstrated the prognostic value of new criteria for evaluating gastro-intestinal stromal tumors (GIST) treated with Imatinib with contrast-enhanced CT scan, assessing not only the diameter of target lesions, but also the enhancement modifications (Table 1). 13
Renal cell carcinoma (RCC) is a highly vascularised neoplasm in which neo-angiogenesis plays a paramount role. 14 Therefore, the commonly used contrast-enhanced CT scan is one of the best examinations for the diagnosis and monitoring of RCC, because tumoral masses usually show increased enhancement compared to benign ones. 15
This fact led some researchers to evaluate different criteria for tumor response for mRCC, to take into consideration not only the dimensions of target lesions, but also modification of the enhancement, which should represent the modifications in tumor vascularization, and in particular the Choi criteria.16-18 Those studies showed a prognostic role of Choi-assessed response, but have been conducted mainly in patients with mRCC treated with TKI, which have a specific inhibitory action on neo-angiogenesis. On the other hand, Choi criteria have been studied for evaluating response patterns in patients affected by advanced melanoma treated with immunotherapy showing that both the increase in diameter and in density (measured in Hounsfield Units - HU) were significantly associated with a higher hazard of dying. 19
The aim of this study was to evaluate the role of enhancement modifications and of Choi criteria in assessing tumor response in patients with mRCC treated with immunotherapy.
Methods
All consecutive patients with mRCC treated with immunotherapy at Istituto Oncologico Veneto in the first (ipilimumab plus nivolumab), second and third line (nivolumab monotherapy) were retrospectively evaluated. Inclusion criteria included histological diagnosis of RCC, treatment with immunotherapy (but excluding TKI-CPI combinations), availability of the contrast-enhanced CT scan images at the baseline and at the first evaluation. Patients who were evaluated with MR or for whatever reason had not done a contrast enhanced CT scan of the thorax, abdomen and pelvis at baseline and at the first assessment were excluded. Clinical data were collected from the clinical charts in an electronic, anonymized database. The baseline and first assessment CT scan were blindly evaluated by an experienced radiologist (EB). Target lesions were described for each CT scan according to the RECIST 1.1 criteria and the Choi criteria. Thus, a tumor response assessment was retrospectively, yet blindly, performed for each patient according to both RECIST and Choi criteria. Radiologic data (maximum diameter and enhancement in the venous phase measured in HU) for all target lesions were acquired.
We compared outcomes of patients according to RECIST and Choi response assessments. PFS was calculated from start of treatment to disease progression (radiological or clinical) or censored at the last follow-up date known without progression. OS was calculated from the start of treatment to death for any cause. Association between nominal variables was analyzed with Chi-squared test, Fisher's exact test or exact test of goodness of fit, whenever appropriate due to the numerosity of subgroups. PFS and OS with Kaplan-Meier method and compared when feasible with log-rank test and Cox proportional hazards method for multivariate analysis. All statistical analyses have been carried out with ‘R’ v.3.6.1. and its ‘survival’ package v.2.44–1.1. At the time of their first visit to our institution, all patients gave their written consent for the use of their clinical data for scientific purposes. The study was conducted according to the Declaration of Helsinki. Data collection was approved by the local Ethical Committee.
Results
Patients’ characteristics and survival outcomes
At the data cut-off, 60 patients were eligible with a total of 154 target lesions. The majority of patients were male (n=42; 70%), median age was 65 years (range=39-83). According to the International Metastatic RCC Database Consortium (IMDC) score, 20 17 patients (28.3%) were classified as good, 36 (60%) as intermediate and four (6.6%) as poor risk. Patient clinical characteristics are shown in Table 2. Most patients received nivolumab monotherapy in the second or third line after a TKI (76.6%), while 14 (23.3%) were treated with the combination of nivolumab and ipilimumab as first line therapy. Median time from the first CPI administration to the first radiologic evaluation was 3.8 months; median follow up time was 40.8 months (range=32.2-56.5). In the whole cohort, median progression free survival was 6.3 months (95% CI=4-16.6 months) and median overall survival was 32.7 months (95% CI=24.1 months – not reached).
Patients’ clinical characteristics at the start of immunotherapy.
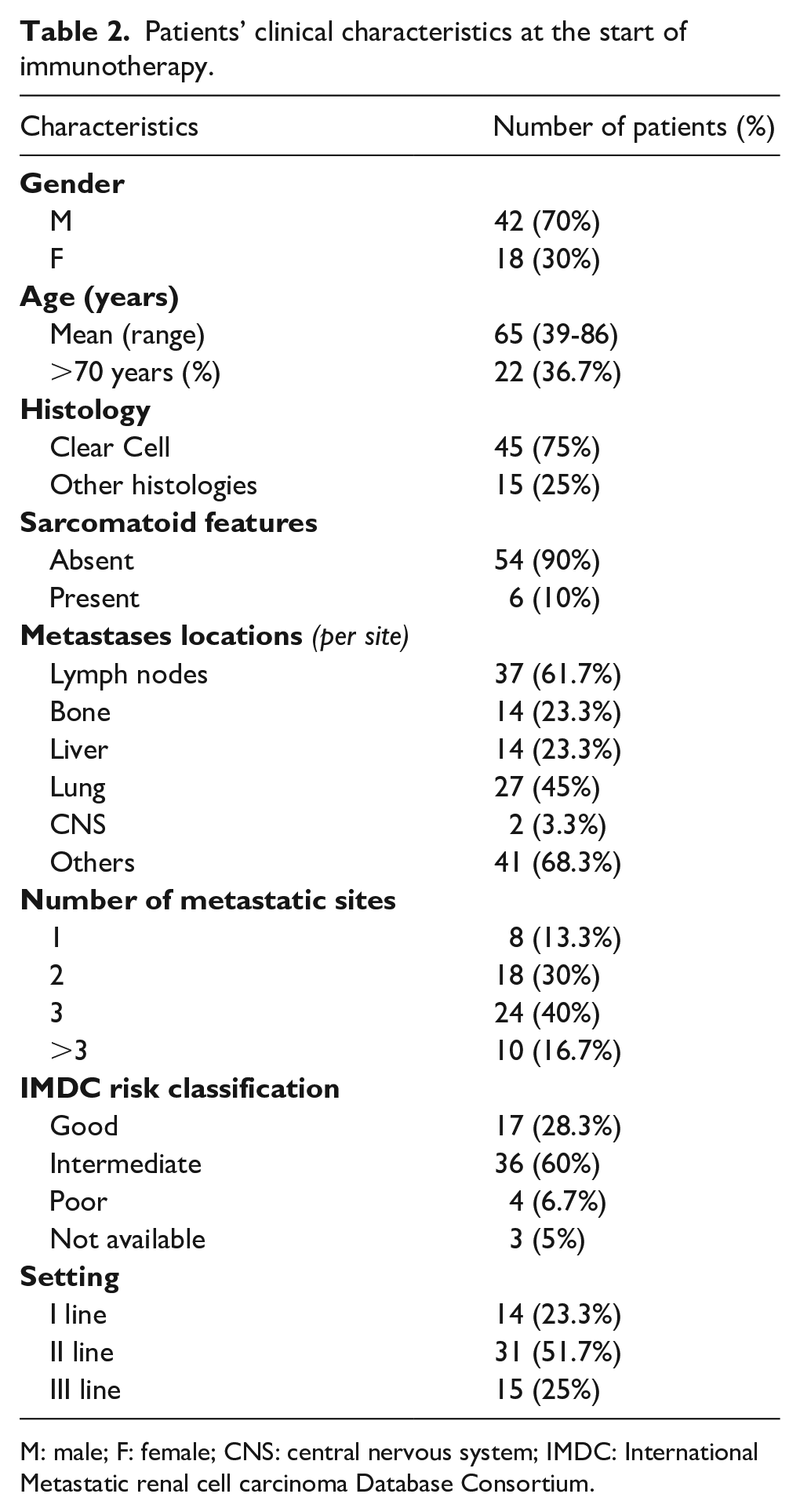
M: male; F: female; CNS: central nervous system; IMDC: International Metastatic renal cell carcinoma Database Consortium.
Response assessment
In the whole cohort, no patient achieved a complete response according to either Choi or RECIST. According to RECIST1.1, the first CT scan documented partial response in 11 patients, stable disease in 31 patients, disease progression in 18 patients. At the same time point, according to Choi criteria, 23 patients achieved partial response, 11 stable disease, 26 disease progression. In the whole cohort, for 39 patients (65%) the two methods were consistent in defining response (Table 3); for the other 21 patients (35%), there was a discordance in response defined using RECIST and response according to Choi. Results are reported in Table 3.
radiological response as assessed using RECIST 1.1 or Choi criteria.

RECIST: Response Evaluation Criteria in Solid Tumors; pts: patients.
Correlation between response and survival outcomes
An objective response according to both evaluation criteria was associated with longer PFS and OS. All the patients (11, 18.3%) achieving partial response according to RECIST 1.1 at the first CT scans are still alive and in follow-up at the time of data cut-off (median OS not reached at data cut-off time). Conversely, non-responders according to RECIST 1.1 showed a median OS of 24.6 months (
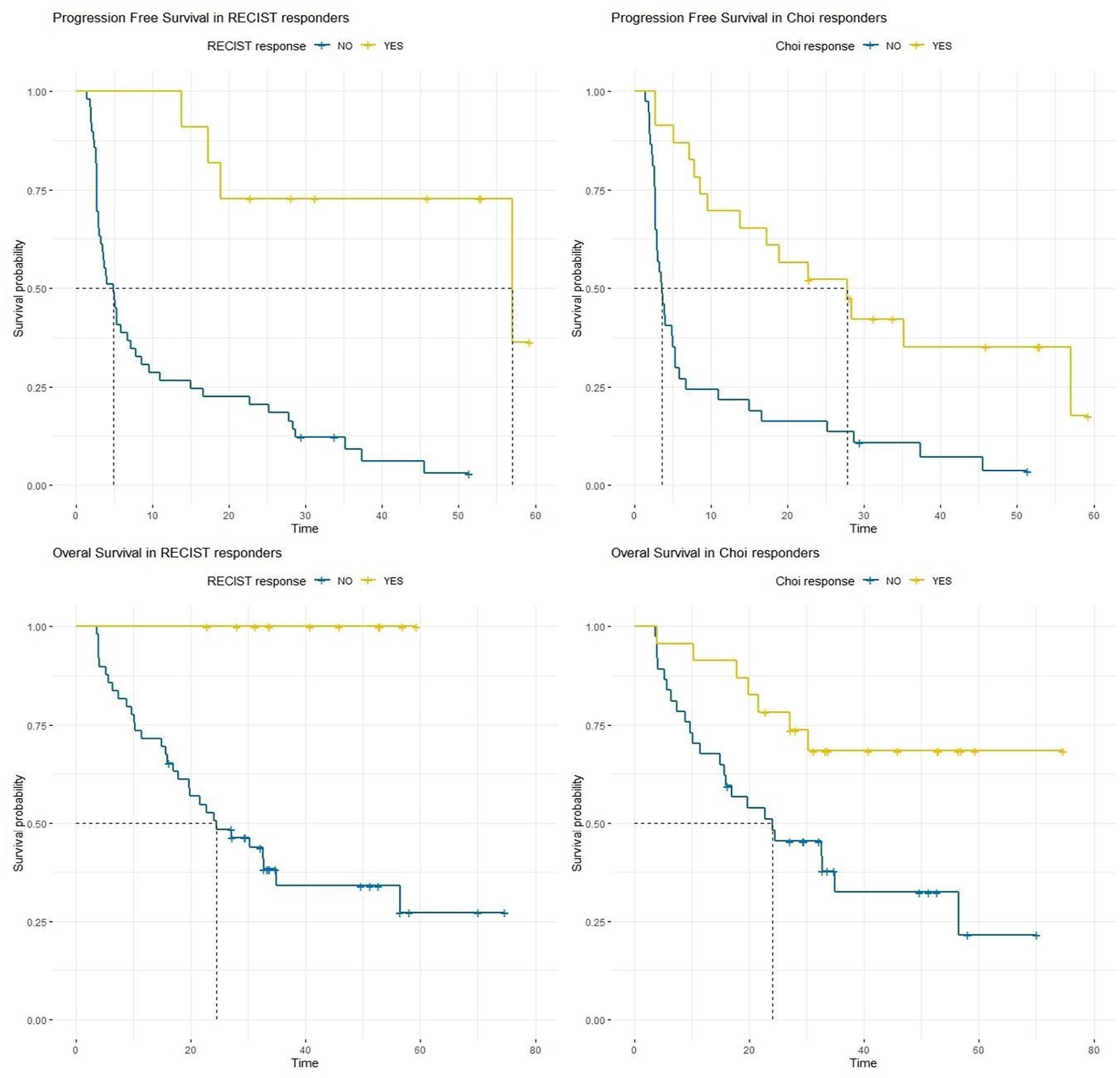
Correlations between response according to RECIST (left charts) and Choi (right charts) with PFS (top) and OS (below).
Objective response rate was higher if evaluated with Choi criteria: 23 patients were classified as responders at first radiologic examination (38.3%). Median OS of responders according to Choi was not reached at time of data cut-off, versus 24.1 months of non-responders (
Lesions responses
Out of 154 target lesions, 18 (11.7%) were localized in the liver, 38 (24.7%) in lymph nodes, 29 (18.8%) in the lung or pleura, 24 (16.2%) in the kidneys or adrenal glands, 25 (16.2%) in the soft tissues and 20 (12.9%) in other organs such as pancreas, thyroid or peritoneum. At the first CT scan, 31 lesions (20.1%) showed a dimensional response of at least 30% of the baseline maximum diameter. On the other hand, 49 lesions (31.8%) demonstrated an enhancement reduction (15% or more reduction in HU). Twenty-four out of the 31 lesions with a dimensional reduction (77.4%) also showed a lower enhancement (an example is reported in Figure 2). Dimensional responses vary depending on the metastasis location, with higher response rate for the lesions in the liver (33.3%) and the lungs (37.9%) (
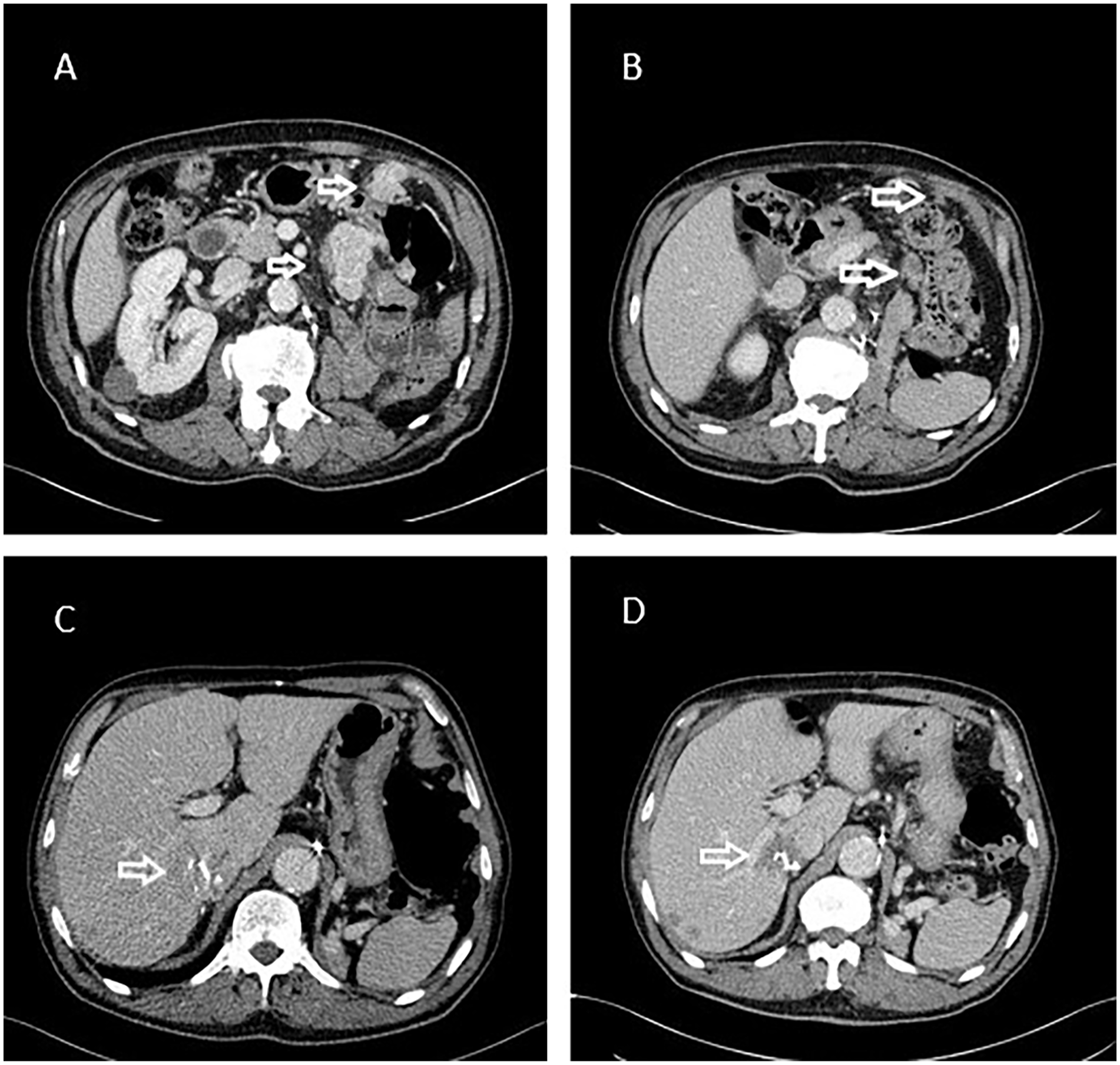
Images of the pre- and post-treatment CT scans of an 82-years old man treated with nivolumab. Baseline CT scan (A, C) showed and two peritoneal nodules (white arrows) and a liver mass. After 14 weeks of treatment (B, D) all the lesions showed, alongside with a dimensional response, a marked reduction in attenuation.
Identification of long-responders and short responders using Choi and RECIST criteria
In the whole cohort, 24 patients (40%) were able to achieve a PFS of more than 12 months. Of those patients, 11 were identified as responders with RECIST criteria at the first evaluation and 16 would have been classified as responders with Choi criteria at the same time point (sensitivity of 66.7%
Discussion
In our cohort, a radiological response assessed with both Choi and RECIST 1.1 criteria was associated with better PFS and OS in patients treated with immunotherapy for renal cell carcinoma.
Lately, many authors have suggested that lesion maximum diameter evaluation alone can underestimate response to novel systemic therapies in patients affected by RCC. Due to the specific mechanism of action of anti-VEGF TKI, one of the first parameters that underwent evaluation was the change in tumor attenuation as a marker of reduction in tumor vascularization and, therefore, drug activity. van der Vald et al. found that Choi criteria could predict, at an early point, patients that could benefit from treatment with Sunitinib. 16 Smith et al. proposed the size and attenuation CT (SACT) and subsequently morphology, attenuation, size, and structure (MASS) criteria, which evaluated changes in dimension, density and morphology of tumoral lesions at CT scan;21,22 the complexity of the method, however, could be problematic in every-day clinical practice.
To the best of our knowledge, this is the first attempt to evaluate changes in tumor density during treatment with immune checkpoint inhibitors for metastatic RCC. Khoja et al. found that response assessed with CHOI criteria was associated with better OS in patients treated with pembrolizumab for metastatic melanoma. 19
In our cohort, the proportion of patients with a significant tumor attenuation reduction, albeit inferior to that reported by other authors during the treatment with TKI (for instance, Van der Veldt et al. 65%, 16 Smith et al. 72% 22 ) was significantly higher than the proportion of patients with radiological response according to RECIST1.1. We think that, due to the peculiar pathogenesis and biology of RCC, being a highly vascularised neoplasm, a reduction in tumor density could arise not only directly from the antiangiogenic action of TKI, but also from the necrosis and tumor growth inhibition provided by any type of systemic treatment. In Figure 2, the example of a male patient that experienced, alongside with a dimensional response of the target lesions, a marked reduction of the attenuation of the tumor, is reported.
In our study, by using Choi criteria we were able to identify more patients that achieved a long-term control of the disease compared to RECIST criteria alone; on the other hand, many responders according to Choi criteria did not achieve a long PFS, whereas all patients with a PR according to RECIST experienced a long disease control.
Many questions remain in the interpretation of the effects of the treatment with immunotherapy in solid malignancies. RECIST 1.1 criteria, that are currently used in all clinical trials, can underestimate response to this type of treatment and have difficulties interpreting novel patterns of response like pseudoprogression or dissociated response. 23
An improvement in the interpretation of radiological response could also help in clarifying the optimal duration of the treatment with immunotherapy, as well as improve the selection of patients that could be treated beyond progression with clinical benefit. In fact, despite in many clinical trials the treatment with anti-PD1 were continued until disease progression,4,5 evidence from studies in other solid malignancies suggests that some patients, after getting a response, could safely discontinue the treatment with a limited risk of subsequent progression; 24 the optimal way to identify those patients, however, is unclear. 25 Another unclear area is the optimal selection of patients that could benefit from a treatment with immunotherapy beyond progression: for instance, in the CheckMate 025 trial 26 48% of patients that progressed to nivolumab continued treatment beyond progression, with the majority of those (59%) achieving subsequent disease control with a median OS of 28.1 months. We believe that improvement in the evaluation of response with radiological exams would reflect in better decision-making in everyday clinical practice; in this field, a significant contribution can come also from artificial intelligence, radiomics analyses and software for automatic analysis of radiological images. Shu et al. showed that radiomic models are able to predict, with a good diagnostic accuracy, Fuhrman grade of clear cell RCC from preoperative CT scans. 27 Smith et al. developed computer software able to automatic quantify the vascular tumour burden (VTB, defined as the sum of the areas of tumor lesions exhibiting enhancement values within specific thresholds) from CT images of patients affected by RCC, with very good inter-observer agreement. 28 Analysing 275 patients treated with sunitinib, they showed that changes in VTB (from baseline to the first post-treatment CT scan) were able to identify patients with significantly different PFS. 28 Moreover, the authors showed that inter-observer agreement for assessment of percentage changes in tumor attenuation was poor. 28
As reported by other authors, 16 even Choi criteria have many limitations. Whereas evaluation of dimensional changes of CT scan is part of routine practice in every oncological radiology department, radiologists must be trained on these new criteria, as well as in a homogeneous method of measurement of lesion density in order to achieve reproducible results. Measurement of enhancement of non-homogeneous lesions (like for instance those with intralesional calcification, or with contrast enhacement (CE) values that differ from CT slice to slice) could be challenging, thus increasing the potential inter-observer (and even intra-observer) variability. 28 The original work from Choi et al. excluded lesions smaller than 15 mm (whereas the limit for RECIST 1.1 criteria is 10 mm, except for lymph nodes); 13 differences in scanning protocol or in timing of infusion of iodinated contrast could also change the apparent density of the same lesion. 16
This study has many limitations. The CT scans were not performed in the same radiological department and, therefore, not with the same scanner. Although all images were evaluated by a single radiologist (blinded to the oncologic outcome of the patients), there was no blinded second review. Due to the low sample size, we were unable to compare survival for patients with PD per RECIST but response per Choi (only one patient achieved such a discordant response); due to lack of information regarding the follow up of some patients, we were unable to compare PFS2. Patients enrolled were treated in different lines of therapy. In addition, all clinical data were collected retrospectively.
Conclusion
In conclusion, conventional RECIST 1.1 may underestimate the benefit of immunotherapy in patients treated with metastatic renal cell carcinoma. Choi criteria, despite some intrinsic limitations in the application in everyday clinical practice, taking into account changes in tumor density could help identify patients with a better prognosis and a longer response. Further research is urgently needed to better understand the underlying mechanisms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received "Ricerca Corrente" funding from the Italian Ministry of Health and funding from Consorzio per la Ricerca Sanitaria CORIS, Padova to cover publication costs.
