Abstract
Introduction:
During the last few years it has been shown that an anaplastic T cell lymphoma can develop as a rare and late sequelae of implant-based breast reconstruction. This malignancy was recognized in the 2017 by WHO and named breast implant associated anaplastic large T cell lymphoma (BIA-ALCL). BIA-ALCL usually presents as abundant effusion around the implant, thus, in addition to cytology smears, its diagnosis also requires immunohistochemistry, T cells clonality and cytometry. Due to the increasing attention of clinicians, it is likely that the number of the BIA-ALCL suspected cases will grow in the future, implying the necessity of a reliable and cost-effective diagnostic procedure.
Methods:
To achieve this goal, we retrospectively analyzed the results of laboratory investigations performed at our Institute (Fondazione IRCCS Istituto Nazionale dei Tumori Milan, Italy) on 44 effusions obtained from 31 women suspected for BIA-ALCL.
Results:
Through cytology, eight out of 44 effusions showed the presence of BIA-ALCL cells. Lymphoma cells were than confirmed in seven samples by immunohistochemistry and/or T cell clonality and/or cytometry. Overall, cytology showed 100% sensitivity, 97% specificity and positive and negative predictive values of 87.5% and 100% respectively. Further analyses were particularly useful in effusions showing small percentages of BIA-ALCL cells. Moreover, an extended cytometric profile that can be applied when fast confirmation of the cytologic result is required was also identified.
Conclusions:
Our results evidenced a central role of cytopathology in the management of BIA-ALCL suspected effusions and suggested that further laboratory investigations might be applied only in cases showing atypical/activated lymphoid cells through cytology.
Introduction
Implant-based breast reconstruction is one of the most frequent kinds of reconstructive surgery in women after mastectomy. Implants are always surrounded by a capsula, which is a fibrous tissue developed by the body itself to isolate the foreign body. The capsula is attached both to the implant and to the adjacent tissues, ensuring the stability of the implant and, in most patients, can be concomitant with a small quantity (few millilitres) of physiological periprosthetic fluid. Several mechanical and biological factors support fluid development: lymphatic surgery, radiotherapy as well as the inflammation generated by the movement of the implant within the periprosthetic space. In some patients this fluid is abundant and viscous and is commonly called seroma that can be distinguished as early (occurring less than one year after breast implants) or delayed. 1 Since the term seroma is technically incorrect, 2 hereafter we will define the fluid present around the breast prosthesis as periprosthetic effusion (PPE), even variably abundant and more or less viscous.
Over the years, a rare anaplastic CD30-positive ALK-negative T cell lymphoma has been associated with the presence of PPE in women with breast implants. This new malignancy was recognized as a provisional entity in the 2017 revised World Health Organization (WHO) lymphoma classification 3 and named breast implant associated anaplastic large cell lymphoma (BIA-ALCL). Although the disease usually presents as a unilateral abundant effusion with a median time from implant of nine years, early (one year) and late (32 years) cases have been reported. 4 A BIA-ALCL lifetime prevalence of one in 30,000 in women with a textured breast implant was reported. 5
Even if large pleomorphic BIA-ALCL cells can be easily detected by May-Grunwald e Giemsa (MGG) and/or Papanicolau (PPN) staining in PPEs smears, the National Comprehensive Cancer Network (NCCN) recommended that, besides MGG and PPN, all the PPEs suspected for BIA-ALCL should undergo CD30 testing by immunohistochemistry (IHC) or flow cytometry (FCM). 6 However, the potential contribution of IHC and FCM to the BIA-ALCL diagnostic workflow (but also T-cell receptor gene rearrangement) is still debated. Some authors consider the morphological analysis of PPE as the ‘central element’ of BIA-ALCL diagnostic work up and proposed that CD30 assessment should be performed only after the identification of atypical cells through cytology and not systematically on all PPEs. 1 On the contrary, other authors suggested that preparation of cell blocks is desirable to allow hematoxylin and eosin staining (H&E), IHC and investigation of T-cell receptor (TCR) gene rearrangement to detect clonality. 7 Furthermore, although FCM was demonstrated to be sensitive and specific in recognizing BIA-ALCL cells,8,9 its role was considered ancillary since compared to IHC, this technology is not available in all laboratories. 10
In the present study, aiming to optimize the BIA-ALCL diagnostic work-up, we retrospectively gathered and analyzed the results obtained in our department (Department of Advanced Diagnostic, Fondazione IRCCS Istituto Nazionale dei Tumori Milan, Italy), on 44 PPEs obtained from 31 women suspected for BIA-ALCL.
Materials and methods
Patients
From 2014 to 2021, a total of 31 patients (Figure 1) who underwent implant-based breast reconstruction, presented with PPE and suspected for BIA-ALCL were retrospectively identified in our database. In all women, the presence of PPE was identified by ultrasound (US) examination and/or magnetic resonance imaging (MRI). PPEs were directly aspirated during US or during surgical removal of the capsule. Due to the PPE recurrence, a total of 44 samples were submitted to our department. This study was performed according with the Declaration of Helsinki and written informed consent for diagnostic purposes was signed by all patients.
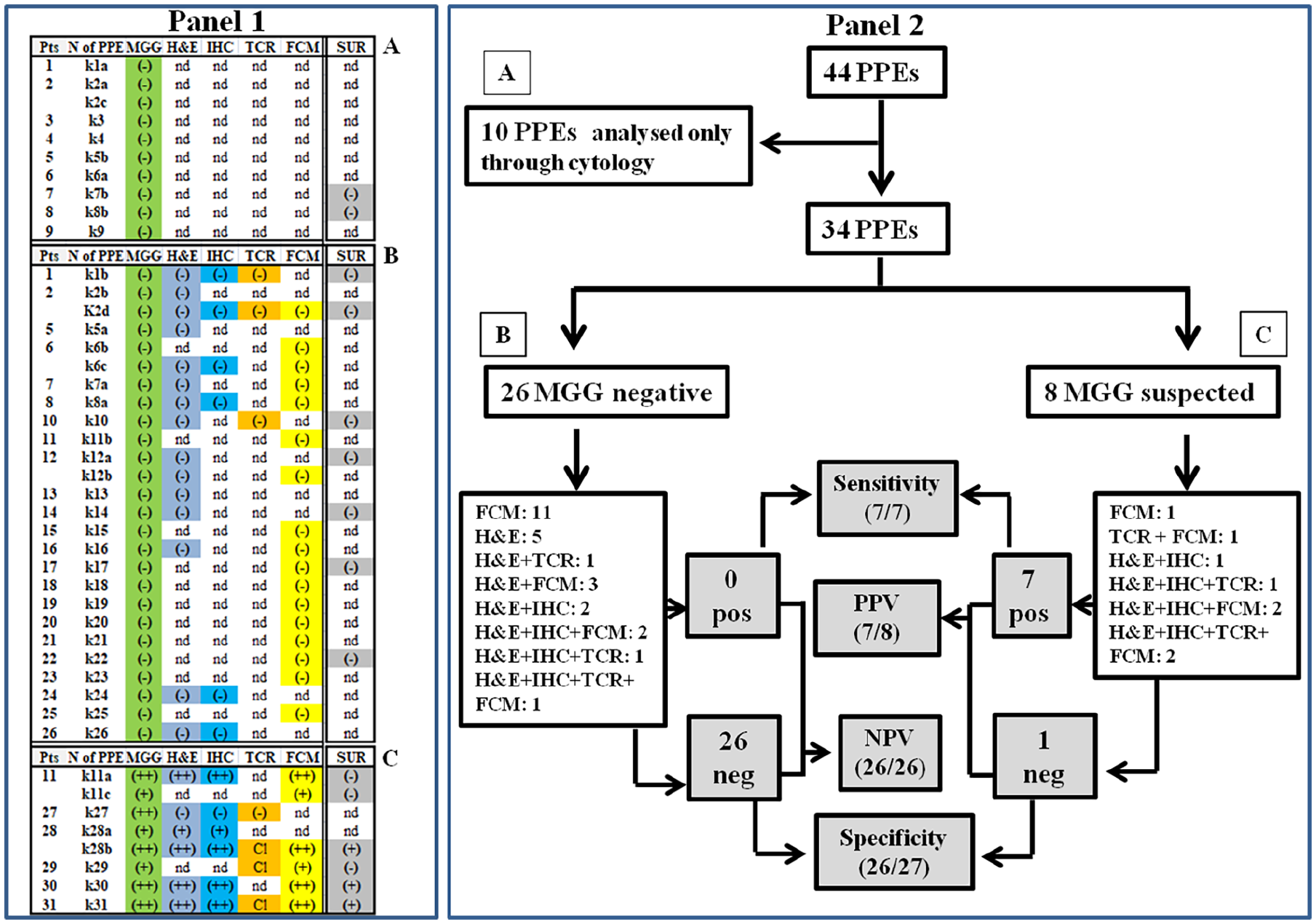
Panel 1: picture resuming the analysis performed on PPEs and capsules. Notes: Each periprosthetic effusion (PPE) was nominated using the lower-case “k” followed by the number of the patients (pts) while the lower- case letter (when present) indicates the sample-timing. The boxes highlighted the performed analysis: cytology May-Grunwald e Giemsa (MGG), hematoxylin and eosin (H&E) staining of formalin fixed paraffin embedded FFPE PPEs, immunohistochemistry (IHC), T cell rearrangement (TCR), flow cytometry (FCM) and Capsule Surgery (Sur). A: cytology-only group (n=10); B and C groups together constituted the integrated PPEs samples (n= 34). (-): negative result (i.e. absence of atypical cells); few (+) and high (++) percentage of atypical cells; Cl: clonal TCR; nd: not done. Panel 2: flow chart showing how the results of IHC, TCR and FCM were used for the assessment of sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) (grey boxes) of cytology. A, B and C were the same groups defined in panel 1. Pos: positive, neg: negative.
Cytological preparations
All the PPEs were smeared and MGG and PPN stained according to standard procedures (Figure 1). A detailed evaluation of cellular types, cytomorphology, nuclear atypia, presence of mitosis and presence of background proteinaceous materials was performed.
Cell block inclusions
Twenty-one PPEs were formalin fixed paraffin embedded (FFPE) for H&E, IHC and/or molecular analyses (Figure 1).
IHC
IHC was carried out on 2 µM FFPE sections of 12 cell blocks (Figure 1) using the following antibodies (Abs): CD2, CD3, CD4, CD7, CD8, CD15, CD20, CD30, CD45, CD68 (clone KP1), CD68 (clone PGM1), Pax5, EMA, CKA1-CKA3, GATA-3 and Claudin 4 (online Supplementary Table 1).
TCR gene rearrangement
DNA was extracted from seven FFPE PPEs (Figure 1) and analyzed using the BIOMED-2 polymerase chain reaction (PCR) assays for TCR (TCRγ and TCRβ rearrangement) genes using the fluorescent multiplex PCR kit from Invivoscribe Technologies (San Diego, CA). PCR products were run on a 3500 DX Genetic Analyzer (Applied Biosystems, Foster City, CA), and data were analyzed with the aid of GeneMapper software (Applied Biosystems). All the results were confirmed on PCR duplicates.
FCM
Twenty-three PPEs samples (Figure 1) were washed with PBS, placed in a 12 x 75 mm round-bottomed polystyrene tube and labelled using the following conjugated Abs: mix 1: CD2, CD3, CD4, CD7, CD8, CD30, CD45 CD56; mix 2 CD25, CD30, HLA-DR and CD45 all from Becton Dickinson (online Supplementary Table 2). The labelling conditions were those routinely employed in diagnostic procedures in our laboratory (ISO9001 certified). All cells were acquired using a FACS-Canto II cytometer, and analyzed using FACS DIVA program. Cellular Forward Scatter (FSC) and Side Scatter (SSC) were defined as high (FSChigh, SSChigh) when augmented compared to those of normal lymphocytes. Likewise, the antigens intensity of expression of BIA-ALCL cells were defined as [+/-] if reduced or heterogeneous, [+] or [++] if comparable or augmented in comparison to normal T cells.
Capsulectomy
Fifteen capsules (Figure 1) were FFPE for H&E staining. IHC was performed as above described for cell blocks.
Results
Clinical presentation
In the present work, we report a mono-institutional retrospective evaluation of the laboratory analyses performed on 44 PPEs received in our department from 2014 to 2021. All women showed the presence of effusion (recurrent in nine cases), sometimes coupled with breast swelling, prosthesis rotation (or rupture) or capsular contracture. In most cases, the presence of PPE was evidenced by US and/or MRI. Fluid withdrawal was performed during US or during surgical procedures related to implanted devices removal or substitution. The time elapsing between capsule or expander implantation and PPE formation for valuable patients ranged from four months to 20 years.
Overview of the performed laboratory investigations
The first NCCN guidelines for BIA-ALCL diagnosis and treatment were published in 2017 11 and then up-dated in 2019. 6 Both papers recommended that suspicious effusions should undergo an integrated diagnostic work up comprising cytology and CD30 staining by IHC and /or FCM. As showed in Figure 1, among 44 samples, 10 (22.7%) had only cytology (Figure 1, Panel 1, group A) and hereafter named cytology-only PPEs while the remaining 34 PPEs (Figure 1, Panel 1, groups B and C), had at least one further investigation (hereafter named integrated PPEs). These results demonstrated that we were not always within the NCCN guidelines. The main reason for this incompliance is likely due to the fact that only the 2019 recommendations suggested that at least 50 ml of effusion should be used for laboratory investigations. In line with that, the majority of cytology-only PPEs were received before 2019 and had a median volume of 8.7 ml (range 0.2-70 ml) making it difficult to set up the other analyses. On the contrary, in the integrated group, the PPEs median volume was 17 ml (range 0.2-140 ml) allowing us to perform a more comprehensive diagnostic procedure. In particular, in addition to cytology, 18 (18/34: 54%), four (4/34: 11%) and 12 (12/34: 35%) had at least one, two or more than two further analyses, respectively (see further details in Figure 1, Panel 2). Finally, 15 capsules were also available.
Cytology results
The cytopathological results can be summarized as follow: eight (8/44: 18%) PPEs contained atypical cells, 28 (28/44: 64%) showed a variable amount of reactive cells (from neutrophil-predominant to lymphocyte or histiocytes predominant), five (5/44: 11%) were haematics and, finally three (3/44: 7%), were devoid of cells (online Supplementary Table 3). Notably, 5/8 BIA-ALCl suspected PPEs showed a great number of atypical cells (k11a, k27, k28b, k30 and k31) while in the remaining 3 (k11c, k28a and k29), only rare atypical cells were evidenced (Figure 1, Panel 1). Secondary features mainly included the presence of proteinaceous materials sometimes encompassing the cellular components (online Supplementary Table 3).
Cytology sensitivity, specificity, positive predictive value and negative predictive value assessment
In order to establish sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of cytology, we took advantage of the investigations performed in fixed (H&E, IHC as well as TCR gene rearrangement) and fresh (FCM) materials enclosed in integrated PPEs samples group (n= 34). Among the 8 samples that contained atypical cells, 7 were confirmed as BIA-ALCL positive by at least one of the further analyses leading to a cytology sensitivity and specificity of 100% (7/7) and 96% (26/27) respectively. Overall, a PPV of 87.5% (7/8) and NPV of 100% (26/26) for cytology can be established (Figure 1, Panel 2). Considering the entire cohort of 44 PPEs, eight (from six patients) showed the presence of BIA-ALCL cells that were then confirmed by H&E, IHC, TCR and FCM (two samples); H&E, IHC and FCM (four samples); H&E and TCR, (one sample); H&E and FCM (one sample) respectively. Altogether, these results confirmed a central role of cytopathology in the management of BIA-ALCL in suspected effusions.
BIA-ALCL cells morphologic features
As already described by other authors,1-4,12-19 (see also online Supplementary Table 4 for an overview of literature data), atypical cells showed an increased amount of cytoplasm coupled with both, pleomorphic nuclei and prominent nucleoli (Figure 2A) in all seven true-positive samples. Rare, enlarged and medium sized lymphoid cells, were also evidenced in the only sample (sample k27, Figure 1, Panel 1) that was not confirmed as BIA-ALCL positive by either IHC and TCR gene rearrangement.
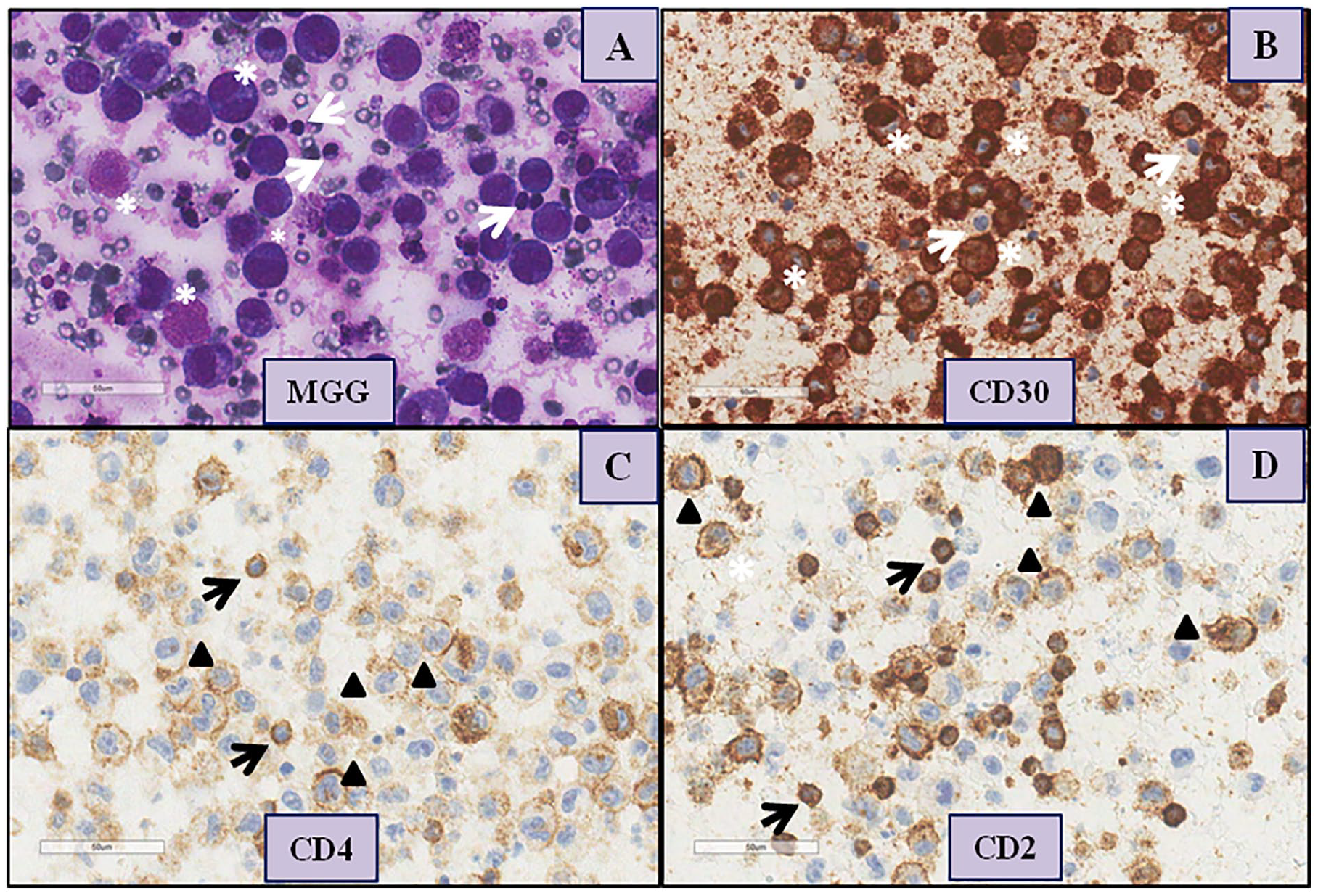
Representative immunohistochemistry (IHC).
BIA-ALCL immunophenotype in cell blocks
Cells from 21 out of 34 PPEs were fixed and paraffin embedded. Six of these cell blocks were obtained from the eight suspected BIA-ALCL PPEs. In five cases, H&E confirmed the presence of atypical cells that, by IHC, showed the following basic immunophenotype: CD30+ CD4+ CD3+ (Figure 2 and online Supplementary Table 5). As described above, in the remaining fixed PPE (sample k27, Figure 1, Panel 1), the atypical lymphocytes found by morphology, in addition to retaining CD2, CD3, CD4, CD7 and CD8 T cell markers, have been shown to be CD30 negative. This immunophenotype, combined with the absence of TCR monoclonal rearrangement, supported an activated nature of lymphocytes rather than the presence of BIA-ALCL cells. In the residual 14 cell blocks, IHC was performed in six cases and confirmed the absence of BIA-ALCL cells. Only one case showed a weak non-specific CD30 immunoreactivity (not shown).
BIA-ALCL FCM immunophenotype
Twenty-three PPEs were subjected to FCM. Cytometry confirmed the results obtained by cytology and extended the BIA-ALCL basic immunophenotype obtained by IHC in PPEs cell blocks. In particular, in all but one (sample k43a, Figure 1, Panel 1 in which cytometry was not performed) positive PPEs, FCM confirmed the presence of enlarged (FSChigh and SSChigh parameters) atypical cells (Figure 3A). The FCM BIA-ALCL phenotype shared between positive samples can be summarized as following: CD2+ (sometimes with weak expression) CD4+ (weak to moderate expression), CD30+ (bright expression), CD45+ (weak expression), CD25+ (bright expression), HLA DR+ (bright expression), surface CD3- (in the majority of samples), CD7- and CD8- (Figure 3A and Table 1). Altogether, the percentage of BIA-ALCL cells ranged from 0.3% to 84% of total leukocytes. Notably, the small percentages of pathologic cells (0.5% and 0.3% of total leukocytes) were measured in two samples (k11c and k29, Table 1) in which only rare, suspected BIA-ALCL cells were evidenced by MGG analysis.
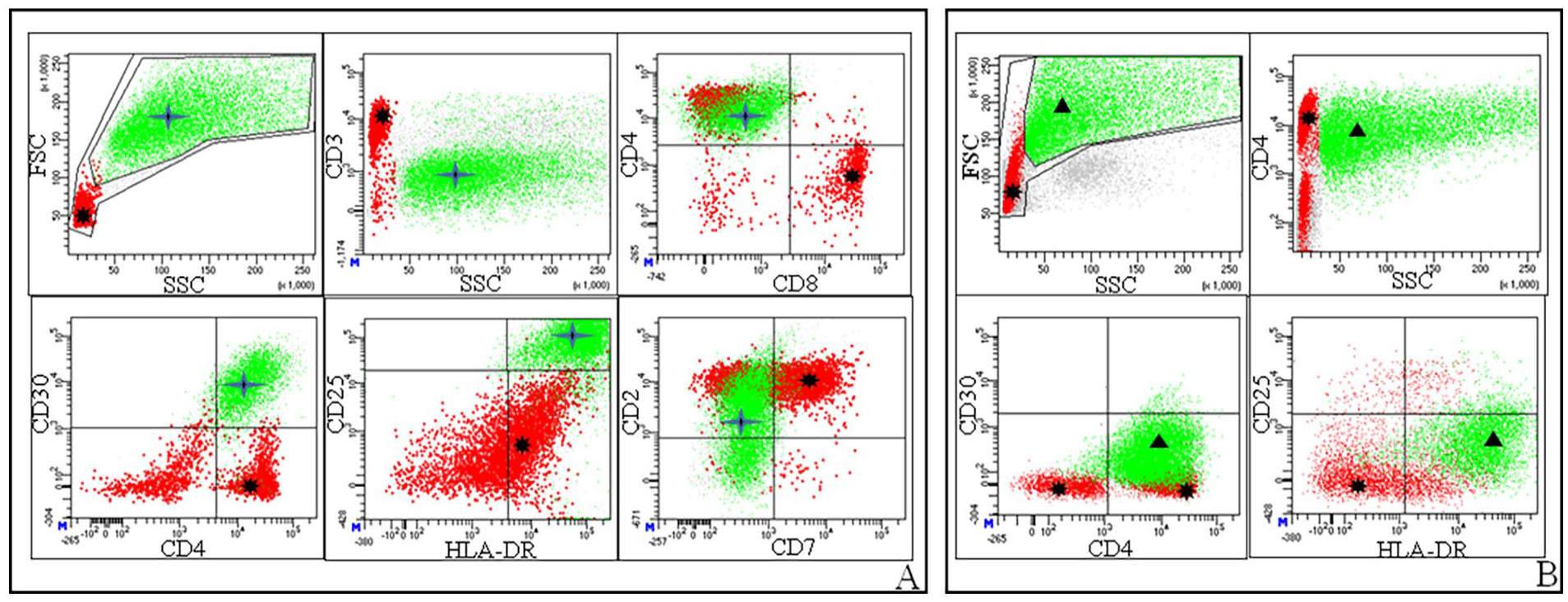
Representative flow cytometry (FCM) results.
Flow cytometric immunophenotype of BIA-ALCL cells obtained in fresh PPE.
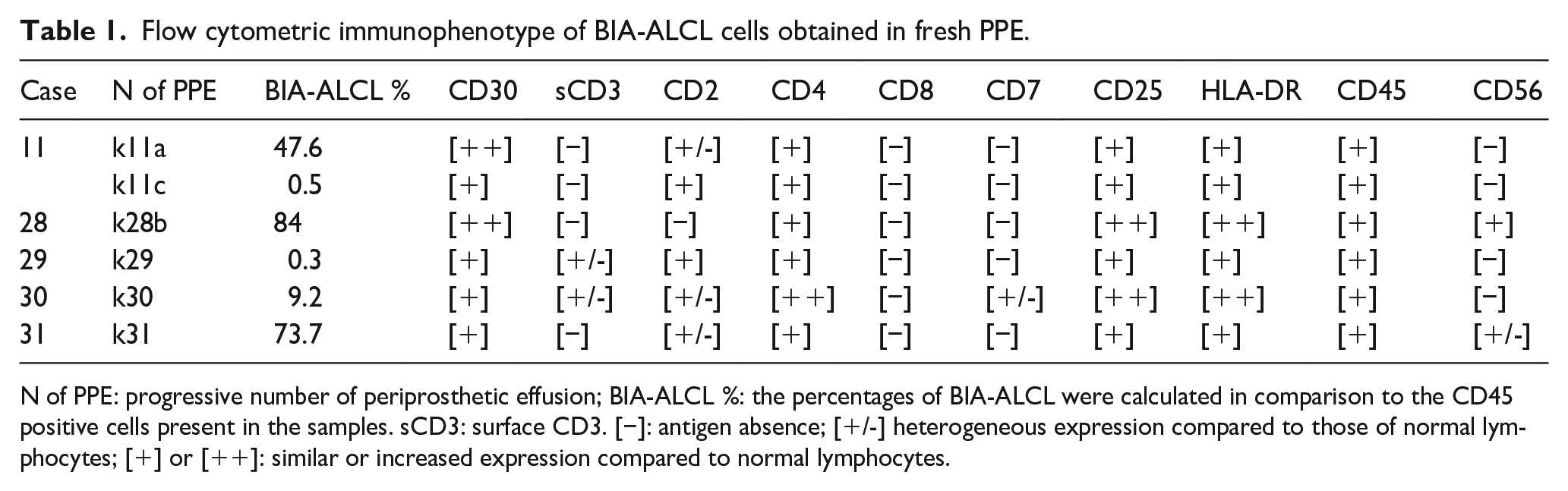
N of PPE: progressive number of periprosthetic effusion; BIA-ALCL %: the percentages of BIA-ALCL were calculated in comparison to the CD45 positive cells present in the samples. sCD3: surface CD3. [−]: antigen absence; [+/-] heterogeneous expression compared to those of normal lymphocytes; [+] or [++]: similar or increased expression compared to normal lymphocytes.
Reactive cells FCM immunophenotype
Beside the detection of BIA-ALCL cells, FCM allowed to precisely quantify the nature of leukocytes present in the effusions. Overall, a reduced percentage of neutrophils in respect to lymphocytes was observed. Furthermore, a prevalence of CD3+ CD4+ in respect to CD3+ CD8+ T lymphocytes was evidenced (online Supplementary Table 6). The PPEs with histiocytes predominant cellular signature at MGG, showed a higher percentage of enlarged CD4+ FSChigh and SSChigh CD30- CD25- HLA-DR+ cells most likely representing histiocytes (Figure 3B).
Capsulectomy results
A total of 15 capsules were submitted for histologic evaluation. Six of them were obtained from BIA-ALCL positive PPEs and three (capsules k28b, k30, and k31) were infiltrated by BIA-ALCL while the remaining showed only inflammatory cells.
Discussion
Although rare and with a favourable prognosis, the BIA-ALCL cases are predicted to increase during next few years, due to the growing number of breast implants used worldwide. In this scenario, the recognition of reliable methodologies able to quickly identify BIA-ALCL cells in PPEs developing after breast implants is imperative. In this report, we provide evidence suggesting that cytology is a sensitive method able to identify all suspected samples and that CD30 IHC, TCR gene rearrangement or FCM can be useful in confirming the presence of BIA-ALCL cells. Furthermore, the use of these additional analyses in cytology-negative samples, does not seem to add value in the BIA-ALCL diagnostic workup. Finally, we also provide a reliable FCM phenotype that can be used for rapid identification of BIA-ALCL cells in PPEs. BIA-ALCL is a recently described T cell lymphoma arising in women with breast implants for reconstructive or aesthetic reasons. BIA-ALCL often presents as a unilateral late (median time nine years after implantation) PPE around the breast implant. 4 In the majority of the reported cases, BIA-ALCL is associated with textured implants and, according to Ann Arbor staging system, 83% of women have clinical stage I, 10% stage II, and 7% stage IV disease at initial diagnosis. 2 Since PPE is the most common BIA-ALCL symptom, cytology constitutes the first assessment performed in all laboratories. The 2019 NCCN guidelines recommended that in addition to cytology, all BIA-ALCL suspected samples should be evaluated also by CD30 IHC or subjected to FCM for CD30+ T cell quantification. 6 However, at odds with suggested guidelines, some authors proposed that cytology constitutes the central element of BIA-ALCL cytopathological work up and the identification of atypical cells by morphology constitute the ‘key’ element for triggering further studies. 1 This fundamental role is mainly supported by some recurrent features of PPEs: i) in comparison with normal T small lymphocytes, BIA-ALCL cells are both, larger in size and with nuclear pleomorphism and ii), in most of the cases, constitute an high percentage of the PPE cellular elements. However, in our series, three samples showed a low number of atypical cells by morphology and two were then confirmed as BIA-ALCL positive by IHC and/or FCM and/or TCR rearrangement. In the remaining PPE, the presence of malignant cells was ruled out by both, CD30 IHC null immunophenotype and TCR gene rearrangement that resulted in polyclonal for γ chain and not informative for β chain. Since medium to enlarged reactive T-cells (accounting for less than 1% of total cellularity) can be found in almost all reactive PPEs, 12 our FCM results showing only 0.3 and 0.5% of BIA-ALCL cells in two samples, suggest the presence of a ‘grey zone’ (characterized by the presence of < 1% of BIA-ALCL cells) in which, in addition to cytology, further investigations are mandatory. Cytology, in addition to identify all positive samples (100% specificity), correctly recognized as negative 33 out of 34 PPEs leading to specificity of 97%. These results (obtained within the integrated group of PPEs samples), prove that no false-negative cases should be present among the 10 PPEs investigated only by cytology and overall support the use of cytology as central examination able to identify the PPEs that must be investigated using other methodologies. Our data indicates that there was no difference in sensitivity between IHC, FCM or TCR. In fact, all the CD30+ BIA-ALCL IHC-positive samples, were confirmed by FCM and/or TCR rearrangement. Overall, this evidence suggests that the choice of methodology could depend on clinical needs. In the presence of ‘suspected PPE’, cell block inclusion followed by CD30 IHC and TCR investigations can be preferred if a low amount of sample is available or if diagnostic questions are beyond those answerable by FCM as previously suggested.7,10 On the contrary, if a rapid support to cytology is requested by clinical conditions, FCM can provide results in few hours. In our hands CD30+ and CD4+ coupled with CD3+ resulted in the IHC core of BIA-ALCL cells. All cases expressed CD30 with strong staining and three (two being non evaluated) expressed CD4. Interestingly, although two samples showed focal or non-homogeneous CD3 staining, we did not observe the loss of CD3 expression reported by others groups.12,20 This discrepancy is in line with the reported heterogeneity of BIA-ALCL cells that are known to express CD3 in about 32% of the cases. 2 In addition to CD30 (with bright expression) and CD4, all BIA-ALCL positive PPEs investigated by FCM, also showed CD25 and HLA-DR while, among others T cell markers, only CD2 (five out of six samples), but not CD3, CD7 or CD8, was recurrently found. FCM was unable to confirm the presence of CD3 evidenced by IHC most probably because this marker is retained in cytoplasm and not exposed in the membrane. Overall, IHC and FCM results confirm BIA-ALCL as an anaplastic T cell lymphoma that, beyond CD30, also express at least one further T cell marker (mainly CD4 and/or CD2 and/or intracellular CD3) coupled with CD25 and HLA-DR. In line with this, both CD25 and HLA-DR have been commonly recognized in both, ALK positive or ALK negative ALCL. 21 Although the bright expression of CD30 in large CD45+ cells is a FCM ‘marker’ of BIA-ALCL cells,8,9 in the absence of surface CD3 it cannot be considered specific.7,10 However the more comprehensive profile FSChigh SSChigh CD30 (bright expression) CD4+ CD25+ HLA-DR+ coupled with CD2 expression can overcome the absence of surface CD3, and can be considered as a reliable FCM pattern useful for rapid identification of BIA-ALCL cells in PPEs in which atypical lymphocytes were already evidenced by cytology. In line with these conclusions, three papers published in literature, in addition to CD30 with bright expression, confirmed the presence of CD4 in 78%, 100% and 75% of PPEs respectively.8,9,22 Furthermore, when evaluated by FCM, CD25, HLA-DR and CD2 were found in 100% of BIA-ALCL positive PPEs. 8 Among the six capsules obtained from BIA-ALCL positive PPEs and submitted for histological evaluation, only three showed the presence of lymphoma. Although this result was probably due to a non-extensive sampling of the capsula, 2 indirectly it underlines that PPE analysis might be more comprehensive and informative than capsule for BIA-ALCL diagnostic management. Due to its retrospective nature, a limitation of our work, is that little new information is provided regarding BIA-ALCL. However, our analyses ‘characterized’ cytology in terms of sensitivity, specificity, PPV and NPV, elements that, to our knowledge, were not present in literature. BIA-ALCL is a rare disease (at the end of 2019, only 573 cases of BIA-ALCL were identified worldwide), 23 but considering both, the large number of breast implants and the augmented attention of clinicians, the number of cases will probably increase during next period implying the necessity of a reliable diagnostic work up. With our work we provide evidence that the recognition of atypical large lymphocytes by cytology in PPEs constitutes the central element of BIA-ALCL diagnostic management. All suspected cases have to be confirmed by the use of other investigations such as IHC, FCM or TCR gene rearrangement. Since no difference in sensitivity between IHC and FCM was evidenced, the choice can be made depending on clinical needs, laboratory-skills or sample quantity. Finally, FSChigh SSChigh CD30+ CD4+ CD25+ HLA-DR+ and CD3- seem to constitute an affordable profile for the identification of BIA-ALCL cells in PPEs by FCM.
Supplemental Material
sj-pdf-1-tmj-10.1177_03008916231157837 – Supplemental material for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup
Supplemental material, sj-pdf-1-tmj-10.1177_03008916231157837 for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup by Laura Vittoria, Laura Sala, Valeria Summo, Iolanda Capone, Elena Conca, Martina Toma, Joseph Ottolenghi, Francesca Testa, Umberto Cortinovis, Biagio Paolini, Antonello Cabras, Antonella Aiello and Fabio Bozzi in Tumori Journal
Supplemental Material
sj-pdf-2-tmj-10.1177_03008916231157837 – Supplemental material for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup
Supplemental material, sj-pdf-2-tmj-10.1177_03008916231157837 for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup by Laura Vittoria, Laura Sala, Valeria Summo, Iolanda Capone, Elena Conca, Martina Toma, Joseph Ottolenghi, Francesca Testa, Umberto Cortinovis, Biagio Paolini, Antonello Cabras, Antonella Aiello and Fabio Bozzi in Tumori Journal
Supplemental Material
sj-pdf-3-tmj-10.1177_03008916231157837 – Supplemental material for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup
Supplemental material, sj-pdf-3-tmj-10.1177_03008916231157837 for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup by Laura Vittoria, Laura Sala, Valeria Summo, Iolanda Capone, Elena Conca, Martina Toma, Joseph Ottolenghi, Francesca Testa, Umberto Cortinovis, Biagio Paolini, Antonello Cabras, Antonella Aiello and Fabio Bozzi in Tumori Journal
Supplemental Material
sj-pdf-4-tmj-10.1177_03008916231157837 – Supplemental material for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup
Supplemental material, sj-pdf-4-tmj-10.1177_03008916231157837 for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup by Laura Vittoria, Laura Sala, Valeria Summo, Iolanda Capone, Elena Conca, Martina Toma, Joseph Ottolenghi, Francesca Testa, Umberto Cortinovis, Biagio Paolini, Antonello Cabras, Antonella Aiello and Fabio Bozzi in Tumori Journal
Supplemental Material
sj-pdf-5-tmj-10.1177_03008916231157837 – Supplemental material for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup
Supplemental material, sj-pdf-5-tmj-10.1177_03008916231157837 for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup by Laura Vittoria, Laura Sala, Valeria Summo, Iolanda Capone, Elena Conca, Martina Toma, Joseph Ottolenghi, Francesca Testa, Umberto Cortinovis, Biagio Paolini, Antonello Cabras, Antonella Aiello and Fabio Bozzi in Tumori Journal
Supplemental Material
sj-pdf-6-tmj-10.1177_03008916231157837 – Supplemental material for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup
Supplemental material, sj-pdf-6-tmj-10.1177_03008916231157837 for Breast implant associated anaplastic large cell lymphoma: Evidence for an efficient diagnostic workup by Laura Vittoria, Laura Sala, Valeria Summo, Iolanda Capone, Elena Conca, Martina Toma, Joseph Ottolenghi, Francesca Testa, Umberto Cortinovis, Biagio Paolini, Antonello Cabras, Antonella Aiello and Fabio Bozzi in Tumori Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
