Abstract
Objective
Peptic ulcer hemorrhage is associated with considerable morbidity and mortality. This study aimed to evaluate the role of improved endoscopic purse-string suturing for the treatment of peptic ulcer hemorrhage.
Methods
This prospective, randomized, controlled, single-blind study was conducted at two institutions; we compared the improved endoscopic purse-string suturing with the technique of monopolar hemostat forceps with soft coagulation for the treatment of peptic ulcer hemorrhage. The primary outcome was the post-procedural re-bleeding rate. The secondary outcomes included initial hemostat success rate, adverse events, volume of transfused blood, cost of hospital stay, and length of hospital stay.
Results
In total, 114 patients were included in the study, and each patient was randomly assigned to 1 of the 2 treatment groups (each group, n = 57). Procedure time, re-bleeding rates, initial hemostat success rates, duration of hospital stay, volume of transfused blood, and cost of hospital stay were similar between the two groups (p > 0.05). Among the 37 patients with duodenal ulcers, the re-bleeding rate was significantly lower in the improved endoscopic purse-string suturing group (0/21) than in the monopolar hemostat forceps with soft coagulation group (3/16) (p < 0.05).
Conclusion
The improved endoscopic purse-string suturing demonstrated a high technical success rate and showed comparable efficacy and safety outcomes to monopolar hemostat forceps with soft coagulation in the treatment of peptic ulcer hemorrhage.
Keywords
Introduction
Upper gastrointestinal hemorrhage (UGIH) is a common cause of hospitalization worldwide. 1 Peptic ulcers (PUs), the most common cause of UGIH, account for 28%–59% of these hospitalizations in large database studies.1–3 The development of effective pharmaceutical therapy has reduced the PU prevalence. However, peptic ulcer hemorrhage (PUH) continues to be associated with considerable mortality, ranging from 5% to 13%.1–3
The Forrest classification of recent hemorrhage stigmata was developed to classify lesions, which would aid in stratifying the risk of further bleeding and identify the most appropriate treatment strategy.1,4–6 Performing early endoscopy (within 24 h) and treating high-risk endoscopic stigmata are key to the successful management of acute PUH and prevention of re-bleeding. 5 Traditional endoscopic therapies for PUH include epinephrine injection, mechanical therapy with clips, and monopolar hemostat forceps with soft coagulation (MHFSC).7–13 However, no single method is superior to another. The safety and efficacy of these methods for high-risk PUH are limited and may result in various adverse events. 7 Furthermore, patients who fail to respond to endoscopic therapies may require emergent arterial embolization or surgery.
Endoscopic purse-string suturing (EPSS), which uses an endoloop and several metallic clips, was initially developed as an endoscopic suturing technique for closing iatrogenic gastrointestinal mucosal defects. 14 A dual-channel endoscope is required to perform the EPSS procedure. However, in China, dual-channel endoscopes are only available in a few large hospitals due to their expensive purchase and maintenance costs. Thus, we adapted an improved EPSS (IEPSS) method for use with a single-channel endoscope to treat PUH. To the best our knowledge, there is a dearth of data on the efficacy and safety of the IEPSS technique for PUH, and we aimed to address this knowledge gap in this randomized, controlled study.
Materials and methods
Study design and approval
This prospective, randomized, controlled, single-blind study compared the efficacy and safety of IEPSS and MHFSC in PUH treatment. Data were collected from all consecutive patients who underwent endoscopy for PUH at two institutions between July 2020 and June 2021. The study design has been described in Figure 1. This study was conducted in accordance with the ethical principles of the Declaration of Helsinki (1975, as revised in 2024). The study protocol was approved by the Clinical Research Ethics Committee of Changshu Affiliated Hospital of Soochow University and Clinical Research Ethics Committee of Zhangjiagang People’s Hospital. Additionally, all experimental procedures and methods described in this study were performed in strict accordance with the relevant guidelines and regulations for biomedical research involving human participants. All patients provided written informed consent prior to study participation. The study was registered with clinicaltrials.gov (ChiCTR2000034015). The reporting of this study conforms to the Consolidated Standards of Reporting Trials (CONSORT) statement. 15
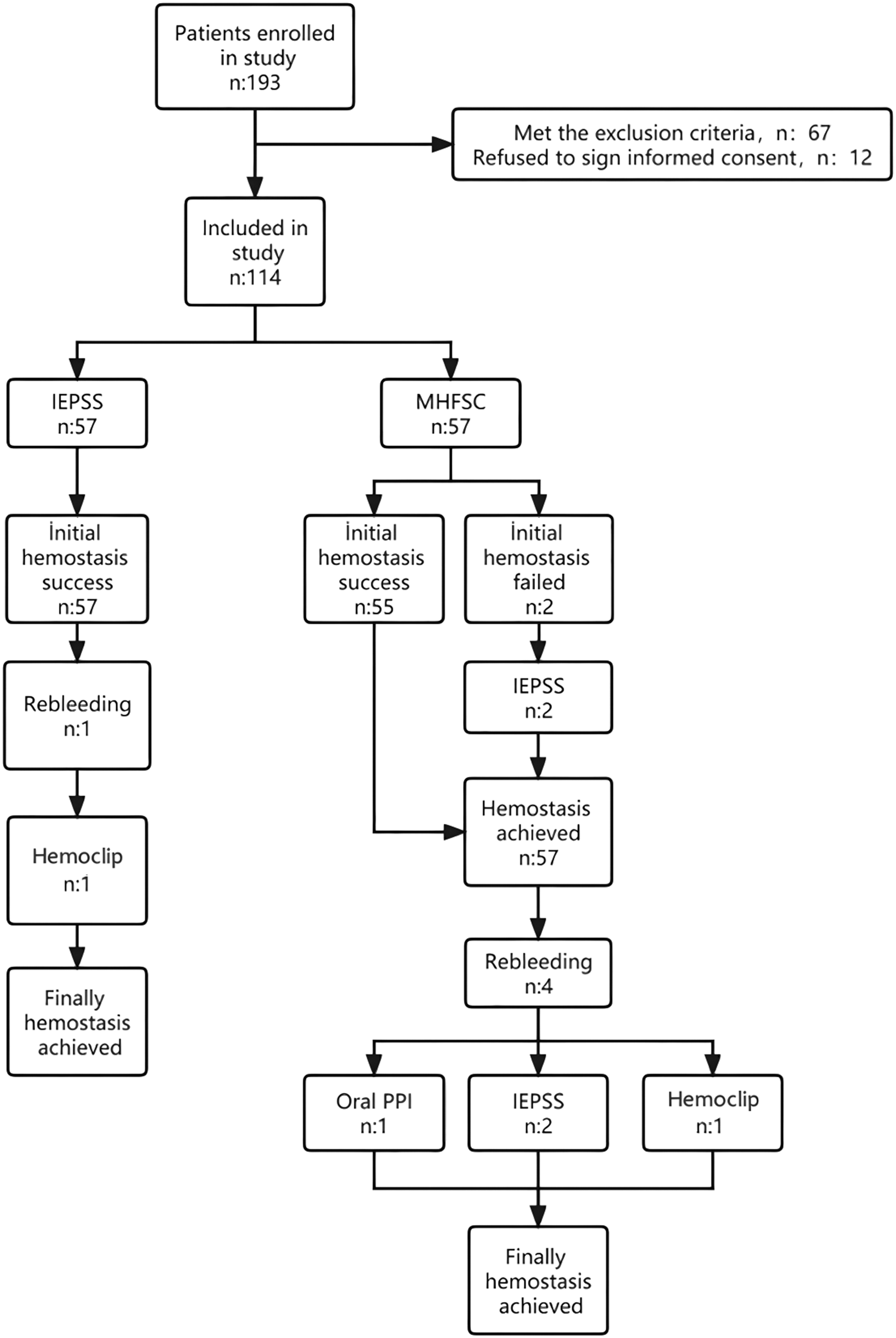
Flowchart of patient selection. IEPSS: improved endoscopic purse-string suturing; MHFSC: monopolar hemostat forceps with soft coagulation; PPI: proton pump inhibitor.
Patients
Consecutive patients with UGIH who had undergone emergent endoscopy (24 h) that confirmed the presence of Forrest classification Ia/Ib/IIa gastric or duodenal ulcer were invited to participate (Figure 1). The exclusion criteria were as follows: (a) malignant ulcer; (b) history of Billroth I/II or Roux-en-Y reconstruction; (c) bleeding diathesis with a platelet count <50.000/mm3, international normalized ratio (INR) >1.5, or requiring continued antiplatelet or anticoagulant therapy; (d) implantation of pacemakers and implantable cardiovascular-defibrillators, severe cardiopulmonary insufficiency, or hepatic or renal failure; and (e) age <18 years, pregnancy, or refusal to provide informed consent.
Randomization
Eligible patients were recruited by investigators who explained the study and obtained written, informed patient consent before the procedure. After PUH (Ia, Ib, and IIa ulcers) was confirmed, each patient was randomly allocated in a 1:1 ratio to either the IEPSS or MHFSC group by using computer-generated numbers placed in sealed envelopes. A nurse who did not participate in the evaluation of the study outcome opened the envelopes immediately before initiation of endoscopic hemostasis, and patients were blinded to the hemostat modalities allocated.
Endoscopic procedure
All endoscopic procedures were performed by two endoscopy specialists (Xu and He), each with >10 years of endoscopic hemostasis experience. Endoscopic therapy was administered to patients with Forrest Ia/Ib/IIa gastric or duodenal ulcers. MHFSC was performed using monopolar hemostat forceps (Coagrasper: FD-410LR; Olympus®, Tokyo, Japan). All vessels (with or without bleeding) on the ulcer were coagulated using hemostat forceps operating in the soft coagulation mode (80 W; VIO 300 D; Erbe Medical Systems®). The closed forceps were gently pressed onto the target and an electric current was applied for 1–2 s. The procedure was repeated until hemostasis was achieved.
IEPSS was performed using a single-channel therapeutic endoscope (Figure 2). Figure 3 illustrates the composition of the endoloop and its operational method in vitro. In the preparation stage, a transparent cap (ND-201-11802, Olympus®, Tokyo, Japan) was attached to the front of the single-channel therapeutic endoscope (GIF-260J, Olympus®, Tokyo, Japan). A rotatable and reopened metallic clip (ROCC-D-26-195, Micro-Tech®, Nanjin, China) was inserted into the biopsy channel of the endoscope. After grasping an endoloop (Loop-15/20/30, LEOmed®, Changzhou, China) in vitro, the clip was retracted into the transparent cap. Then, the endoloop pusher leaned against the outside of the endoscope. In the operation stage, the chief endoscopist inserted the endoscope and the endoloop pusher simultaneously into the gastric cavity. The endoloop was loosened by controlling the handle of the pusher and anchored onto the distal margin of the ulcer with the clip followed by the insertion of three to six additional clips to anchor the endoloop to different sides of the margin. Finally, the endoloop was tightened by slight pulling of all the clips together. Additional clips were used if any clip was not accurately positioned or the purse-string suture was not tight.

Endoscopic purse-string suturing of peptic ulcer. (a) An ulcer located in the antrum of the stomach, Forrest classification IIa, diameter >2 cm. (b) Endoscopic purse-string suturing procedure. (c) Ulcer oversewing completed and (d) visualization of the ulcer with second-look endoscopy.

The composition of the endoloop and its operational method in vitro. (a) Endoloop. (b) The pushing device of the endoloop. (c) The hook at the tip of the pushing device. (d) Engagement of the endoloop by the pusher hook. (e) Application of moderate tension via the pusher to stabilize the endoloop. (f) A titanium clip is extended through the endoscopic biopsy channel. (g) Clip grasping the endoloop and (h) retraction of the clip into the endoscope channel, positioning the pusher parallel to the endoscope shaft.
All procedures were performed under general anesthesia. Intravenous general anesthesia was administered by an anesthesiologist, with routine endotracheal intubation for airway protection. This ensured that patients were unconscious and immobile during the procedure, providing a stable operating condition for the endoscopist. Consequently, although the IEPSS technique involves an increased diameter during passage through the pharynx under general anesthesia with airway protection, no patient experienced discomfort or complications related to the procedure itself; furthermore, there were no insertion-related injuries (e.g. pharyngeal mucosal injury).
If the allocated method failed, additional hemostasis methods, including the alternative modality (MHFSC or IEPSS) or application of clips, were employed, according to the endoscopist’s judgment. If hemostasis was still not achieved, intervention radiology or surgical treatment was planned. After endoscopic hemostasis surgery, both groups of patients were administered the same pharmacological treatment, which mainly included the use of proton pump inhibitors (PPIs) and hemostatic drugs as well as correction of electrolyte imbalance and fluid replacement therapy.
Outcome measures and study definitions
Re-bleeding was defined as patient complaint of recurrent tarry stools or hematemesis within 30 days after initial hemostasis and was confirmed when emergency endoscopy showed active bleeding from the ulcers. To ensure complete follow-up, all patients were contacted by telephone 30 days after the initial endoscopic hemostasis. During the call, patients or their families were systematically asked about any recurrent symptoms such as hematemesis, melena, and hematochezia. If re-bleeding was suspected, the patient was advised to return to the hospital immediately for confirmatory emergency endoscopy and appropriate management. Initial hemostasis success was defined as no active bleeding from the target lesion after treatment with the single allocated method and hemostasis confirmation on a repeat endoscopy performed 24 h later. Adverse events were classified according to the American Society for Gastrointestinal Endoscopy (ASGE) guidelines. 16
The primary outcome of the present study was the re-bleeding rate associated with the two allocated methods. Secondary outcomes were the initial hemostat success rate, adverse events, volume of transfused blood, cost of hospital stay, and length of hospital stay.
Statistical analyses
All statistical analyses were performed based on “intention-to-treat” (ITT) analysis. In our study, the experimental group comprised patients who received the IEPSS treatment, while the control group included those treated with MHFSC. Intergroup comparisons were made to evaluate the efficacy of IEPSS relative to that of the standard MHFSC treatment. We compared treatment effects between the groups using risk difference (RD) and its 95% confidence intervals (CIs). We also calculated the risk ratios (RRs) and their 95% CIs from study probabilities. Chi-square test or Fisher’s exact test was used to compare categorical variables between the experimental and control groups, while the unpaired Student’s t-test or nonparametric Mann–Whitney U test was used to compare continuous variables. All tests were two-tailed. A p-value <0.05 was considered statistically significant.
Results
In total, 193 consecutive eligible patients were recruited at our hospital and randomly assigned to either the IEPSS or MHFSC group. A total of 67 patients were excluded because they failed to meet the inclusion criteria, and 12 were excluded because they refused to sign the informed consent form. Thus, 114 patients were finally included in the study, and each was randomly assigned to 1 of the 2 groups (n = 57 per group) (Figure 1).
Baseline data and endoscopic procedure characteristics
The two groups were comparable in terms of baseline characteristics, including Rockall scores. The ulcer characteristics observed on the first endoscopic examination were also similar (Table 1).
Baseline characteristics of patients and peptic ulcers.

IEPSS: improved endoscopic purse-string suturing; MHFSC: monopolar hemostat forceps with soft coagulation; NSAID: non-steroidal anti-inflammatory drug; H. pylori: Helicobacter pylori; PU: peptic ulcer.
Primary and secondary outcomes
In total, five patients, one (1.8%) in the IEPSS group and four (7.2%) in the MHFSC group (RD: −0.053, 95% CI: −0.257 to 0.152 and RR: 0.167, 95% CI: 0.021 to 1.316; p = 0.3600), experienced re-bleeding during the 30-day follow-up period (Table 2).
Comparison of the treatment results between the two groups.

IEPSS: improved endoscopic purse-string suturing; MHFSC: monopolar hemostat forceps with soft coagulation; RMB: people’s renminbi; CNY: Chinese Yuan; CI: confidence interval.
Initial hemostasis was attained in 57 (100%) patients treated with IEPSS and 55 (96.5%) treated with MHFSC (RD: 0.018, 95% CI: −0.0184 to 0.219 and RR: 1.036, 95% CI: 0.676 to 1.590; p = 0.4756). In two patients for whom hemostasis was not achieved after MHFSC, IEPSS was subsequently performed, and hemostasis was finally achieved. No adverse events such as perforation, apnea, cardiac arrest, and worsening of bleeding due to the hemostat procedure were observed in either group.
The procedure time (p = 0.7789), duration of hospital stay (p = 0.1392), and cost of hospital stay (p = 0.2348) were similar between the two groups. The mean volume of transfused blood (number of units of blood) after endoscopic procedure was 3.34 ± 1.65 units in the IEPSS group and 2.91 ± 1.77 units in the MHFSC group (p = 0.1824) with no difference between the two groups either before or after endoscopic treatment.
The re-bleeding rates according to the characteristics of the ulcer are shown in Table 3. There were no differences in the re-bleeding rates of patients with gastric ulcer between the two groups. Among 37 patients with duodenal ulcer, the re-bleeding rate for patients who underwent IEPSS (0/21, 0%) was lower than that for those who underwent MHFSC (3/16, 18.7%, RD: −0.188, 95% CI: −0.554 to 0.179, p = 0.0385). No differences were detected in the Forrest classification between the two groups.
Recurrent bleeding ulcer characteristics.
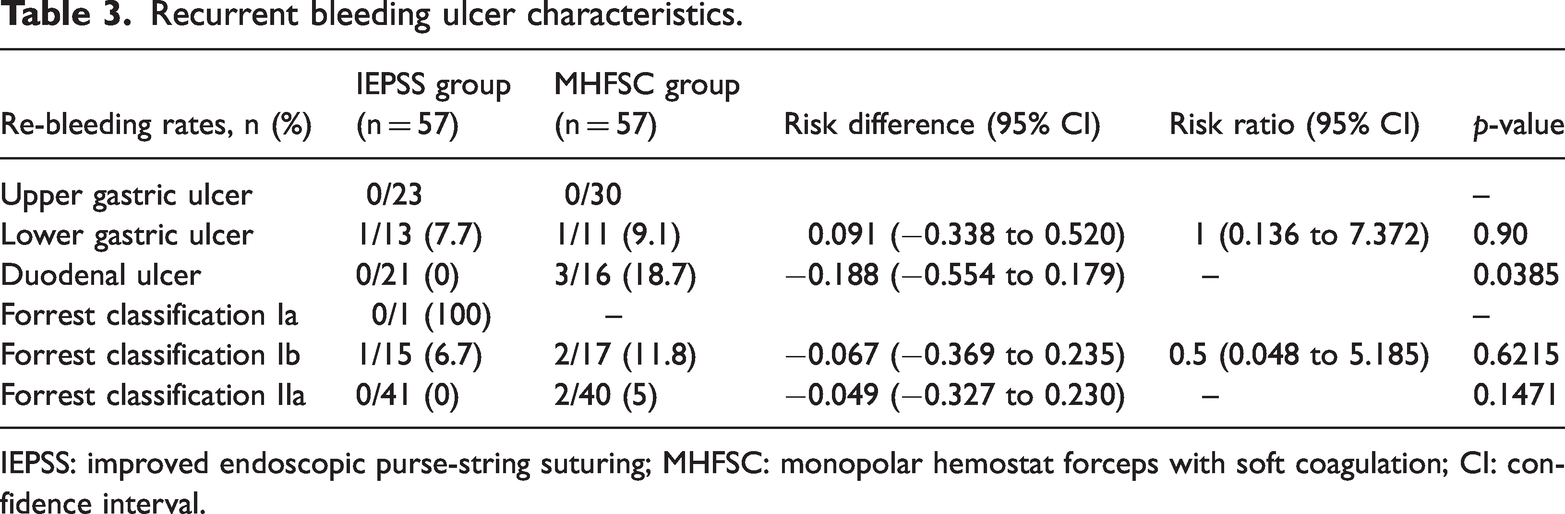
IEPSS: improved endoscopic purse-string suturing; MHFSC: monopolar hemostat forceps with soft coagulation; CI: confidence interval.
Re-bleeding occurred in one patient in the IEPSS group and was successfully managed using hemoclips during emergency endoscopy. In the MHFSC group, four patients experienced re-bleeding; of these, one responded to conservative treatment with oral PPIs, two were treated successfully with salvage IEPSS, and one with hemostatic clips. All re-bleeding events were resolved using these endoscopic or medical measures, and no patient required radiological or surgical intervention.
Discussion
Conventional hemostasis modalities are not always able to achieve hemostasis. 17 For a fibrotic ulcer base, application of hemoclips is technically difficult, and repeated application of the heater probe is associated with a risk of thermal injury and perforation because scar tissue may adhere to the heater probe and detach upon probe removal. 18 MHFSC is a relatively new, affordable method that does not involve such risks.8,11–13,19–21 Compared with methods involving other contact thermal devices, MHFSC is performed at a lower voltage that may not cause coagulation when the outer surface of the bleeding vessel is covered with excessive residual blood or fluids such as the water instilled through the working channel of the endoscope.
Recently, three case reports22–24 and one pilot study 18 have described the endoscopic suturing device (Overstitch TM, Apollo Endosurgery, Austin, TX, United States) as a novel endoscopic modality for PUH treatment that has a high technical success rate and provides desirable clinical results in terms of achievement of hemostasis and prevention of recurrent bleeding. Limitations of the Overstitch device include high costs as well as the need for a double-channel endoscope and expert endoscopic skills. Therefore, we performed EPSS under single-channel endoscopy and reserved the OverStitch for treatment of high-risk PUH. To the best of our knowledge, the current prospective randomized study is the first to evaluate the efficacy and safety of IEPSS compared with those of MHFSC for PUH treatment. Purse-string suturing was technically successful in all cases. In case of duodenal ulcers, the re-bleeding rate was significantly lower with IEPSS than with MHFSC. We speculate that this phenomenon is associated with the characteristic anatomy and function of the duodenum, which is exposed to frequent reflux of pancreatic juice and bile. In theory, purse-string suturing achieves permanent closure of the ulcer, keeps the bleeding site relatively fixed, and prevents corrosion caused by digestive fluids.
The 100% technical success rate achieved with IEPSS in duodenal ulcers is particularly noteworthy. The duodenum presents unique anatomical and physiological challenges—its relatively fixed position, proximity to the pancreas and bile ducts, and constant exposure to pancreaticobiliary secretions can adversely impair the efficacy of conventional hemostatic methods. Thermal probes may be less effective due to tissue friability and fluid interference, while clip application can be technically difficult in fibrotic or posterior wall ulcers. IEPSS, by mechanically approximating the ulcer margins using an endoloop and clips, creates a stable, watertight closure that physically protects the bleeding site from luminal content and digestive fluids. This method that creates a mechanical barrier is likely to provide greater resistant to the erosive effects of bile and pancreatic juice compared with coagulation-based methods. Therefore, the high success and low re-bleeding rates observed in our duodenal ulcer subgroup suggest that IEPSS is particularly suited, and perhaps considered a primary endoscopic option, for bleeding duodenal ulcers, especially those with high-risk stigmata or at unfavorable locations.
Upon further analysis, it is evident that the total cost of the procedure is an important consideration when evaluating treatment options for PUH. In our study, the IEPSS method not only demonstrated a high technical success rate but also offered significant economic advantages compared with the MHFSC technique. Specifically, the cost of materials used in the IEPSS procedure, including 1 endoloop and 3–6 metallic clips, ranges from 1000 to 2000 Chinese Yuan (CNY), which is considerably lower than the cost of the monopolar hemostat forceps used in MHFSC, priced at approximately 2500 CNY. This cost difference makes IEPSS a more economically viable option, especially in resource-limited settings, where cost-effectiveness is a crucial factor in healthcare decision-making. In conclusion, IEPSS not only provides a technically successful treatment option for PUH but also involves a significantly lower total procedure cost.
Several new endoscopic methods have been developed for PUH treatment. One is the over-the-scope clip (OTSC) system, which has been described as an effective and safe rescue therapy for refractory ulcer bleeding at anatomic locations that are difficult to treat using traditional methods. 25 However, OTSC is technically challenging in case of a large or fibrotic ulcer and must only be used for small, shallow, and nonfibrotic ulcers. 26 Hemostasis spray (HS; Cook Medical, Winston–Salem, NC, United States), also known as HS or TC-325, is an absorptive inorganic powder that coalesces and adheres to the bleeding site, forming a mechanical barrier. 27 Although hemospray is reported to be effective in controlling immediate bleeding, with hemostasis rates ranging from 76% to 96%, high re-bleeding rates of up to 29%–38% in benign nonvariceal upper gastrointestinal bleeding (NUGVB) have been reported.28–30 The ideal role for hemospray would be as part of combination or rescue therapy to optimize bleeding control. The European Society of Gastrointestinal Endoscopy (ESGE) recommends transcatheter arterial embolization (TAE) or surgery for patients with recurrent bleeding following failure of a second endoscopic attempt, supported by a high level of evidence. 1 In a multicenter, retrospective, real-life study, 31 TAE has demonstrated very high technical and clinical success rates in managing severe bleeding caused by PUs that had not responded to endoscopic hemostasis. However, TAE requires the use of specialized equipment and techniques and the support of experienced interventional radiologists who are trained to perform the procedure safely and effectively. Therefore, not all medical institutions are able to offer TAE as a treatment option to their patients.
The parallel insertion technique described in this study, although effective, does entail a distinct learning curve and requires specific technical proficiency. Simultaneously maneuvering the endoscope and the external pusher demands coordinated bimanual dexterity and spatial awareness, which differs from the ergonomics involved in conventional through-the-scope (TTS) methods. In our experience, endoscopists already skilled in advanced therapeutic procedures (e.g. endoscopic submucosal dissection) may adapt more readily. We recommend that clinicians interested in adopting IEPSS undergo dedicated simulation training and initial proctoring to safely master the hand-eye coordination and instrument handling required to perform this technique, thereby minimizing the procedure time and potential technical complications likely during the early learning phase.
The main limitation of this study is that it was a pilot study, and we had no previous data to calculate the sample size. This may have resulted in a sample size that is too small to provide sufficient power. Additionally, there are certain limitations associated with the use of IEPSS in PUH. The major concern is the requirement of extensive expertise and training for performing the oversewing procedure. When less experienced endoscopists perform the procedure, it may prolong the procedure time and raise the risk of adverse events.
This study demonstrated that the novel endoscopic hemostasis method of IEPSS offers a high technical success rate and appears as reliable and safe as MHFSC in PUH treatment. This modality reduces the re-bleeding rate of duodenal ulcers compared with MHFSC. Our findings show that IEPSS can be used as the initial endoscopic option for PUH in duodenal ulcers. We recommend that larger, controlled, multi-site studies be performed to further explore the safety and effectiveness of the IEPSS technique for PUH.
Footnotes
Acknowledgments
None.
Author contributions
Jie Xu, Bin He, and Xiaodan Xu contributed to data collection and writing. Luojie Liu and Xiaodan Xu were responsible for statistical analyses. Luojie Liu contributed to revising this dissertation. Luojie Liu and Xiaodan Xu managed this project. All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
Consent to publish declaration
Not applicable.
Consent to participate
All participants provided informed consent to participate in this study.
Data availability statement
The datasets generated and/or analyzed during the current study are not publicly available due to concerns regarding patient privacy and the sensitive nature of the medical data collected. The data contain detailed patient information, including clinical histories and personal identifiers, which require strict confidentiality and protection under applicable data protection laws and ethical guidelines. However, the datasets are available from the corresponding author on reasonable request, subject to the completion of a data use agreement and adherence to all relevant ethical and legal requirements. This ensures that the data are used responsibly and only for legitimate research purposes.
Disclosures
Jie Xu, Bin He, Xiaodan Xu, and Luojie Liu disclose no conflicts of interest for this article.
Ethics statement
The study protocol was approved by the Institutional Review Board of Changshu Affiliated Hospital of Soochow University and the Institutional Review Board of Zhangjiagang People’s Hospital. Informed consent was obtained from all patients; all authors had access to the study data and reviewed and approved the final manuscript. The study was registered with ClinicalTrials.gov (ChiCTR2000034015). The study data were maintained with confidentiality to ensure the privacy of all participants.
Funding
This study was supported by the Changshu Science and Technology Program (CY202339); Suzhou Youth Science and Technology Project for the Advancement of Science, Education, and Health (KJXW2023067); Specialized Project for Diagnosis and Treatment Technologies of Key Clinical Diseases in Suzhou City (LCZX202220); and Suzhou 23rd Batch of Science and Technology Development Plan (Clinical Trial Institution Capability Enhancement) Project (SLT 2023006).
