Abstract
Background:
Very little is currently known about molecular alteration of matrix-producing carcinoma of the breast. However, the morphological similarity with other neoplasm with a myxo-chondroid component is remarkable. In this pilot study we evaluated the molecular alterations involving PLAG1 and MYC genes in 12 cases of matrix producing carcinoma.
Methods:
We evaluated PLAG1 rearrangements as Break-Apart and Gene Copy Gain, and MYC as amplification and polysomy in 12 cases of matrix producing carcinoma using a FISH method.
Results:
Among the 12 cases of matrix producing carcinomas we found that the three cases harboring MYC amplification were all negative for PLAG1 break-apart; four cases with MYC polysomy were associated to PLAG1 break-apart and high Gene Copy Number; among four cases wild type for MYC, three showed a PLAG1- break-apart signal and of them two died with disease. One of the deceased patients showed an amplification of MYC with PLAG1- wild-type and the other showed a PLAG1 break-apart (6%) and a MYC wild-type.
Conclusion:
This is the first report to the best of our knowledge that shows a possible correlation between a matrix producing carcinoma with PLAG1 and MYC involvement in the development and progression of this kind of tumor. We can suppose that MYC amplification behaves in an aggressive way together with PLAG1- break-apart in the cases of matrix producing carcinoma presented here. The gene copy gain is a useful diagnostic tool in the case of difficult diagnosis because an increase was observed in more than 50% of cases.
Keywords
Introduction
Metaplastic carcinoma of the breast is a rare “special type” of tumor including different subtypes, all of which are extremely aggressive and with a poor prognosis.
The most common is metaplastic carcinoma with squamous differentiation, with components showing mesenchymal differentiation (chondroid, muscular or fibrous). With lower frequency the mesenchymal overgrowth can simulate a true sarcomatous appearance.
One type of metaplastic carcinoma is composed of a chondromyxoid background resembling the pleomorphic adenoma of the parotid gland (PA) and/or carcinoma ex pleomorphic adenoma (Ca-ex-PA) or a myxoid lipoblastoma in children. The authors discuss the role of PLAG1 and MYC involvement in development and aggressiveness of this tumor.
Material and methods
Between 2015 and 2017, we identified and followed-up 12 cases of matrix producing carcinoma (MPC). Fluorescence in-situ Hybridization (FISH) was performed using a dual-color break-apart (BA) PLAG1 probe from Empire Genomics (Buffalo, NY, USA). The PLAG1 probe is a dual-color break-apart probe consisting of a 265-kb 3′ centromeric side labeled in green and a 289-kb 5 ′ telomeric side labeled in orange.
Formalin-fixed paraffin-embedded sections (FFPE) 4 μm thick, were air-dried and oven baked for 30 minutes at 60°C. FISH was performed in accordance with the manufacturer’s technical instructions; 10 μl of PLAG1 mix probe was added, and the slides were cover slipped and sealed. Slides were then denatured for three minutes at 83°C and hybridized overnight at 37°C on a Thermo-Brite (DAKO Hybridizer). Following hybridization, coverslips were removed by placing the slides in a room temperature bath of 2XSSC, moved and heated to 0.4xSSC 0,3% IGEPAL 72°C for two minutes and then rinsed in 2xSSC 0,1% IGEPAL at room temperature. A two-fusion signal pattern indicates no rearrangement involving the PLAG1 gene, whereas a distant separation between orange and green signals indicates PLAG1 involvement. A positive FISH score, for PLAG1, was reported
The cases with high Gene Copy Gain (GCG) for PLAG1, were also analyzed using the LSI MYC Dual Color Break-Apart Rearrangement Probe (Abbott Molecular Inc., Des Plaines, IL, USA). This kit contains a mixture of LSI-MYC Spectrum-Orange probe and LSI-MYC Spectrum-Green probe. Briefly, 4-μm–thick, FFPE sections were air-dried and oven baked for 30 minutes at 60°C and then processed following the manufacturer’s recommendations on the Abbott Vysis. After the slides were processed, 10 μl of MYC break-apart probe (Abbott Molecular, Inc.) were added, and the slides were cover slipped and sealed. Slides were then denatured for 10 minutes at 80°C and hybridized overnight at 37°C on a Thermo-Brite. Following hybridization, coverslips were removed by placing the slides in a room temperature bath of 2 min 2X SSC and then moved to a second bath heated to 73°C for three minutes. They were then rinsed in Wash Buffer. DAPI (4’,6-Diamidino-2-Phenylindole, Dihydrochloride) was then applied to each slide, and a coverslip was applied. For amplification of MYC the threshold limit of MYC/CEP8 ratio was ⩾2 was used following the international guideline. Orange staining was used to evaluate the MYC locus at 8q24 while green staining was used to evaluate the centromeric 8 chromosome (8-CEP).1,2 All cases underwent surgical procedures as mastectomy or quadrantectomy following our Institutional Guidelines and an adjuvant or neoadjuvant CT was performed in almost all cases.
Results
This type of carcinoma showed a frequency up to 5% in our series and rarely expressed hormonal receptors (estrogen, progesterone and Her2/Neu).3,4
The patients ranged from 40 to 78 years old with a mean of 64.7 years, and the tumor size ranged from 1.1 to 5 cm (mean 2 cm). Only one case showed an ER and PgR receptor expression (both in 90% of cells) with negative Her-2/Neu expression by IHC (Table 1). From a morphological point of view, the neoplasm was composed of epithelial cells with anaplastic features, aggregated in poorly cohesive nests or strands intermingled with a chondromyxoid matrix, and we also detected foci of coagulative necrosis (Figure 1). All tumors had a high mitotic rate and a high proliferative index evaluated with Ki-67 labeling index (average 54.7%) (Table 1). We observed the classic rearrangement as BA (Break-Apart) of PLAG1 in eight cases (BA From 2% to 29%), four cases did not show a BA (Table 2). We observed an increase of GCG for PLAG1 from 1.7 up to 6.1 signal for cells. At times there was an increase of red 5’-signal (average gain 3.7 signal for cell) and a high level of green 3’-signal (average gain 22.4 signal for cells) (Table 2, Figures 2 and 3). Cases with a PLAG1 rearrangement showed a GCG (34.7%) and an increased green signal (68%). This unexpected observation suggested more involvement of 8-CHR. To ascertain whether or not this represented a true polysomy, we evaluated MYC status. Since MYC is present in 8-CHR, MYC evaluation is more accurate than PLAG1 for ascertaining polysomy. A true polysomy for MYC (threshold value >3) was found in a high percentage of cases (33.3%). An amplification of MYC, was found in three cases with MYC/CEP-8 ratio ⩾ 2. only one case showed a borderline value of 1.9 (case no.11) (Table 2).
General feature of MPC in our cases.
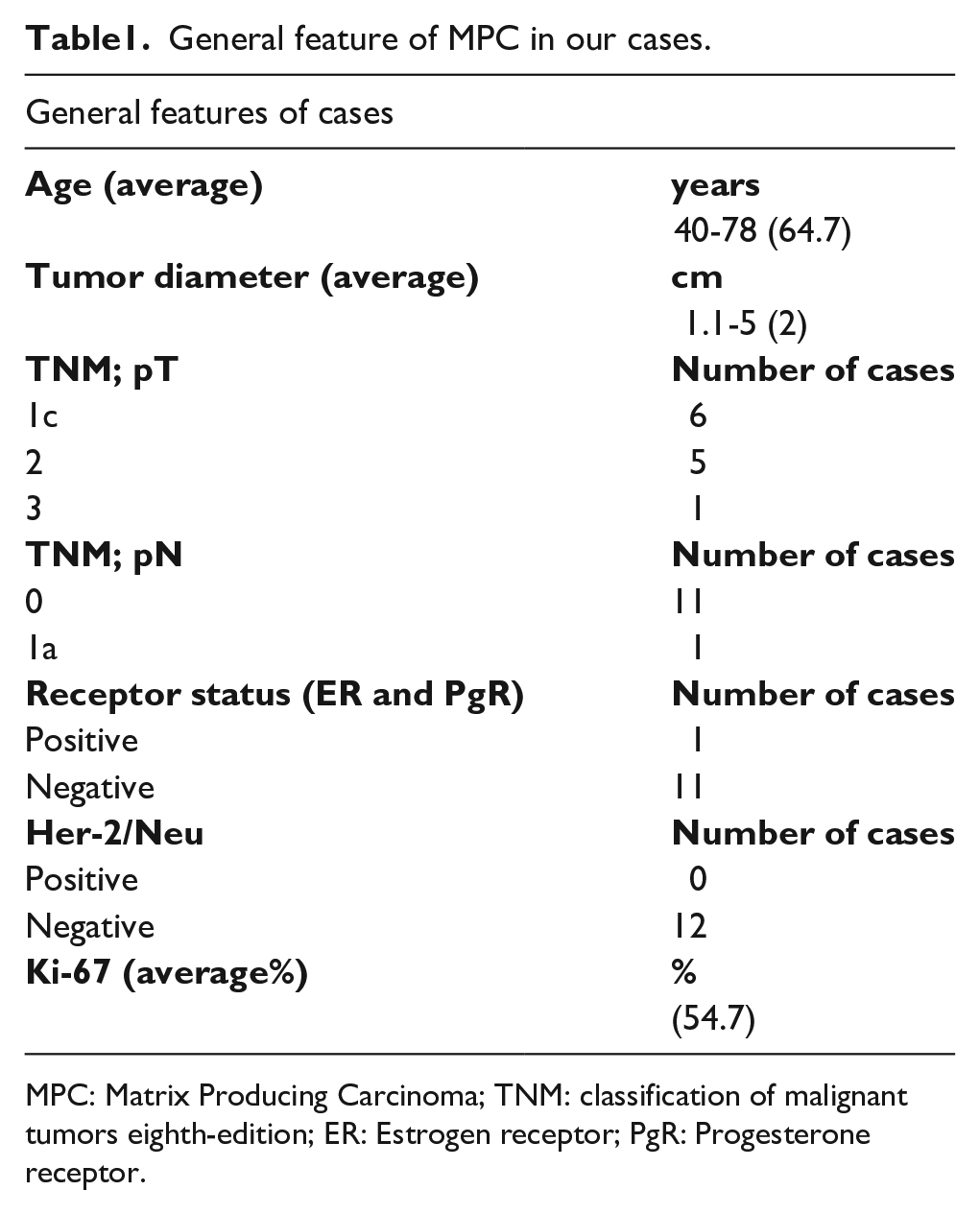
MPC: Matrix Producing Carcinoma; TNM: classification of malignant tumors eighth-edition; ER: Estrogen receptor; PgR: Progesterone receptor.
Scoring the PLAG1 as break-apart (BA), GCN (gene Copy Number), single extra Red or Green signal and MYC genes alteration observed in MPC.
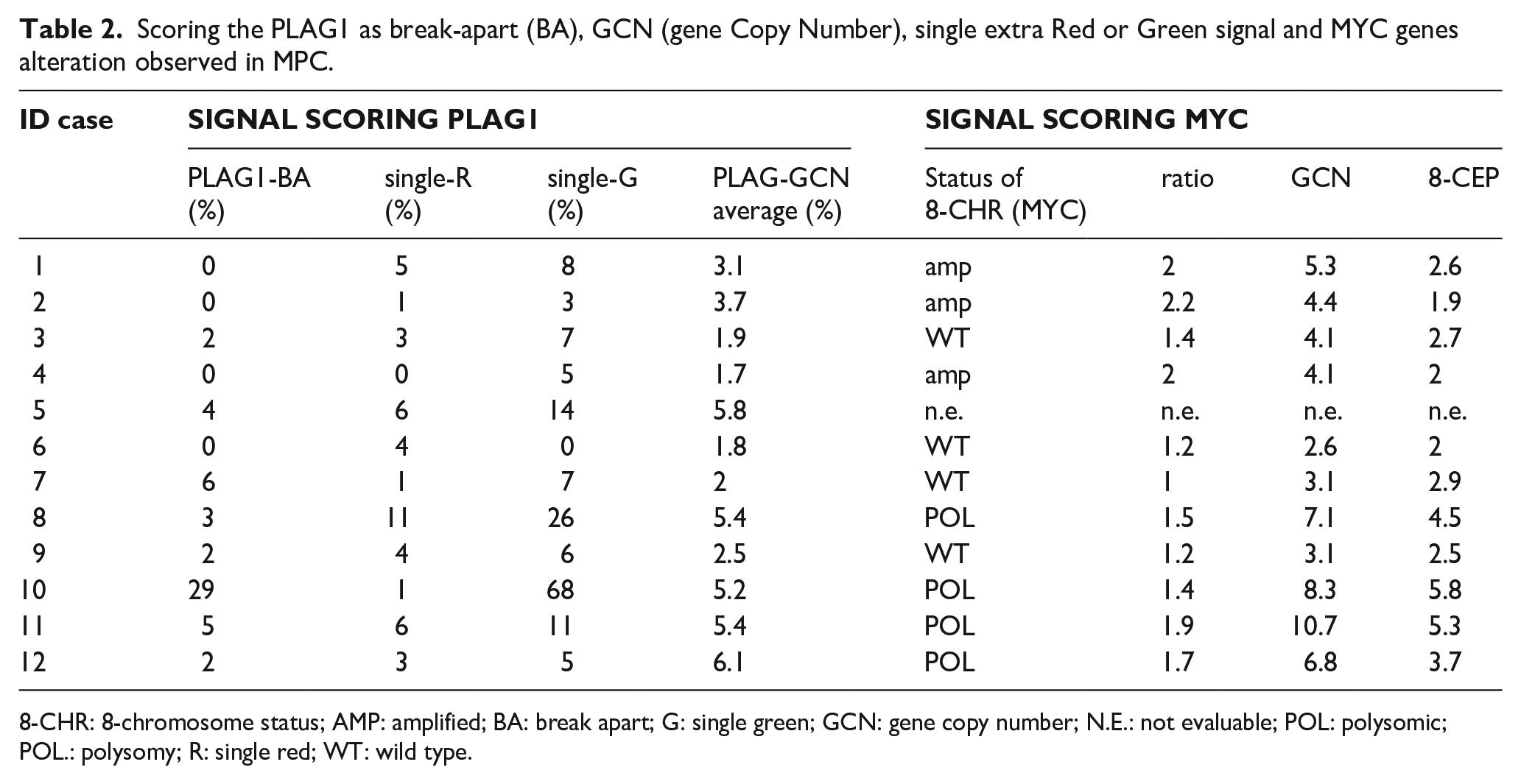
8-CHR: 8-chromosome status; AMP: amplified; BA: break apart; G: single green; GCN: gene copy number; N.E.: not evaluable; POL: polysomic; POL.: polysomy; R: single red; WT: wild type.
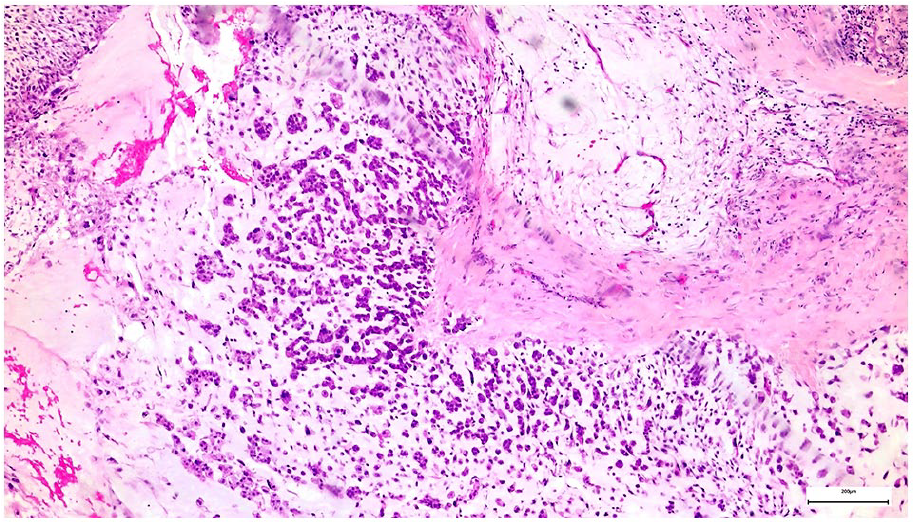
H&E stain of MPC characterized by isolate plasmocytoid-epithelioid cells intermingled with chondromyxoid matrix.
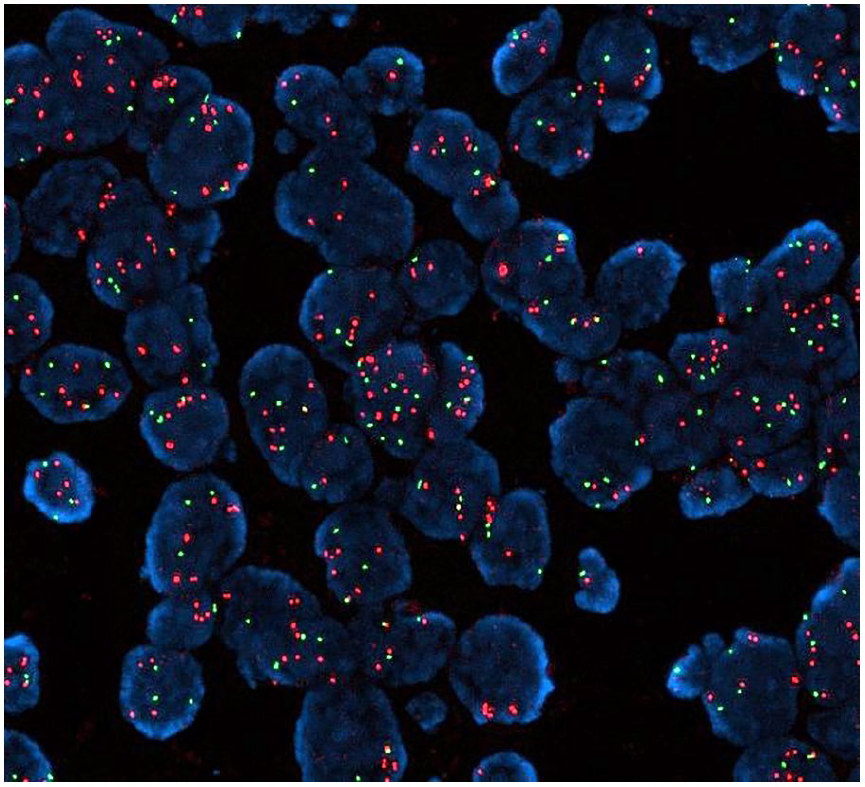
Amplification of MYC by FISH, red shows the gene and green is an 8CEP.
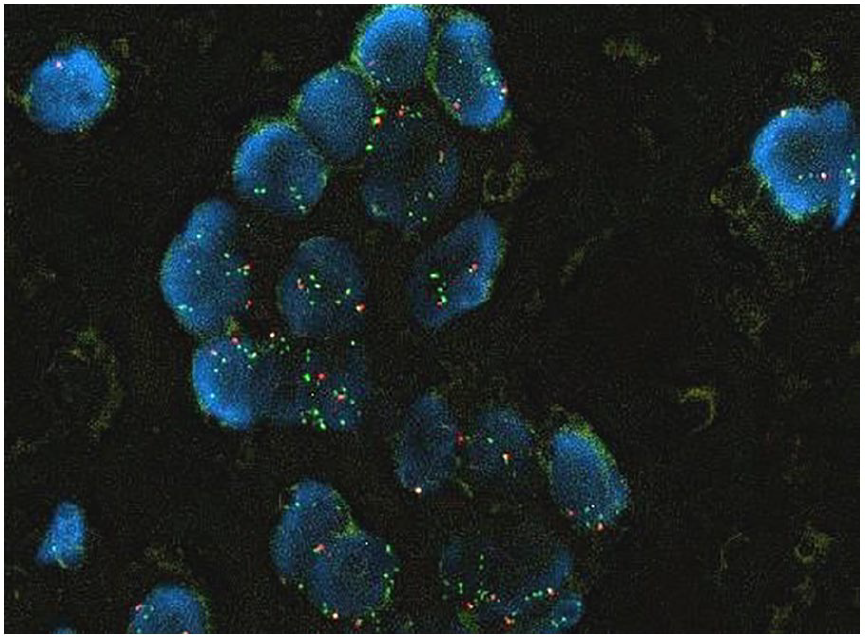
Extra-green signal for PLAG-1 observed in many of our cases by FISH.
In two cases, a neoadjuvant therapy (NeoA-Therapy) was used. In one case with EC (case no.3), and the other with a taxane-based drug following an IEO trial Protocol (data not shown) (case no.2). Due to disease progression, the second case was excluded from the protocol and an Adjuvant therapy (Adj-Therapy) was carried out with EC.
At the last follow-up, only two cases died with disease (DWD; case no. 2 and 7) (Table 3) and the others were NED (not evidence of disease) (Table 3; Figure 4).
Physical and surgical procedure with Follow Up (last FU*) of our cases.
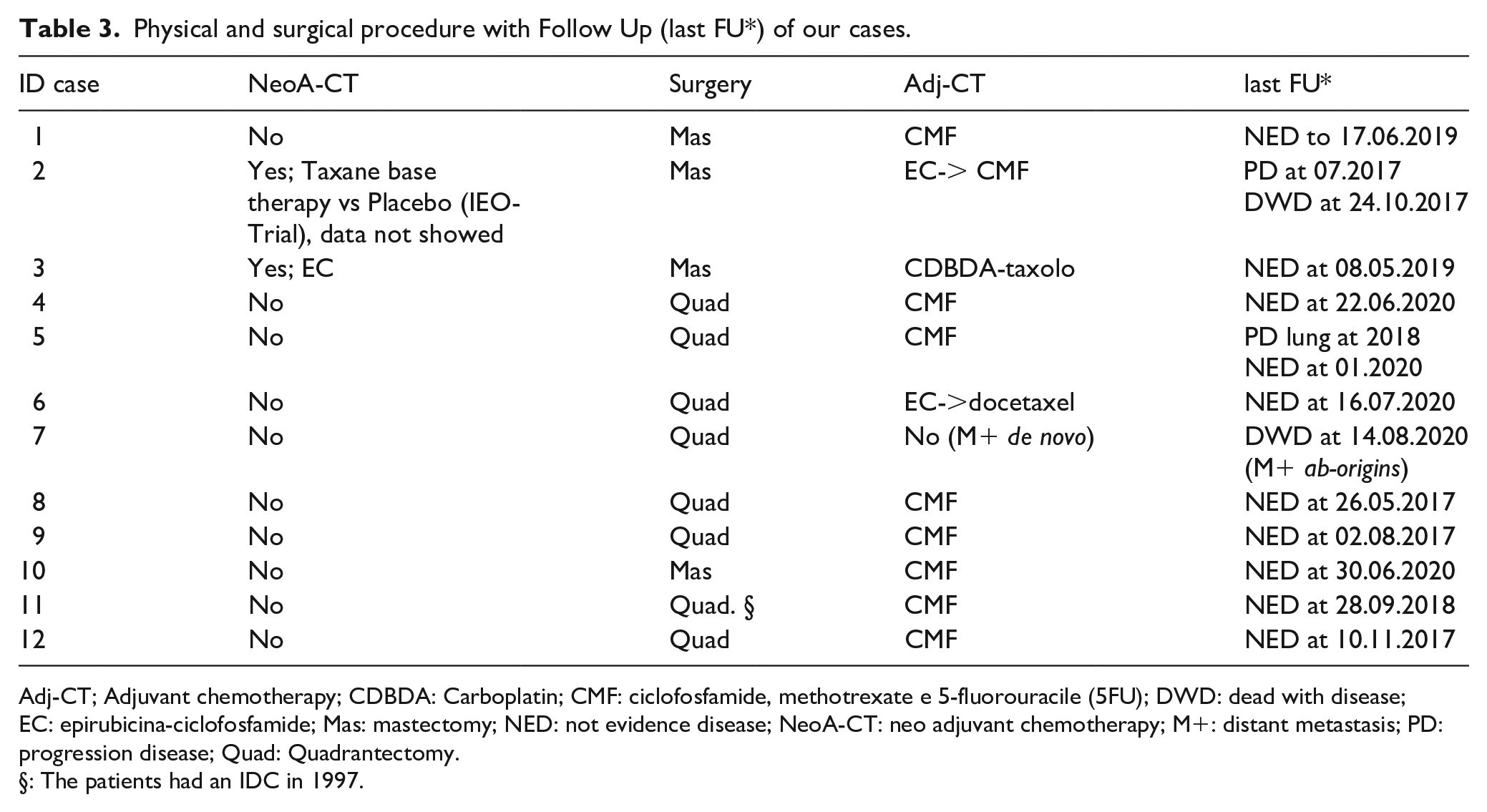
Adj-CT; Adjuvant chemotherapy; CDBDA: Carboplatin; CMF: ciclofosfamide, methotrexate e 5-fluorouracile (5FU); DWD: dead with disease; EC: epirubicina-ciclofosfamide; Mas: mastectomy; NED: not evidence disease; NeoA-CT: neo adjuvant chemotherapy; M+: distant metastasis; PD: progression disease; Quad: Quadrantectomy.
The patients had an IDC in 1997.
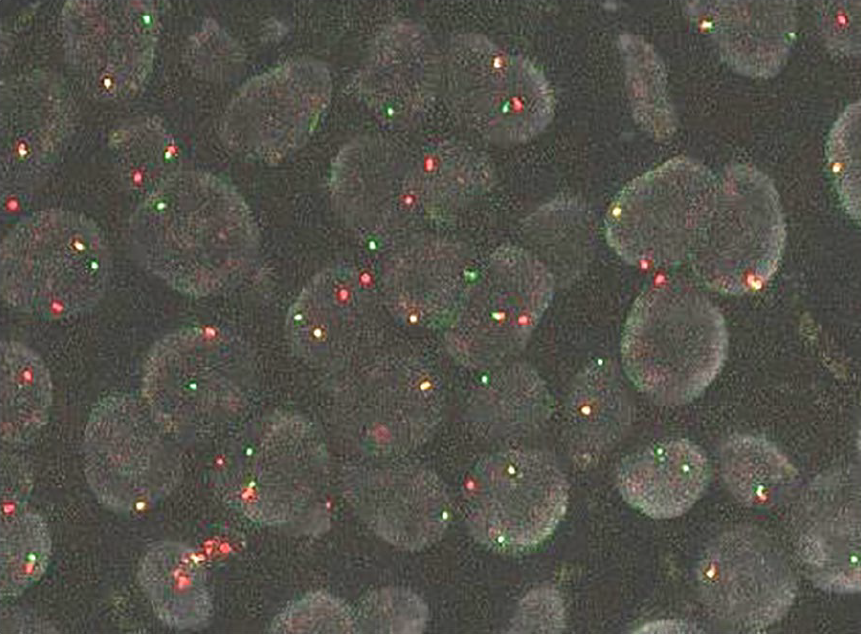
Break-Apart split red and green signals observed for PLAG1 gene evaluated by FISH.
The cases with MYC amplification did not show PLAG1-BA, while all the cases polysomic for MYC showed PLAG1-BA. Among Wild Type (WT) cases of MYC (four cases), one was negative for PLAG1-BA rearrangement while others were rearranged with PLAG1-BA (Table 2).
Discussion
Pleomorphic Adenoma Gene-1 PLAG1, located in CHR 8q12.1, encodes a nuclear zinc finger protein that is rearranged in many tumors while MYC is a transcriptional protein factor (bHLH transcription factor) linking many other genes and acquiring a pro-oncogene activity. This gene observed in CHR 8q24.21 plays a pivotal role in regulation, progression, cellular transformation and apoptosis in great number of tumors, hematological and solid ones. PLAG1 were observed rearranged in many cases of PA and/or Ca-ex-PA (frequency of 80% of cases), in myoepithelial tumor of salivary gland, in myxoid-lipoblastoma in children and in some mesenchymal tumors of the uterus. MPC is a rare breast tumor that shares a similar morphology with those tumors. Following these morphological oddities, our hypothesized that MPC could share the same PLAG1 alteration.5,6
To the best of our knowledge, no association has been published in the biomedical literature between PLAG1 and MYC rearrangements and MPC. We observed a rearrangement of PLAG1, involving 8-CHR, not only as a BA signal, but also as Gene Copy Gain (GCG). These alterations were observed and reported in salivary tumors and myxoid lipoblastoma.7,8 MYC involves the same chromosome of PLAG1 and we tried to understand that PLAG1-GCG was a true polysomy or not. We observed that the cases with PLAG1-BA showed a MYC polysomy, while the cases without PLAG1-BA showed MYC amplified, moreover, cases with high value PLAG1-GCG showed a MYC-polysomy, while the cases with low value of PLAG1-GCG showed an amplification of MYC (Table 2). These observations suggest that PLAG1-GCG, in MPC, is due to a polysomy. PLAG1-GCG was observed in salivary gland tumor together with PLAG1 deletion. The PLAG1-GCG without a BA signal suggest a chromosomic rearrangement of CHR 8q12 followed by PLAG1 upregulation in a same manner used in lymphomas and due to an oncogenic mutation of promoted region of MYC. This is a cause of polysomy of 8-CHR in non rearranged PLAG1 cases.9-11 The association between PLAG1-GCG and MYC amplified-polysomic was observed in salivary glands. In cases reported in literature the amplification of MYC give an aggressive behavior in a subgroup of salivary tumors, this can also happen in MPC.8,12,13
MYC were amplified even in a subgroup of inflammatory breast carcinomas with poor prognosis that showed a high-expression of hormonal receptor (Estrogen and Progesteron) and absence of Her-2/Neu. 14
In our cases it was difficult to identify this association because so few cases were studied. Among the two cases DWD one showed a MYC amplification without PLAG1-BA and the others one was MYC-WT with PLAG1-BA in 6% of neoplastic cells (cases n.2 and n.7; Table 2). We suppose that MYC amplification gives an aggressive behavior together with PLAG1-BA in the cases of MPC discussed here.9-11
Rearrangement of PLAG1 in salivary gland tumors does not change the prognosis, in fact, PLAG1 was observed in benign and malignant salivary tumor with the same frequency. Only MYC changes the prognosis. Among MYC-WT cases we observed a PLAG1-BA rearrangement, suggesting a difference between salivary tumors and MPC with involvement of PLAG1-BA in the prognosis and aggressiveness of neoplasms.9,10
Despite morphological similarities between MPC and other salivary and soft tissue myxoid tumors, its behavior is different.9,10
Because of the few cases analyzed we cannot show a statistical correlation between prognosis and the PLAG1 and MYC alterations. In literature this is the first report that shows an involvement of PLAG1 and MYC in prognosis and behavior of breast MPC. So, this is should be a bases for a future studies.
Footnotes
Acknowledgements
We would like to thank our librarian Russel William for the language review and critical article revision and also our laboratory technicians Flavia Giarratano, Mila Schiavi, Marilisa Valente and Francesco Spinelli, for their work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
