Abstract
Background:
With the availability of multiple treatment options for metastatic castration-resistant prostate cancer (mCRPC), new real-world data on disease management and drugs’ performance are needed.
Methods:
We described characteristics, management and clinical outcomes of patients receiving first-line mCRPC treatment within the Italian cohort of the real-world, prospective, international Prostate Cancer Registry. Patients were enrolled consecutively (2013-2016) in 32 Italian sites and followed for 3 years.
Results:
238 patients were included: 157 received first-line abiraterone acetate plus prednisone (“abiraterone” thereafter) and 70 first-line docetaxel; 11 patients receiving other treatments were not considered. Compared with docetaxel-treated patients, those receiving abiraterone were significantly older (age ⩾75: 63.7% vs 38.6%), less frequently had a Gleason score >8 (48.2% vs 67.6%, p<0.005) at initial diagnosis, and more frequently an ECOG score ⩾1 (52.7% vs 36.2%, p<0.05) and comorbidities (76.4% vs 57.1%, p<0.05) at baseline; they reported a lower analgesic use (15.3% vs 30%, p<0.005). In the abiraterone group (median follow-up 22.1 months), median time to progression (TTP) and progression-free survival (PFS) were, respectively, 14.4 months (95% confidence interval, CI, 10.6-18.0) and 13.0 months (95% CI, 9.1-16.8); median overall survival (OS) was not reached, and 3-year OS was 59.1%. In the docetaxel treatment group (median follow-up 25.3 months), median TTP, PFS and OS were, respectively, 8.2 months (95% CI, 6.1-10.3), 8.2 months (95% CI, 5.8-10.3) and 33.2 months (95% CI, 19.2-not estimable).
Conclusion:
This investigation provided valuable information on the overall mCRPC treatment pattern and the effectiveness of first-line abiraterone and docetaxel in a population representative of everyday practice.
Keywords
Introduction
Worldwide, prostate cancer ranks second in cancer incidence and fifth in cancer mortality in men, with about 1.4 million new cases and 375,000 associated deaths estimated to have occurred in 2020. 1
The large majority of newly diagnosed prostate cancer patients (>90%) present with localized or locally advanced disease and have good prognosis and treatment options. Patients progressing to metastatic disease, 2 despite the dramatic initial response to androgen deprivation therapy (ADT), 3 inevitably progress to castration-resistant prostate cancer (CRPC) within a median of 2-3 years, with subsequent poor prognosis and increased mortality. 4
Since 2004, docetaxel-based chemotherapy has been the standard of care for patients with metastatic CRPC (mCRPC).5-7 In the last 10 years, however, the advent of novel agents with varying mechanisms of action, including androgen receptor (AR)-targeted agents, novel chemotherapies and radiopharmaceuticals, has dramatically enriched the therapeutic armamentarium of mCRPC.8,9 In particular, the androgen biosynthesis inhibitor abiraterone acetate (abiraterone, henceforth) and the antiandrogen enzalutamide have become widely used in clinical practice, both in the post-docetaxel and post-ADT settings, 10 because of their excellent antitumor effects, good safety profile over docetaxel, and ease of administration.
With the availability of multiple treatment options for mCRPC and taking into consideration the clinical heterogeneity of the disease and the possible cross-resistance among drugs, it is relevant to clarify how these treatments fit into the current treatment paradigm for mCRPC and to evaluate their performance in unselected patients from clinical practice.
The Prostate Cancer Registry (NCT02236637) is the first and largest prospective, international, observational study collecting real-world data on mCRPC patients. 11 The Prostate Cancer Registry aims to describe the characteristics, management, and outcomes of patients with mCRPC in routine clinical practice. Since it collected data on over 3000 patients with mCRPC from 16 European countries ad hoc and prospectively, it represents a unique opportunity to improve the understanding and the management of the disease. Recently, the Registry published results on the parallel effectiveness of first-line abiraterone acetate plus prednisone, enzalutamide and docetaxel in mCRPC, indicating reasonably good patient outcomes even in subgroups of patients with cardiovascular diseases or diabetes. 11
The present investigation presents the baseline characteristics, treatment pattern and clinical outcomes of 227 patients receiving their first treatment for mCRPC in the Italian population of the Prostate Cancer Registry. To our knowledge, our study represents the largest prospective investigation on the overall mCRPC pattern of treatment in a real-life Italian scenario.
Methods
This study is based on the Italian cohort of the Prostate Cancer Registry, a prospective, international, non-interventional, observational study of patients with mCRPC initiated in 2013. 11 The Prostate Cancer Registry methods were previously reported. 11 Briefly, the Registry collected real-world data on over 3000 mCRPC patients from 199 centers specialized in the treatment of prostate cancer across Europe. The Registry aims to document the characteristics, management, and outcomes of a broad range of mCRPC patients treated in routine clinical practice with any mCRPC treatment.
Eligible patients were men aged ⩾18 years with histologically confirmed diagnosis of adenocarcinoma of the prostate with mCRPC, i.e., documented metastatic prostate cancer and progression of prostate cancer despite surgical or medical castration (defined as a testosterone level <50 ng/dL), under surveillance according to clinical practice or who started a systemic treatment for mCRPC after the baseline data collection. Patients were enrolled consecutively, at any time after diagnosis. The study lasted 5.5 years (study end date 9 July 2018), and patients were followed for a maximum of 3 years, independently from the treatment received. The investigation was approved by the Ethics committees of the participating centers and was conducted according to the principles of the Declaration of Helsinki and the Good Clinical Practice guidelines. All patients provided written informed consent to participate in the study.
In Italy, enrollment took place between 2013 and 2016 in 32 participating centers. The present investigation is based on patients who received their first treatment for mCRPC within the Italian cohort of the Registry. We considered only patients receiving abiraterone acetate plus prednisone or prednisolone (“abiraterone”, thereafter) or docetaxel (95.4%); patients who started other treatment for mCRPC (e.g., enzalutamide, radium-223 or other chemotherapy) were excluded from the analysis because numbers were too low (n=11).
Information of interest was collected at baseline and prospectively every ̴3 months during routine follow-up over the 3-years study period. Only data available as per clinical practice were recorded, extracted primarily from the medical records of the enrolled patients. At baseline, data on patient demographics, co-morbid medical conditions, concomitant medications, prior disease history, disease management, and clinical data (including selected biological parameters, performance status, Tumor, Node, Metastasis [TNM] stage and Gleason score) were collected. Follow-up data included clinical assessment, serum total prostate-specific antigen (PSA), performance status according to Eastern Cooperative Oncology Group (ECOG PS) and all therapies for mCRPC (local and systemic anticancer therapies, including reasons for starting, choosing and discontinuing such therapies). At the time of study start, abiraterone acetate was a relatively new drug for the treatment of chemotherapy-naïve mCRPC patients in the country (approval in 2014), and adverse events were collected according to regulatory requirements. The study did not attempt to collect adverse events associated to docetaxel, i.e., a standard regimen with a well-known safety profile.
The study outcomes were the duration of the initial therapy and the reason for stopping, overall survival (OS), progression-free survival (PFS), and time to progression (TTP).
Statistical analysis
The current investigation aims to describe patients’ characteristics and the clinical outcomes of patients receiving first-line treatment for mCRPC in clinical practice. Continuous variables were presented as mean values ± standard deviation (SD) or median (ranges), and categorical variables as numbers and percentages. Baseline characteristics of patients receiving abiraterone and docetaxel were compared using the chi-square test for categorical variables or either the t-test or the Mann–Whitney U test for normally distributed and non-normally distributed continuous variables, respectively. The distribution of time-to-event variables was estimated using standard survival analysis methods, including Kaplan-Meier product-limit survival curve. The median time to event with 2-sided 95% confidence interval (CI) was also calculated.
Results
A total of 238 patients with no previous therapy for mCRPC started an mCRPC treatment within the Italian part of the Prostate Cancer Registry. Abiraterone acetate plus prednisone or docetaxel as first-line treatments were received, respectively, by 157 and 70 patients, who constituted the study population of the present investigation; 11 patients starting other treatments were not considered in the present analysis Median time from initial prostate cancer diagnosis to study inclusion was 3.9 years (range: 0-27) in the abiraterone acetate group and 2.2 years (range: 0-17) in the docetaxel group. Most patients (n=213 out of 227) were enrolled in the Registry at the time of starting their first mCRPC treatment (the remaining were enrolled while in surveillance).
Baseline characteristics
Patients starting abiraterone acetate plus prednisone were significantly older than those starting docetaxel (age ⩾75: 63.7% vs 38.6%, respectively, p<0.001) (Table 1). At initial prostate cancer diagnosis, a significantly higher proportion of patients treated with docetaxel than abiraterone had a Gleason score ⩾8 (67.6% vs 48.2%, p<0.05), and 34.8% of patients in the abiraterone acetate plus prednisone group and 46% of patients in the docetaxel group had de novo metastases. At baseline, a significantly higher proportion of abiraterone-treated than docetaxel-treated patients had an ECOG-PS ⩾1 (52.7% vs 36.2%, p<0.05). Bone and visceral metastases were detected, respectively, in 76.1% and 12% of abiraterone-treated patients and 84.1% and 15.8% of docetaxel-treated patients. In particular, 5 patients (3.5%) in the abiraterone acetate plus prednisone group and 5 (7.9%) in the docetaxel group had liver metastases. A significantly higher proportion of patients receiving abiraterone acetate plus prednisone had comorbidities compared to patients receiving docetaxel (76.4% vs 57.1%, p<0.05), in particular cardiovascular conditions (70.1% vs 51.4%, p<0.05) (mainly hypertension). In the abiraterone and docetaxel groups, 14.0% and 12.9% of patients had diabetes. Respiratory (abiraterone: 7.6%, docetaxel: 7.1%), renal (5.7% and 4.3%), hepatic (4.5% and 3.8%) and neurological diseases (3.8% and 5.7%) were reported by less than 10% of patients in both groups. In the docetaxel group there was significantly more frequent use of analgesics (30% vs 15%, p<0.05); the prevalence of strong acting opioids use was 12.9% in the docetaxel and 8.3% in the abiraterone plus prednisone group.
Patient demographics and disease and biological characteristics at study inclusion among patients starting a first-line therapy for mCRPC in the Italian cohort of the Prostate Cancer Registry.
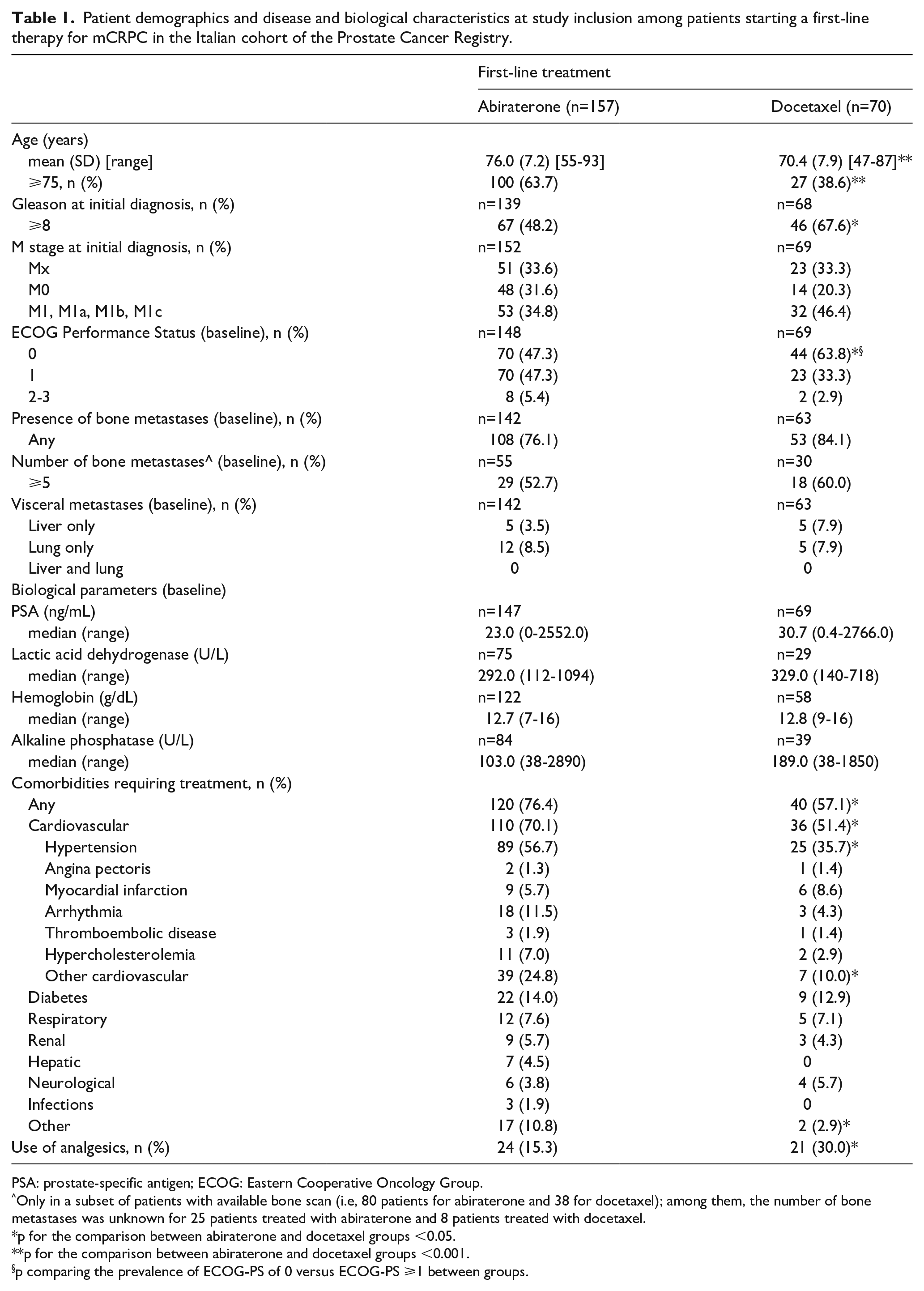
PSA: prostate-specific antigen; ECOG: Eastern Cooperative Oncology Group.
Only in a subset of patients with available bone scan (i.e, 80 patients for abiraterone and 38 for docetaxel); among them, the number of bone metastases was unknown for 25 patients treated with abiraterone and 8 patients treated with docetaxel.
p for the comparison between abiraterone and docetaxel groups <0.05.
p for the comparison between abiraterone and docetaxel groups <0.001.
p comparing the prevalence of ECOG-PS of 0 versus ECOG-PS ⩾1 between groups.
Almost half of the patients in both groups underwent radical prostatectomy and/or prostate-directed radiotherapy after the initial diagnosis of prostate cancer, and all the patients in the abiraterone acetate group and 94% of those in the docetaxel group had received prior systemic anticancer therapy (mostly endocrine therapy).
Treatment exposure and outcomes of patients treated first-line with abiraterone acetate plus prednisone
The median follow-up time for abiraterone acetate-treated patients was 22.1 months and the estimated median duration of the treatment was 14.5 months (95% CI, 11.0 – 18.5). The most common reason for stopping treatment was disease progression (n=74 out of 107 with available information, i.e., 69.2%); 12 patients (11.2%) discontinued abiraterone acetate because of toxicity and 17 (15.9%) for other reasons. Three patients (2.8%) died during treatment. Sixty-five patients (41.4%) received a subsequent therapy during the study period; among them, 39 switched to docetaxel, 11 to enzalutamide, and the remaining to other regimens (e.g., other chemotherapies, radium 223). The estimated median time to the next therapy was 21.9 months (95% CI, 19.3-not estimable).
Figure 1 shows TTP (Panel A), PFS (Panel B), and OS (Panel C) for mCRPC patients treated in first-line with abiraterone acetate plus prednisone. The estimated median TTP was 14.4 months (95% CI, 10.6-18.0). Median PFS was 13.0 months (95% CI, 9.1-16.8), and PFS rates were 51% (95% CI, 42.8-58.7) at 1 year and 20.3% (95% CI, 13.4-28.3) at 3 years. Median OS was not reached, and the 1- and 3-year OS rates were, respectively, 84.0% (95% CI, 77.1-89.0) and 59.1% (95% CI, 49.7-67.4).
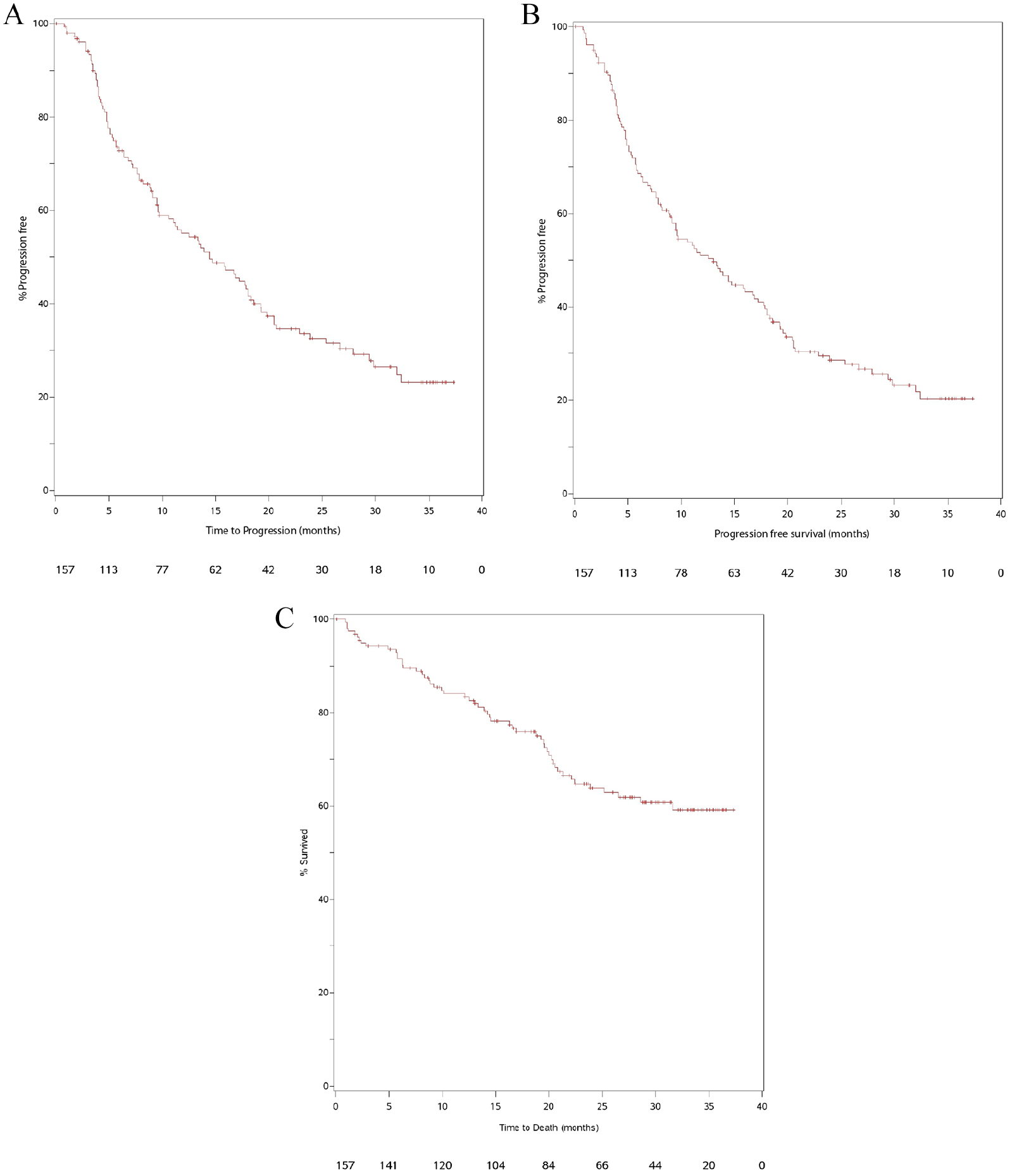
Kaplan-Meier curves for time to progression (Panel A), progression-free survival (Panel B) and overall survival (Panel C) for patients with metastatic castration resistant prostate cancer treated first-line with abiraterone within the Italian Prostate Cancer Registry.
Fifty-seven patients (43.5% of those with available information) achieved a PSA decline >50% within 6 months of abiraterone acetate plus prednisone treatment.
Seventy-one patients (45.2%) experienced treatment-emergent adverse events (TEAE), requiring temporary drug interruption in 27 patients (38%), drug withdrawn in 3 (4%) and dose reduction in 1 patient (1.4%). In 44 patients, the TEAE resolved (or was resolving at the last documented follow-up); in 16 patients the TEAE did not resolve in study and in 11 patients was fatal for the individual. 31 patients (19.7%) had at least one serious TEAE. The most common serious TEAE were cardiovascular events in 8 patients (2 patients had atrial fibrillation, 2 cardiac failure, 1 cardiac arrest, 1 cardiac failure congestive, 1 myocardial infarction and 1 supraventricular tachycardia).
Treatment exposure and outcomes of patients treated first-line with docetaxel
Patients receiving docetaxel as first-line treatment for mCRPC were followed for a median of 25.3 months. In this population, the estimated median treatment duration was 5.1 months (95% CI, 4.2 – 5.6), for typically 6-10 cycles. Thirty-one patients (45.6% of those with available information) completed the therapy; 29 patients (42.6%) stopped docetaxel use because of disease progression and 6 (8.8%) because of toxicity. Two patients died during treatment. Fifty-five patients (78.6%) received a subsequent therapy; among them, 28 were administered abiraterone acetate plus prednisone, 11 enzalutamide, 11 cabazitaxel, and the remaining other therapies. The estimated median time to the next therapy was 9.8 months (95% CI, 7.9-12.2).
Figure 2 shows the Kaplan-Meier curves for TTP (Panel A), PFS (Panel B) and OS (Panel C) for mCRPC patients treated in first-line with docetaxel. The estimated median time to progression was 8.2 months (95% CI, 6.1-10.3). Median PFS was 8.2 months (95% CI, 5.8-10.3), and PFS rates were 29.9% (95% CI, 19.2- 41.5) at 1 year and 5.2% (95% CI, 1.2-14.1) at 2 years. The corresponding estimates for OS were, respectively, 33.2 months (95% CI, 19.2-not estimable), 76.2% (95% CI, 64.1-84.7) and 42.6% (95% CI, 29.1-55.5).
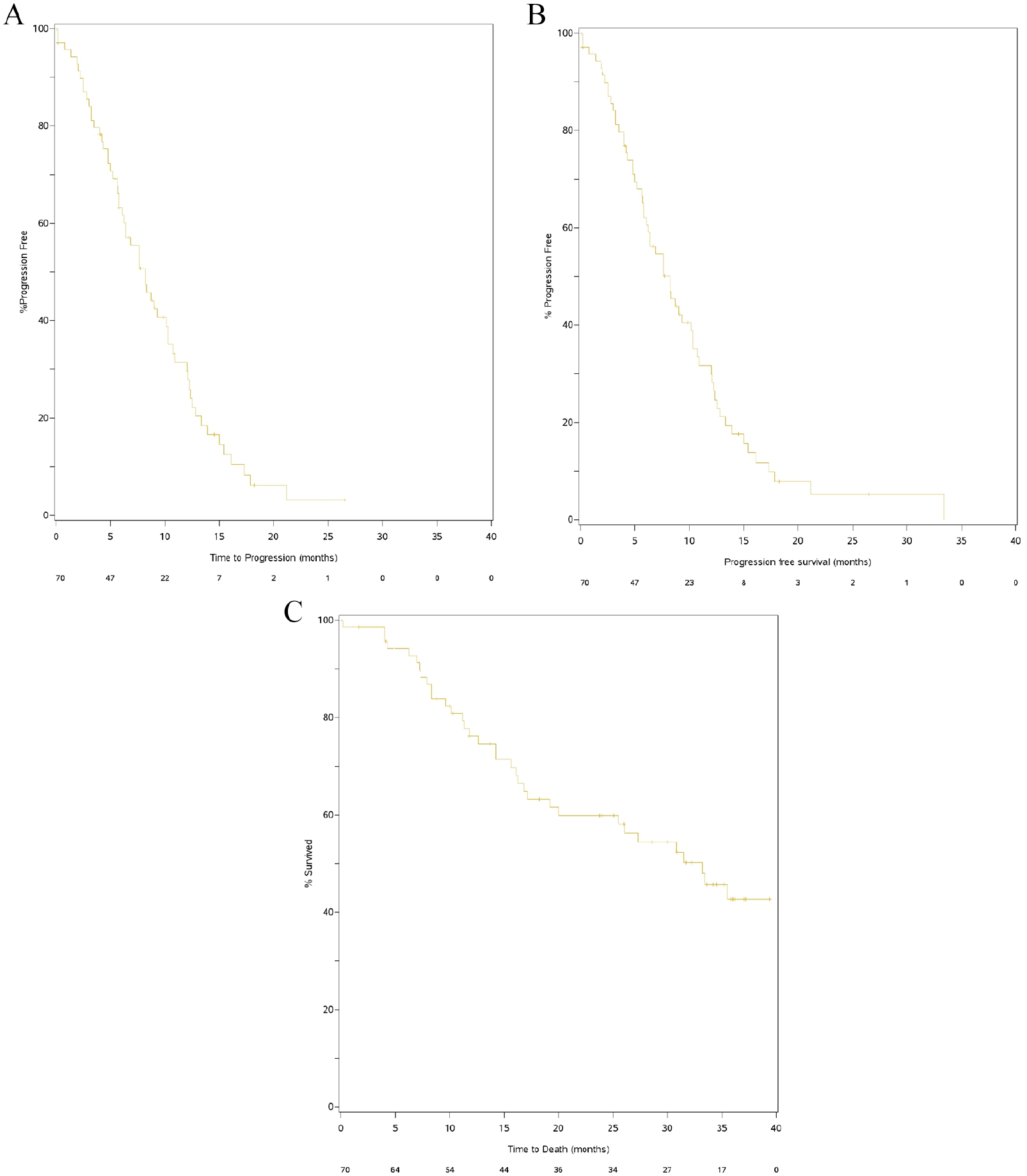
Kaplan-Meier curves for time to progression (Panel A), progression-free survival (Panel B) and overall survival (Panel C) for patients with metastatic castration resistant prostate cancer treated first-line with docetaxel within the Italian Prostate Cancer Registry.
In this cohort, a PSA decline of >50% within 6 months after starting docetaxel was observed in 32 patients (50.8% of those with available information).
Discussion
The present analysis within the Prostate Cancer Registry provides original and interesting real-world data on characteristics, treatment pattern and clinical outcomes of a large cohort of Italian mCRPC chemotherapy-naïve patients, largely contributing to the limited evidence on the issue from Italian real-life settings.12,13
Our data support the shift in the treatment of mCRPC observed in the last decade 10 when the approval of new therapeutic options for the disease leads to a progressive decline in the use of cytotoxic drugs in favor of highly effective novel antiandrogen agents. Because of the timeline of commercial availability of enzalutamide in Italy for the treatment of mCRPC, especially for chemotherapy-naïve patients, only a very few patients had received the drug in the present cohort; such patients were then not included in the current analysis.
According to the observational nature of the Registry, treatment choice depended on the physician preferences and the routine clinical practice of the participating centers, following approved local prescribing information. An unbalanced distribution of the baseline characteristics in the two treatment groups is therefore not unexpected. In the present study, docetaxel was mainly given to young patients with good performance status who could tolerate chemotherapy and who had aggressive disease. In any case, a considerable proportion of patients with analgesic use received first-line abiraterone acetate plus prednisone.
In this unselected cohort of chemotherapy-naïve mCRPC patients, which included subjects with comorbid conditions and poor prognostic features, median OS was not reached among patients treated with first-line abiraterone acetate plus prednisone (over a median follow-up of 22.1 months) and was 33.2 months among those treated with docetaxel (over a median follow-up of 25.3 months). Median TTP and PFS were, respectively, 14.4 and 13 months in the abiraterone acetate group and 8.2 and again 8.2 months in the docetaxel group. Because of the descriptive nature of the study, we did not conduct any formal comparison of the effectiveness of the two different treatments.
Although extreme caution is required in comparing our results to those from pivotal RTCs because of the different study setting and patients’ population, clinical outcomes of our patients receiving first-line abiraterone acetate plus prednisone in routine clinical practice were similar to those observed in the registrative COU-AA-302 trial, supporting the effectiveness of first-line abiraterone acetate plus prednisone for mCRPC in the real-life. Over a median follow-up of ̴49 months, the COU-AA-302 trial estimated a median OS of 34.7 months and 1- and 3-year OS rates of ~90% and ~50%, respectively; 14 in an analysis over a median follow-up duration of 27 months, the median radiographic PFS was 16.5 months. 15 Noteworthy, the COU-AA-302 trial was based on asymptomatic or mildly symptomatic men with good PS (ECOG PS 0-1), no visceral metastases and no relevant comorbidities, while, in the present study, over 75% of patients receiving abiraterone acetate plus prednisone had comorbidities and 15% reported analgesic use. In addition, 12% of our patients treated with abiraterone acetate plus prednisone had visceral metastases at treatment initiation. Although a negative prognostic role of visceral disease has been observed regardless of the treatment received, abiraterone acetate showed a survival benefit in RTCs in post-chemotherapy mCRPC 16 and metastatic hormone-sensitive prostate cancer. 17 In the overall analyses of the Prostate Cancer Registry, however, the median OS in first-line abiraterone acetate-treated patients was 27.1 in the overall cohort and 17.4 months in the subgroup of patients with visceral metastases 11 . Several real-world investigations from different countries, including Italy,12,13 supported the effectiveness of abiraterone acetate plus prednisone for the treatment of chemotherapy-naïve mCRPC patients outside the clinical trial setting. 18 In particular, the present results are in agreement with those from a large prospective Italian cohort of first-line mCRPC patients followed 3 years, in which abiraterone acetate plus prednisone showed good clinical (and patients’ reported) outcomes. 19
Our results on docetaxel are broadly comparable to those observed in a trial comparing the drug given as first-line treatment for mCRPC against cabazitaxel, which estimated a median OS of 24.3 months and a median PFS of 5.3 months in the docetaxel arm; 20 any comparison with the two 2004 landmark trials5,7 is likely to be misleading, because of the limited available treatment options for mCRPC and the different patients’ characteristics and disease management at the time of trials conduction.
A relatively high proportion of our patients had metastatic disease at initial diagnosis. According to the most recent international guidelines, ADT monotherapy is not recommended anymore in such patients.21,22 Nowadays the standard of care of the metastatic hormone-sensitive population is based on a more aggressive treatment approach besides long-term ADT before the development of castration resistance. 23 The addition of docetaxel, 24 abiraterone acetate plus prednisone,25,26 enzalutamide 27 or apalutamide 28 to ADT in the upfront treatment of hormone-sensitive metastatic prostate cancer is recommended due to a substantial improvement in patients’ survival.
Major strengths of the study include the prospective design, the robust monitoring, and the ad hoc collection of a large amount of data in a dedicated case report form, which limited inaccuracies and incompleteness of the information and ensured the standardization of data collection across the participating centers. The involvement of several major centers across the country and the inclusion of all consecutive patients meeting eligibility criteria, irrespective of their treatment, reduced selection bias and ensured a patient population representative of the typical mCRPC patients seen in the Italian clinical practice. In addition, the study was conducted during a period of remarkable progress in mCRPC treatment and has the potential to improve the understanding of the real-world performance of the newly approved agents for mCRPC.
Among the limitations, the present non-experimental study aims to descriptively document the characteristics and outcomes of patients receiving their first-line treatment for mCRPC in routine clinical practice and, although the baseline characteristics of patients were compared between treatment groups, no comparative effectiveness analysis was performed. An additional limitation is the lack of data on enzalutamide. The drug was made commercially available in Italy for the treatment of mCRPC in the chemotherapy-naïve setting in 2016, and we were therefore not able to provide reliable data on the outcomes of patients receiving enzalutamide in first-line.
In conclusion, the present investigation has provided original and valuable information on the effectiveness of abiraterone acetate plus prednisone and docetaxel in first-line mCRPC treatment, based on a patients’ population representative of routine daily clinical practice.
Footnotes
Acknowledgements
The authors acknowledge Carlotta Galeone, Ph.D., from Statinfo srl, for medical writing and editorial assistance based on the authors’ input and direction; writing assistance was funded by Janssen EMEA.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A. Sbrana: Novartis Pharma (editorial work); P. Beccaglia is an employee of Janssen SpA. All other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Janssen EMEA. Janssen EMEA contributed to the study design, the collection, analysis, and interpretation.
