Abstract
The cancer stem cell model hopes to explain solid tumour carcinogenesis, tumour progression and treatment failure in cancers. However, the cancer stem cell model has led to minimal clinical translation to cancer stem cell biomarkers and targeted therapies in solid tumours. Many reasons underlie the challenges, one being the imperfect understanding of the cancer stem cell model. This review hopes to spur further research into clinically translatable cancer stem cell biomarkers through first defining cancer stem cells and their associated models. With a better understanding of these models there would be a development of more accurate biomarkers. Making the clinical translation of biomarkers into diagnostic tools and therapeutic agents more feasible.
Keywords
Introduction
When the cancer stem cell hypothesis first emerged in the field of haematology it captured the imagination of many solid tumour oncologists and scientists. This hypothesis provided an explanation for the development of solid tumour cancers, more importantly it provided a target for scientists and doctors to focus on when advancing existing treatments.
Unfortunately, there remains a relative paucity of clinically translatable drugs or therapies despite many years of basic science and clinical research into solid tumour cancer stem cells (CSC). Multiple reasons may have contributed to this, but an important reason was the unclear definition of stemness in solid tumours. An unclear definition of stemness’ defining traits complicates CSC isolation in experiments and impacts development of biomarkers for targeted therapies.
This unclear definition of stemness in solid tumours may in part reflect a natural situation whereby not all cancers subscribe to the classical CSC hierarchical model seen in haematology. Therefore, it is important to re-examine the definition of CSC and their associated hypothetical models. At its most basic is to first understand the concept of a stem cell. Stem cells are defined as self-renewing cells that can generate differentiated daughter cells. Three main groups of stem cells exist: totipotent, pluripotent and multipotent stem cells. Their degree of differentiation can be seen in the Venn diagram (Figure 1).
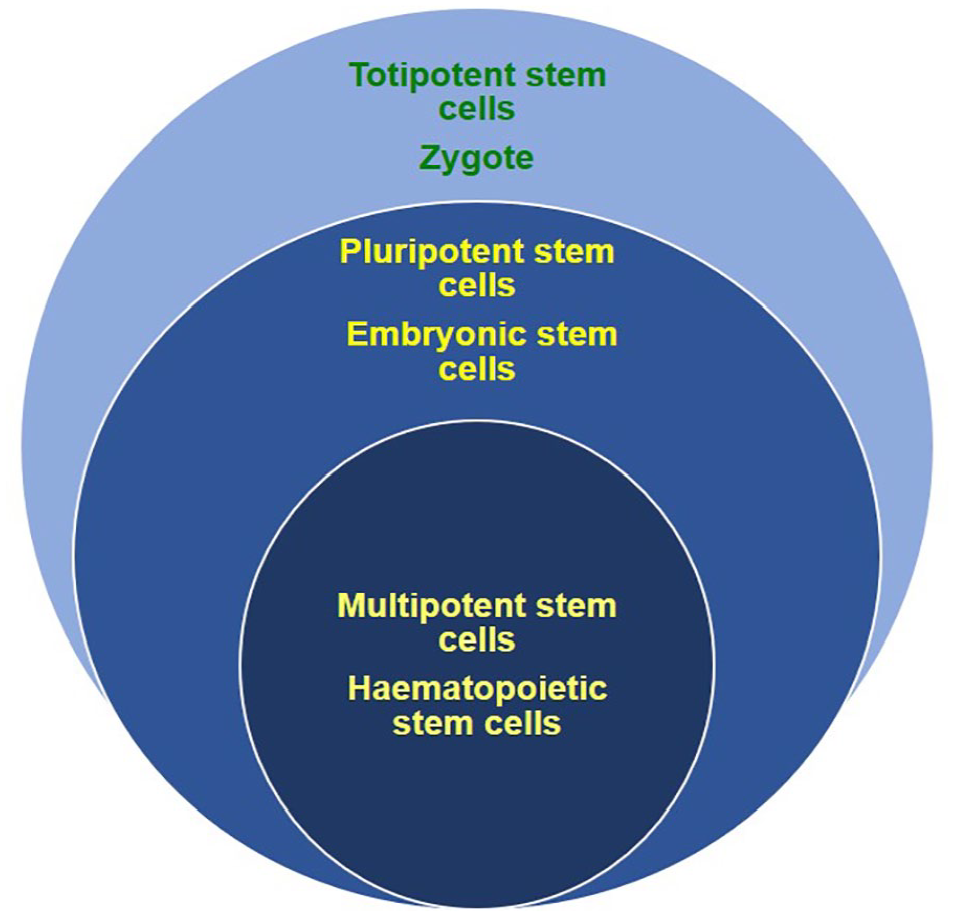
Diagram indicating degrees of differentiation possible for each stem cell type. Totipotent stem cells are represented by the largest subset as they can differentiate and reconstitute embryonic and extra-embryonic tissues. A slightly more differentiated cell is the pluripotent stem cells that can only differentiate into embryonic tissues which still implies it can regenerate any tissue or organ in an organism. Multipotent stem cells however are more differentiated and are already committed to certain cell lineages for specific cell lines, therefore it is represented by the smallest subset.
Challenges of defining an ideal CSC model
The classical CSC model derived from haematology states that cancer cell populations are heterogeneous with a small subset of cancer cells providing a reservoir for tumour self-renewal. 1 An important point to highlight is that CSCs must not only possess the capability for tumour initiation but also self-renewal which is not the case for tumour initiation cells. 2 Analogous to normal stem cells providing differentiated cells for tissue and organ renewal, CSCs provide malignant tumours with cancer cell progeny. These CSCs possess the ability to divide asymmetrically to ensure long term clonal maintenance of the CSC pool while providing progeny which can differentiate into non-tumorigenic ‘progenitor’ cancer cells which compose the majority of a tumour. 1 This model may explain the reasons for tumour resistance and cancer recurrence despite aggressive chemotherapy and surgery. Current therapies may be insufficient to eradicate CSCs which form the source of tumour cells. 1 Despite the attractiveness of the theory, several studies have shown that isolating solid tumour CSC clinically and experimentally remains problematic. Based on this model the key feature of CSC is its ability to reconstitute their parent tumour in vivo, hence the gold standard when experimentally defining CSCs is through serial xenotransplantation of cultured or primary cancer cell lines. However experimentally isolating CSCs proves challenging with presumptive CSCs proving elusive to detection. This is due to the phenotypic plasticity of CSCs which can limit the sensitivity and utility of CSC markers. For example, primary cancer cells were able to reconstitute their parent tumour in vivo and also possess the capacity for self-renewal and generate subsequent tumour growth. This was seen in breast cancer, glioblastoma, colon cancer and melanoma cell lines irrespective of their CSC marker positivity. 3 This observed phenomenon could be due to the assumed solid tumour CSC behaviour not adhering to the strictly defined classical CSC model. Historically, the CSC model was derived from malignant haematology, and this process reflects a dysfunctional form of haematopoiesis, whereby a strict hierarchy of rare CSC populations serves as the tumour source. Arguments for such a model were seen in experiments where multipotent stem cells progressively acquired mutations or epigenetic alterations that converted them into CSCs. 4
To explain such incongruence with the expected classical CSC model, alternative models have been proposed to explain tumour origin, maintenance, progression, and heterogeneity. One such example is the stochastic or clonal evolution (CE) model.5,6 The CE model states that tumours contain genotypically and phenotypically heterogenous subpopulations. Thus, when various selection pressures are applied this translates to different survival and replication rates amongst competing subpopulations. Over time, there is a selection for genetically and phenotypically advantageous traits, thereby altering the composition of tumour subpopulations and leading to an emergence of a dominant tumour subpopulation. The CE model partly explains the clinical phenomenon of chemotherapeutic resistance and the aggressivenes of recurrent cancers linking the observed heterogenity of tumour subpopulations and the tumour’s ability for self-maintenance. These heterogenous subpopulations are able to continually support tumour growth as chemotherapeutic agents select for dominant subpopulations that are able to maintain the tumour. More importantly is the presence of experimental evidence showing that external stressors such as chemotherapy can epigenetically induce non-CSC to acquire a CSC-like phenotype. Chemotherapy can alter the tumour microenvironment through hypoxia, oxidative stress and starvation, providing contextual signals for CSC conversion. In a patient-derived xenograft glioblastoma recurrence model, temozolomide increased the rate of single-cell phenotypic and functional conversion from a non-stem glioma cell into a stem-like state.7,8 It was believed that therapy-induced hypoxia-inducible factors (HIF) HIF1α and HIF2α were responsible for glioma cells acquiring CSC properties.7,8 Experiments such as these argue against a strict unidirectional CSC hierarchical model.
Using breast cancer as an example, based on studies of adolescent women exposed to radiation following the Hiroshima and Nagasaki atomic blasts, these young ladies had the highest susceptibility of breast cancer 20 to 30 years later compared with women of other age groups. 9 This suggests that adult mammary stem cells accumulate genetic changes that predispose to development of solid tumours. 9 Therefore, a model of tumour formation and development must account for breast stem cells as primary targets for mutation in the development of breast cancer. However, it is possible for early or late progenitor cells to also undergo malignant transformation through mutations or epigenetic change where they acquire CSC characteristics of self-renewal. Human CD44+, CD24-/low, Lin-expression breast CSCs, could form tumours when mice models were injected with as few as 100 cells, while 20,000 cells without this phenotype were unable to form a tumour when injected into the mice. 9
Another important disease model for solid tumour CSCs would be that of intestinal stem cells, intestinal epithelial cells and colorectal cancer. The intestinal lining consists of epithelial cells which are tightly regulated and yet have an immense turn-over capacity. 10 It is believed that intestinal stem cells reside in the bottom region of the epithelial cell crypts, while progenitors and differentiated cells are found more towards the top of crypts and villi. 10 The differentiated epithelial cells and their progenitors display great regenerative capacity upon injury due to their ability to easily switch between differentiation states. 10 It also appears that the microenvironment of the epithelial cells and intestinal stem cells are a contributing factor in their acquisition of malignant characteristics, for example an inflammatory environment installs stem cell like functionality and malignant transformation capacity of differentiated cells. 10 It is likely that the CSC state is not a fixed entity but a rather highly dynamic cellular state driven by environmental cues and intrinsic factors. 10
To help bridge the apparent gap between the classical CSC hierarchical model and the CE model. Hybrid models have emerged that blend aspects of both models. 11 One such model is the plasticity model as proposed by Marjanovic et al. 11 it developed from evolving evidence which supports a new model of tumorigenicity, in which considerable plasticity exists between the non-CSC and CSC compartments. This allows for non-CSCs to reacquire a CSC phenotype where there is a bidirectional, dynamic equilibrium between CSCs and their progeny. Therefore, a more plastic CSC model (Figure 2) with a fluid hierachy being more reflective of experimental and clinical findings. Hence the elusiveness of CSCs in some solid tumours may be explained by a refinement of the existing CSC model.
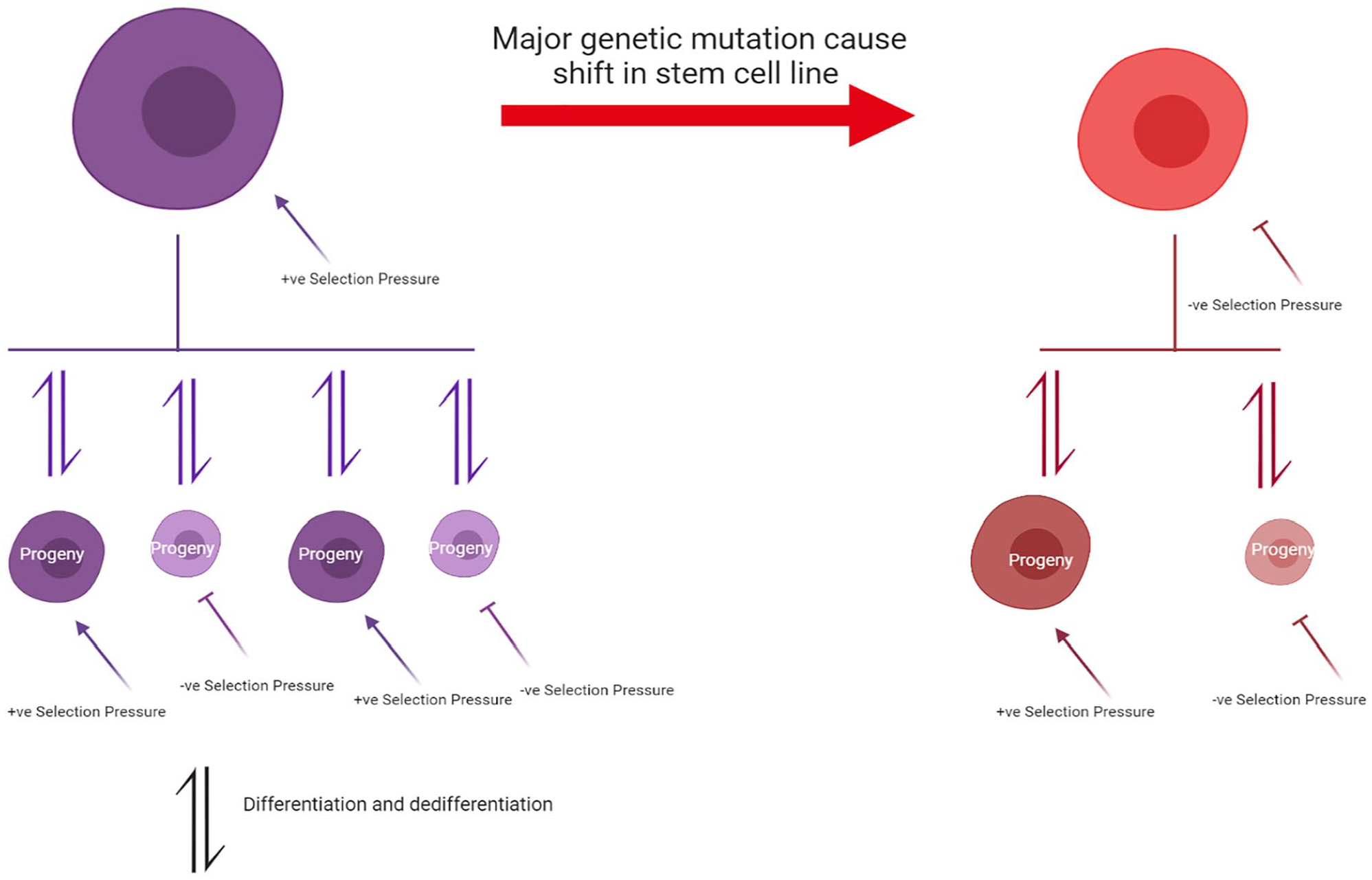
A revised CSC model that integrates stem cell hierarchy seen in the classical CSC theory while also incorporating elements of clonal evolution. Each clonal CSC acts as an evolutionary unit which experiences a selection pressure forcing it to undergo clonal evolution. CSCs with beneficial traits are better able to survive the extreme selection pressures and hence pass down these traits to their progeny. The selection pressure is consistently present and is exerted across all the clones, both the progenitor CSC and its progeny. The CSC also have the ability to differentiate and dedifferentiate to more primitive forms or committed tumour cell lines.
The author of this current study suggest that in the plastic CSC model, CSCs would serve as the primary unit of selection which incorporates CE mechanism of evolutionary selection, propogation and adapation. 12 The strict unidirectional hierachical CSC model may be more specific for haematological malignancies. In contrast, solid tumours may be suited to a plastic CSC model. With a more accurate model, it would be easier to understand specific solid tumour CSC traits.
Identifying CSCs via common properties with stem cells
Current evidence shows overlapping traits between CSCs and normal stem cells. Current pathways established to be important in the maintenance of self-renewal in stem cells have also been identified in pathways associated with oncogenesis. Examples of such pathways are Notch, Sonic Hedgehog (Shh) and Wnt signalling pathways. Identifying key common CSC pathways may be helpful for isolating solid tumour CSCs, as within a cancer type, there exists multiple levels of intra-tumoral heterogeneity. For example, exome and whole-genome sequencing of breast cancer have identified that the majority of these tumours have multiple driver mutations, with a large proportion of abnormalities affecting passenger genes. 13 The large variety of genetic abnormalities could cause the development of different subpopulations within each tumour with different combinations of genes for growth and survival within the tumour microenvironment. 13
Replicative immortality is a key characteristic of stem cells and may potentially be linked to CSCs. One possible explanation is that fewer mutations or epigenetic alterations may be required to maintain replicative immortality in stem cells rather than reactivating it in well-differentiated cells. Due to the long-lived nature of stem cells, there are more opportunities for normal stem cells to accumulate mutations or epigenetic alterations. This is relative to differentiated cells which presumably must undergo a process of dedifferentiation to become CSCs. Wnt signalling modulates stem cell/progenitor cell self-renewal in epidermal and gut progenitors, β-catenin a downstream activator of Wnt increases epidermal stem cell proliferation and decreases stem cell differentiation. 14 Mutation of the APC gene is thought to be responsible for familial adenomatous polyposis (FAP) a hereditary cancer syndrome. Mutation of APC leads to β-catenin becoming stabilised, forming adenomas via the Wnt signalling. 15 Through the Wnt signalling, the adenomas can acquire self-renewal capabilities and survive for many years allowing for accumulation of further mutations leading to colon cancer. 15
Another approach to consider is focusing on important CSC characteristics that facilitate tumour reconstitution when attempting to identify CSCs. Characterisation of key secondary traits of stemness involved in the CSC’s tumour reconstitution ability can unravel the underlying complex processes of CSCs tumour reconstitution and assist in solid tumour CSC isolation. For example, genomic instability has been noted in induced stem cells and are characterised by genomic deletions and amplifications. These alterations to the genome are believed to be due to oncogene-induced DNA replication stress which are significantly dependent on c-Myc expression. 16 Apoptosis resistance is another characteristic of CSCs. In a model using human non-small cell lung carcinoma CSC cell lines, gemcitabine was used to induce irreparable damage to CSCs’ genetic material but this had minimal effect on CSC death. This was likely due to the apoptotic resistance as a result of altered DNA damage response pathways in CSCs. 17 This apoptotic resistance was overcome by combining gemcitabine with a CHK1 kinase inhibitor that promoted CSC apoptosis. 17 CHK1 kinase is a key element in the DNA damage response pathway, it functions by inducing the S-phase checkpoint and causing cell cycle arrest and allowing time for DNA repair. Hence by inhibiting CHK1 there is a synergistic effect of overriding the checkpoint defence of CSCs against the lethal damage induced by DNA-directed chemotherapeutic agents. 17
Somatic cells under the right conditions are also able to dedifferentiate, and this cellular plasticity may be another trait to focus on when developing CSC biomarkers. As pluripotent stem cells differentiate, they undergo DNA methylation and chromatin remodelling which silences stem cell factors which regulate their pluripotency. However, the process of differentiation has been proved to be reversible through the transfection of somatic cells with the four Yamanaka factors Sox2, OCT4, Klf4, and c-Myc which have generated induced pluripotent stem (iPS) cells. 18 These four transcription factors are linked to a poorly understood network of regulators of chromatin which allow for reprogramming to occur. This network includes NANOG, transcription factors, Poly-comb complexes, microRNAs and histone modification enzymes. 19 Interestingly this phenomenon of dedifferentiation was also observed in cancer cells where by cancer cells possessed stem cell like properties after being exposed to hypoxic conditions. Subsequent experiments identified the presence of SOX-2, OCT4, KLF-4 in hypoxic glioma, lung cancer and hepatoma cells. 20 This phenomenon of dedifferentiation was characterised by sphere formation, asymmetric cell division and cell cycle arrest. This suggests that cancer cells have the capacity to acquire stem-cell traits under hypoxic microenvironments, through the reactivation of pluripotency-related genes. 20
Apart from simply focusing on the CSC itself it may be worthwhile to also better understand the malignant environmental niche that supports it. Better understanding of the niche would aid in identifying and separating bystander biomarkers from true biomarkers of CSCs. A stem cell niche is the surrounding microenvironment that nurtures the stem cell enabling it to retain its differentiation capacity. The niche consists of cells such as fibroblasts, endothelial cells and immune cells, and extracellular components such as matrices and secreted factors. These secreted factors bind to stem cell surface receptors stimulating intracellular signalling cascades directing stem cell fates by controlling self-renewal and differentiation. Likewise, in the case of cancer cells, it is postulated that a similar tumour microenvironment can also cause the conversion of stem cells into CSCs. 21 It is believed that a microenvironment that has chronic inflammation may enhance the production of cytokines, chemokines, growth factors, proangiogenic factors, and extracellular matrix-modifying enzymes. These factors are likely to induce signal transductions essential for cell survival and proliferation, facilitating chromosomal instability. Cytokines such as TNF, IL-1, and IL-6 and activated transcription factors like NF-κB and STAT3 have key roles in chronic inflammation but also play key roles in tumour initiation.22,23 A microenvironment of chronic inflammation can also induce epigenetic changes causing irregular activation or silencing of genes. 24 Some of these genes may critically contribute to tumour initiation resulting in cancer associated gene expression. 24 Chronic inflammation can also cause oxidative damage to a cell’s genetic material. Specifically NF-κB and STAT3, can induce parenchymal cells to produce excess amount of reactive oxygen species and reactive nitrogen species, which induces genomic instability and DNA mutations. 25
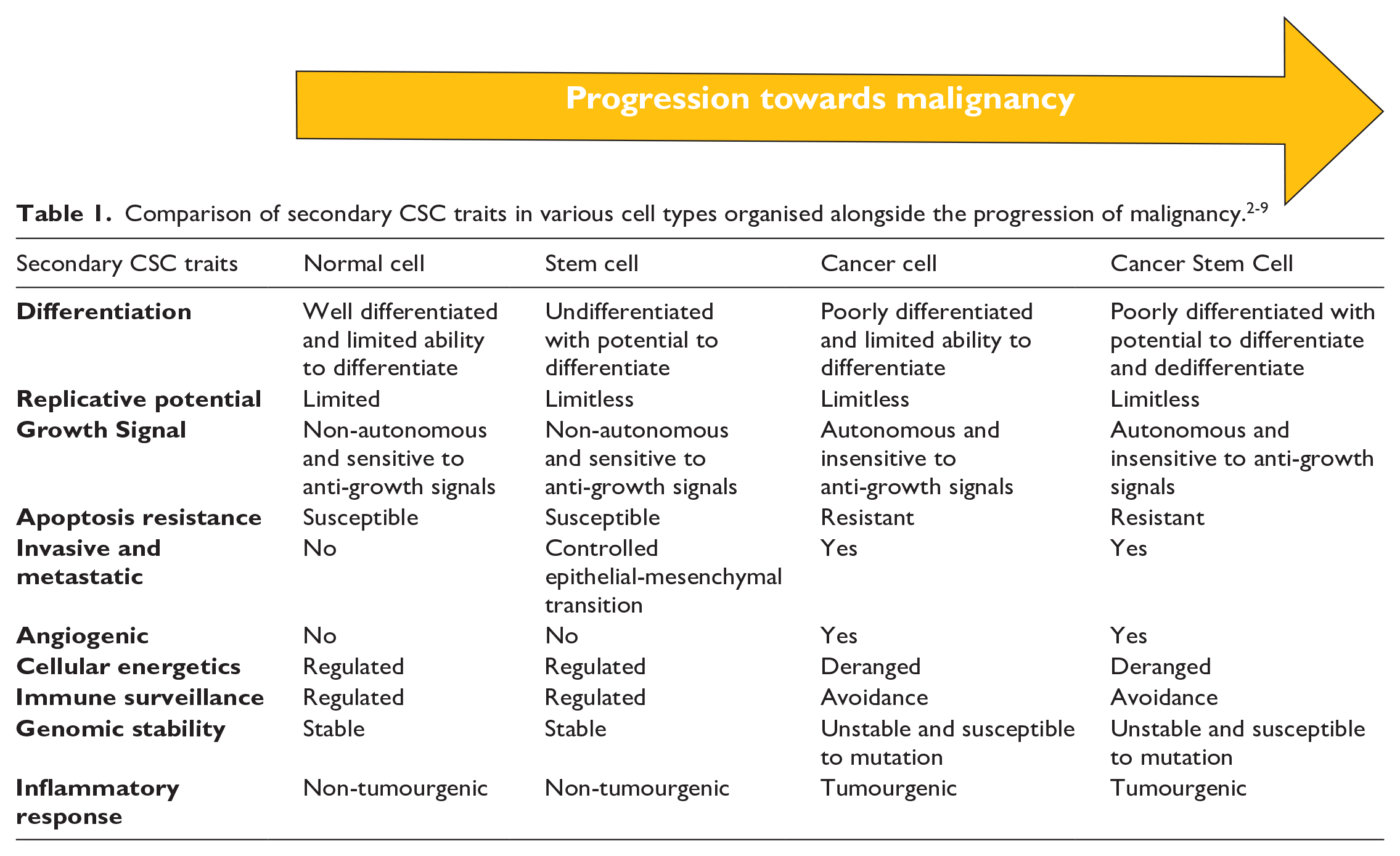
Table 1 highlights key secondary CSC characteristics such as apoptosis resistance, invasiveness and genomic instability which promotes CSC survival and tumour reconstitution. These key secondary CSC characteristics have been contrasted against normal cells, stem cells and cancer cells. A combination of biomarkers to these secondary CSC characteristics may help unravel the mystery of CSC’s tumour reconstituting ability.

Exploring immunophenotypic, genetic and epigenetic biomarkers
Current studies suggest overlapping traits between stem cell biomarkers and CSC biomarkers. This postulate stems from common properties observed in both stem cells and CSCs. One example is replicative immortality, these overlapping traits suggest the existence of common cellular pathways. Therefore, current stem cell biomarkers may be able to assist in isolating CSCs from heterogenic tumour populations. Proof of this concept is the relative success in identifying CSCs using immunophenotypic biomarkers for haematological malignancies. However, this situation has not been the case for solid tumours.
CD133, a potential prostate CSC immunophenotypic biomarker, faced some controversy in light of conflicting data. The data suggested that CD133 expression is not unique to prostate CSCs as it can be found in human and rodent luminal epithelial cells.26-29 Furthermore, CD133 was unable to select for cells which were able to recapitulate their parent tumour - i.e in vivo tumorigenicity which is a key CSC property. 30 To overcome the challenge of lack of specificity when developing immunophenotypic biomarkers, it is critical for the biomarkers to be linked to CSC’s underlying pathophysiology. One such example would be hypoxic tumour microenvironments in rapidly growing dysplastic tumour vasculature. Investigations into biomarkers of hypoxia have yielded Carbonic Anhydrase 9 (CA9) a protein responsible for intracellular pH regulation. This is significant as it established an in vivo pathophysiological link between CA9 enriched tumours and increased cervical CSCs in primary cervical cancer xenografts. 31 CA9 were also clinically associated with poorer patient outcome for all endpoints this includes overall survival hazard ratio, disease-free survival, locoregional control, disease-specific survival, metastasis-free survival and progression-free survival. 32
Understanding the immune system’s interaction with CSCs is also important for biomarker discovery. PDL-1 an important protein involved in immune checkpoint signalling appears to also confer CSC like characteristics to cancer cells. Upregulation of OCT4 and NANOG transcription factors via PI3K/AKT pathway which are critical for pluripotency and tumorigenesis have been associated with PDL-1. 33 Specifically PDL-1 has been closely associated with epithelial-mesenchymal transition (EMT) which is a CSC hallmark ability, which enables cells to dedifferentiate into more primitive tissue types to promote tumour invasiveness and metastasis. EMT appears to be associated with PDL-1, as PDL-1 was indirectly knocked down in lung adenocarcinoma cells using microRNA-200/ZEB1 which suppress EMT characteristics in lung cancer cells. 34
It is also worthwhile noting that environmental stressors such as hypoxia can epigenetically cause non-CSCs to acquire a CSC-like phenotype, which includes the ability to differentiate and recapitulate tumours in vivo.7,8 Hence when identifying biomarkers which capture CSC traits, genetic/epigenetic biomarkers should also be considered beyond that of traditional immunophenotypic biomarkers. CSC genetic and epigenetic signatures can potentially serve as biomarkers enabling the isolation of rare CSC niches, this would require identifying genetic and epigenetic alterations specific for the CSC. The challenge is that CSCs possess many genetic and epigenetic alterations commonly seen in malignancy, 35 many are non-specific to the CSC and are secondary bystander mutations that can occur in malignancy due to faulty gene expression mechanisms. 36
To overcome this challenge, initial screening for genetic signatures can be focused on genes involved in cell cycle pathways associated with normal stem cell function. Shh, Notch and Wnt pathways have been previously implicated in stem cell biology. For example, the Lgr5 (leucine-rich-repeat-containing G-protein-coupled receptor 5) which is part of the Wnt pathway has been discovered in intestinal crypt base columnar cells. 37 Lgr5+ columnar cells in mice can generate gastrointestinal epithelial lineages such as paneth cells, enteroendocrine cells, goblet cells, tuft cells, columnar cells and M cells. 38 Lgr5 can possibly be used as gastrointestinal epithelial stem cell marker. Other important stem cell linked pathway are the Shh pathway, which when dysregulated promotes tumorigenesis.39,40 The Ptch1 gene which codes for the receptor for Shh has been implicated in basal cell carcinomas in Gorlin syndrome patients which is a rare neurocutaneous syndrome associated with the development of multiple tumours. 41 The Notch pathway has also been implicated in EMT in pancreatic and prostate cancers.42,43
Epigenetic changes observed in stem cells have also been identified in induced pluripotent stem cells reconstituted from fibroblasts. 44 Similar global epigenetic alterations have also been consistently observed in tumours. 45 Histone hypoacetylation and hypermethylation induces a transcriptional repression of their associated genes. In the case of cancer cells, it appears that tumour suppressor genes may be consequently suppressed with histone modification. 45 Histone modification patterns may serve as a new epigenetic biomarker of CSCs and a possible target for cancer therapy. 45
It is likely that to identify CSCs, future biomarkers would encompass immunophenotypic, genetic and epigenetic data. Nodal is one such biomarker with integrated immunophenotypic, genetic and epigenetic information, nodal is an embryonic morphogen belonging to the TGF-β superfamily and is important for the maintenance of pluripotent cells and for proper embryonic development. 46 Nodal protein and RNA expression has been identified in human embryonic tissues, embryonic stem cells and cancer cells. It was also noted that melanoma cells express similar nodal protein as human embryonic stem cells. 46 This is in stark contrast to melanoma’s normal counterparts, such as melanocytes, in which Nodal was not detected. Further embryological work in mice coupled with bioinformatic tools have identified that nodal expression is largely restricted to very early progenitor and reproductive cell types and re-emerges later during tumorigenesis. 46
Linking pathophysiology to biomarker discovery in uncommon cancers
Research into genetic and epigenetic sarcoma CSC biomarkers has identified SOX2 as a sarcoma CSC biomarker. 47 Sarcomas are rare connective tissue cancers whose tissue of origin can range from cancellous bone, cartilage, fat, muscle and vascular tissue. Clinical evidence has revealed that sarcomas possess CSC-like characteristics such as self-renewing properties, chemotherapy resistance and rapid metastasis. 48 SOX2 gene is part of the SRY gene family, it codes for a transcription factor critical in regulating early embryonic development and stem cell properties. 48 SOX2 has been identified in synovial sarcoma cells, reinforcing the notion that synovial sarcoma formation is driven by epigenetic processes similar to those operating within pluripotent stem cells. 49 SOX2 is also associated with other stem cell related markers OCT4, NANOG and Myc. 50 Co-transfection of SOX2 with OCT4 into adult fibroblasts was able to generate pluripotent stem cells. 51 While microRNA depletion of SOX2 reduced sarcoma cell migration and invasiveness and prevented formation of osteosphere colonies. 52
Apart from sarcomas, SOX2 has been implicated in other human cancers most notably glioblastoma. Glioblastoma is a common and deadly primary brain tumour with a poor prognosis of median time between 12–15 months. 53 Glioblastoma CSC are able to self-renew and differentiate into distinct lineages. These glioblastoma CSCs can also initiate and propagate tumours in xenograft models. 54 These CSCs are also capable of generating vascular pericytes to support vessel function and tumour growth, these CSCs are recruited toward endothelial cells via the SDF-1/CXCR4 axis and induced to become pericytes predominantly by TGF-β molecule. 55 SOX2 has been discovered to be expressed in glioblastoma CSC and as previously mentioned SOX2 plays a critical role in regulating stem-cell-like properties of stem cells. 54 Therefore, SOX2 may play a prominent role in regulating growth, promoting treatment resistance and glioblastoma recurrence. 56 Epigenetic and genetic biomarkers have also been identified in more common cancers such as breast cancer. Pro-opiomelanocortin expression and G protein-coupled estrogen receptor protein phosphorylation were also discovered in primary breast CSCs and was correlated with a poor prognosis. 57
Recent work by Niu and team highlighted the ability of polyploid giant cancer cells (PPGC) in acquiring embryonic-like-stemness and initiating tumour formation. 58 Morphologically, the differentiated PPGCs were indistinguishable from early human embryos. 58 Moreover, the PGCCs also showed time and space dependent expression of embryonic stem cell genetic markers such as OCT4, NANOG, SOX2 and SSEA1. 58 Subsequently the embryonic PGCCs were then able to differentiate into all three germ layers in vitro this also includes germ cell tumours and carcinomas. 58 This embryonic-like-stemness of the embryonic PGCCs was associated with nuclear accumulation of YAP, a key mediator of the Hippo pathway, a pathway associated with cell proliferation and apoptosis. When YAP was inhibited, the embryonic PGCCs were unable to differentiate into different tissue types. 58
Functional plasticity has also been described in aggressive melanoma cells which enables it to express genes from a variety of cell types. 46 For example, E-Cadherin expression is essential for formation of tumour-derived vascular networks that are key in supporting rapidly growing tumours via a paravascular perfusion pathway, while keratin expression is associated with enhanced invasion and metastasis. 46 Evidence of plasticity is demonstrated via in vitro inhibitory experiments with metastatic C8161 melanoma cells and general inhibitors of TGF-β/Activin/Nodal signalling which led to a reduction in tumour cell invasion. 46 Further evidence was demonstrated in animal experiments using morpholino antisense oligonucleotides to specifically knockdown Nodal gene expression in C8161 melanoma cells resulted in decreased tumorigenesis in nude mice and downregulation of VE-Cadherin and keratin. C8161 melanoma cells treated displayed less in vitro as well as in vivo tumour migration. 46 Collectively, these studies suggest that Nodal may play a key role in maintaining melanoma cell plasticity, invasiveness and tumorigenicity.
Therefore, it is important to understand underlying mechanisms driving CSC tumorigenesis, this will enable biomarkers of greater clinical significance to be identified. A flow chart (Figure 3) highlights the inter-relationship between clinical, in vitro and in vivo research involved in CSC biomarker discovery. An overview of the timeline of the discovery of various important CSC biomarkers and their relationship with the CSC model can also be seen in the flow chart (Figure 4).
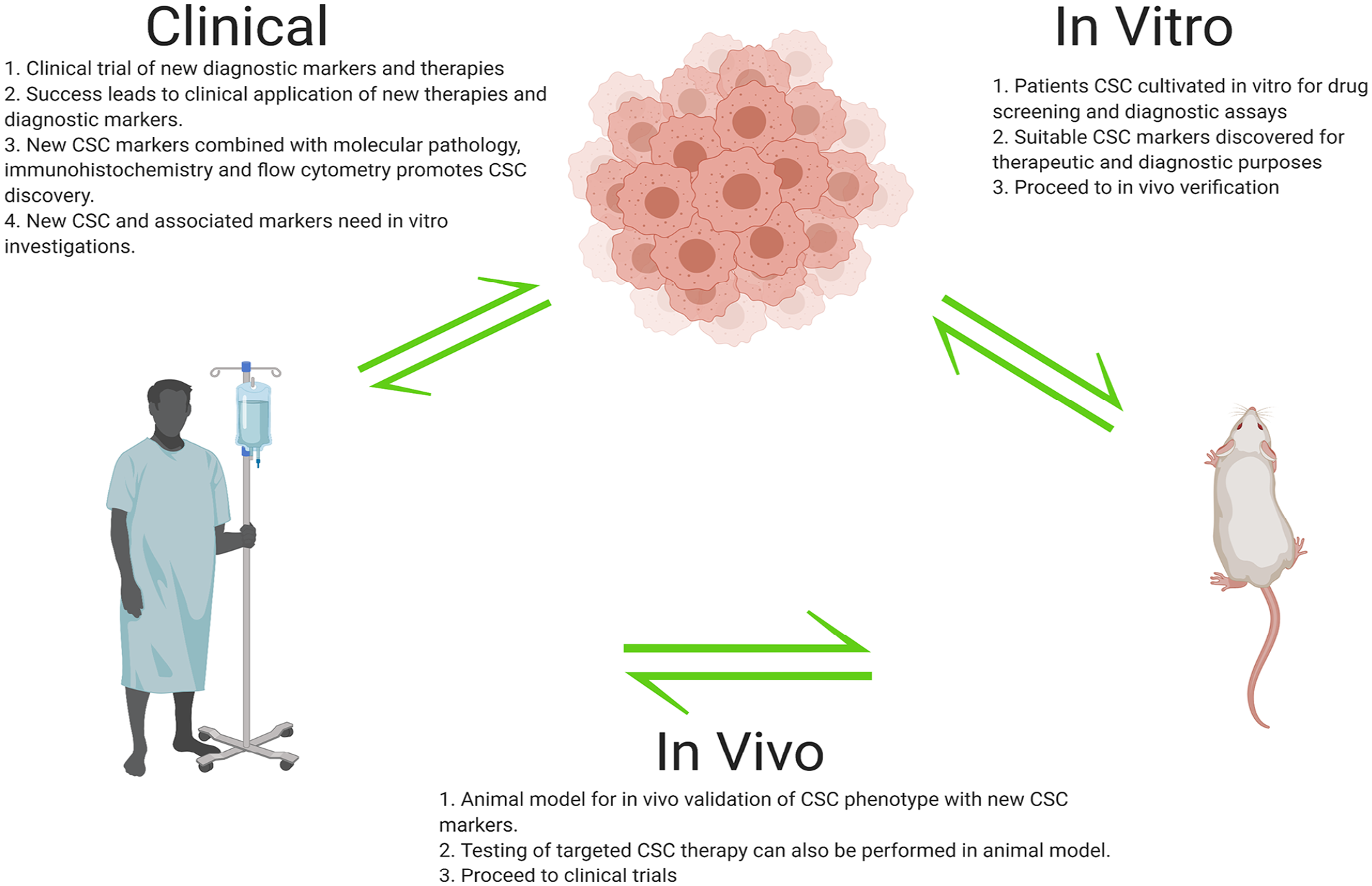
Flowchart highlighting the identification and clinical application of CSC markers. Experimentally identified CSC markers need to be verified via in vitro and in vivo experiments before proceeding to clinical trials. After successful clinical trials, CSC markers are integrated into clinical practice alongside existing clinical diagnostic tools. Remaining clinically unidentified cells are referred to experimental laboratories for future discovery of new CSC markers. This flowchart highlights the positive feedback loop between clinical and experimental laboratories that drives the discovery of CSC markers.
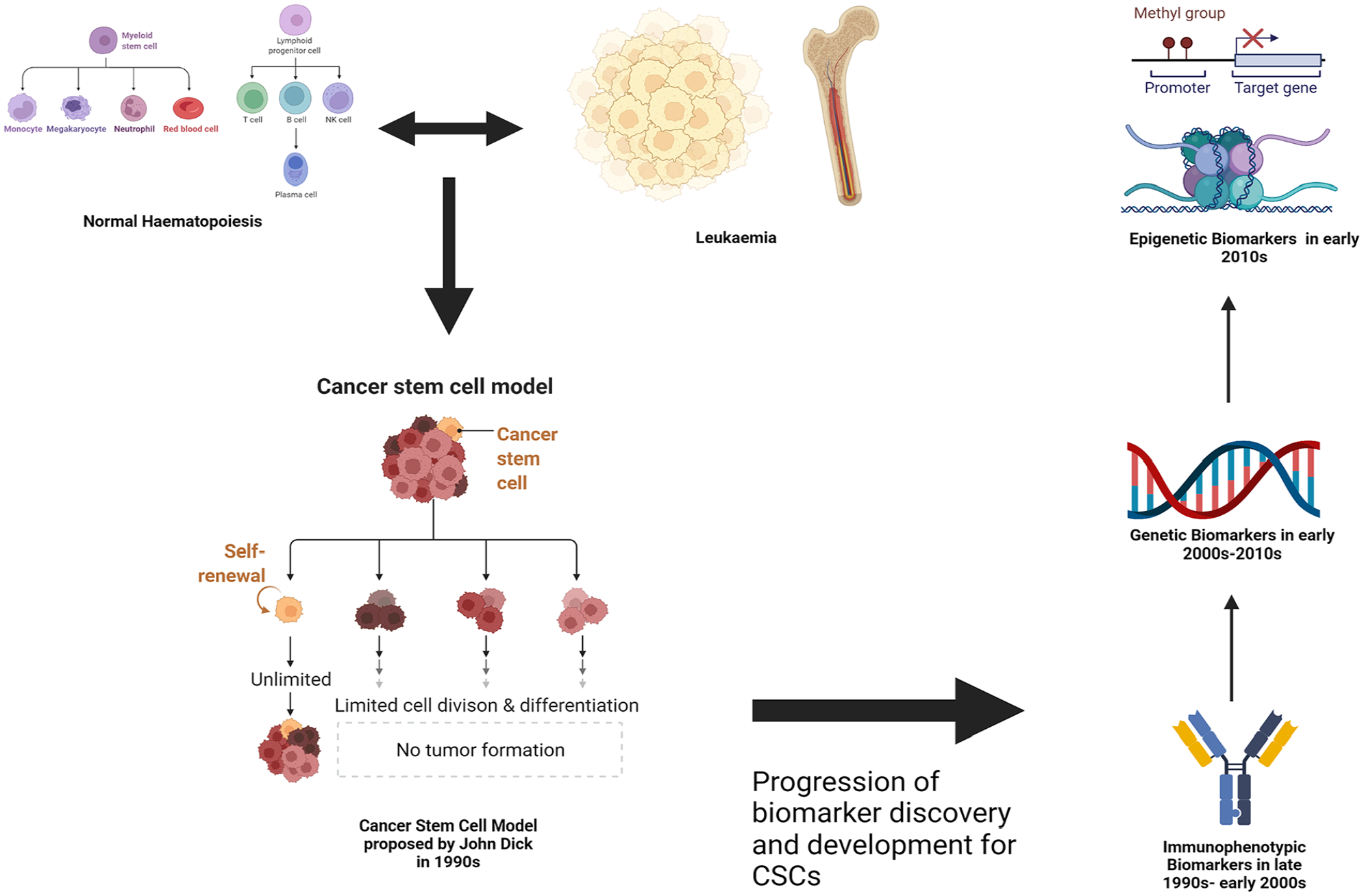
Flowchart highlighting the rough timeline and links between the development of the CSC model and the research/discovery of CSC associated biomarkers. Theories and experiments by Dick 59 regarding an aberrant version of haematopoiesis causing leukaemia led to developments of the CSC model. This proved a useful foundation for the research of CSC biomarkers initially focusing on the immunophenotypic markers, followed by the genetic and then epigenetic markers.
Clinical application of CSC biomarkers
For CSC biomarkers to have clinical applicability, they have to add value to clinical decision making. Diagnostic CSC biomarkers would identify the type, presence and amount of CSC present in the tumour refining the initial cancer diagnosis and prognosis. The presence of primitive undifferentiated CSCs would suggest that the tumour possesses self-renewal and high proliferation capacity. Additionally, CSC presence suggests greater differentiation potential with increased risk for tissue invasion/ metastases and likely resistance to traditional anti-neoplastic therapies. A systematic review of CSC biomarker expression in head and neck cancers identified ALDH1 which is a cytoplasmic enzyme responsible for catalysing oxidation of toxins into carboxylic acids was closely associated clinically with increased tumour size, presence of regional lymph node metastases and higher histopathological grade. 60 Similar results were also seen in Sox2 and OCT4, and it is believed that they are involved in the EMT process. 60
With a refinement in understanding of the tumour’s aggressiveness and subsequent risk stratification, overall clinical prognosis can be better evaluated. Potential CSC targeted therapies can then be explored and offered to the patient. CD44 overexpression, particularly CD44 isoforms have been common in various solid tumours and has potential prognostic and therapeutic value. A metanalysis of 2403 patients with gastric cancer, identified that CD44 expression was associated with higher TNM stage, greater lymph node, and distal metastasis and reduced overall survival rates. 61 Moreover, the presence of CD44 positive circulating tumour cells was an independent predictor of gastric cancer recurrence. 61 Due to CD44’s role in the cross-talk with the tumour microenvironment and regulation of CSC it is also currently being evaluated as a therapeutic target. 61 Promising animal data led to the development of bivatuzumab a humanized monoclonal antibody against CD44 which showed anti-tumour effects. 61 However, further clinical evaluation was halted after a patient’s death due to the immunogenic accumulation of the antibody in nontumor keratinocytes. 61 Despite this setback, it does highlight the clinical potential of CSC biomarkers for prognostic and therapeutic purposes.
CSCs serving as a backbone to cancer treatment resistance
Current treatment modalities against cancer fall under five broad categories: radiotherapy, chemotherapy, targeted therapy, immunotherapy and surgery. CSCs are believed to be a source of resistance against these current treatment modalities. Surgical treatment is at times ineffective despite early detection and clear surgical margins, this is due to the possible presence of CSCs.62,63 Despite removal of the bulk of the tumour, small amounts of CSCs may be present allowing for local cancer recurrence. For the treatment of hepatocellular carcinoma, surgical resection is a well-accepted therapy. 63 But patients frequently develop cancer recurrence which is the main cause of death in the long term. 63 Radiotherapy is currently the most effective available therapy for glioblastoma, despite this treatment failure is still a fairly common occurrence hence the poor prognosis. 64 CSCs have been observed to support radiotherapy resistance through activation of DNA damage checkpoints in response to irradiation to promote DNA repair. 65 CSC radiotherapy resistance was reduced through inhibition of Chk1 and Chk2 checkpoint kinases.66,67 CSC initiated DNA repair is an important target for neoadjuvant chemotherapy, which would enhance the effectiveness of radiotherapy treatments in glioblastoma. Chemotherapeutic resistance provided by CSCs is another obstacle to overcome for cancer treatment. In a colorectal CSC model, 5-fluouracil and oxaliplatin was trialled in a dose escalating fashion to reach clinically relevant doses. From the study, it appeared that cells resistant to chemotherapy were enriched with both CD133 and CD44 markers which is consistent with the colorectal CSC phenotype. 68 It was interesting to note that Insulin-like Growth Factor-I Receptor (IGF-IR) was also upregulated in the CSCs, and upon inhibition with IGF-IR monoclonal antibodies, there was a greater growth inhibition. 69 Usage of adjuvant immunotherapy to target CSCs may provide synergistic benefits to conventional chemotherapy.
CSCs also appear to be able to modify their local environment to form a tumour microenvironment to support tumour growth. This tumour microenvironment can provide an immunosuppressive effect, allowing for further cancer progression through invasion and metastasis which would normally be halted by the immune system. 70 Glioma CSCs appear to induce immunosuppressive macrophages and microglia. 71 The glioma CSCs secreted cytokines such as MΦ inhibitory cytokine-1 (MIC-1) and transforming growth factor (TGF-β1).72,73 Additionally, they were also able to alter the tumour microenvironment to impede the phagocytic ability of recruited monocytes. 72 CSCs are believed to be a key regulator in the tumour microenvironment, promoting cancer growth and metastasis. The tumour microenvironment acts as a barrier, shielding the cancer cells from chemotherapy and radiotherapy. Recent work on osteosarcoma (OS) revealed that mesenchymal stem/stromal cells (MSCs) have also been implicated in the tumour microenvironment supporting OS cells. The OS cells are supported through the MSCs via extracellular vesicles which contain cytokines like IL-6, growth factors like VEGF, metabolites like lactate and even microRNAs. 74
Approaches to targeting CSC
CSC serve as attractive targets for future cancer therapy as they enable a targeted approach against cancers minimising collateral damage to healthy cells while enabling tumour source control. This allows for efficacious cancer treatment with minimal side effects. Multiple reasons may be responsible for lack of clinical translation, chief amongst them is the gap in knowledge in identifying and isolating CSCs. This problem is largely attributed to the paucity of specific CSC defining biomarkers which itself may be attributed to an inappropriate CSC model or the failure to incorporate key secondary CSC traits when investigating CSC biomarkers.
Even if targeted therapy against CSCs were to be developed, there may be concerns for intratumoral and intertumoral CSC heterogeneity which may limit successful eradication of tumours. This tumour heterogeneity may be the result of tumour evolution, as CSC cell lines resistant to specific therapies are selected for due to vulnerable cell lines being eradicated over time.7,8 This variability has generated therapeutic challenges in using molecularly targeted and CSC specific immunotherapy. As incomplete eradication of CSCs risks tumour resistance and recurrence. CSC’s may also be able to overcome targeted immunotherapy, cancer stemness regulators such as b-catenin in the Wnt signalling pathway appear to possess immunomodulating capabilities. 75 c-Myc a pluripotency transcriptions factors, upregulates both the innate immune inhibitor CD47 and adaptive immune checkpoint molecule PD-L1. 76 This suggests that CSCs might not only evade immune attacks but also actively suppress immune responses. Additionally, stemness regulators such as SOX2 and EZH2 are able to confer lineage plasticity, allowing CSCs to survive anti-tumour therapies.77,78 Thus, this intrinsic heterogeneity and adaptive resistance of CSCs to stress confers resistance to antitumor therapies.
Despite the challenges posed when treating CSC, there is still sustained interest to develop CSC specific treatments due to possibility of sustained tumour eradiation. Additionally, with further refinement in CSC identification, the efficacy of CSC targeted therapy would improve. For example, AQP5 which is commonly identified in primary intestinal and diffuse subtypes of gastric cancer has been found to enrich CSCs key to the early formation of gastric cancer. 79 Robust studies such as these have incorporated in vitro/ in vivo genomic and transcriptomic data and integrated it with clinical phenotypic data which are key to clinical translation.
Cancer immunotherapy is a broad term which can involve biologics such as antibodies to modified chimeric antigen receptor T cells. One method being explored would be antibodies designed to bind to CSCs while ignoring the normal stem cell population. There is some interest in generating monoclonal antibodies to CD133, which is a 120 kDa highly glycosylated 5-span transmembrane protein belonging to the pentaspan/ prominin family. 80 CD133 is found on haematopoietic progenitor cells and a variety of CSCs. To increase the selectivity of cytotoxic drugs, CD 133 antibody-cytotoxic drug conjugates were synthesised and tested on hepatocellular, pancreatic and gastric cancer cell lines overexpressing CD133. 80 The antibody-drug conjugate was internalised by the cancer cell in vitro and resulted in cancer cell death. 80 A more recent example would be the usage of Notch1 monoclonal antibodies tagged with docetaxel in patient derived triple negative breast CSC which was evaluated by a reduction in mammosphere formation and CD44+/CD24-/lo cell population. 81 Additionally there was also a decreased tumour incidence upon re-implantation and delay in tumour recurrence. 81 Due to the important role that Nodal plays in maintaining the melanoma tumorigenic phenotype there has been interest in treating melanoma or other Nodal-expressing human cancers through nodal inhibiting agents. Function-blocking antibodies have been preliminarily explored, which shows human melanoma cells treated with function-blocking antibodies against Nodal demonstrating a significant reduction in their capacity to engage in vasculogenic mimicry in vitro. 46 More interestingly, function-blocking anti-Nodal antibody was demonstrated to reduce the ability of metastatic melanoma cell (C8161) to colonise nude mice lungs in an in vivo tumour colonisation assay. 46 The nude mice were injected intra peritoneally with either a function-blocking anti-Nodal antibody or an isotype control antibody (control IgG). Tumour cell colonies on the lung surface were macroscopically less evident in anti-Nodal antibody-treated mice. 46 Additionally, at a cellular level, melanoma cells in the lungs of anti-Nodal antibody treated mice showed signs of cellular distress, such as cytoplasmic swelling and vacuolisation, apoptosis and decreased immunohistochemical expression of Nodal. 46 This suggests that apart from being a diagnostic/ prognostic marker, Nodal can potentially serve as a therapeutic target in metastatic melanoma cells. Unfortunately, despite promising in vitro and in vivo work there remains a lack of clinically approved solid tumour specific CSC antibody markers due to their lack of specificity. 30
Another aspect of immunotherapy would be the development of cancer vaccines, which isolates CSC specific biomarkers and highlights them to the immune system to selective target the CSCs. Prostate stem cell antigen (PSCA) is one such CSC biomarker being considered for a cancer vaccine. The peptide fragments of PSCA are presented by major histocompatibility molecules as recognition targets for CD8 T cells. 82 This will allow the body to mount a systemic immune response against the prostate cancer using cytotoxic CD8 T cells. This is assuming that the CD8 T cells have not been clonally deleted or peripherally tolerated. 83 Dannul et al. managed to generate a PSCA specific T cell response from a human lymphocyte culture. 84 The cytotoxic PSCA specific T cells managed to recognise three prostate cancer lines. 84 Building upon this concept is the development of Chimeric Antigen Receptor (CAR) T cells to retarget T lymphocytes to PSCA positive prostate CSCs. 85
Apart from creating new therapies, CSC biomarkers can improve current treatments such as radiotherapy by identifying CSC niches within a tumour. This allows for high-dose fraction of stereotactic radiotherapy to be delivered to the CSC while sparing healthy surrounding tissue.86,87 Radioactive-tracers conjugated to antibodies specific to CSC markers can also localise CSCs and simultaneously eradicate them, research in this area is developing particularly in the realm of ‘theranogstics’ which is a portmanteau of therapeutics and diagnostics. 88 Apart from identifying CSC niches, CSC biomarkers can identify potential radiosensitising agents to augment the effectiveness of radiotherapy. Treatment failure for head and neck cancers (HNC) with radiotherapy is believed to be a consequence of CSCs. 89 Alltrans-retinoic acid (ATRA) was shown to induce CSC differentiation, cell cycle redistribution and CSC radiosensitisation. To overcome the issues of radiation resistance in HNC and strengthen the level of evidence of ATRA’s effects, an in silico model was developed to evaluate the synergistic effect of ATRA and radiotherapy. 89 ATRA’s cell arrest property showed the strongest influence on CSC subpopulation evidenced by the exponential behaviour of the dose-response curve. 90 While ATRA induced apoptosis showed a linear correlation between apoptotic cells and radiation dose required to eradicate CSCs. 90 ATRA can be a potent CSC targeting agent and when combined with radiotherapy shows promise in tumour control. 90
Another approach is targeting the epigenetic biomarkers of CSCs by interfering with RNA transcription and methylation of oncogenes. RNA interference and other forms of epigenetic changes have been also associated with cancer. Previous groups have shown that by antisense RNA silencing of tumour suppressor gene p15, involved in DNA methylation and heterochromatin formation is associated with tumorigenesis. 91 Perhaps the processes that promote tumour formation can also be harnessed to treat cancer. Selectively targeting the epigenome might be another avenue for development of therapeutics, recent in vivo work with CSCs from testicular germ cell tumours has found that the CSCs are sensitive to DNA methyltransferase inhibitor, 5-azadeoxycytidine (5-aza). 92 Building upon that work, Albany et al. utilised clinically optimised second generation demethylating agent guadecitabine (SGI-110) on CSCs in an animal model reflecting cisplatin refractory testicular cancer. 93 Results highlighted that guadecitabine mediated transcriptional reprogramming of p53 targets while repressing pluripotency genes, however this was contingent on high levels of DNA methyltransferase 3B in the CSCs. 93 Guadecitabine was also able to abolish progression and induced regression in the cisplatin resistant tumour xenografts. 93 Gene silencing is also another potential new area for developing anti-neoplastic agents, RNA interference has been able to knockdown β2-microglobulin and OCT4 gene expression in CSCs. 94 RNA interference was then applied to colorectal CSCs in a mice model leading to a markedly attenuated tumour growth and lung metastasis. 95 This technique has also been applied to luminal and basal-A triple-negative breast cancer mice models. 96 This result opens the possibility of using RNA interference to silence oncogenes or other tumorigenic genes in the context of CSCs. A possible issue for the clinical application of RNA interference is site specific delivery, which may be solved with the usage of nanoparticles. 97
Targeted therapy can also focus on various cell receptor pathways, genome-wide analysis studies have highlighted that active p53 and immune-related pathways are strongly associated with the observed in vivo anti-tumour activity. 98 Bruton’s tyrosine kinase (BTK) has been strongly associated with B cell malignancies, but has recently been implicated in solid tumours like ovarian and prostate cancer. 99 BTK regulates key signalling pathways involved in cellular growth, differentiation, apoptosis and recent data shows even cytoskeletal reorganisation and cell motility. 100 In vitro inhibition of BTK in glioblastoma multiforme (GBM) cell lines resulted in reduced ability in colony formation, migration and GBM sphere formation. Additionally, ex vivo data using tissue microarray analysis of patient samples revealed higher BTK staining in malignant tumours compared to normal brain tissues. Mechanistically, BTK inhibited cells also displayed a concomitant reduction in CD133 and Akt/mTOR signalling. Finally, in vivo data revealed that BTK inhibitor treated xenograft mice had reduced tumourgensis. This has great implications for developing pioneering therapeutic agents for GBM by targeting the BTK pathway, these findings may also imply that ibrutinib may be used as an adjuvant for GBM. 101
Conclusion
Due to the complexity of CSCs, it is likely that a panel of biomarkers incorporating immunophenotypic, epigenetic and genetic biomarkers would be needed to isolate CSCs. It is possible that two broad categories of biomarker panels will emerge. One category of biomarkers serving as highly sensitive but not specific biomarkers for cancer presence and aggressiveness, this is based on the concept of common mutations and pathways found in CSCs that promote tumour survival under evolutionary selection pressures. A current example would be overexpression of Tripartite motif-containing protein 28 (TRIM28) being identified in 14 different tumour types, including solid and haematological tumours. 102 The second category of biomarkers would be cancer specific, this would be identifying the cell of origin for the cancer allowing a specific diagnosis and subclassification for prognosis. Current literature has identified CSCs from different cancer categories such as carcinomas, sarcomas and haematological tumours possessing different properties,103-105 further refinement in the future would enable single cell tumour identification with developing molecular techniques. Apart from aiding the initial diagnosis, both categories of biomarker panels would also be useful for conveniently screening at risk individuals and detecting residual disease post treatment.
The refinement of solid tumour CSC models and the development of new biomarkers is an ongoing positive feedback loop. With new scientific discoveries, the CSC model is refined and a better definition of a CSC would enable more focused and targeted research into CSC biomarkers. Likewise with new CSC biomarkers emerging, it would ease the identification of CSCs whose pathophysiological/biological properties can be better understood and a more accurate CSC model can be established. This continual process will help ensure that future CSC biomarkers and their associated CSC models will be tightly linked and will hopefully have greater clinical translatability/utility for both diagnostic and therapeutic purposes.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.