Abstract
Objective
To evaluate the utility of intraoperative somatosensory evoked potential monitoring for detecting position-related brachial plexus injury during cerebellopontine angle tumor resection, identify independent risk factors, and assess the effectiveness of somatosensory evoked potential–guided interventions.
Methods
This retrospective cohort study included 45 patients who underwent cerebellopontine angle microsurgery in the lateral or semilateral position. Median nerve somatosensory evoked potentials were continuously monitored. Patients were stratified according to somatosensory evoked potential changes. Clinical characteristics, surgical variables, and protective positioning measures were compared. Cox regression was performed to identify independent risk factors.
Results
Somatosensory evoked potential alterations occurred in 14 patients (31.1%). Following repositioning, 13 patients recovered intraoperatively; 9 patients returned to baseline within 46–110 min (mean, 64 ± 20.5 min) and 4 patients within 2 h. Changes in latency and threshold across time points were statistically significant (p
Conclusions
Our findings suggest that routine somatosensory evoked potential monitoring during lateral decubitus cerebellopontine angle tumor resection may help mitigate brachial plexus injury and improve surgical safety.
Keywords
Introduction
The retrosigmoid approach is a commonly used surgical technique for treating lesions in the cerebellopontine angle (CPA).1–3 For this approach, patients with CPA lesions are most frequently placed in the lateral or lateral-prone position to optimize surgical exposure and facilitate instrument maneuverability. Compared with the sitting position, the lateral or lateral-prone position offers distinct advantages, including reduced venous pressure in the operative field, thereby minimizing venous bleeding, and a significantly lower risk of paradoxical air embolism, thus enhancing overall procedural safety. 4 However, maintaining these positions for prolonged durations is associated with inherent risks. Sustained compression of the axillary region or excessive traction resulting from improper limb positioning can induce mechanical stress and ischemic insult to the brachial plexus.5,6 In severe cases, this may result in complications such as pressure ulcers, traction injuries, and compressive neuropathies. Among these, brachial plexus injury is a particularly consequential position-related complication, potentially resulting in postoperative upper limb pain, sensory disturbances, muscle weakness, or functional deficits, all of which can adversely affect patient’s quality of life and rehabilitation. 7 Reported incidences of position-related brachial plexus injury in posterior fossa and cervicothoracic surgeries range from approximately 0.5%–10%, with risk notably elevated in lateral and prone positions. However, the exact incidence varies across studies due to differences in surgical type, duration of positioning, and use of monitoring modalities. Such injuries frequently result in prolonged recovery, lasting functional impairment, and potential medicolegal consequences. Consequently, their prevention remains a critical priority in intraoperative safety protocols in neurosurgical and anesthesiology practice.
To mitigate these risks, the implementation of objective and continuous intraoperative functional monitoring is essential. Upper limb somatosensory evoked potential (SEP) monitoring is a well-established neurophysiological technique that evaluates the integrity of sensory pathways by recording evoked potentials in the central nervous system following peripheral nerve stimulation.8,9 Its noninvasive nature, ability to provide real-time continuous monitoring, and high sensitivity to early neural ischemia and hypoxia have led to its widespread adoption in spinal procedures for the early detection of intraoperative nerve injury. Despite its established utility, there remains a paucity of focused clinical research evaluating the specific role of upper limb SEP monitoring in predicting brachial plexus injury during lateral or lateral-prone CPA surgeries. Key unanswered questions pertain to its monitoring efficacy, the optimal timing for intervention based on its readings, and the associated risk factors in this specific surgical context.
Therefore, this study aimed to investigate the early warning value of continuous upper limb SEP monitoring for detecting position-related brachial plexus injury during CPA tumor resection in patients placed in the lateral or semilateral position. Furthermore, we sought to analyze the associated risk factors contributing to such injuries and to evaluate the effectiveness of timely interventions guided by SEP findings. The ultimate objective was to generate robust evidence to inform strategies that enhance the safety profile of these complex surgical procedures.
Materials and methods
Study population
This single-center retrospective cohort study consecutively enrolled patients with CPA lesions who underwent microsurgery in the lateral or semilateral position with intraoperative SEP monitoring between March 2021 and March 2024. Inclusion criteria were as follows: (a) age ≥18 years; (b) elective surgery for CPA lesions in the specified positions; and (c) intraoperative bilateral median nerve SEP monitoring with stable, reproducible baseline waveforms. Exclusion criteria included the following: (a) pre-existing upper limb neurological deficits (e.g. brachial plexus injury and carpal tunnel syndrome); (b) systemic diseases affecting peripheral nerve conduction (e.g. diabetic neuropathy and chronic renal failure); or (c) incomplete SEP data due to equipment failure. Throughout the perioperative period (preoperative, intraoperative, and postoperative phases), continuous monitoring of SEPs of the upper limb median nerve was performed. Complete neurophysiological monitoring data were obtained from 45 patients (Table S1), comprising 28 males and 17 females, with a mean age of 56.38 years (range: 37–73 years), mean height of 1.66 m (range: 1.53–1.79 m), and mean body mass index (BMI) of 24.41 kg/m2 (range: 18.99–29.38 kg/m2). The mean operative time was 5.53 h (range: 3.9–7.2 h). All patients had no pre-existing upper limb neurological abnormalities. All patient data were anonymized to ensure that individual identities could not be ascertained. This study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 10 The study was conducted in accordance with the Declaration of Helsinki (1975), as revised in 2024, and approved by the Medical Ethics Committee of Nanfang Hospital, Southern Medical University (Approval Number: NFEC-2025-198). The requirement for written informed consent was waived.
Surgical positioning
Patients were positioned as follows: the lower arm on the affected side was extended and fixed on an armrest (parallel to the arm under pressure), whereas the upper arm was placed on an arm support (aligned with the shoulder peak). The arm support was positioned approximately 10 cm from the bed end on the same side armpit, preventing compression of the brachial plexus and vessels, with both arms forming a right angle with the body. A small soft pillow (approximately 5–7 cm thick, leaving 2–3 cm clearance under the armpit) or an air cushion wrapped in a cloth sheet was placed under the chest to reduce pressure on the lower arm. The lower leg was kept straight, and the upper leg was slightly flexed with a 4 cm thick cushion under the thigh (parallel to the femoral axis) to reduce vascular torsion and expand the pressure-bearing surface for better pressure distribution (Figure 1).
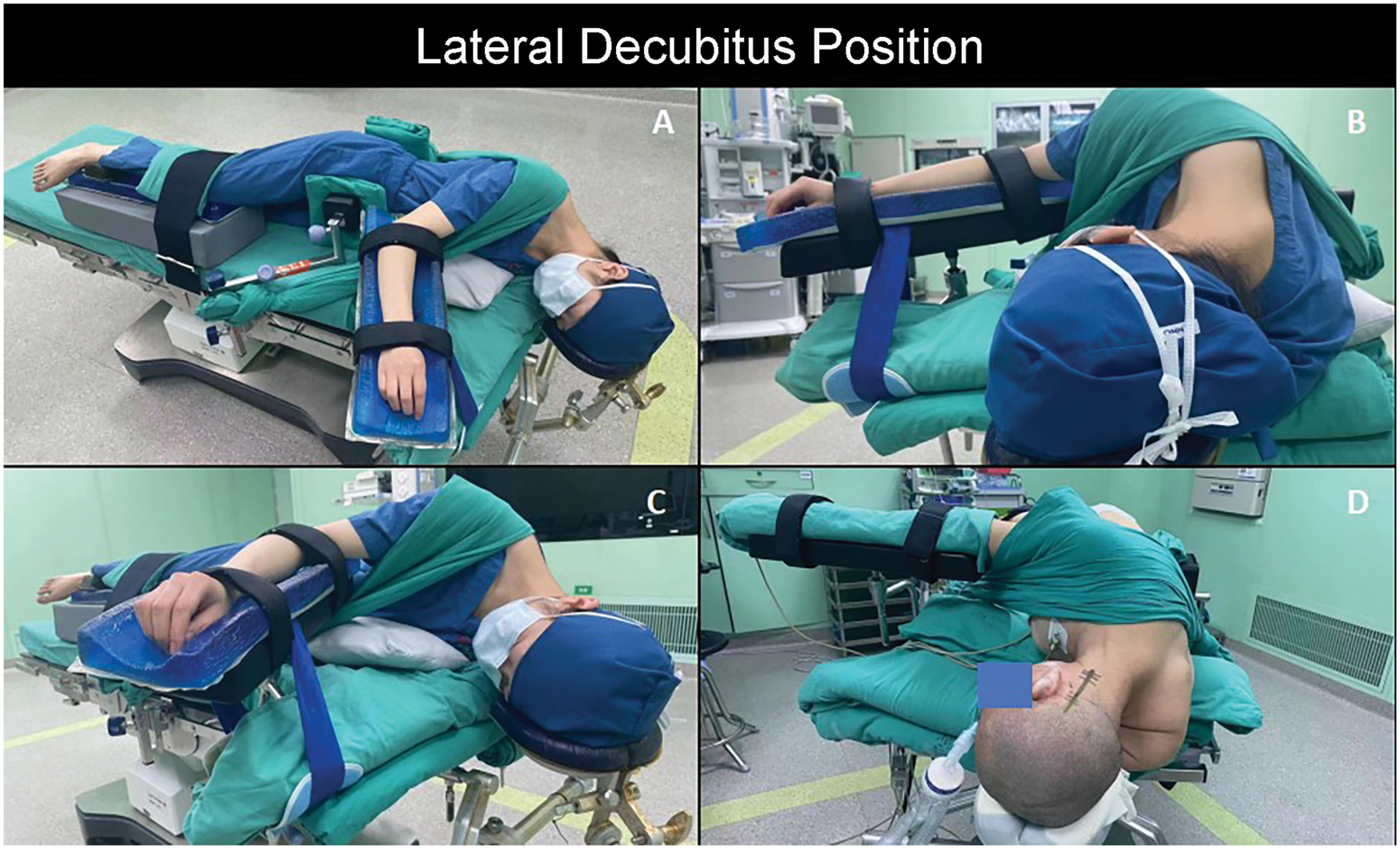
Schematic illustration of the lateral decubitus position for CPA surgery. Key steps in patient positioning are shown to minimize brachial plexus compression. (a) The lower leg was straightened, and the upper leg was slightly flexed with a cushion under the thigh to optimize pressure distribution. (b) The lower arm on the affected side was extended and fixed on an armrest, whereas the upper arm was supported on an armboard aligned with the shoulder. The armboard was positioned at approximately 10 cm from the bed end toward the axilla to prevent neurovascular compression, with both arms abducted at a right angle to the body. (c) A soft pillow or air cushion was placed under the chest to offload pressure, maintaining 2–3 cm clearance under the axilla. (d) Schematic diagram of operative positioning for patients undergoing CPA surgery. CPA: cerebellopontine angle.
Neurophysiological monitoring
Continuous monitoring of SEPs of the upper limb median nerve was performed using a neurophysiological monitoring system (Cadwell; USA). After positioning, electrodes were connected and baseline measurements were obtained. Bipolar electrodes were placed at the central inner wrist of the upper limb to stimulate the median nerve (constant-current square-wave pulses; pulse width, 0.2 ms; frequency, 2.1 Hz; intensity, 25 mA). Surface skin electrodes were used for recording, with electroencephalography (EEG) International 10–20 system positions C3, C4, and Fz as reference points for recording cortical responses to median nerve stimulation. Relevant parameters were recorded accordingly.
Criteria for evaluation
According to the classic 50/10 rule, a positive change was defined as a decrease in peak-to-peak amplitude >50% and/or an increase in latency >10% compared with baseline values. 11 The time from the start of surgery to the onset of SEP decline, as well as the time required for SEP recovery to baseline levels following intervention, were observed and recorded.
Patient grouping and data collection
Patients were categorized into groups based on the occurrence of acute brachial plexus injury. Baseline characteristics, including sex, age, operative duration, BMI, presence of intraoperative protective measures, and intraoperative temperature, were collected and compared between the injury group and noninjury group. This approach enabled a comprehensive evaluation of neurophysiological responses during lateral-position surgery, with the aim of identifying risk factors associated with brachial plexus injury and evaluating the effectiveness of SEP-guided interventions.
Intervention measures
Upon detection of positive monitoring indicators, prompt adjustment of patient positioning was undertaken. When direct compression was identified, measures such as removing padding, slightly elevating the shoulder or elbow, and performing passive limb movement were implemented. When monitored values failed to normalize, surgical procedures were temporarily paused to readjust the upper limb position, ensuring avoidance of excessive traction. Adjustment of shoulder supports and elbow positioning was performed to relieve upper limb pressure until monitoring values returned to normal.
Statistical analysis
Statistical analysis was conducted using Statistical Package for Social Sciences (SPSS) 27.0 (IBM Corp.; USA). Descriptive statistics for quantitative data were presented as mean ± SD. Comparison of SEP thresholds and latency periods before and after intervention were performed using paired t-tests or one-way analysis of variance. A significance level of p
Results
Clinical characteristics of the study cohort and comparison between SEP-positive and SEP-negative groups
This retrospective analysis included 45 patients who underwent CPA surgery in the lateral or semilateral position. The demographic and baseline clinical characteristics of the entire Nanfang Hospital (NFH) of Southern Medical University cohort are summarized in Table S1. The cohort comprised 68.9% males and 31.1% females (Figure 2(a)), with a mean age of 56.38 ± 8.98 years and a mean BMI of 24.41 ± 2.58 kg/m2. The mean operative time was 5.53 ± 0.84 h.

Comparison of clinical characteristics between SEP-positive and SEP-negative groups. (a–b) Sex distribution (M: male, F: female) in the overall cohort (n = 45) and SEP-positive subgroup (n = 14); (c) age comparison between the two groups; (d) weight comparison between the two groups; (e) height comparison between the two groups; (f) BMI comparison between the two groups; (g) operative time comparison between the two groups; (h) intraoperative body temperature comparison between the two groups. Statistical significance (p values) is indicated for each parameter, highlighting differences between patients with and without SEP changes. SEP: somatosensory evoked potential; BMI: body mass index.
Based on intraoperative median nerve SEP monitoring, patients were stratified into two groups: those exhibiting significant SEP changes (SEP-positive group, n = 14) and those without such changes (SEP-negative group, n = 31). Within the SEP-positive group, the majority were male (71.4%, Figure 2(b)). Comparative analysis between the groups revealed several statistically significant differences. Patients in the SEP-positive group were significantly older than those in the SEP-negative group (63.79 ± 5.56 vs. 53.03 ± 8.23 years, p
Comparison of SEP thresholds and latency periods
To quantitatively evaluate the impact of positional intervention on nerve function, we analyzed the SEP parameters (latency and threshold) across three key time points: pre-operation (baseline), pre-intervention (at the onset of SEP decline), and post-intervention. The analysis demonstrated statistically significant alterations in both parameters following positional compromise (Table S2). The latency of upper limb muscle potentials varied significantly across the three time points (p

Temporal changes in SEP parameters during surgery. (a) Latency (ms) measured at pre-operation, pre-intervention, and post-intervention time points; (b) threshold (μV) changes across the three time points.
Duration of upper limb muscle potential decrease and recovery time
The onset and reversal of SEP abnormalities were recorded to evaluate the dynamics of nerve compromise and recovery. Among the 14 patients who exhibited significant SEP amplitude decreases, the initial manifestation occurred at a mean duration of 3.30 ± 0.87 h (range: 1.5–5 h) after surgery initiation (Table S3). Upon detection of these changes, targeted interventions, primarily positional adjustments, were promptly implemented. This resulted in successful restoration of SEP signals to baseline levels in 13 of the 14 patients (92.9%). In most cases (n = 9, 64.3%), recovery was achieved within a mean time of 64 ± 20.54 min (range: 46–110 min) post-intervention. An additional 4 patients demonstrated complete recovery within 2 h. Notably, all 13 patients with SEP recovery exhibited no postoperative neurological deficits, confirming the functional effectiveness of the intervention (Figure 4(a)). One patient, who presented with a severe amplitude decrease of 90%, was an exception. Despite intervention, SEP signals did not normalize intraoperatively, and the patient subsequently developed transient postoperative brachial plexus symptoms, underscoring the correlation between the severity of intraoperative neurophysiological changes and clinical outcomes. Representative cases are shown in Figure 4(b).
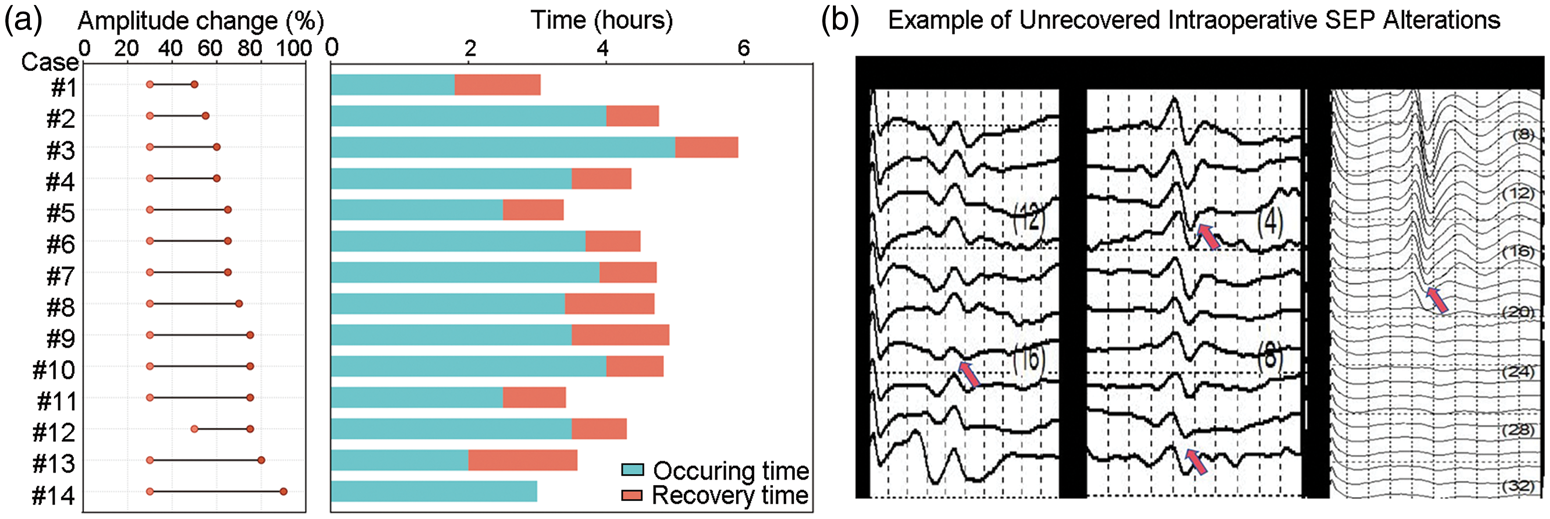
Monitoring SEP responses to intraoperative intervention. (a) Recovery timeline of SEP amplitude following positional adjustment in 14 cases; (b) representative waveforms from a patient in whom SEP did not recover intraoperatively. SEP: somatosensory evoked potential.
Univariate and multivariate analysis of intraoperative brachial plexus injury in patients undergoing surgery in lateral decubitus position
To identify predictors of intraoperative brachial plexus injury, we first performed a univariate analysis comparing patients with and without SEP changes. This initial screening revealed several factors significantly associated with injury, including advanced age (≥60 years), elevated BMI (≥25 kg/m2), prolonged operative time (≥5 h), the absence of intraoperative protective positioning measures, and intraoperative hypothermia (core temperature <36.5°C). In contrast, sex was not significantly associated with injury.
Factors demonstrating statistical significance in the univariate analysis were subsequently included in the multivariable Cox analysis, with variables coded as follows: operative duration (0, <5 h; 1, ≥5 h); BMI (0, <25 kg/m²; 1, ≥25 kg/m²); presence of intraoperative protective positioning measures (0, yes; 1, no); and intraoperative temperature (0, ≥36.5°C; 1, <36.5°C). The results indicated that operative duration ≥5 h, BMI ≥25 kg/m2, absence of intraoperative protective measures, and intraoperative temperature <36.5°C were independent risk factors for intraoperative brachial plexus injury in patients undergoing surgery in lateral decubitus position (p
Discussion
The CPA refers to the triangular region between the cerebellum, pons, medulla oblongata, and the petrous part of the temporal bone (internal auditory meatus). 12 It is a common site for intracranial tumors. Due to its deep location and numerous important structures, surgical treatment of CPA lesions remains challenging in neurosurgery. Patient positioning during CPA surgery is particularly specialized, with the most commonly used positions being the lateral decubitus or lateral-prone position, typically at 90°.1,2,6,12 In these positions, pressure is concentrated around the axilla and forearm. Given the prolonged duration of neurosurgical procedures, there is a risk of direct compression of the brachial plexus or compression of the axillary artery, resulting in ischemia of the brachial plexus. Prolonged compression may result in brachial plexus injury.5,13,14 In accordance with surgical positioning principles, clinical staff take precautions to avoid excessive traction and pressure on the upper limbs to prevent brachial plexus injury. However, in practice, some patients may still experience overstretching of the upper limbs, incorrect placement, or pressure on certain parts of the body, ultimately resulting in brachial plexus injury. Affected patients typically experience reduced muscle strength and function, significantly impacting their daily lives. Postoperatively, extensive rehabilitation is often necessary. Therefore, preventing brachial plexus compression in patients undergoing surgery in lateral decubitus or lateral-prone position is critically important.7,9 Current clinical strategies to prevent brachial plexus compression include the following: preoperative evaluation of shoulder joint and upper limb function to ensure full range of motion during positioning;15,16 adjustment of head frame height and position according to the physiological curvature of the neck to prevent excessive rotation and spinal cord injury; ensuring that the elbows are naturally flexed and that arm boards are appropriately positioned with arm abduction <90°, avoiding any weight bearing on the axillae, forearms, and hands; using cotton padding under the forearms to partially suspend the elbows; and actively moving the limbs during surgery to promote blood circulation and alleviate muscle spasms and tension, maintaining normal joint and muscle extension. Intraoperative nursing staff generally rely on their experience to assess whether specific body areas are subjected to excessive pressure; however, this approach is subjective and often lacks objective, scientifically based judgment.16–22
SEP reflect spinal nerve conduction function and can detect subtle or latent neurological abnormalities. 8 In clinical practice, SEP monitoring has become a primary modality for assessing nerve and spinal cord function. Studies have shown that multimodal intraoperative neurophysiological monitoring, including SEP combined with motor evoked potentials (MEPs), can effectively reduce the risk of position-related nerve injury during surgery.9,23 For example, Schwartz et al. 24 reported 1121 cases of spinal deformity correction using multimodal monitoring with SEP and MEP, achieving a sensitivity of 100%. Labrom et al. 25 demonstrated that intraoperative SEP monitoring of the bilateral median nerves during scoliosis surgery enabled prompt detection of position-related brachial plexopathy, and timely repositioning reversed SEP abnormalities and prevented postoperative neurological deficits. In our study, the 31.1% rate of SEP changes exceed typical clinical injury rates, likely attributable to the high sensitivity of continuous monitoring in detecting reversible dysfunction, the high-risk profile of our cohort (e.g. older age and higher BMI), and additional mechanical stress from prolonged CPA surgery positioning. Furthermore, studies suggests that simultaneous monitoring of SEP and MEP during surgery reveals significant changes in SEP waveforms preceding changes in MEP waveforms when neurological abnormalities occur.9,23,26–28 Therefore, comprehensive monitoring of upper limb SEPs is essential for the objective assessment of potential brachial plexus injury during CPA surgery. Upon detection of abnormal changes, prompt intervention is essential. Evaluation criteria typically include observing differences in SEP peak latency (P40) and asymmetric patterns in left and right P40 latencies. 29 According to Chiappa’s standards, abnormal SEP is defined as unilateral or bilateral waveform disappearance, prolonged peak latency, or asymmetrical peak latency.9,11,27,29 In our study, continuous SEP monitoring of the bilateral median nerves was introduced to objectively assess brachial plexus function throughout surgery (preoperative, intraoperative, and postoperative phases). Significant SEP decreases were most frequently observed 1.5–5 h after surgery initiation, suggesting a progressive process rather than an immediate event of brachial plexus compression or ischemia. Real-time monitoring of upper limb SEP is crucial during this process. Monitoring should be continued when upper limb SEP amplitude decreases >30% to effectively assess brachial plexus function. A decrease exceeding 50% in upper limb SEP amplitude suggests positive changes in neurological function. Our study identified 14 cases of position-related brachial plexus injuries based on SEP monitoring, where amplitude reductions ranged from 30% to 100%. Prompt positional adjustments and passive limb movements during surgery restored upper limb SEP to baseline levels in 64.29% of cases within 46–110 min following intervention, with remaining cases recovering within 2 h without symptomatic brachial plexus injuries. However, patients with a 90% decrease in SEP amplitude required longer recovery despite intervention and developed predominant postoperative symptoms, such as arm pain and weakness. With comprehensive rehabilitation and medical management, full neurological recovery was achieved within 3–6 months. Therefore, intraoperative SEP monitoring facilitates timely detection of position-related brachial plexus injuries during lateral decubitus positioning, preventing delays in diagnosing nerve injuries due to subjective impressions. Our findings underscore that position-related brachial plexus injury during CPA microsurgery is typically a transient and reversible nerve impairment. Despite longer recovery times in some cases, timely detection and appropriate intervention can effectively restore brachial plexus function in patients.
Upper limb median nerve SEP monitoring enables timely, accurate, and objective detection of nerve compression. Real-time adjustments can be made during repositioning upon detection of abnormal waveforms. For example, direct compression may necessitate removing padding or slightly elevating the shoulder. When monitoring values fail to normalize, suspending the patient with a frame adjusted forward until the chest is no longer compressed typically restores normal values. The position of shoulder pads during repositioning is closely associated with the degree of brachial plexus compression. The use of objective monitoring data for assessment and adjustment reduces reliance on subjective or experience-based practices. In this study, statistically significant differences were observed in preoperative, pre-intervention, and post-intervention upper limb MEP thresholds. This approach has been used in multiple cases, with only one case of minor brachial plexus injury that resolved rapidly and achieved satisfactory clinical outcomes. Identification of risk factors for brachial plexus injury in patients undergoing surgery in lateral decubitus position is essential for developing effective prevention strategies and informing preventive protocols. This retrospective comparative study identified several risk factors for intraoperative brachial plexus injury during lateral decubitus position surgery, including older age (≥60 years), BMI ≥25 kg/m2, and prolonged operative duration. Particularly, operative duration ≥5 h and BMI ≥25 kg/m² were significantly associated with injury occurrence. The underlying mechanism whereby these risk factors induce brachial plexus injury during lateral decubitus position surgery include the following: (a) advanced age (≥60 years) results in reduced bodily functions and organ performance, thereby decreasing tolerance to external forces; (b) BMI ≥25 kg/m2, which marks the threshold for overweight in adults, combined with the patient’s own weight, increases concentrated pressure during lateral decubitus positioning, thereby increasing the risk of pressure-related injuries; and (c) prolonged surgery duration is directly associated with a higher risk of intraoperative brachial plexus injury because of the extended compression time on the upper limbs, promoting ischemic reactions and exacerbating pressure-related injuries. Patients with intraoperative hypothermia are more likely to experience brachial plexus injury because hypothermia activates the sympathetic nervous system and causes vasoconstriction in the skin and subcutaneous tissue. Prolonged immobility and compression of local tissues can result in tissue hypoxia, necrosis, and subsequent injury. Our findings also offer a nuanced perspective on risk assessment. Although male patients constituted the majority of those with SEP changes, statistical modeling indicated that the observed risk was attributable to associated physiological factors (e.g. higher BMI) rather than sex itself. This underscores the importance of focusing preventive strategies on modifiable and quantifiable risk factors.
This study has several limitations. First, as a single-center retrospective observational study, it is inherently subject to selection and measurement biases. Although consecutive enrollment and strict inclusion and exclusion criteria were applied, selection bias related to surgeon preference, variability in monitoring protocols, or incomplete records cannot be entirely excluded. In addition, SEP waveforms were interpreted in real-time by intraoperative neurophysiologists without blinding, which may have introduced measurement bias. Second, the absence of standardized bilateral upper limb SEP baseline data acquired after patient positioning but before skin incision precluded comparison to rule out potential subclinical compression prior to surgery. Finally, the relatively small sample size and lack of long-term postoperative neurological follow-up limited comprehensive prognostic analysis. These limitations highlight the need for multicenter, prospective, blinded cohort studies to validate these findings.
Conclusion
This single-center retrospective cohort study indicates that intraoperative upper limb SEP changes are associated with factors such as age, BMI, and operative duration in patients undergoing CPA tumor resection in the lateral decubitus position. Most monitoring abnormalities were reversible after intervention, suggesting that SEP monitoring may serve as an early indicator of position-related brachial plexus injury. These findings provide preliminary evidence supporting the role of SEP monitoring as an early indicator of position-related brachial plexus injury. Larger prospective studies are warranted to confirm its clinical utility and to establish standardized intervention protocols.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261436656 - Supplemental material for Somatosensory evoked potential monitoring for position-related brachial plexus injury during cerebellopontine angle tumor resection: A retrospective cohort study
Supplemental material, sj-pdf-1-imr-10.1177_03000605261436656 for Somatosensory evoked potential monitoring for position-related brachial plexus injury during cerebellopontine angle tumor resection: A retrospective cohort study by Xian Jiang, Yanyi Yin, Xiaomin Hou, Jun Liu and Peng Li in Journal of International Medical Research
Footnotes
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest relating to this study.
Funding
This research received no funding support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
